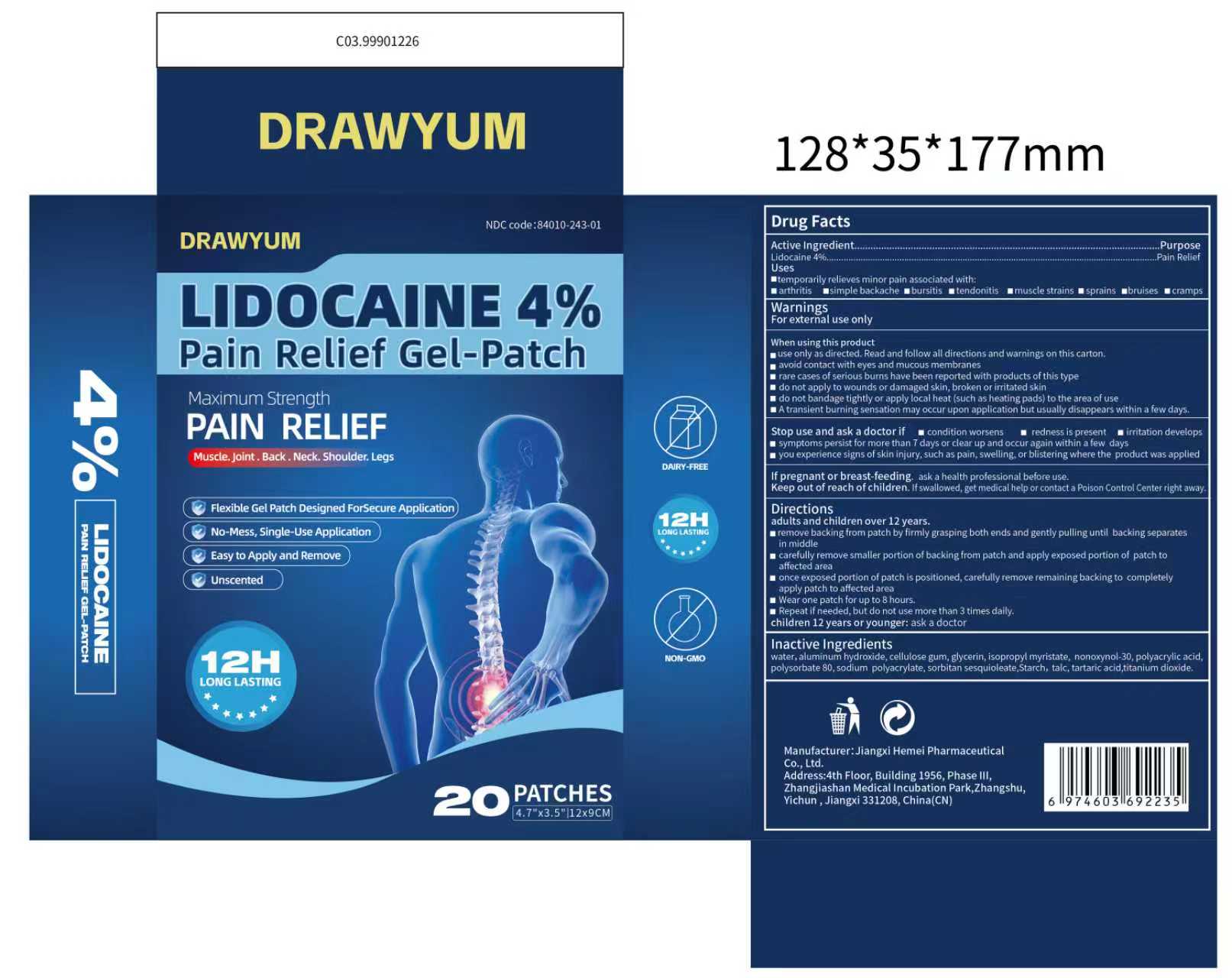
DRAWYUM Pain Relief Gel-Patch by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-243complete
DRAWYUM Pain Relief Gel-Patch by
Drug Labeling and Warnings
DRAWYUM Pain Relief Gel-Patch by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DRAWYUM PAIN RELIEF GEL-PATCH- lidocaine 4% pain relief gel-patch patch
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-243complete
Use
▆temporarily relieves minor pain associated with: ▆arthritis ▆simple backache▆bursitis ▆tendonitis ▆muscle strains ▆sprains▆bruises ▆cramps
Do not use
▆use only as directed. Read and follow all directions and warnings on this carton. ▆avoid contact with eyes and mucous membranes ▆rare cases of serious burns have been reported with products of this type ▆ do not apply to wounds or damaged skin, broken or irritated skin ▆do not bandage tightly or apply local heat (such as heating pads) to the area of use ▆ A transient burning sensation may occur upon application but usually disappears within a few days.
When Using
▆use only as directed. Read and follow all directions and warnings on this carton. ▆avoid contact with eyes and mucous membranes ▆rare cases of serious burns have been reported with products of this type ▆ do not apply to wounds or damaged skin, broken or irritated skin ▆do not bandage tightly or apply local heat (such as heating pads) to the area of use ▆ A transient burning sensation may occur upon application but usually disappears within a few days.
Stop Use
▆condition worsens ▆redness is present ▆irritation develops▆symptoms persist for more than 7 days or clear up and occur again within a few dsys ▆you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied If pregnant or breast-feeding. ask a health professional before use
Ask Doctor
▆condition worsens ▆redness is present ▆irritation develops▆symptoms persist for more than 7 days or clear up and occur again within a few dsys ▆you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied
Keep Oot Of Reach Of Children
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away
Directions
adults and children over 12 years. ▆remove backing from patch by firmly grasping both ends and gently pulling until backing separate in middle ▆carefully remove smaller portion of backing from patch and apply exposed portion of patch to affected area ▆once exposed portion of patch is positioned, carefully remove remaining backing to completely apply patch to affected area ▆Wear one patch for up to 8 hours. ▆Repeat if needed, but do not use more than 3 times daily. children 12 years or younger: ask a doctor
| DRAWYUM PAIN RELIEF GEL-PATCH
lidocaine 4% pain relief gel-patch patch |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-243) | |