1.INDICATIONSAND USAGE
SILDENAFIL ORAL FILM is indicated for the treatment of erectile dysfunction.
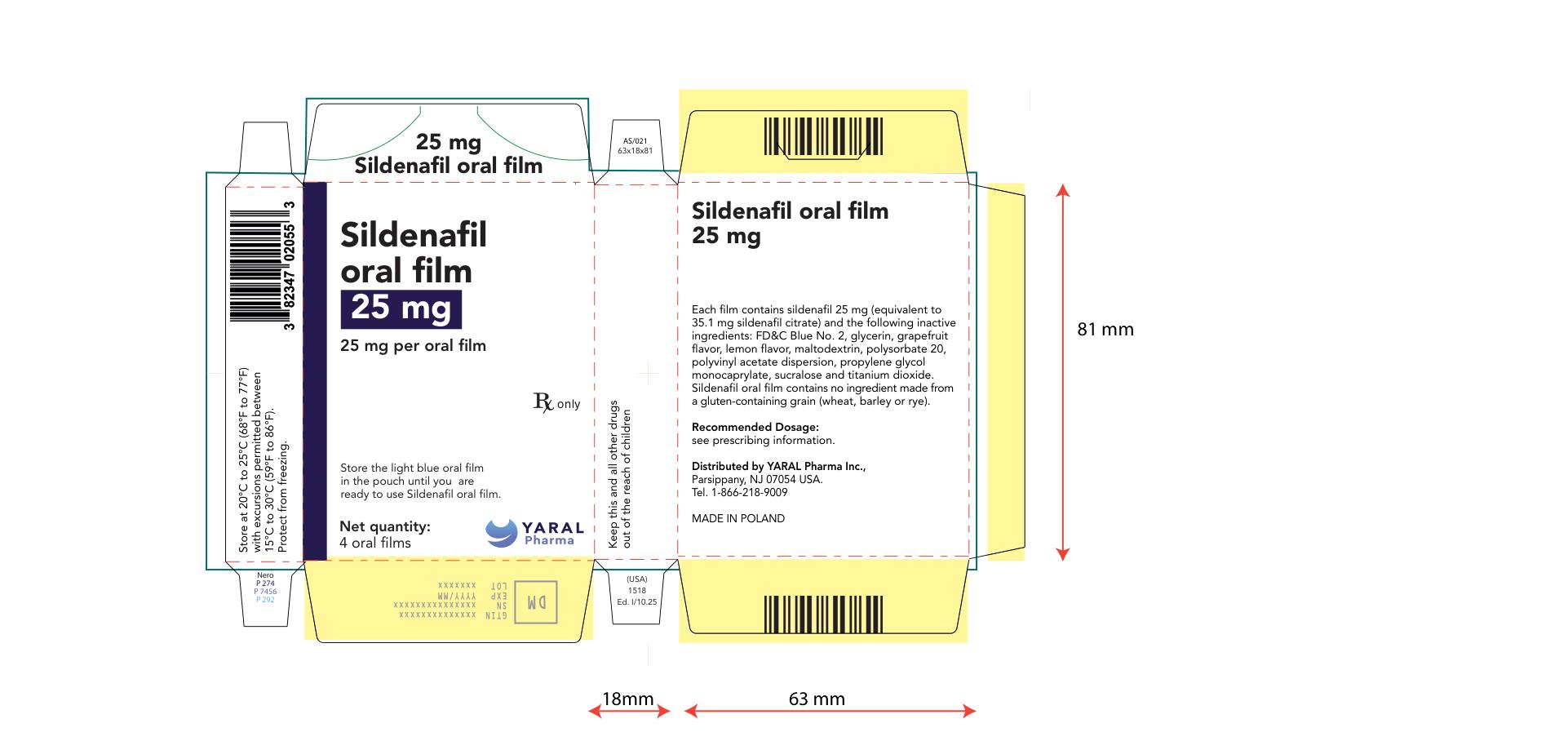
PRINCIPAL DISPLAY PANEL
NDC: 43328-020-04
25 mg
Sildenafil oral film
25 mg per oral film
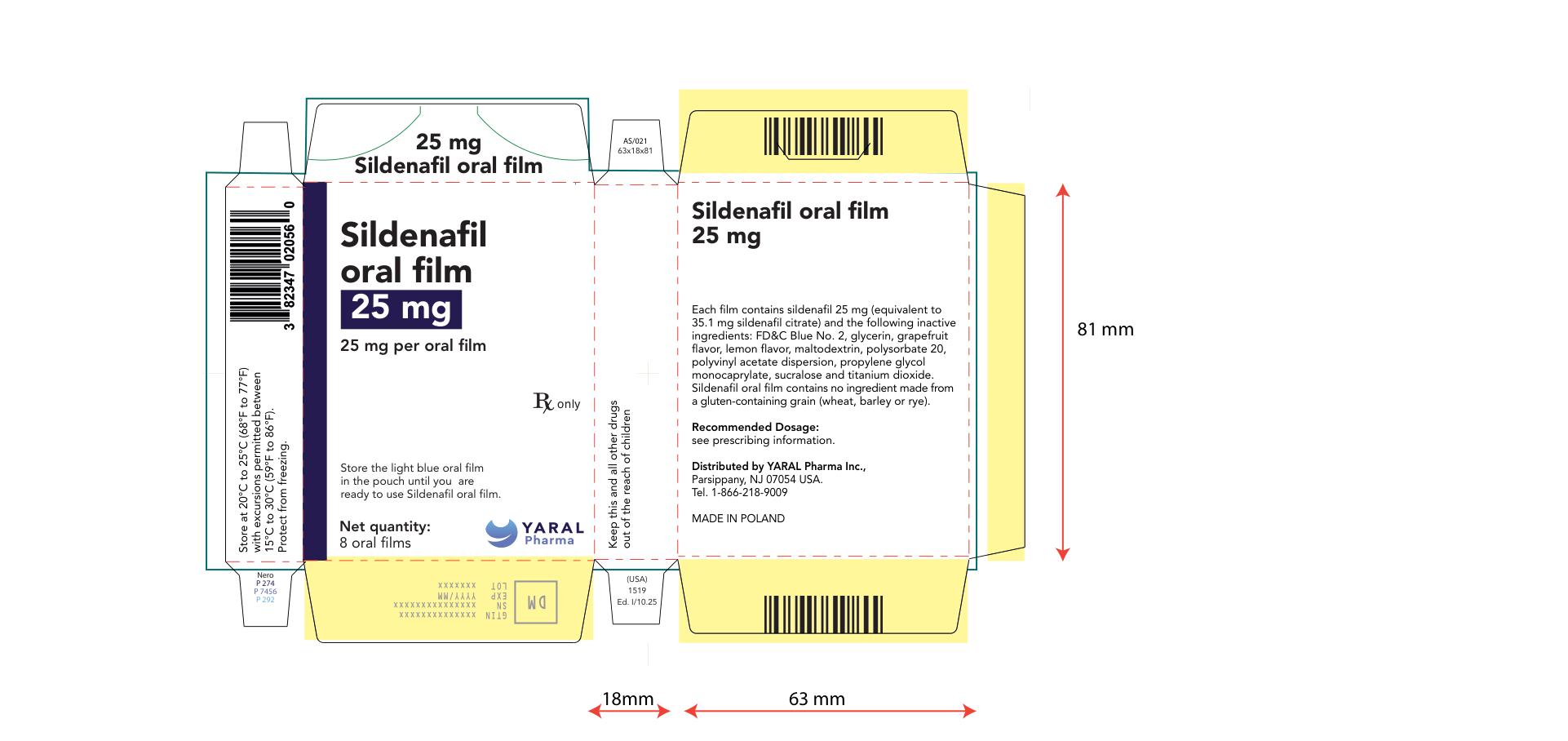
PRINCIPAL DISPLAY PANEL
NDC: 43328-020-08
25 mg
Sildenafil oral film
25 mg per oral film
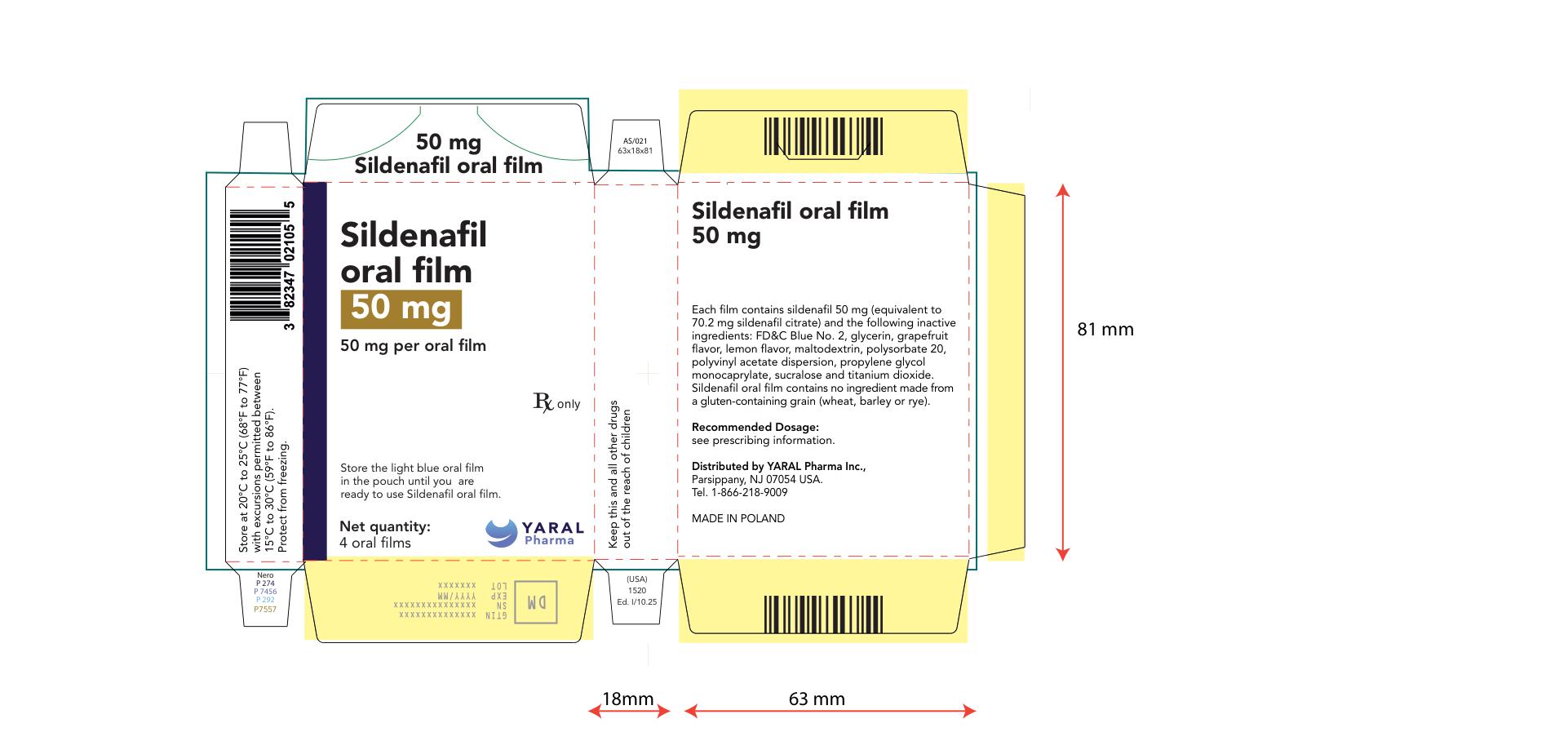
PRINCIPAL DISPLAY PANEL
NDC: 43328-021-04
50 mg
Sildenafil oral film
50 mg per oral film
PRINCIPAL DISPLAY PANEL
NDC: 43328-021-08
75 mg
Sildenafil oral film
75 mg per oral film
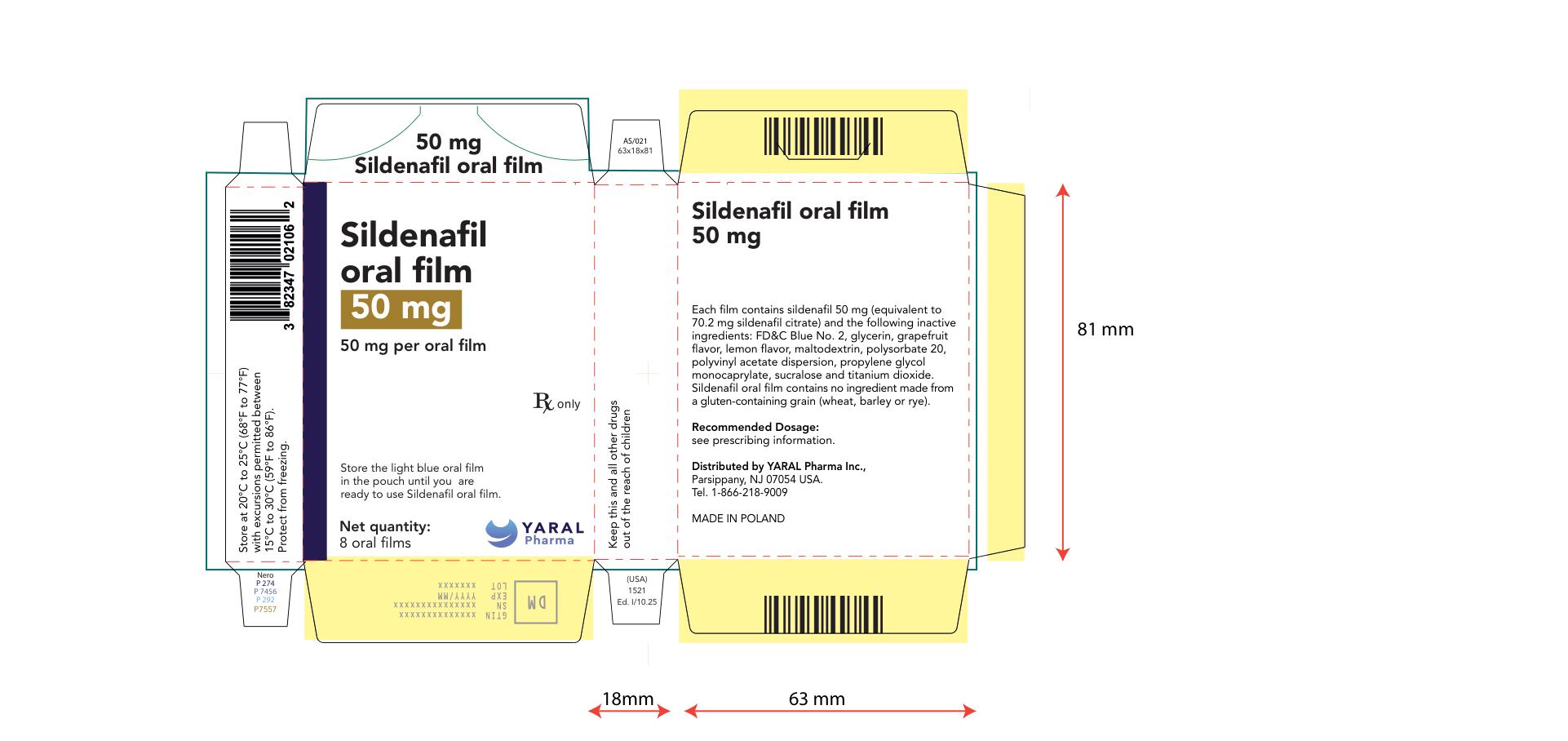
PRINCIPAL DISPLAY PANEL
NDC: 43328-022-04
75 mg
Sildenafil oral film
75 mg per oral film

PRINCIPAL DISPLAY PANEL
NDC: 43328-020-08
75 mg
Sildenafil oral film
75 mg per oral film

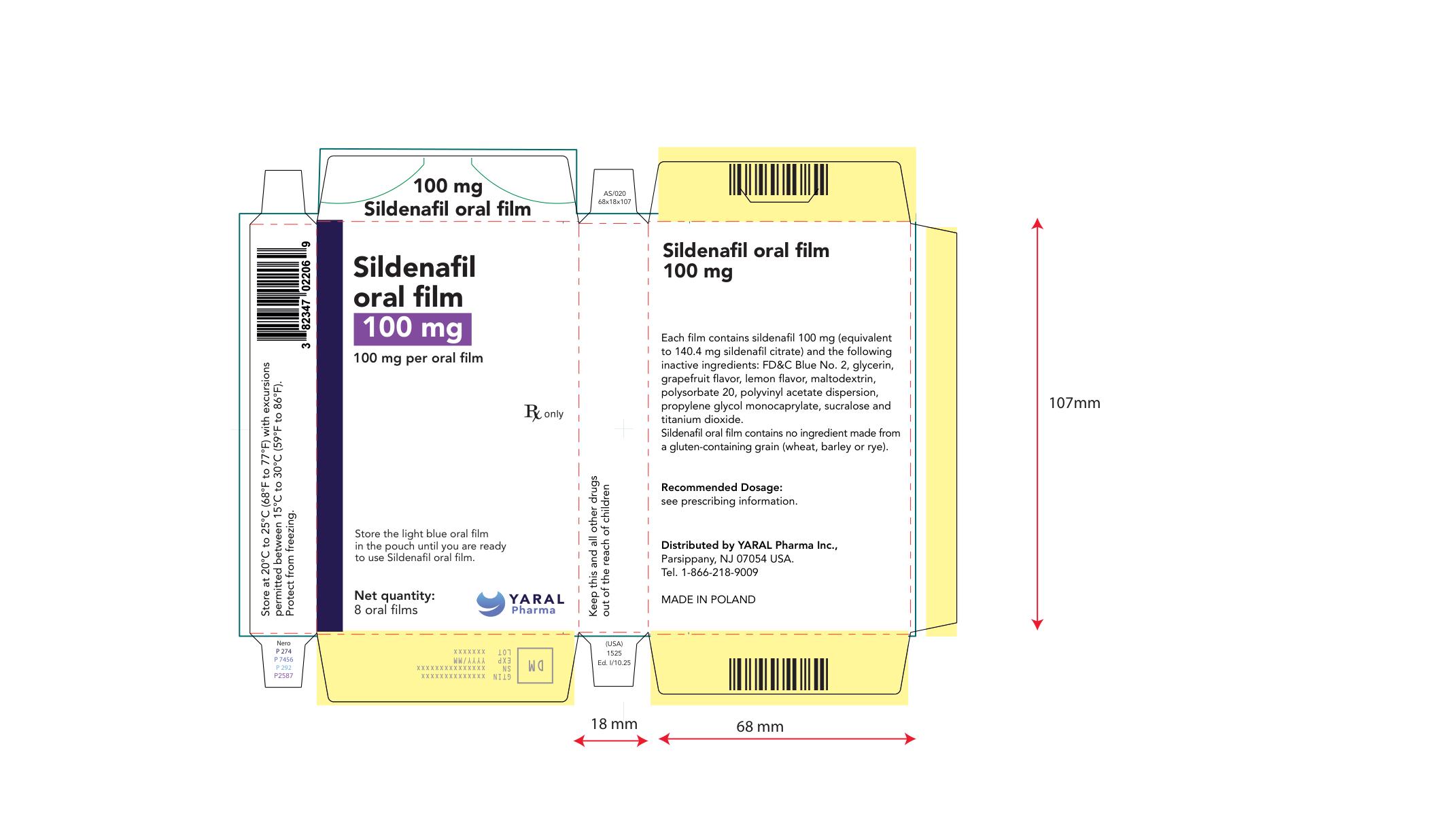
PRINCIPAL DISPLAY PANEL
NDC: 43328-023-04
100 mg
Sildenafil oral film
100 mg per oral film
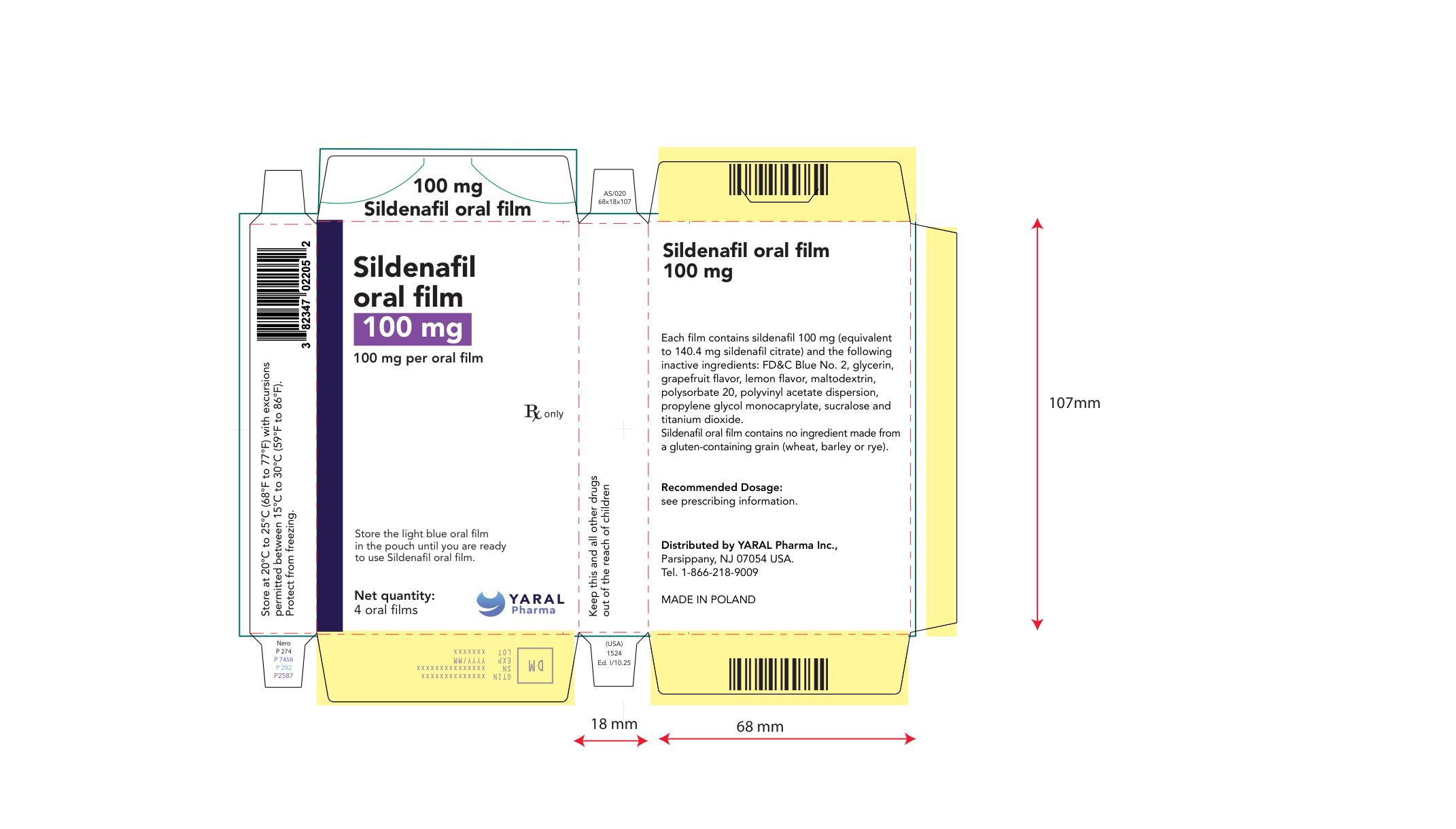
PRINCIPAL DISPLAY PANEL
NDC: 43328-023-08
100 mg
Sildenafil oral film
100 mg per oral film