LOPERAMIDE HYDROCHLORIDE- loperamide hydrochloride oral solution
Loperamide Hydrochloride by
Drug Labeling and Warnings
Loperamide Hydrochloride by is a Otc medication manufactured, distributed, or labeled by Bionpharma Inc., OrBion Pharmaceuticals Private Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each 7.5 mL)
- Purpose
- Use
-
Warnings
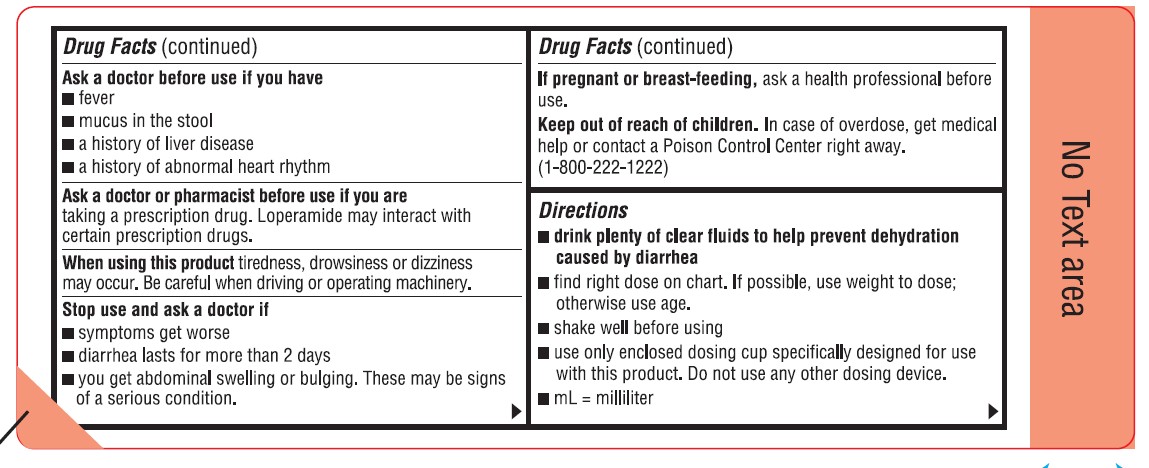
Ask a doctor before use if you have
- fever
- mucus in the stool
- a history of liver disease
- a history of abnormal heart rhythm
Ask a doctor or pharmacist before use if you are taking a prescription drug. Loperamide may interact with certain prescription drugs.
When using this product tiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
-
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise use age.
- shake well before using
- use only enclosed dosing cup specifically designed for use with this product. Do not use any other dosing device.
- mL = milliliter
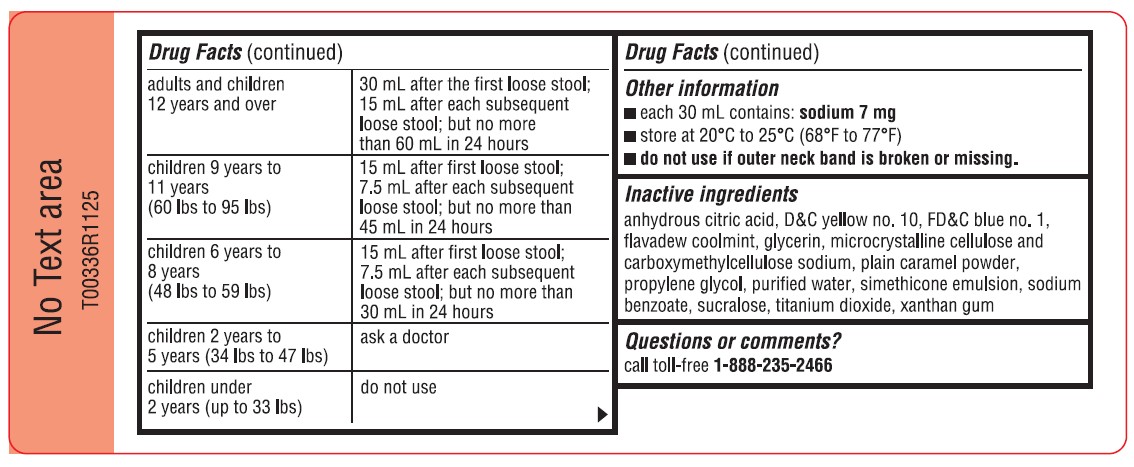
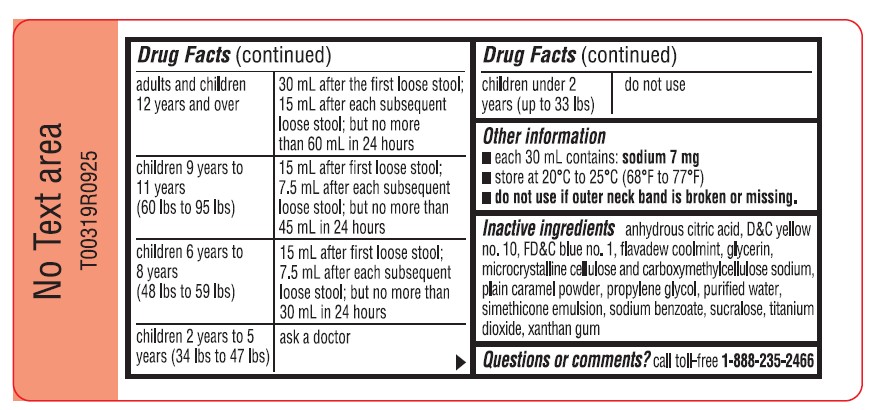
adults and children 12 years and over 30 mL after the first loose stool; 15 mL after each subsequent loose stool; but no more than 60 mL in 24 hours children 9 years to 11 years (60 lbs to 95 lbs) 15 mL after first loose stool; 7.5 mL after each subsequent loose stool; but no more than 45 mL in 24 hours children 6 years to 8 years (48 lbs to 59 lbs) 15 mL after first loose stool; 7.5 mL after each subsequent loose stool; but no more than 30 mL in 24 hours children 2 years to 5 years (34 lbs to 47 lbs) ask a doctor children under 2 years (up to 33 lbs) do not use - Other information
- Inactive ingredients
- Questions or comments?
-
SPL UNCLASSIFIED SECTION
Do not use if outer neck band is broken or missing
PEEL OPEN TO READ COMPLETE DIRECTIONS AND WARNINGS BEFORE PURCHASE
*This product is not manufactured or distributed by the owners of Imodium ®A-D.
Code: TN/Drugs/TN00002222/2006
L0001188 R1125 948006945
Distributed by:
BIONPHARMA INC.
Princeton, NJ 08540MADE IN INDIA
- PRINCIPAL DISPLAY PANEL-8 fl oz
- PRINCIPAL DISPLAY PANEL-4 fl oz
-
INGREDIENTS AND APPEARANCE
LOPERAMIDE HYDROCHLORIDE
loperamide hydrochloride oral solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 69452-405 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 1 mg in 7.5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GLYCERIN (UNII: PDC6A3C0OX) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) CARAMEL (UNII: T9D99G2B1R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) SODIUM BENZOATE (UNII: OJ245FE5EU) SUCRALOSE (UNII: 96K6UQ3ZD4) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color Score Shape Size Flavor MINT (flavadew coolmint) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69452-405-55 120 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/02/2026 2 NDC: 69452-405-93 240 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/03/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA220100 03/02/2026 Labeler - Bionpharma Inc. (079637826) Registrant - Bionpharma Inc. (079637826) Establishment Name Address ID/FEI Business Operations OrBion Pharmaceuticals Private Limited 854403569 manufacture(69452-405)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.