MECLIZINE HYDROCHLORIDE- meclizine hydrochloride tablet MECLIZINE HYDROCHLORIDE- meclizine hydrocloride tablet
Drug Labeling and Warnings
Drug Details [pdf]
-
DESCRIPTION
Chemically, Meclizine HCl is 1-(p-chloro-α-phenylbenzyl)-4-(m-methylbenzyl) piperazine dihydrochloride monohydrate.
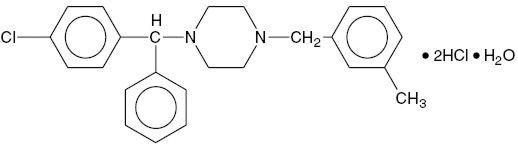
C25H27ClN2. 2HCl. H2O M.W. 481.88
Meclizine HCI Tablets, USP are available in two different strengths: 12.5 mg and 25 mg. In addition each tablet contains the following inactive ingredients: Colloidal Silicon Dioxide, Croscarmellose Sodium, Lactose Monohydrate, Magnesium Stearate, Microcrystalline Cellulose. Also, Meclizine HCI Tablets USP, 12.5 mg contains FD&C Blue #1 Aluminum Lake (11-13%) and Meclizine HCI Tablets USP, 25 mg contains D&C Yellow #10 Aluminum Lake (15-20%). -
CLINICAL PHARMACOLOGY
Meclizine Hydrochloride is an antihistamine which shows marked protective activity against nebulized histamine and lethal doses of intravenously injected histamine in guinea pigs. It has a marked effect in blocking the vasodepressor response to histamine, but only a slight blocking action against acetylcholine. Its activity is relatively weak in inhibiting the spasmogenic action of histamine on isolated guinea pig ileum.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Since drowsiness may, on occasion, occur with use of this drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery.
Patients should avoid alcoholic beverages while taking this drug.
Due to its potential anticholinergic action, this drug should be used with caution in patients with asthma, glaucoma or enlargement of the prostate gland. -
PRECAUTIONS
PREGNANCY, Teratogenic Effects
Pregnancy Category B. Reproduction studies in rats have shown cleft palates at 25-50 times the human dose. Epidemiological studies in pregnant women, however, do not indicate that medicine increases the risk of abnormalities when administered during pregnancy. Despite the animal findings, it would appear that the possibility of fetal harm is remote. Nevertheless, meclizine, or any other medication, should be used during pregnancy only if clearly necessary.
Pediatric Use
Clinical studies establishing safety and effectiveness in children have not been done; therefore, usage is not recommended in children under 12 years of age. - ADVERSE REACTIONS
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Meclizine HCI Tablets, USP are available in the following strengths and package sizes:
12.5 mg (Blue, oval-shaped, scored, debossed with TL122)
NDC: 35356-901-30 Bottles of 30NDC: 35356-901-60 Bottles of 60
NDC: 35356-901-90 Bottles of 90
25 mg (Yellow, oval-shaped, scored, debossed with TL121)NDC: 35356-914-20 Bottles of 20
NDC: 35356-914-30 Bottles of 30NDC: 35356-914-60 Bottles of 60
NDC: 35356-914-90 Bottles of 90
Store at 20-25°C (68-77°F) (See USP Controlled Room Temperature].Manufactured By:
Jubilant Cadista Pharmaceuticals Inc.
Salisbury, MD 21801, USA.Revised 03/11
- Image of 12.5mg Label
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MECLIZINE HYDROCHLORIDE
meclizine hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 35356-901(NDC:59746-122) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Meclizine Hydrochloride (UNII: HDP7W44CIO) (Meclizine - UNII:3L5TQ84570) Meclizine Hydrochloride 12.5 mg Inactive Ingredients Ingredient Name Strength Silicon Dioxide (UNII: ETJ7Z6XBU4) Croscarmellose Sodium (UNII: M28OL1HH48) Lactose Monohydrate (UNII: EWQ57Q8I5X) Magnesium Stearate (UNII: 70097M6I30) Cellulose, Microcrystalline (UNII: OP1R32D61U) Fd&c Blue No. 1 (UNII: H3R47K3TBD) Aluminum Oxide (UNII: LMI26O6933) Product Characteristics Color BLUE Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code TL122 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 35356-901-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 10/11/2019 2 NDC: 35356-901-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 10/11/2019 3 NDC: 35356-901-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 10/11/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040659 06/04/2010 10/11/2019 MECLIZINE HYDROCHLORIDE
meclizine hydrocloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 35356-914(NDC:59746-121) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Meclizine Hydrochloride (UNII: HDP7W44CIO) (Meclizine - UNII:3L5TQ84570) Meclizine Hydrochloride 25 mg Inactive Ingredients Ingredient Name Strength Silicon Dioxide (UNII: ETJ7Z6XBU4) Croscarmellose Sodium (UNII: M28OL1HH48) Lactose Monohydrate (UNII: EWQ57Q8I5X) Magnesium Stearate (UNII: 70097M6I30) Cellulose, Microcrystalline (UNII: OP1R32D61U) D&c Yellow No. 10 (UNII: 35SW5USQ3G) Aluminum Oxide (UNII: LMI26O6933) Product Characteristics Color YELLOW Score 2 pieces Shape OVAL Size 13mm Flavor Imprint Code TL121 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 35356-914-20 20 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 06/01/2018 2 NDC: 35356-914-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 3 NDC: 35356-914-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 10/11/2019 4 NDC: 35356-914-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2013 10/11/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040659 06/04/2010 Labeler - Lake Erie Medical DBA Quality Care Products LLC (831276758) Establishment Name Address ID/FEI Business Operations Lake Erie Medical DBA Quality Care Products LLC 831276758 repack(35356-901, 35356-914)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

