Vybrique is indicated for erectile dysfunction
Vybrique 25 mg 2 pack sample
NDC: 43328-016-02
Vybrique 25mg
sildenafil oral film
2 pack sample
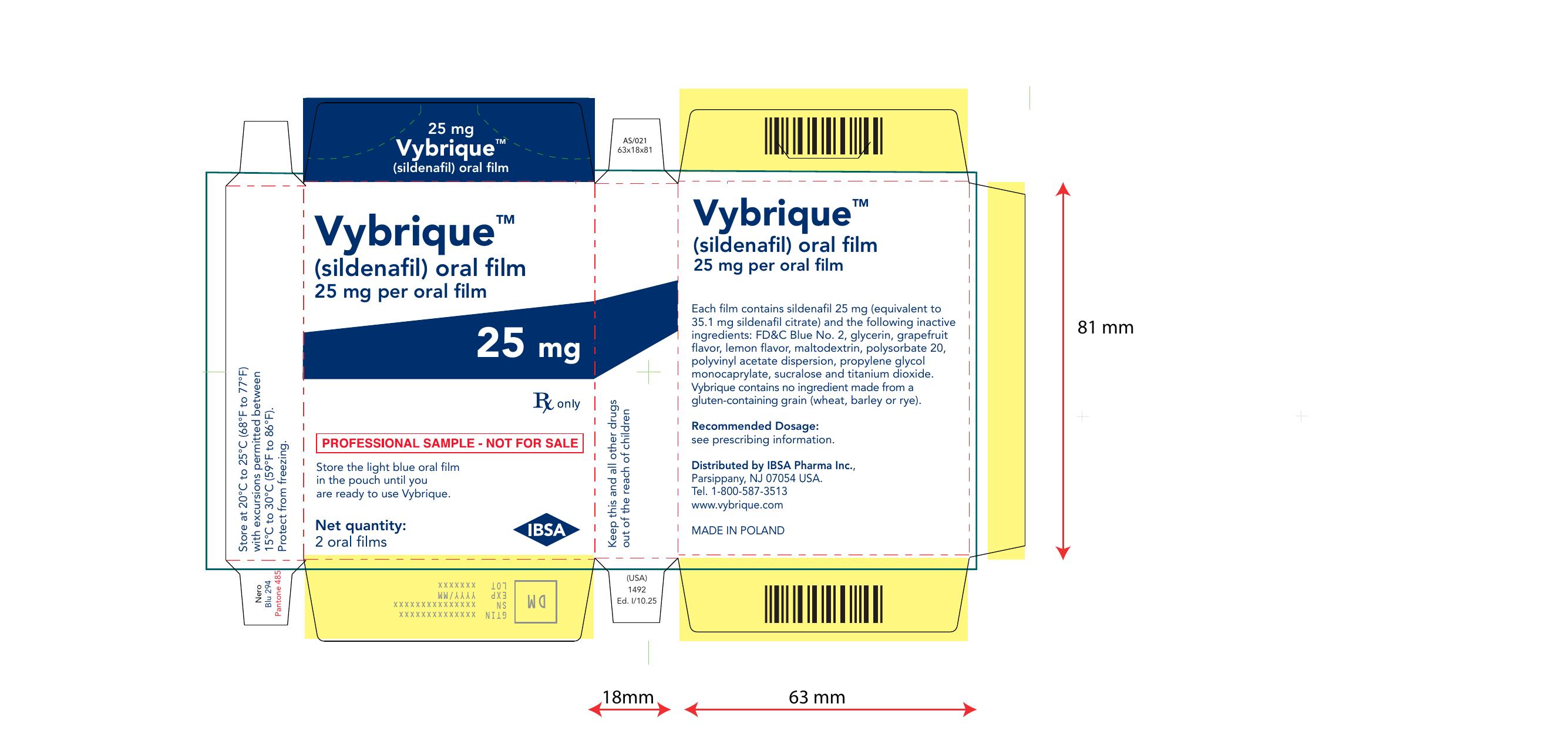
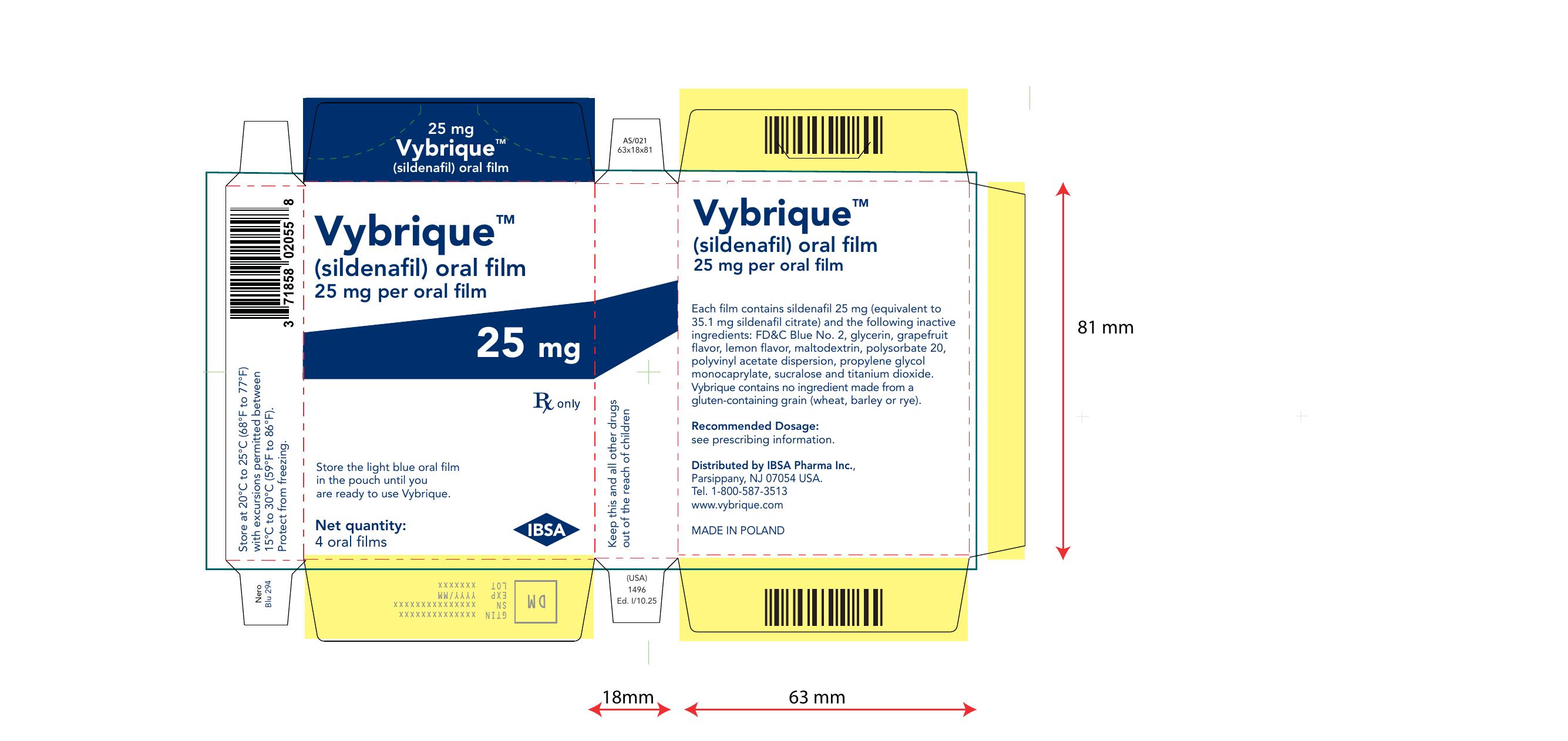
Vibrique 25 mg 4 pack
Vybrique
Sildeanfil Oral Film
4 pack
Vybrique 25 mg 8 pack
NDC: 43328-016-08
Vybrique 25 mg
sildenafil oral film
8 pack

Vybrique 50 mg 2 pack sample
NDC: 43328-017-02
Vybrique 50 mg
sildenafil oral film
2 pack sample
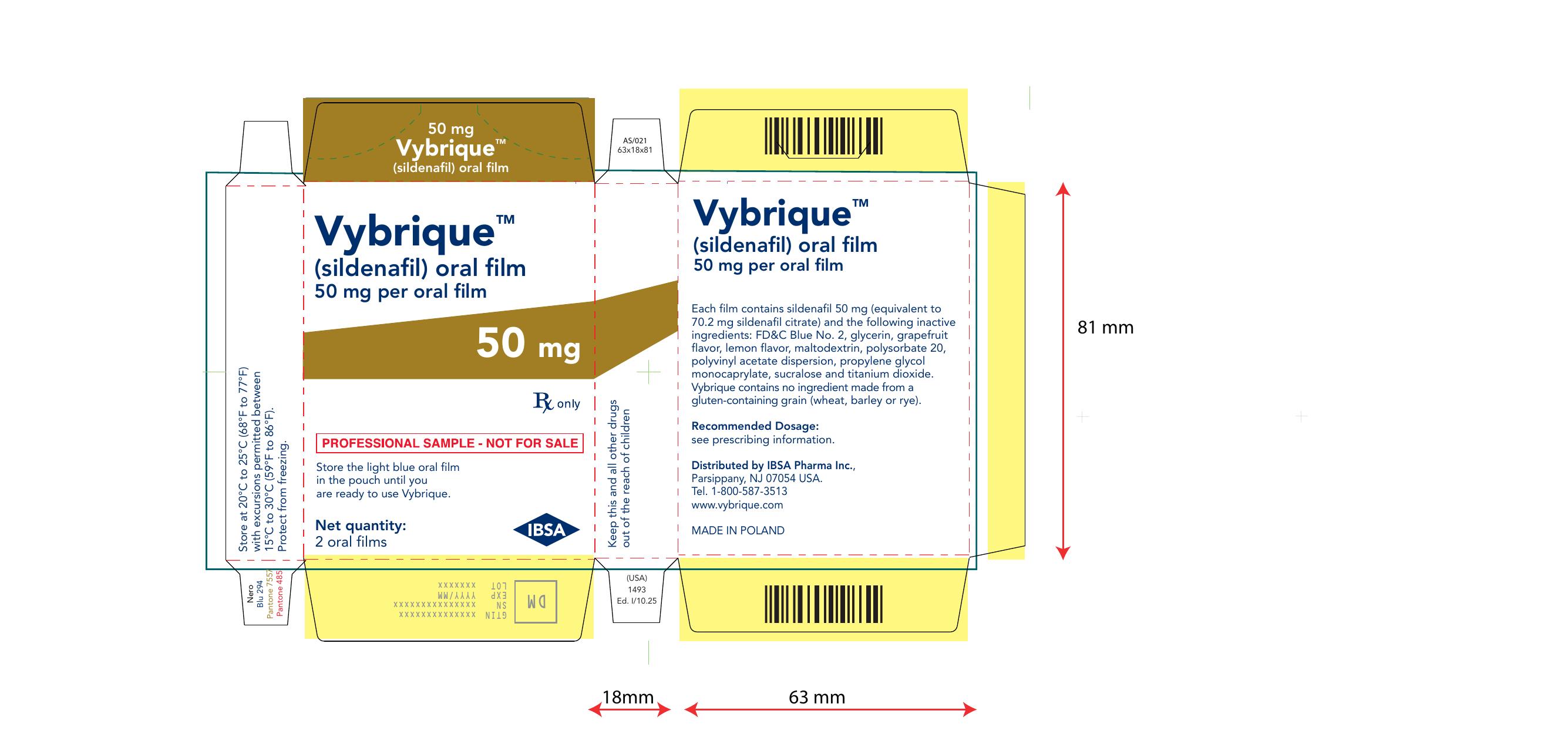
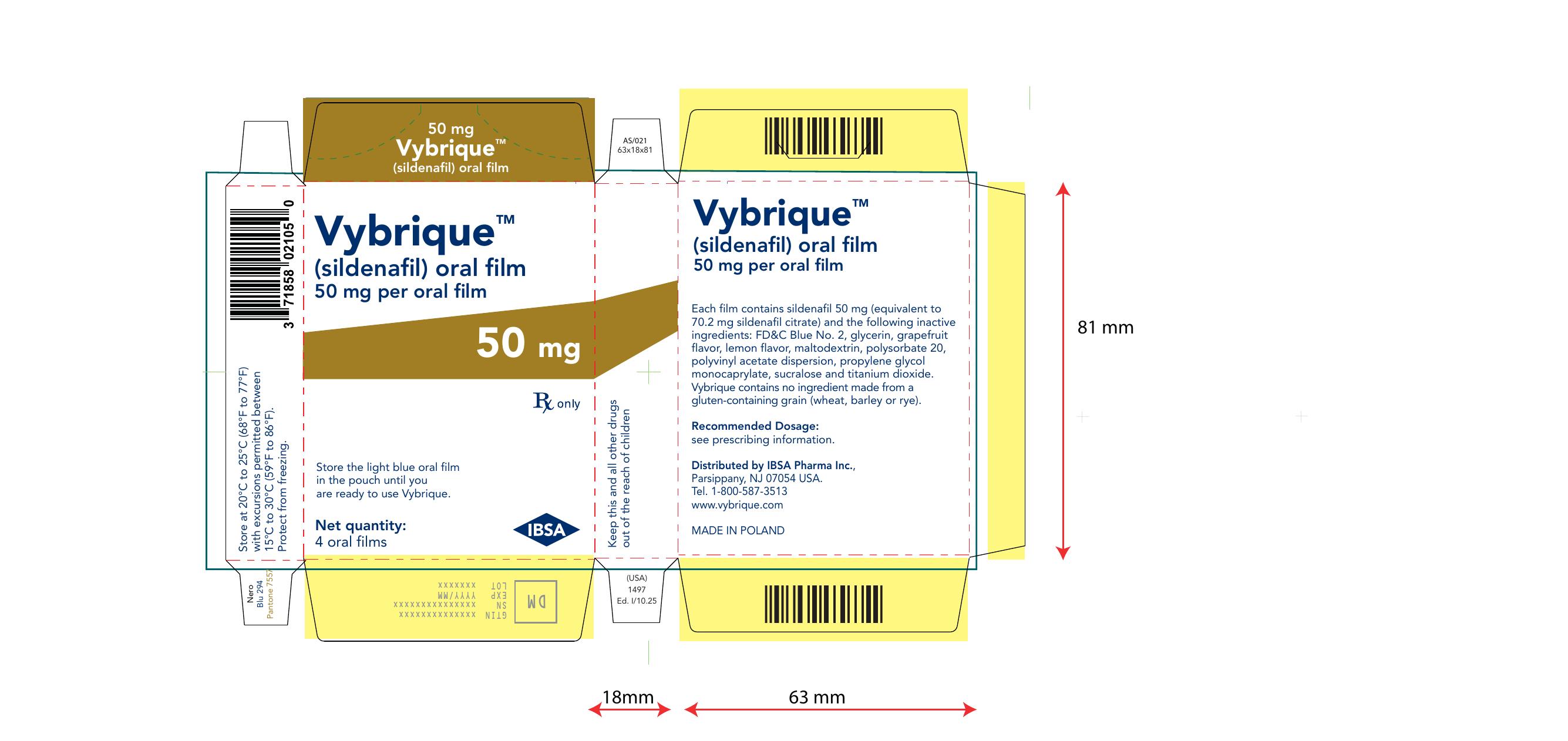
Vybrique 50 mg 4 pack
NDC: 43328-017-04
Vybrique 50 mg
sildenafil oral film
4 pack
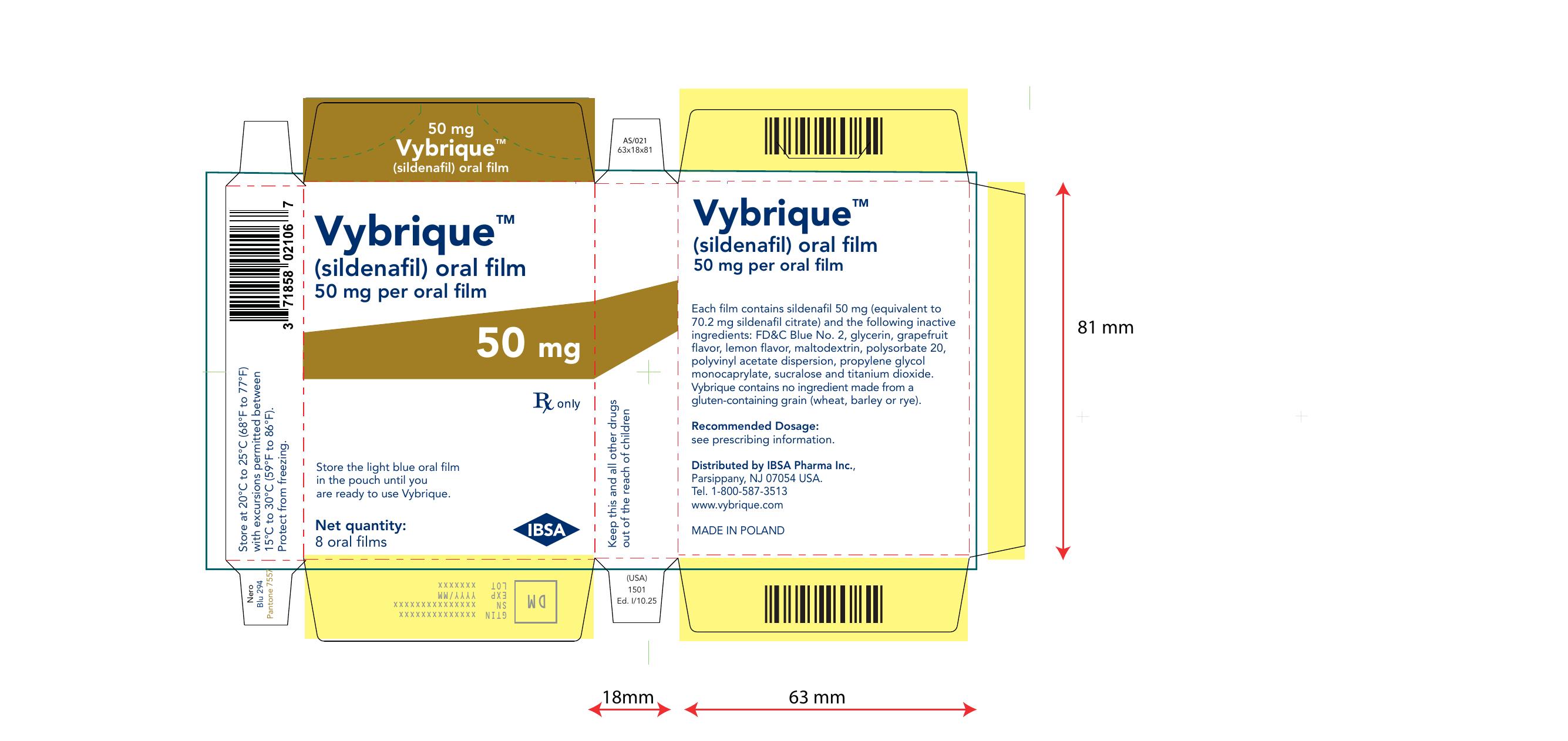
Vybrique 50 mg 8 pack
NDC: 43328-017-08
Vybrique 50 mg
sildenafil oral film
8 pack
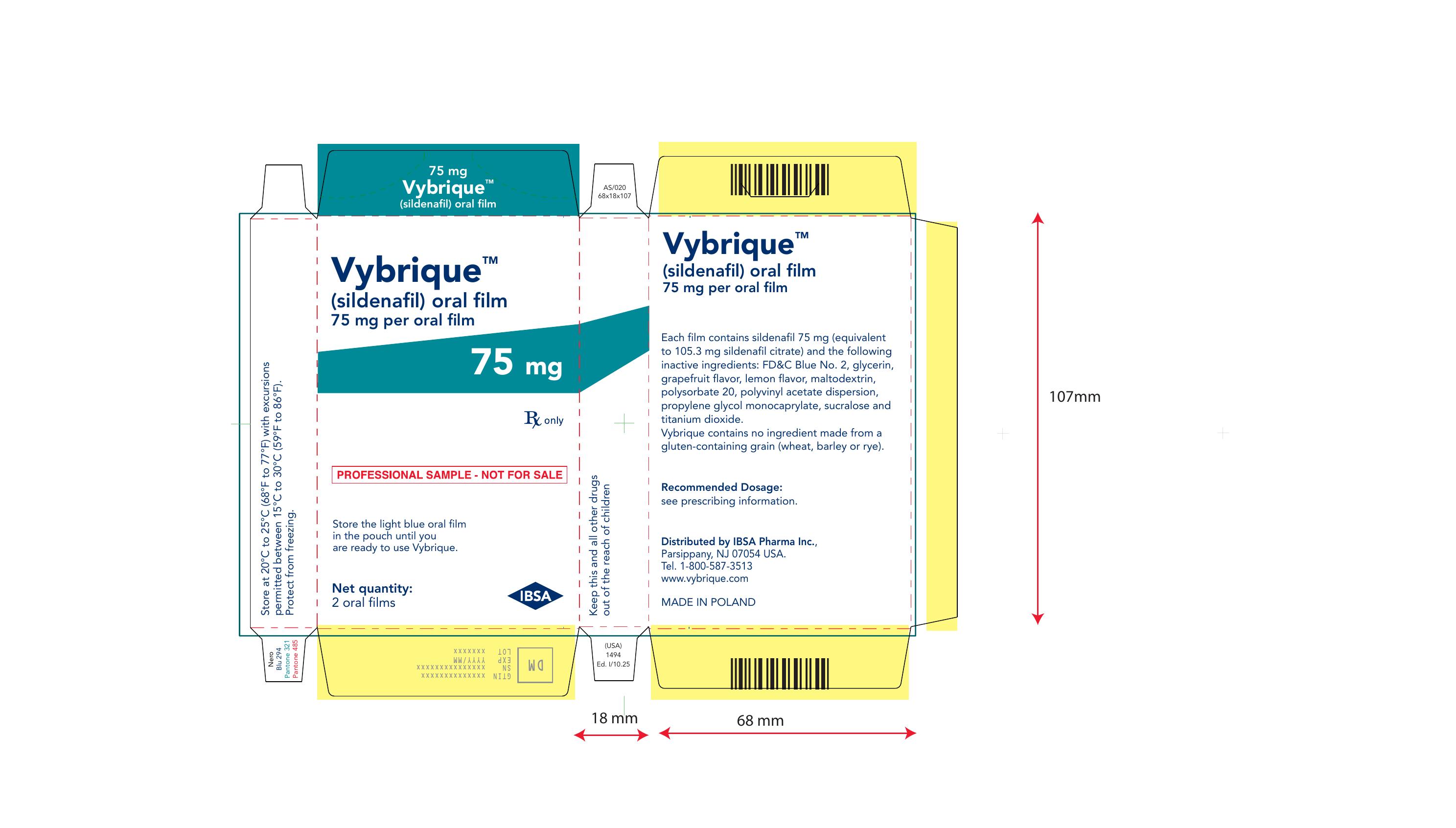
Vybrique 75 mg 2 pack sample
NDC: 43328-018-02
Vybrique 75 mg
sildenafil oral film
2 pack sample
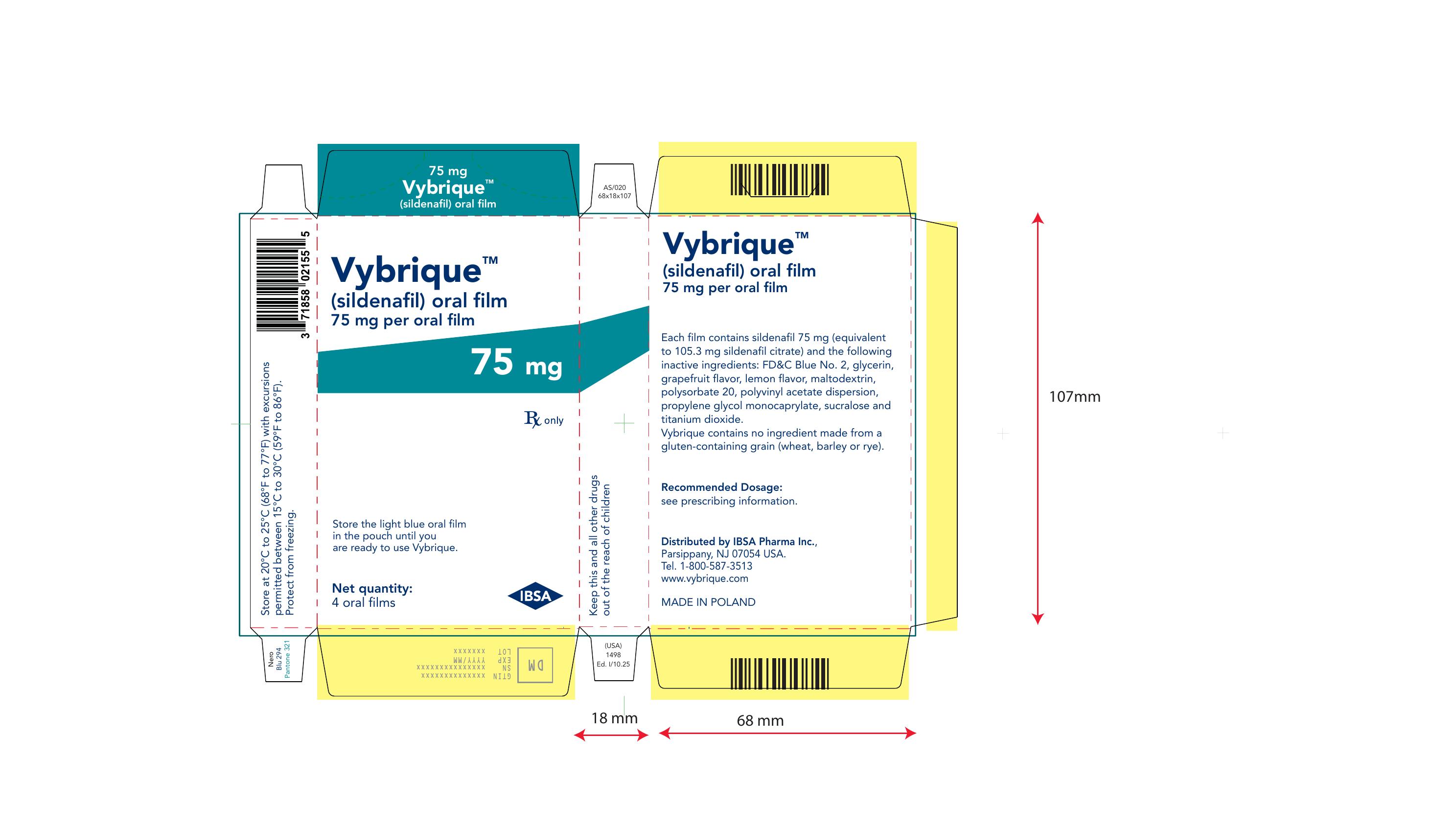
Vybrique 75 mg 4 pack
NDC: 43328-018-04
Vybrique 75 mg
sildenafil oral film
4 pack
Vybrique 75 mg 8 pack
NDC: 43328-018-08
Vybrique 75 mg
sildenafil oral film
8 pack
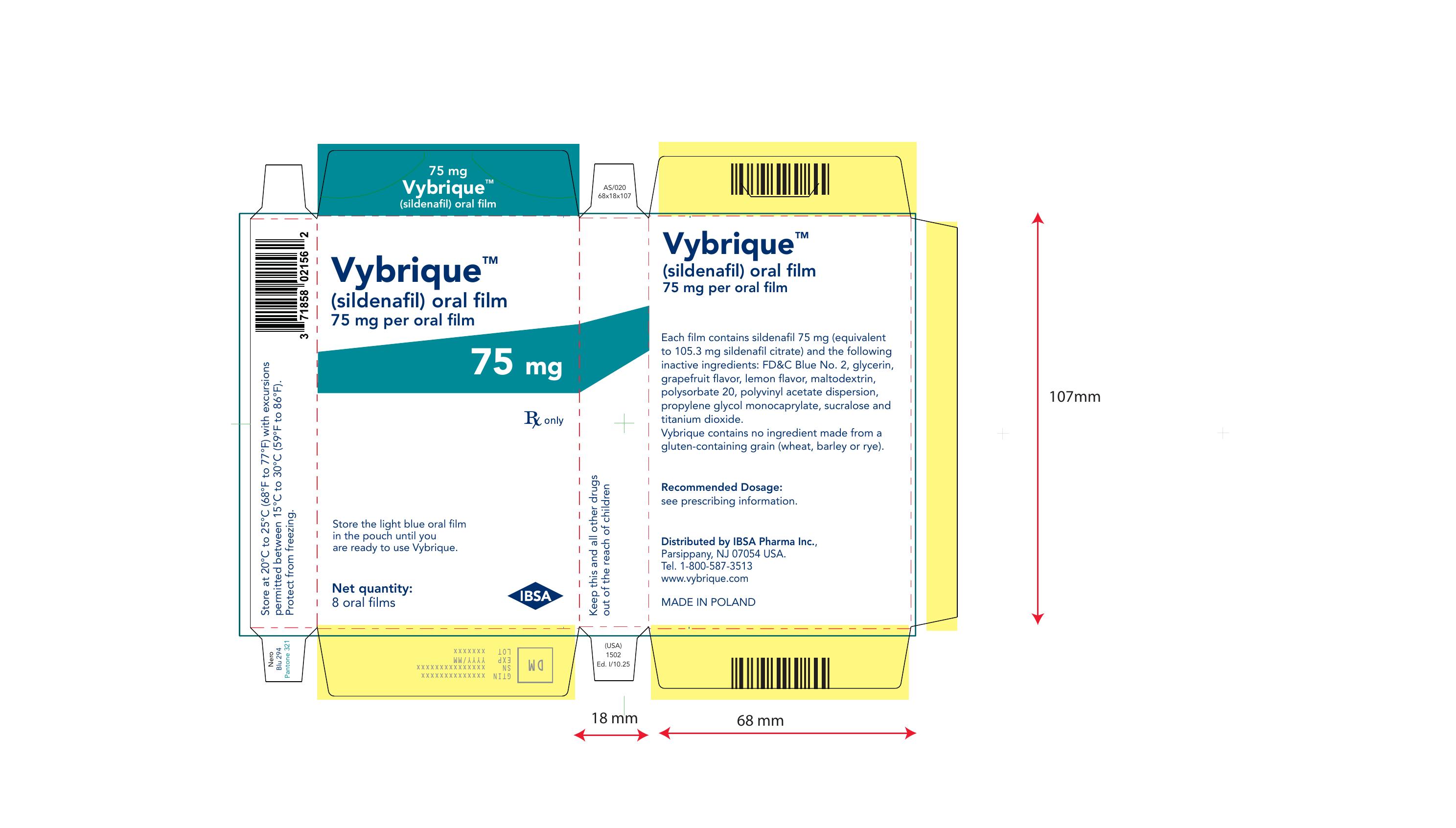
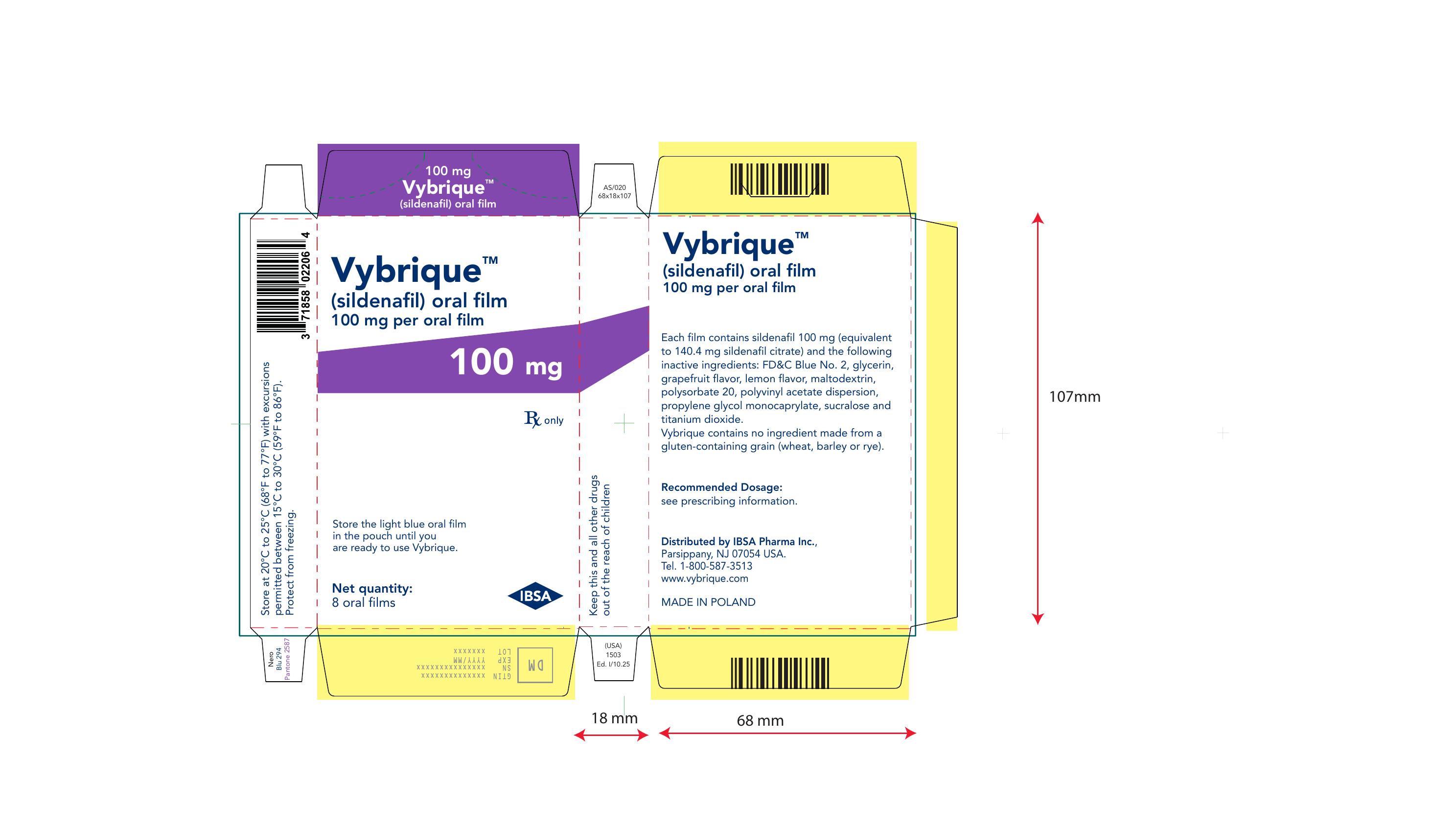
Vybrique 100 mg 2 pack sample
NDC: 43328-019-02
Vybrique 100 mg
sildenafil oral film
2 pack sample
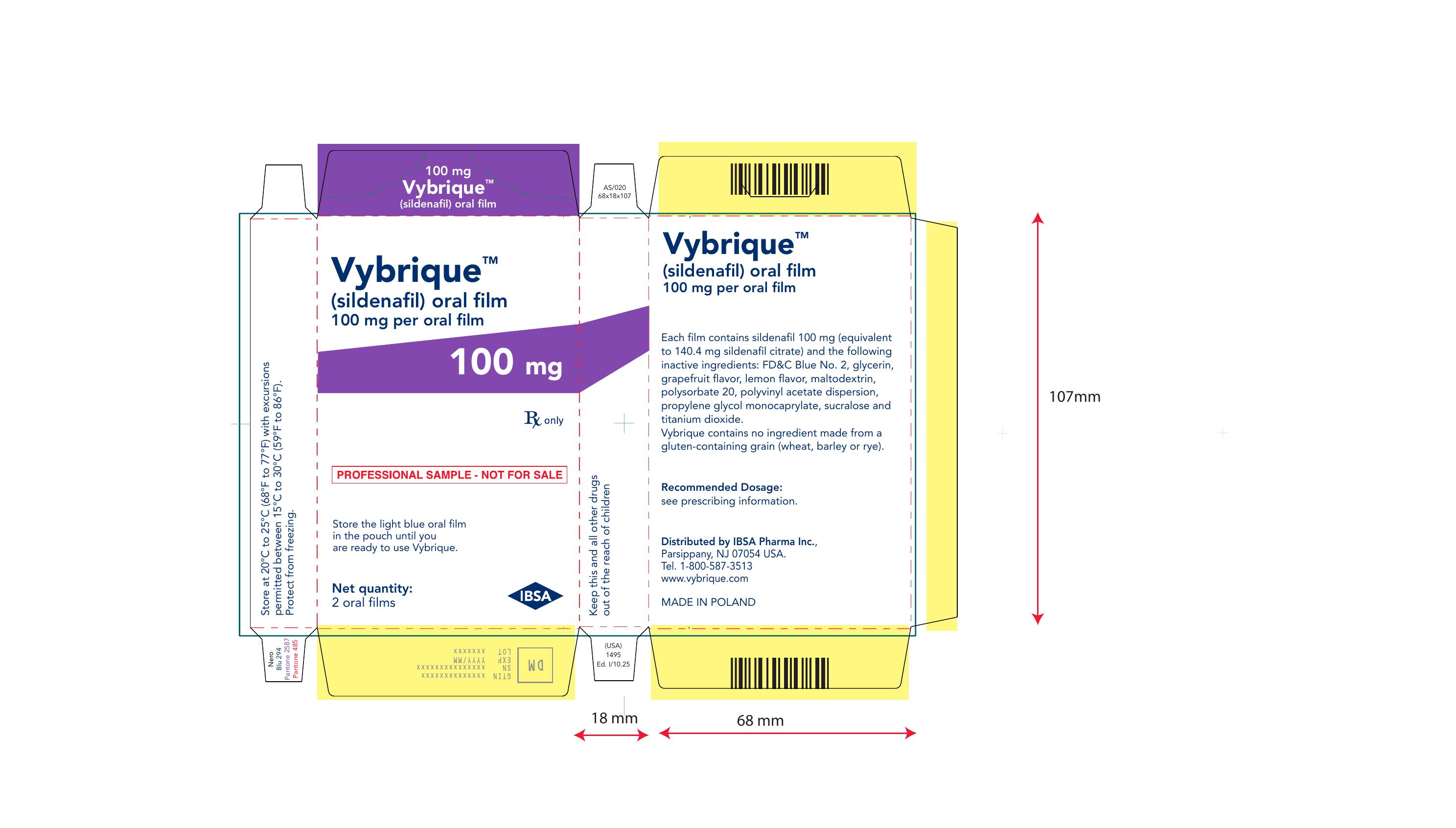
Vybrique 100 mg 4 pack
NDC: 43328-019-04
Vybrique 100 mg
sildenafil oral film
4 pack
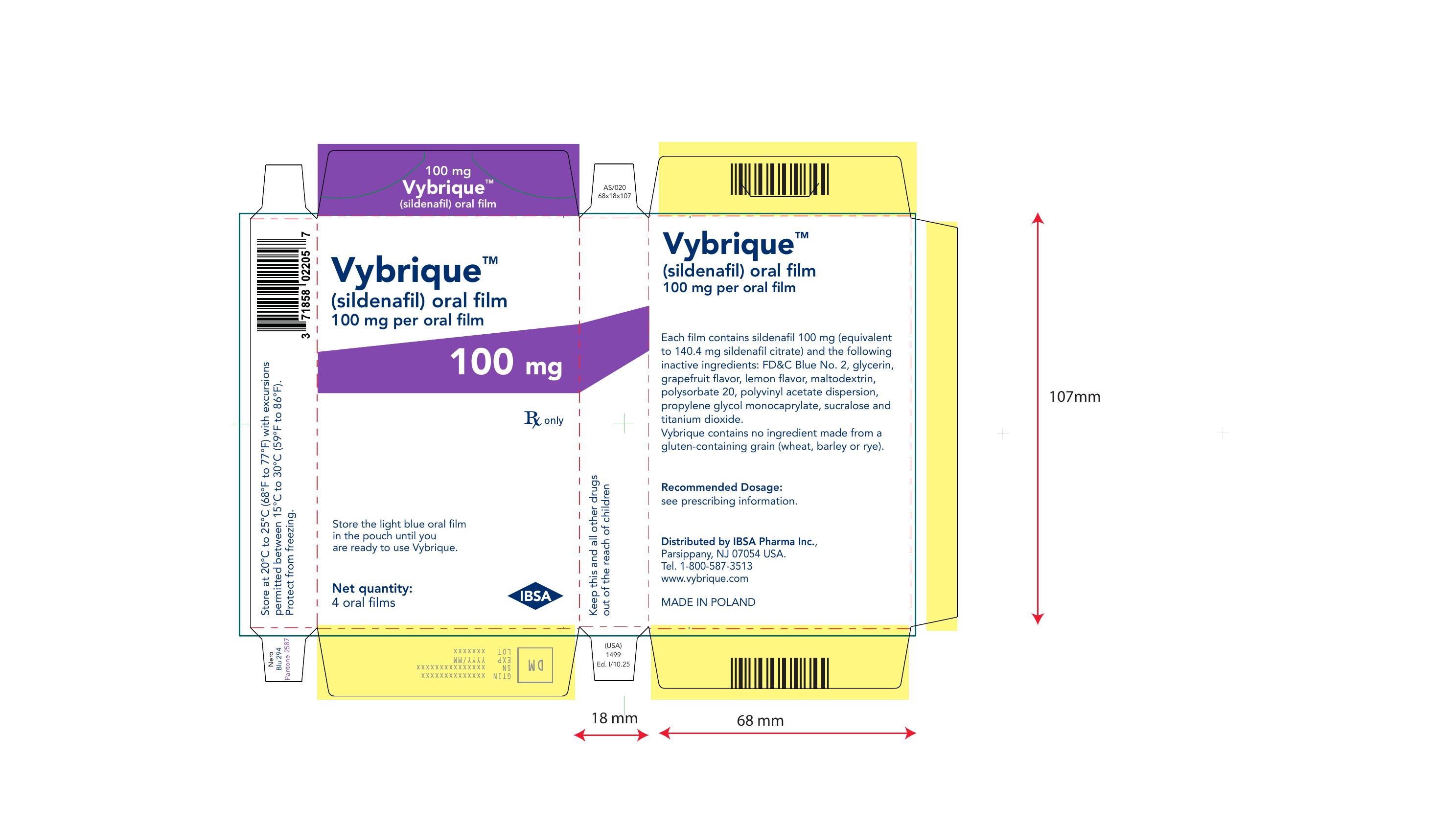
Vybrique 100 mg 8 pack
NDC: 43328-019-08
Vybrique 100 mg
sildenafil oral film
8 pack