NUMELVI- atinvicitinib tablet
Numelvi by
Drug Labeling and Warnings
Numelvi by is a Animal medication manufactured, distributed, or labeled by Merck Sharp & Dohme Corp.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- WARNINGS
- DESCRIPTION
- VETERINARY INDICATIONS
-
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATION:
NUMELVI should be administered orally, once daily, at a dose of 0.36 to 0.54 mg atinvicitinib/lb (0.8 to 1.2 mg atinvicitinib/kg) body weight according to the Dosing Table below.
NUMELVI should be administered with food.
Dosing Table
Body Weight
Ranges (lb)Body Weight
Ranges (kg)Number of Tablets to be Administered 4.8 mg tablets 7.2 mg tablets 21.6 mg tablets 31.6 mg tablets 4.4 - 6.5 2 - 2.9 0.5 6.6 - 9.5 3 - 4.3 0.5 9.6 - 13.3 4.4 - 6 1 13.4 - 19.9 6.1 - 9 1 20 - 29.8 9.1 - 13.5 0.5 29.9 - 42.6 13.6 - 19.3 0.5 42.7 - 58.4 19.4 - 26.5 1 58.5 - 87 26.6 - 39.5 1 87.1 - 118.9 39.6 - 54 1.5 119 - 174.2 54.1 - 79 2 ≥ 174.3 ≥79.1 Administer the appropriate combination of tablet strengths -
WARNINGS:
User Safety Warnings:
This product is not for human use. Keep this and all drugs out of reach of children. Wash hands thoroughly with soap and water immediately after handling tablets. In case of accidental ingestion, seek medical attention immediately.
Animal Safety Warnings:
Dogs should be monitored for the development of infections because NUMELVI may increase susceptibility to opportunistic infections, including demodicosis and interdigital furunculosis (see Target Animal Safety).
NUMELVI is not for use in dogs with serious infections.
NUMELVI is a JAK inhibitor. New neoplastic conditions (benign and malignant) have been reported in dogs treated with other JAK inhibitors.
Consider the risks and benefits of treatment prior to initiating NUMELVI in dogs with a history of recurrent demodicosis.
NUMELVI modulates the immune system.
NUMELVI is not for use in dogs less than 6 months of age (see Target Animal Safety).
Keep NUMELVI in a secure location out of reach of dogs, cats, and other animals to prevent accidental ingestion or overdose.
-
PRECAUTIONS:
The effectiveness and safety of NUMELVI have not been evaluated in a field study beyond 28 days (see Adverse Reactions and Effectiveness).
The safe use of NUMELVI has not been evaluated in breeding, pregnant, or lactating dogs. Decreased mean testes weight was observed in a laboratory safety study (see Target Animal Safety).
The safe use of NUMELVI has not been evaluated in combination with glucocorticoids, cyclosporine, or other systemic immunosuppressive agents.
-
ADVERSE REACTIONS:
In a masked field study assessing effectiveness and safety of NUMELVI for the control of pruritus associated with allergic dermatitis in dogs, 144 NUMELVI-treated dogs and 144 placebo-treated dogs were evaluated for safety for up to 28 days. Adverse reactions seen during the field study are summarized in Table 1 below.
Table 1. Adverse Reactions through Day 28 Adverse Reaction NUMELVI (N=144)
Number of dogs (%)Placebo (N=144)
Number of dogs (%)Vomiting or nausea 10 (6.9%) 6 (4.2%) Otitis externa 9 (6.3%) 8 (5.6%) Hematuria (without urinary tract infection) 7 (4.9%) 6 (4.2%) Anorexia 6 (4.2%) 5 (3.5%) Bacterial skin infection 6 (4.2%) 10 (6.9%) Diarrhea 6 (4.2%) 15 (10.4%) Crystalluria 5 (3.5%) 2 (1.4%) Lethargy 5 (3.5%) 5 (3.5%) Urinary tract infection 5 (3.5%) 5 (3.5%) Upset stomach, including flatulence, retching, and bloating 3 (2.1%) 0 Neurological disorder (e.g., tremors, ataxia) 2 (1.4%) 1 (0.7%) Ocular discharge 2 (1.4%) 1 (0.7%) Coughing 1 (0.7%) 0 Granuloma 1 (0.7%) 0 Increased urination 1 (0.7%) 0 Abnormal hematology results likely related to NUMELVI administration included leukopenia, neutropenia, eosinopenia, monocytopenia, and lymphocytosis.
Abnormal serum chemistry results likely related to NUMELVI administration included increased alanine aminotransferase (ALT), aspartate aminotransferase (AST), and symmetric dimethylarginine (SDMA), and hypercholesterolemia.
Three NUMELVI-treated dogs withdrew from the study early due to an adverse reaction, two of which were considered likely related to NUMELVI treatment (i.e., diarrhea). Two placebo-treated dogs also withdrew from the study early due to an adverse reaction (i.e., diarrhea).
-
CONTACT INFORMATION:
For technical information or to report a suspected adverse event, please contact Merck Animal Health at 1-800-224-5318 or https://www.merck-animal-health-usa.com. Safety Data Sheets (SDSs) can be found at https://www.merck.com/products/safety-data-sheets/#.
For additional information about reporting adverse drug experiences for animal drugs, contact FDA at 1-888-FDA-VETS or https://www.fda.gov/reportanimalae.
-
CLINICAL PHARMACOLOGY:
Mechanism of Action
Atinvicitinib is a Janus kinase (JAK) inhibitor. It inhibits the function of a variety of pruritogenic, pro-inflammatory and allergy related cytokines that are dependent upon JAK enzymes. Atinvicitinib is more selective for JAK1 compared to JAK2, JAK3, and tyrosine kinase 2 (TYK2). Atinvicitinib is not a corticosteroid or an antihistamine.
Pharmacokinetics
In a laboratory margin of safety study in healthy 6-month-old dogs (see Target Animal Safety), after repeat daily oral administration of atinvicitinib at 1.2 mg/kg for 6 months, the atinvicitinib mean (coefficient of variation % [CV%]) maximum concentration (Cmax) was 214 ng/mL (42%) with a median Tmax (time to Cmax) of 2 (range 1-6) hours. The mean area under the curve from the time of dosing to the last quantifiable plasma concentration (AUClast) and half-life were 888 h*ng/mL (27%) and 2.3 hours (27%), respectively. Minimal accumulation was observed between Days 1 and 182 with geometric mean accumulation ratios for Cmax and AUClast of 1.0 and 1.2. AUClast and Cmax increased in a linear but less than proportional manner where a 5-fold increase in dose resulted in a 4.5-fold (95% confidence interval [CI]: 3.6-5.3) and 3.7-fold (95% CI: 3.1-4.2) increase in AUClast and Cmax , respectively.
Following a single oral or intravenous administration of atinvicitinib, the geometric mean oral bioavailability based on AUClast was 45.5%.
The mean systemic clearance following intravenous administration is 1074 mL/h/kg with a harmonic mean terminal half-life of 1.1 hours and a mean volume of distribution of 1651 mL/kg. Following a single oral administration of atinvicitinib at 1.2 mg/kg, Cmax and AUClast were 280% and 120% higher, respectively, in the fed state as compared to the fasted state (n=16). Atinvicitinib was found to be mainly excreted in the feces while urinary elimination was a minor route of excretion. Atinvicitinib has moderate protein binding with 82.3% bound in fortified canine plasma at a concentration of 1802 ng/mL (5 μM).
-
SPL UNCLASSIFIED SECTION
EFFECTIVENESS:
A masked, 28-day, placebo-controlled study was conducted at 26 veterinary clinics in the US, enrolling 288 client-owned dogs diagnosed with allergic dermatitis and having at least moderate pruritus. The allergic dermatitis was attributed to one or more of the following conditions (in the order of frequency): atopic dermatitis, unspecified allergic dermatitis, food hypersensitivity, flea allergy dermatitis, and contact dermatitis. Dogs were randomized to once daily treatment with NUMELVI at 0.8 - 1.2 mg/kg or placebo, at a ratio of 1:1 respectively. Other medications that could affect the evaluation of effectiveness were not allowed during the study, such as corticosteroids, antihistamines, and cyclosporine. Treatment success for each dog was defined as a ≥ 50% reduction from baseline in owner-assessed pruritus scores on the Pruritus Visual Analog Scale (PVAS) on at least 5 out of the first 7 days of treatment. The proportion of dogs in the NUMELVI group that were treatment successes was greater than and significantly different compared to the placebo group on Day 7 (Table 2, below).
Table 2. Estimated Proportion of Dogs Achieving Treatment Success Treatment Group Estimated Proportion of Success* N = number of dogs - * Based on back-transformed least squares means.
- † Placebo vs. NUMELVI p = 0.0128
NUMELVI (N = 125) 0.23 Placebo (N = 125) 0.07† Mean owner-assessed PVAS scores were lower in the NUMELVI group after Day 0 (Figure 1, below).
Figure 1: Mean owner-assessed PVAS Scores by Treatment for Days 0-7
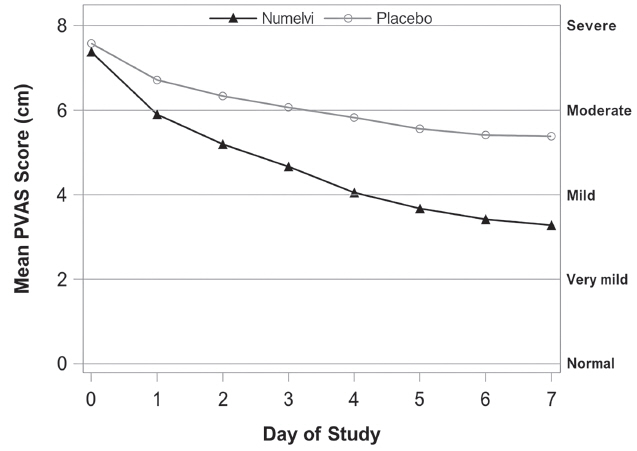
Veterinarians used a Dermatitis Visual Analog Scale (DVAS) to assess each dog's dermatitis. Veterinarian-assessed DVAS scores were lower for the NUMELVI group compared to the placebo group at all time points through Day 28.
-
TARGET ANIMAL SAFETY:
Margin of Safety Study
NUMELVI was administered orally to 24 healthy, 6-month-old Beagle dogs at 1X, 3X, and 5X (8 dogs per group) the maximum labeled dose of 1.2 mg/kg once daily for 6 months. The 8 dogs in the control group were sham dosed. Dogs were dosed in the fed state. One dog in the 5X group developed progressive alopecia, fever, lethargy, generalized lymphadenitis, interdigital furunculosis (cysts), interdigital swellings, and pustules on the border of the oral cavity. The dog had a declining clinical condition under continued treatment with NUMELVI and was euthanized on Day 175. The lesions were histologically consistent with generalized demodicosis including marked, chronic, active dermal inflammation, and minimal, multifocal, neutrophilic infiltration in the mandibular lymph node and were considered due to NUMELVI-induced immunosuppression. The dog demonstrated greater systemic exposure to NUMELVI (higher Cmax and AUClast values) compared to other dogs in the 5X group. One dog in the 3X group developed a single interdigital cyst with no Demodex canis mites identified in the hair follicles. Male dogs in the NUMELVI treatment groups had decreased mean testes weight compared to the control dogs. Microscopic pathology findings in one 1X dog with pre-existing unilateral cryptorchidism and one 3X dog included unilateral, focal, minimal testicular tubular hypoplasia. There were no NUMELVI-related effects on clinical pathology.
Vaccine Response Study
NUMELVI was administered orally to 10 healthy, 6-month-old, vaccine naïve Beagle dogs at 3X the maximum labeled dose of 1.2 mg/kg once daily for 84 days. The dogs in the control group (10) received placebo tablets. All dogs were dosed in the fed state. After the last dose on Day 84, all dogs were observed for an additional 84 days. All dogs received a multivalent modified live canine adenovirus type-2 (CAV), canine distemper virus (CDV), canine parvovirus (CPV) vaccination and a killed rabies virus (RV) vaccination on Day 28. An additional multivalent modified live CAV, CDV, CPV vaccination was administered on Day 56. All dogs in the NUMELVI treatment group demonstrated an adequate serological immune response to the modified live CAV, CDV, CPV, and killed RV vaccination on Day 84. There were no NUMELVI-related adverse clinical effects.
- STORAGE CONDITIONS:
- HOW SUPPLIED:
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 4.8 mg Bottle Carton
- PRINCIPAL DISPLAY PANEL - 7.2 mg Bottle Carton
- PRINCIPAL DISPLAY PANEL - 21.6 mg Bottle Carton
- PRINCIPAL DISPLAY PANEL - 31.6 mg Bottle Carton
-
INGREDIENTS AND APPEARANCE
NUMELVI
atinvicitinib tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 0061-1434 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATINVICITINIB (UNII: C238YE66P2) (ATINVICITINIB - UNII:C238YE66P2) ATINVICITINIB 4.8 mg Product Characteristics Color WHITE (White to off-white) Score 2 pieces Shape OVAL (Oblong shape) Size 11mm Flavor Imprint Code S;S Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0061-1434-01 1 in 1 CARTON 1 30 in 1 BOTTLE, PLASTIC 2 NDC: 0061-1434-02 1 in 1 CARTON 2 90 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141596 02/25/2026 NUMELVI
atinvicitinib tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 0061-1435 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATINVICITINIB (UNII: C238YE66P2) (ATINVICITINIB - UNII:C238YE66P2) ATINVICITINIB 7.2 mg Product Characteristics Color WHITE (White to off-white) Score 2 pieces Shape OVAL (Oblong shape) Size 12mm Flavor Imprint Code M;M Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0061-1435-01 1 in 1 CARTON 1 30 in 1 BOTTLE, PLASTIC 2 NDC: 0061-1435-02 1 in 1 CARTON 2 90 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141596 02/25/2026 NUMELVI
atinvicitinib tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 0061-1436 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATINVICITINIB (UNII: C238YE66P2) (ATINVICITINIB - UNII:C238YE66P2) ATINVICITINIB 21.6 mg Product Characteristics Color WHITE (White to off-white) Score 2 pieces Shape OVAL (Oblong shape) Size 17mm Flavor Imprint Code L;L Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0061-1436-01 1 in 1 CARTON 1 30 in 1 BOTTLE, PLASTIC 2 NDC: 0061-1436-02 1 in 1 CARTON 2 90 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141596 02/25/2026 NUMELVI
atinvicitinib tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 0061-1437 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATINVICITINIB (UNII: C238YE66P2) (ATINVICITINIB - UNII:C238YE66P2) ATINVICITINIB 31.6 mg Product Characteristics Color WHITE (White to off-white) Score 2 pieces Shape OVAL (Oblong shape) Size 20mm Flavor Imprint Code XL;XL Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0061-1437-01 1 in 1 CARTON 1 30 in 1 BOTTLE, PLASTIC 2 NDC: 0061-1437-02 1 in 1 CARTON 2 90 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141596 02/25/2026 Labeler - Merck Sharp & Dohme Corp. (001317601)
Trademark Results [Numelvi]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NUMELVI 98773651 not registered Live/Pending |
Intervet Inc. 2024-09-27 |
 NUMELVI 90842291 not registered Live/Pending |
Intervet Inc. 2021-07-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.



