DIFAMILAST- adquey ointment
Difamilast by
Drug Labeling and Warnings
Difamilast by is a Prescription medication manufactured, distributed, or labeled by Acrotech Biopharma Inc, Contract Pharmaceuticals Limited, Contract Pharmaceutical Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ADQUEY safely and effectively. See full prescribing information for ADQUEY.
ADQUEY™(difamilast) ointment, for topical use
Initial U.S. Approval: 2026
INDICATIONS AND USAGE
ADQUEY is a phosphodiesterase 4 inhibitor indicated for the topical treatment of adults and pediatric patients 2 years of age and older with mild to moderate atopic dermatitis. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Ointment: 1%. (3)
CONTRAINDICATIONS
None. (4)
ADVERSE REACTIONS
The most common adverse reaction occurring in ≥1% of subjects is nasopharyngitis. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Acrotech Biopharma Inc. at 1-888-292-9617 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS & USAGE
2 DOSAGE & ADMINISTRATION
3 DOSAGE FORMS & STRENGTHS
4 CONTRAINDICATIONS
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
6.2 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS & USAGE
- 2 DOSAGE & ADMINISTRATION
- 3 DOSAGE FORMS & STRENGTHS
- 4 CONTRAINDICATIONS
-
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ADQUEY was assessed in two double-blind, vehicle-controlled clinical trials (Trial 2 and Trial 3) that enrolled 532 adult and pediatric subjects 2 years of age and older in Japan with mild to moderate atopic dermatitis (AD). Subjects applied ADQUEY or vehicle ointment topically twice daily for 4 weeks [see Clinical Studies (14)]. Adverse reactions reported by ≥1% of ADQUEY-treated subjects and more frequently than in subjects receiving vehicle are listed in Table 1.
Table 1: Adverse Reactions Occurring in ≥1% of Adult and Pediatric Subjects 2 Years of Age and Older Treated with ADQUEY for Mild to Moderate Atopic Dermatitis (and Greater than Vehicle) through Week 4 in Trials 2 and 3
Adverse Reaction ADQUEY
(N=267)
n (%)Vehicle
(N=265)
n (%)
Nasopharyngitis 16(6) 10(4) Less common (<1%) adverse reactions in subjects treated with ADQUEY in Trials 2 and 3 included application site folliculitis, contact dermatitis, application site rash, and molluscum contagiosum.
In Trial 1, a vehicle-controlled dose ranging trial, 43 subjects 10 years of age and older in the United States, Australia, and Poland received ADQUEY topically twice daily for 8 weeks and the safety profile was consistent with Trials 2 and 3.
In two additional vehicle-controlled dose ranging trials (Trial 4 and Trial 5), 92 subjects 2 years of age and older in Japan received ADQUEY topically twice daily for 4 weeks (Trial 4) and twice daily for 8 weeks (Trial 5) and the safety profile was consistent with Trials 2 and 3.
In open-label trials of both Japanese and United States (US) subjects, 857 adult and pediatric subjects continued twice-daily treatment with ADQUEY for up to 52 weeks. The following application site adverse reactions occurred that led to drug discontinuation: pain, pruritus, vesicles, blistering, erythema, burning and contact dermatitis.6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ADQUEY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
General disorders and administration site condition: application site swelling. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The available data on the use of topical difamilast during pregnancy are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes.In animal reproduction studies, difamilast administered subcutaneously to pregnant rats and rabbits during the period of organogenesis produced no fetal structural abnormalities at doses up to 30 and 3 times the maximum recommended human dose (MRHD), respectively. Difamilast induced increased post-implantation loss, decreased fetal weight, retarded ossification and increased visceral abnormalities in rats at subcutaneous dose 263 times the MRHD. Difamilast induced increased skeletal variations in rabbits at subcutaneous doses 14 times the MRHD (see Data). The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss, and other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In an embryo-fetal development study, pregnant rats were dosed subcutaneously during the period of organogenesis with up to 100 mg/kg/day difamilast (263 times the MRHD based on AUC comparison). Increased post-implantation loss, decreased fetal weight, retarded ossification, and increased visceral abnormalities (membranous ventricular septum defect) were noted at 100 mg/kg/day (263 times the MRHD based on AUC comparison). No embryo-fetal toxicity was observed at 10 mg/kg/day (30 times the MRHD based on AUC comparison).In an embryo-fetal development study in rabbits, pregnant does were dosed subcutaneously during the period of organogenesis with up to 3 mg/kg/day difamilast (14 times the MRHD based on AUC comparison). Increased skeletal variations (increased supernumerary lumbar vertebra) were observed at 3 mg/kg/day (14 times the MRHD based on AUC comparison). No embryo-fetal toxicity was observed at 1 mg/kg/day (3 times the MRHD based on AUC comparison).
In a pre- and post-natal development study in rats, dams were dosed subcutaneously with up to 3 mg/kg/day difamilast during the periods of organogenesis and lactation. No treatment-related adverse effects on reproductive functions of dams, or pre- and post-natal development of offspring were observed at 3 mg/kg/day (13 times the MRHD based on AUC comparison).8.2 Lactation
Risk Summary
There are no data on the presence of difamilast or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production.
Difamilast and/or its metabolites are excreted into the milk of lactating rats (see Data). When a drug is present in animal milk, it is likely that the drug will be present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ADQUEY and any potential adverse effects on the breastfed infant from ADQUEY or from the underlying maternal condition.Clinical Considerations
To minimize potential infant exposure, advise breastfeeding women not to apply ADQUEY directly to the nipple or areola. If applied to the patient's chest, avoid exposure via direct contact with the infant's skin.
Data
When 14C-difamilast was subcutaneously administered to lactating rats at a single dose of 3 mg/kg, the concentration of radioactivity in milk was higher than that in blood (with milk/blood ratios at Cmax and AUC∞ of 13.7 and 5.4, respectively). The concentration of difamilast in animal milk does not necessarily predict the concentration of drug in human milk.8.4 Pediatric Use
The safety and effectiveness of ADQUEY for the topical treatment of mild to moderate atopic dermatitis have been established in pediatric patients 2 years of age and older. Use of ADQUEY in this age group is supported by data from five vehicle-controlled trials, which included 236 subjects 2 to less than 17 years of age, of whom 119 received ADQUEY and 117 received vehicle [see Adverse Reactions (6.1), Clinical Pharmacology (12.3) and Clinical Studies (14)].
The safety and effectiveness of ADQUEY have not been established in pediatric patients younger than 2 years of age.8.5 Geriatric Use
In clinical trials of ADQUEY in subjects with mild to moderate atopic dermatitis, 2 subjects were 65 years of age or older, and 0 were aged 75 years and older. Clinical trials of ADQUEY did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger adult subjects.
-
11 DESCRIPTION
ADQUEY (difamilast) ointment contains difamilast as the active ingredient. Difamilast, is a phosphodiesterase-4 (PDE-4) inhibitor.
Difamilast is described chemically as N-({2-[4-(Difluoromethoxy)-3-(propan-2-yloxy)phenyl]-1,3-oxazol-4-yl}methyl)-2-ethoxybenzamide. The empirical formula is C23H24F2N2O5 and the molecular weight is 446.44 g/mol.
The structural formula is represented below: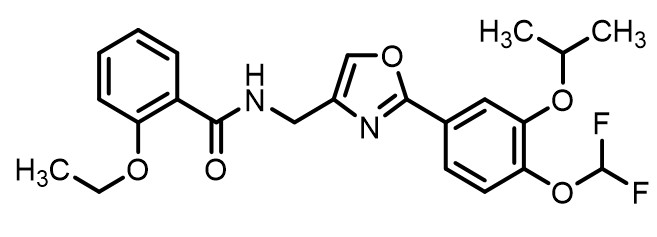
Difamilast occurs as white (or practically white) crystals or crystalline powder. It is very soluble in N-methylpyrrolidone, freely soluble in methanol and ethanol (99.5), and practically insoluble in water. It shows crystal polymorphism.
Each gram of ADQUEY contains 1% difamilast (w/w) in a wax-based, white to off-white ointment containing the following inactive ingredients: mineral oil, paraffin, propylene carbonate, white petrolatum, and white wax.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Difamilast is an inhibitor of phosphodiesterase-4 (PDE-4). Difamilast’s inhibition of PDE-4 (a major cyclic adenosine monophosphate (AMP)-metabolizing enzyme) activity leads to accumulation of intracellular cyclic AMP and decreased productions of various cytokines and chemokines. However, the specific mechanism(s) by which difamilast exerts its therapeutic action is not well defined.
12.2 Pharmacodynamics
The pharmacodynamics of ADQUEY ointment in the treatment of atopic dermatitis is not known.
Cardiac Electrophysiology
ADQUEY ointment is not expected to cause clinically significant QTc interval prolongation at the recommended dosages.12.3 Pharmacokinetics
Absorption
The pharmacokinetics (PK) of ADQUEY were investigated in 31 pediatric subjects 2 years of age and older with moderate to severe atopic dermatitis and a mean ± SD body surface area (BSA) involvement of 44 ± 13% (range 25% to 80%). In this trial, subjects applied an average of approximately 4.3 g of ADQUEY ointment, 1% (dose range was 1.7 to 11.7 g per application) twice daily for 2 weeks.
Plasma concentrations were quantifiable in all the subjects. On Day 15, the mean ± SD maximum plasma concentration (Cmax) and area under the concentration time curve from 0 to 8 hours post dose (AUC0-8) for difamilast were 16.9 ± 21.9 ng/mL and 86.2 ± 79.6 ng*h/mL, respectively. Systemic concentrations of difamilast were at steady state by Day 15, with no evidence of accumulation.
The PK of ADQUEY were investigated in 31 adult subjects with mild to moderate atopic dermatitis and a mean ± SD BSA involvement of 6 ± 3% (range 3% to 19%). Subjects applied an average of approximately 0.9 g of ADQUEY ointment, 1% (dose range was 0.1 to 3.8 g per application) twice daily for 4 weeks. On Day 29, the mean ± SD Cmax and AUC0-12 for difamilast were 0.76 ± 1.16 ng/mL and 6.10 ± 8.85 ng*h/mL, respectively.Distribution
Difamilast serum protein binding is 99% and is not concentration-dependent, in vitro.
Elimination
Metabolism
Difamilast is substantially metabolized to form three major metabolites in the plasma via CYP3A4-mediated O-deethylation (Metabolite 1), CYP1A2-mediated hydroxylation (Metabolite 2), and enzymatic hydrolysis (Metabolite 3).Excretion
After both single dosing and twice daily administration of difamilast ointment 1% in healthy Japanese subjects for 2 weeks, difamilast and Metabolite 1 were undetectable in urine. The urinary excretion ratios of all the other metabolites were less than 0.1% of the administered dose.Specific Populations
No dedicated clinical trials have been conducted to assess the impact of intrinsic factors on the PK of difamilast. Based on cross-study analyses, no substantial differences in the PK of difamilast were observed based on age (2-70 years), sex, race, mild or moderate renal impairment, or mild or moderate hepatic impairment. The effect of severe renal impairment (eGFR < 30 mL/min), or severe (Child-Pugh Class C) hepatic impairment on difamilast PK is unknown.Pediatric Patients
The plasma difamilast trough concentration corrected by dose in pediatric patients was 1.3 to 1.9 times higher than that in adults (on Day 1 and Day 15, respectively).
Drug Interaction Studies
Clinical Studies
No clinical drug interaction trials have been conducted with topical difamilast.In Vitro Studies
CYP450 Enzymes: Difamilast is a substrate of CYP3A4 and CYP1A2. Difamilast is not expected to inhibit CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4 at clinically relevant concentrations. Induction of CYP1A2, CYP2B6, CYP2C9, and CYP3A4 by difamilast is expected to be low at clinically relevant concentrations.
Transporter Systems: Difamilast is a substrate of breast cancer resistance protein (BCRP),but is not a substrate of P-glycoprotein (P-gp), OATP1B1, or OATP1B3. Difamilast is not expected to inhibit P-gp, BCRP, OAT1, OAT3, OCT1, OCT2, OATP1B1, OATP1B3, MATE1, or MATE2-K at clinically relevant concentrations. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
In a 2-year dermal carcinogenicity study in rats, no test article-related neoplastic findings were observed in male or female rats that received difamilast at dermal doses up to 3% ointment applied at 0.008 ml/cm2 (4 times the MRHD based on AUC comparison).
In a 2-year dermal carcinogenicity study in mice, no test article-related neoplastic findings were observed in male or female mice that received difamilast at dermal doses up to 3% ointment applied at 0.008 ml/cm2 (4 times the MRHD based on AUC comparison).
Difamilast was not genotoxic in an in vitro bacterial reverse mutation (Ames) test, an in vitro mammalian cell mutation test in mouse lymphoma cells, or an in vivo rat bone marrow micronucleus test.
In a fertility and early embryonic development study in rats, irregular estrus cycles, sperm abnormalities, increased preimplantation loss, and decreased copulation and fertility indexes were observed at a subcutaneous dose of 100 mg/kg/day difamilast (116 times the MRHD for males and 263 times the MRHD for females, respectively, based on AUC comparison). No treatment-related adverse effects on fertility or early embryonic development were observed at 10 mg/kg/day for males and females (20 times the MRHD for males and 30 times the MRHD for females, respectively, based on AUC comparison). -
14 CLINICAL STUDIES
The efficacy of ADQUEY for the treatment of mild to moderate atopic dermatitis was assessed in three multicenter, randomized, double-blind, parallel-group, vehicle-controlled trials (Trial 1 [NCT02068352], Trial 2 [NCT03908970], and Trial 3 [NCT03911401]) that treated a total of 612 subjects. Trial 1 enrolled adult and pediatric subjects 10 years of age and older in the United States, Australia, and Poland. Trial 2 enrolled adult and pediatric subjects 15 years of age and older in Japan, and Trial 3 enrolled pediatric subjects 2 to 14 years of age in Japan. In these trials, subjects were randomized 1:1 to receive ADQUEY or vehicle ointment, applied topically to the entire treatment area twice daily for at least 4 weeks.
The trials enrolled subjects with an Investigator's Global Assessment (IGA) score of 2 (mild) or 3 (moderate) atopic dermatitis and affected body surface area (BSA) (excluding the face, neck, and head [Trial 1]; excluding the scalp [Trials 2 and 3]) of ≥ 5% and ≤ 40%. The IGA included an overall assessment of atopic dermatitis (erythema, induration/papulation, and oozing/crusting) on a severity scale of 0 to 4.
At baseline, 29%, 15%, and 15% of the subjects had an IGA of mild, and 71%, 85%, and 85% had an IGA of moderate in Trials 1, 2, and 3, respectively. Concomitant treatment for atopic dermatitis was prohibited.
In Trial 1, 56% of the subjects were female, 73% were White, 20% were Black or African American, and 4% were Asian; for ethnicity, 18% of the subjects identified as Hispanic or Latino. In Trials 2 and 3, the majority of the subjects were male (54% in Trial 2 and 58% in Trial 3), 100% were Asian, and 100% identified as Not Hispanic or Latino. The median age of enrolled subjects at baseline was 30 years (range 10 to 67 years), 30 years (range 15 to 65 years), and 7 years (range 2 to 14 years) in Trials 1, 2, and 3, respectively.
The primary efficacy endpoint for all trials was the proportion of subjects who achieved IGA success, defined as an IGA grade of clear (0) or almost clear (1) and with a 2-grade or greater improvement from baseline, at Week 4.
The efficacy results at Week 4 are shown in Table 2.
Table 2: Efficacy Results at Week 4 in Adult and Pediatric Subjects 2 Years of Age and Older with Mild to Moderate Atopic Dermatitis in Trials 1, 2, and 3Trial 1
(Multinational‡)Trial 2
(Japan)
Trial 3
(Japan)
ADQUEY
(N = 43)Vehicle
(N = 37)ADQUEY
(N = 182)
Vehicle
(N = 182)ADQUEY
(N = 85)
Vehicle
(N = 83)
IGA Success†, n (%) 9 (21)
1 (3)
70 (38)
23 (13)
40 (47)
15 (18)
Difference, %
from Vehicle (95% CI)
18
(5, 31)
26
(17, 34)
29
(15, 42)
† IGA success is defined as an IGA score of 0 or 1 and with ≥ 2-grade improvement from baseline.
‡ Trial 1 enrolled subjects from the United States, Australia, and Poland.
Subjects with missing IGA scores were imputed as failures. -
16 HOW SUPPLIED/STORAGE AND HANDLING
How supplied
ADQUEY (difamilast) ointment, 1% is a white to off-white ointment supplied in the following packages:- 27 g tube (NDC: 72893-017-08)
- 85 g tube (NDC: 72893-017-09)
Storage and Handling
Store at 20°C–25°C (68°F–77°F); excursions permitted to 15°C–30°C (59°F–86°F). [see USP Controlled Room Temperature].
Store away from direct sunlight, heat, and moisture.
Keep tube tightly closed. -
17 PATIENT COUNSELING INFORMATION
Advise patients or caregivers to read the FDA-approved patient labeling (Patient Information).
Administration
Advise patients or caregivers that ADQUEY is for topical use only and is not for ophthalmic, oral, or intravaginal use [see Dosage and Administration (2)].
Lactation
Advise breastfeeding women not to apply ADQUEY directly to the nipple or areola to minimize potential infant exposure. Instruct patients to avoid inadvertent contact of treated areas with the infant’s skin [see Use in Specific Populations (8.2)].
(Made in Canada)
Manufactured for:
Acrotech Biopharma Inc
East Windsor, NJ 08520 USA
PATIENT INFORMATION
ADQUEYTM (ahd kway)
(difamilast)
ointment, for topical useImportant information: ADQUEY is for use on the skin (topical use) only. Do not use ADQUEY near or in your eyes, mouth, or vagina. What is ADQUEY?
ADQUEY is a prescription medicine used on the skin (topical) to treat adults and children 2 years of age and older with mild to moderate atopic dermatitis.
It is not known if ADQUEY is safe and effective in children younger than 2 years of age.Before using ADQUEY, tell your healthcare provider about all your medical conditions,
including if you:
- are pregnant or plan to become pregnant. It is not known if ADQUEY will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if ADQUEY passes into your breast milk or if it may harm your baby. Talk to your healthcare provider about the best way to feed your baby during treatment with ADQUEY.
Breastfeeding women using ADQUEY should not apply ADQUEY directly to the nipple or areola to avoid contact with your baby. Avoid contact between your baby and any skin areas treated with ADQUEY.
How should I use ADQUEY?
- Use ADQUEY exactly as your healthcare provider tells you to use it.
- Apply a thin layer of ADQUEY to the affected areas 2 times a day. Rub the ointment in completely until you no longer see it on your skin.
- Avoid applying ADQUEY to areas of the skin that are red, warm, swollen or painful.
- Wash your hands after applying ADQUEY. If someone else applies ADQUEY for you, they should wash their hands after applying it.
What are the possible side effects of ADQUEY?
The most common side effects of ADQUEY include: pain or swelling in the nose and throat (nasopharyngitis).
These are not all of the possible side effects of ADQUEY.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store ADQUEY?
- Store ADQUEY at room temperature between 68°F to 77°F (20°C to 25°C).
- Store ADQUEY away from direct sunlight, heat and moisture.
- Keep ADQUEY tube tightly closed after each use.
General information about the safe and effective use of ADQUEY.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ADQUEY for a condition for which it was not prescribed. Do not give ADQUEY to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ADQUEY that is written for health professionals.What are the ingredients in ADQUEY?
Active ingredient: difamilast
Inactive ingredients: mineral oil, paraffin, propylene carbonate, white petrolatum, and white wax.
(Made in Canada)
Manufactured for:Acrotech Biopharma Inc, East Windsor, NJ 08520 USA
For more information, call 1-888-292-9617 or go to www.ADQUEY.com.This Patient Information has been approved by the U.S. Food and Drug Administration.
Issued: 02/2026
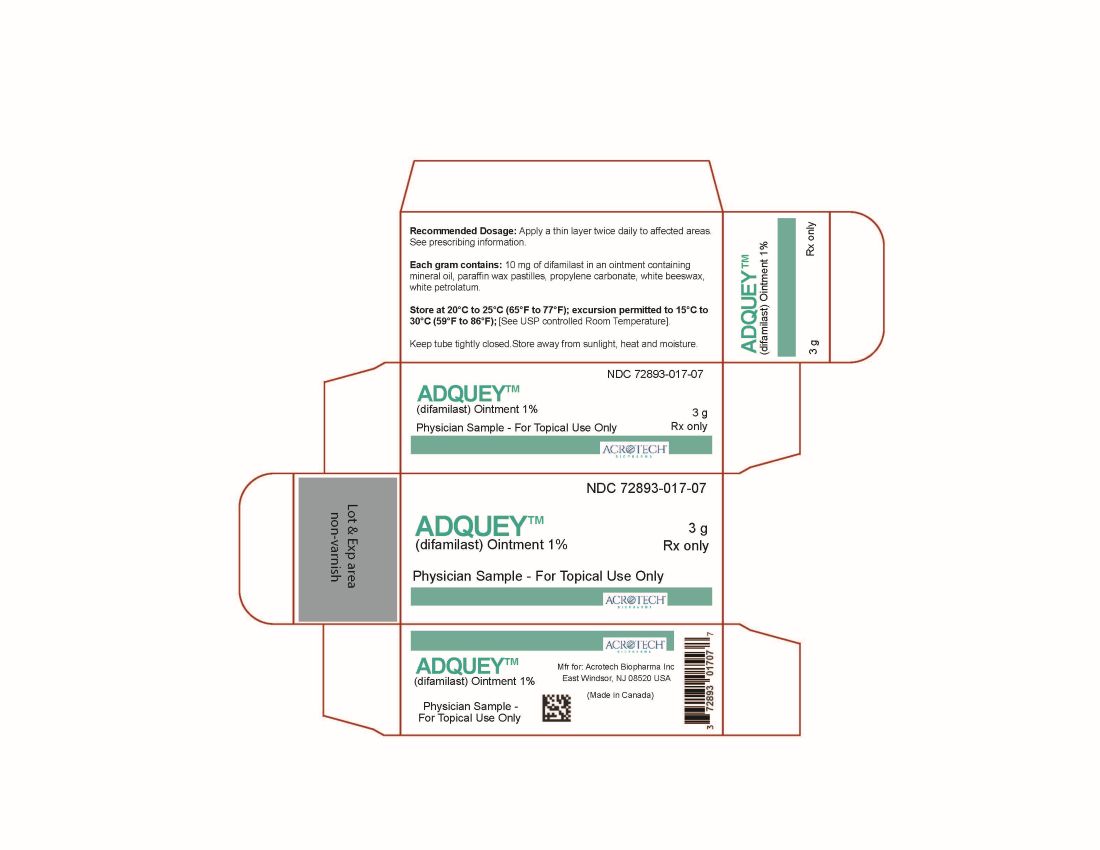
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIFAMILAST
adquey ointmentProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 72893-017 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Difamilast (UNII: T3U32GLJ0F) (Difamilast - UNII:T3U32GLJ0F) Difamilast 10 mg in 100 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) PARAFFIN (UNII: I9O0E3H2ZE) PROPYLENE CARBONATE (UNII: 8D08K3S51E) WHITE PETROLATUM (UNII: B6E5W8RQJ4) WHITE WAX (UNII: 7G1J5DA97F) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 72893-017-09 85 g in 1 TUBE; Type 0: Not a Combination Product 02/25/2026 2 NDC: 72893-017-08 27 g in 1 TUBE; Type 0: Not a Combination Product 02/25/2026 3 NDC: 72893-017-07 3 g in 1 TUBE; Type 0: Not a Combination Product 02/25/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219474 02/25/2026 Labeler - Acrotech Biopharma Inc (116965616) Establishment Name Address ID/FEI Business Operations Contract Pharmaceuticals Limited 248761249 MANUFACTURE(72893-017) , PACK(72893-017) Establishment Name Address ID/FEI Business Operations Contract Pharmaceutical Limited 202733655 ANALYSIS(72893-017)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.