LAURA GELLER SHINE-N-SHIELD LIP BALM SPF 15- GOING COCONUT
LAURA GELLER X BLISS SHINE - N - SHIELD LIP BALM SPF 15 Sunset Sands by
Drug Labeling and Warnings
LAURA GELLER X BLISS SHINE - N - SHIELD LIP BALM SPF 15 Sunset Sands by is a Otc medication manufactured, distributed, or labeled by COSMAX USA, CORP. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LAURA GELLER X BLISS SHINE - N - SHIELD LIP BALM SPF 15 SUNSET SANDS- zinc oxide lipstick
COSMAX USA, CORP
----------
LAURA GELLER SHINE-N-SHIELD LIP BALM SPF 15- GOING COCONUT
Warnings:
For external use only.
DIRECTIONS
- Apply liberally 15 minutes before sun exposure. Reapply:
- After 80 minutes of swimming or sweating
- Immediately after towel drying
- After eating or drinking
- As often as necessary
INACTIVE INGREDIENTS: DIISOSTEARYL MALATE, SILICA DIMETHYL SILYLATE, POLYBUTENE, CAPRYLIC/CAPRIC TRIGLYCERIDE, LECITHIN, ISOSTEARIC ACID, POLYGLYCERYL-3 POLYRICINOLEATE, POLYHYDROXYSTEARIC ACID, HYDROGENATED POLYISOBUTENE, BUTYLOCTYL SALICYLATE, MICROCRYSTALLINE WAX, BIS-DIGLYCERYL POLYACYLADIPATE-2, SORBITAN OLIVATE, ISONONYL ISONONANOATE, SORBITAN ISOSTEARATE, VP/HEXADECENE COPOLYMER, MENTHONE GLYCERIN ACETAL, DEHYDROACETIC ACID, FLAVOR, TITANIUM DIOXIDE (CI 77891), IRON OXIDES (CI 77491, CI 77492, CI 77499), YELLOW 5 LAKE (CI 19140), RED 7 LAKE (CI 15850).
| LAURA GELLER X BLISS SHINE - N - SHIELD LIP BALM SPF 15 SUNSET SANDS
zinc oxide lipstick |
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - COSMAX USA, CORP (010990210) |
| Registrant - COSMAX USA, CORP (010990210) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| COSMAX USA, CORP | 010990210 | manufacture(68577-221) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
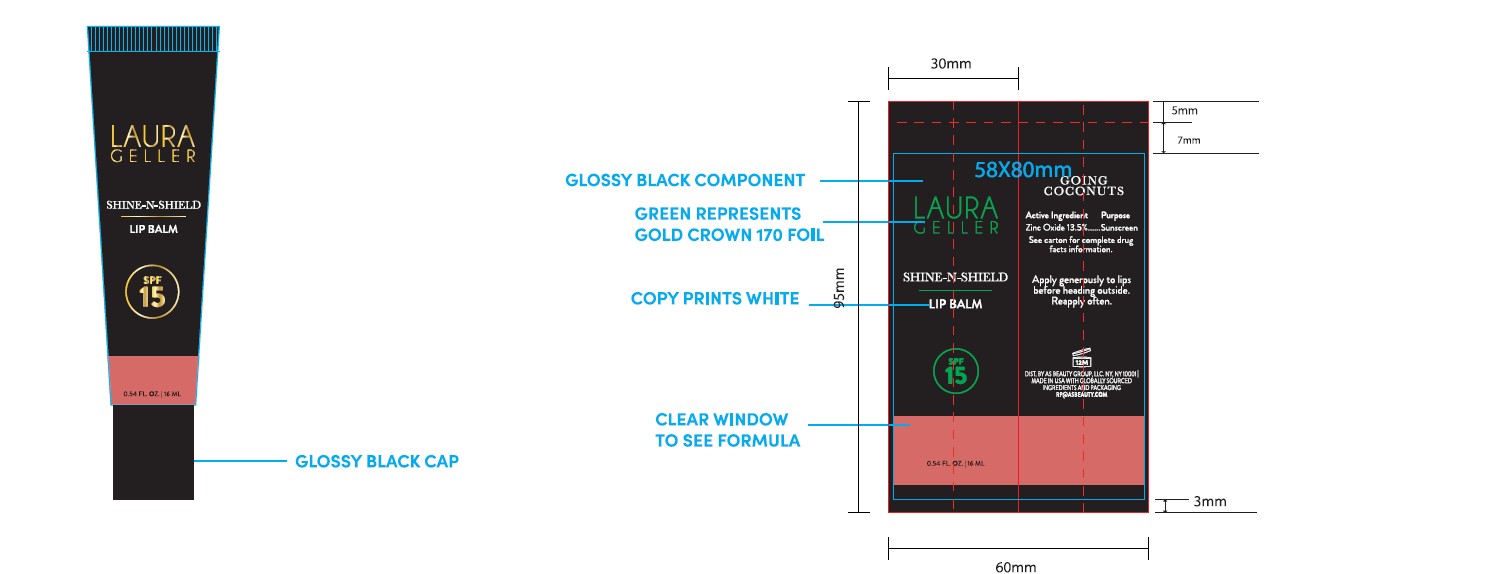
 Laura Geller Shine-n-Shield Lip Balm SPF 15 0.54 fl.oz./16ML
Laura Geller Shine-n-Shield Lip Balm SPF 15 0.54 fl.oz./16ML