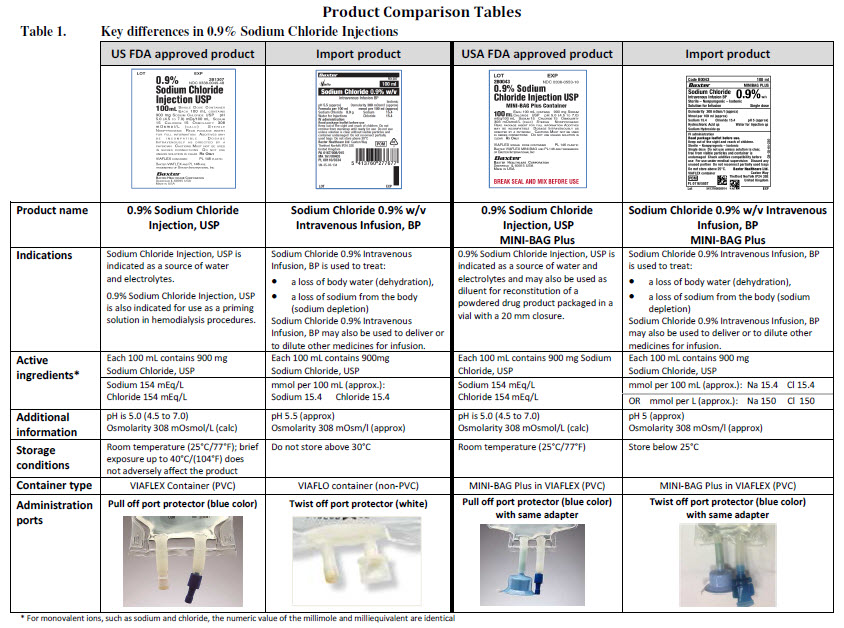
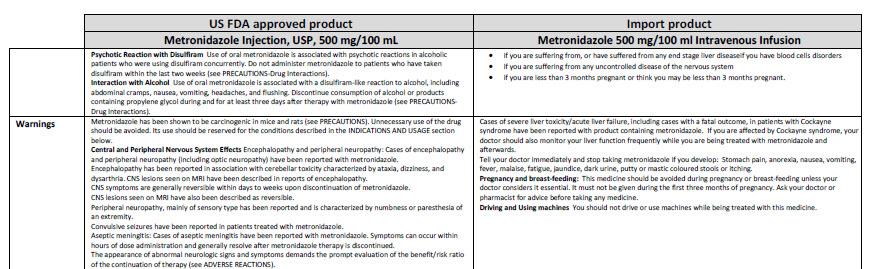
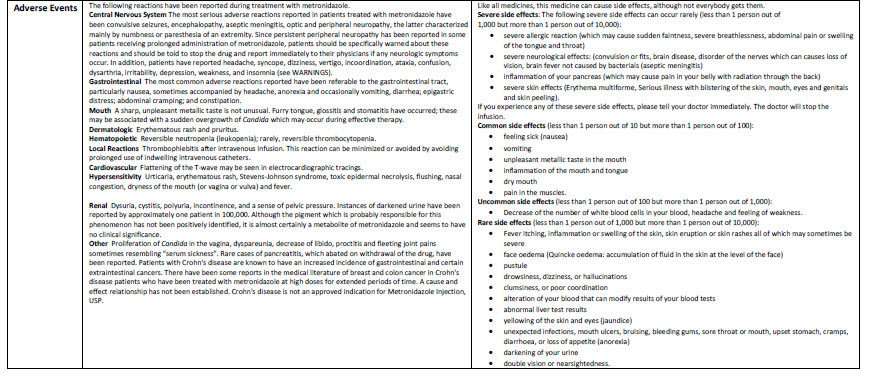
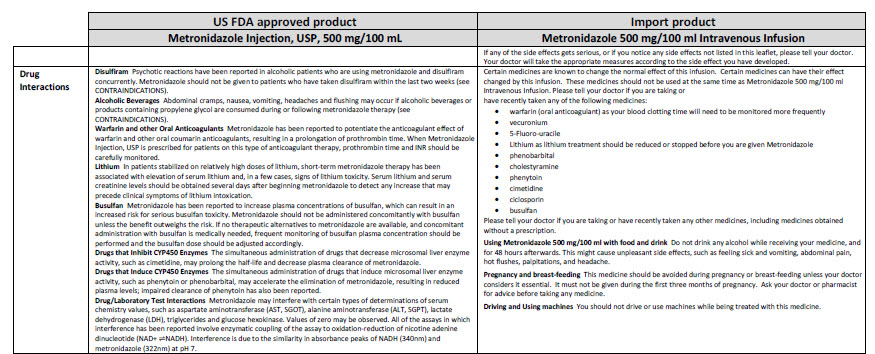
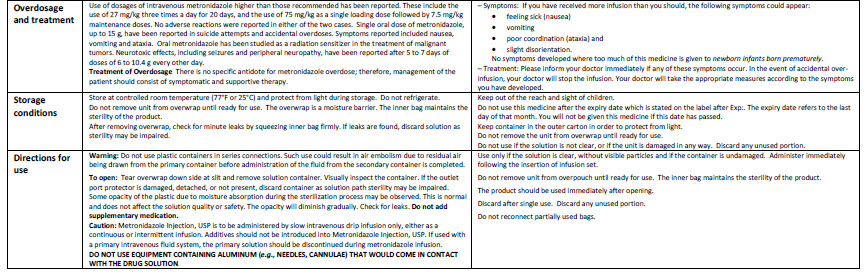
Metronidazole 500 mg/100 ml Intravenous Infusion
Metronidazole by
Drug Labeling and Warnings
Metronidazole by is a Prescription medication manufactured, distributed, or labeled by Baxter Healthcare Corporation, Baxter Healthcare S.A.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
METRONIDAZOLE- metronidazole injection, solution
Baxter Healthcare Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Metronidazole 500 mg/100 ml Intravenous Infusion
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
Baxter Logo
Viaflo Logo
FE3400
Metronidazole
500 mg/100 ml
Intravenous Infusion
100 ml
Iso-osmotic
Sterile nonpyrogenic
Single dose
Osmolarity
Solution for infusion
pH 4.5 – 6.0 (approx)
308 m0smol/l (approx.)
Each 100 ml contains
Metronidazole 500 mg and also includes Citric acid
monohydrate Disodium phosphate dodecahydrate, Sodium
chloride and Water for Injections
For intravenous administration
Store out of sight and reach of children Do not use unless
solution is clear and container is undamaged Additions should
not be made to this solution unless compatibility is known For
use under medical supervision Discard any unused portion Do
not use in a series of connection Do not reconnect partially
used bags Store in the original overpouch Do not remove unit
from the overpouch until ready to use Protect from light
Keep container in the outer carton
Baxter Healthcare Ltd.,
Thetford
Norfolk, England
PL00116/0353
POM Symbol
UN-35-03-268
Bar Code
5 413760277752
Lot
Expiry
| METRONIDAZOLE
metronidazole injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Baxter Healthcare Corporation (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Healthcare S.A. | 988899845 | ANALYSIS(0338-9554) , MANUFACTURE(0338-9554) , LABEL(0338-9554) , STERILIZE(0338-9554) | |