NUZOLVENCE- zoliflodacin for suspension
NUZOLVENCE by
Drug Labeling and Warnings
NUZOLVENCE by is a Prescription medication manufactured, distributed, or labeled by La Jolla Pharmaceutical Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use NUZOLVENCE safely and effectively. See full prescribing information for NUZOLVENCE.
NUZOLVENCE® (zoliflodacin) for oral suspension
Initial U.S. Approval: 2025INDICATIONS AND USAGE
NUZOLVENCE is a spiropyrimidinetrione bacterial type II topoisomerase inhibitor indicated for the treatment of uncomplicated urogenital gonorrhea due to Neisseria gonorrhoeae in adults and pediatric patients 12 years of age and older, weighing at least 35 kg. (1.1)
Usage to Reduce Development of Drug-Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of NUZOLVENCE and other antibacterial drugs, NUZOLVENCE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.2)
DOSAGE AND ADMINISTRATION
- Pregnancy Testing: Obtain a pregnancy test in females of reproductive potential prior to initiating NUZOLVENCE. (2.1)
- NUZOLVENCE must be mixed with water before administering. (2.2)
- Do not mix NUZOLVENCE with other liquids or sprinkle on food. (2.2)
- Administer the entire dose within 15 minutes of mixing. If the dose is not administered within 15 minutes of mixing, a new dose of NUZOLVENCE must be prepared. (2.2, 2.4)
- Adults and pediatric patients 12 years of age and older, weighing at least 35 kg: Recommended dose is 3 g (one packet) administered as a single dose orally. (2.3)
- Patients weighing 35 kg to less than 50 kg: Administer NUZOLVENCE on an empty stomach, 1 hour before or 2 hours after food. (2.3)
- Patients weighing greater than or equal to 50 kg: Administer NUZOLVENCE with food. (2.3)
- See full prescribing information for complete details on preparation and administration of NUZOLVENCE. (2.4)
DOSAGE FORMS AND STRENGTHS
For oral suspension: 3 g of zoliflodacin in each unit-dose packet of NUZOLVENCE. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Embryo-Fetal Toxicity: Potential Risk for Pregnant Females: May cause fetal harm when administered during pregnancy based on data from animal studies. Advise pregnant females about the potential risk to the fetus with maternal exposure to NUZOLVENCE. Avoid use of NUZOLVENCE during pregnancy. (5.1, 8.1, 8.3)
- Embryo-Fetal Toxicity: Potential Risk Related to Males with Female Partners of Reproductive Potential: Advise males with female partners of reproductive potential to use effective contraception for at least 3 months after administration of NUZOLVENCE. (5.2, 8.3, 13.1)
- Testicular Toxicity and Risks to Male Fertility: May cause testicular toxicity and impair male fertility based on data from animal studies. An assessment of spermatogenesis has not been conducted in humans. Advise males of the potential risk. (5.3, 8.3, 13.1)
- Hypersensitivity Reactions: Hypersensitivity reactions, including rash and pruritus, have been reported in patients receiving NUZOLVENCE. Discontinue NUZOLVENCE and institute appropriate supportive measures, if an allergic reaction occurs. (5.4)
- Clostridioides difficile Infection: Evaluate if diarrhea occurs. (5.5)
ADVERSE REACTIONS
The most common adverse reactions including laboratory abnormalities (incidence ≥2%) with NUZOLVENCE are neutropenia, headache, leukopenia, dizziness, nausea, and diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Entasis Therapeutics, Inc. at 1-800-651-3861 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 12/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Uncomplicated Urogenital Gonorrhea
1.2 Usage to Reduce Development of Drug-Resistant Bacteria
2 DOSAGE AND ADMINISTRATION
2.1 Pregnancy Testing in Females of Reproductive Potential
2.2 Important Administration Instructions
2.3 Recommended Dosage in Adults and Pediatric Patients 12 Years of Age and Older Weighing at Least 35 Kg
2.4 Preparation and Administration Instructions for NUZOLVENCE for Oral Suspension
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Embryo-Fetal Toxicity: Potential Risk for Pregnant Females
5.2 Embryo-Fetal Toxicity: Potential Risk Related to Males with Female Partners of Reproductive Potential
5.3 Testicular Toxicity and Risks to Male Fertility
5.4 Hypersensitivity Reactions
5.5 Clostridioides difficile Infection
5.6 Development of Drug-Resistant Bacteria
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on NUZOLVENCE
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Uncomplicated Urogenital Gonorrhea
NUZOLVENCE is indicated for the treatment of uncomplicated urogenital gonorrhea due to Neisseria gonorrhoeae in adults and pediatric patients 12 years of age and older, weighing at least 35 kg [see Clinical Studies (14)].
1.2 Usage to Reduce Development of Drug-Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of NUZOLVENCE and other antibacterial drugs, NUZOLVENCE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
-
2 DOSAGE AND ADMINISTRATION
2.1 Pregnancy Testing in Females of Reproductive Potential
Obtain a pregnancy test in females of reproductive potential prior to initiating treatment with NUZOLVENCE [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1, 8.3)].
2.2 Important Administration Instructions
NUZOLVENCE must be mixed with water before administering. Do not mix NUZOLVENCE with other liquids or sprinkle on food. Do not take NUZOLVENCE in the dry form. Administer the entire dose within 15 minutes of mixing. If the dose is not administered within 15 minutes of mixing, a new dose of NUZOLVENCE must be prepared [see Dosage and Administration (2.4)].
2.3 Recommended Dosage in Adults and Pediatric Patients 12 Years of Age and Older Weighing at Least 35 Kg
The recommended dose of NUZOLVENCE in adults and pediatric patients 12 years of age and older weighing at least 35 kg is 3 grams (g) (one packet) administered as a single dose orally.
See Table 1 for instructions on whether to administer NUZOLVENCE with or without food based on body weight [see Clinical Pharmacology (12.3)].
Table 1: Administration of NUZOLVENCE With or Without Food Based on Weight Dose of NUZOLVENCE Body Weight (kg) Administration of NUZOLVENCE Relative to the Ingestion of Food 3 g administered as a single, oral dose 35 kg to less than 50 kg Take on an empty stomach, 1 hour before or 2 hours after food Greater than or equal to 50 kg Take with food 2.4 Preparation and Administration Instructions for NUZOLVENCE for Oral Suspension
- NUZOLVENCE must be mixed with water before administering.
- Do not mix NUZOLVENCE with other liquids or sprinkle on food.
- Accurately measure 60 mL of water into the provided mixing container.
- Add the entire contents of one unit-dose packet of NUZOLVENCE to the mixing container and immediately secure the provided lid onto the mixing container. Shake vigorously for at least 60 seconds. Continue to shake until all granules are suspended and there is a uniform suspension.
- Once NUZOLVENCE is mixed to achieve a uniform suspension, administer the entire contents of the mixing container immediately.
- To ensure the full dose of NUZOLVENCE is consumed, add an additional 60 mL of water to the same mixing container, shake, and administer the entire additional 60 mL of water.
- Administer the entire dose within 15 minutes of mixing. If the dose is not administered within 15 minutes of mixing, a new dose of NUZOLVENCE must be prepared.
- See the Instructions for Use for additional details on the preparation and administration of NUZOLVENCE for oral suspension.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
NUZOLVENCE is contraindicated in:
- patients with a known history of hypersensitivity to NUZOLVENCE [see Warnings and Precautions (5.4)].
- patients who use concomitant moderate or strong CYP3A4 inducers because concomitant use is predicted to result in decreased plasma concentrations of zoliflodacin and may reduce the efficacy of NUZOLVENCE [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Embryo-Fetal Toxicity: Potential Risk for Pregnant Females
Based on data from animal studies, NUZOLVENCE may cause fetal harm when administered to a pregnant female at clinically relevant doses. Reproductive and developmental toxicity studies at AUC exposures 1.6-fold (mice) and 8.5-fold (rats) the maximum recommended human dose (MRHD) of zoliflodacin administered to pregnant rodents during organogenesis resulted in fetal malformations (exencephaly) and increased embryo-fetal loss [see Use in Specific Populations (8.1)].
Advise pregnant females about the potential risk to the fetus with maternal exposure to NUZOLVENCE. Avoid use of NUZOLVENCE during pregnancy. Obtain a pregnancy test prior to initiation of treatment with NUZOLVENCE in females of reproductive potential [see Dosage and Administration (2.1) and Use in Specific Populations (8.1, 8.3)].
5.2 Embryo-Fetal Toxicity: Potential Risk Related to Males with Female Partners of Reproductive Potential
Based on data from an animal toxicity study, the risk of early pregnancy loss may be increased in female partners of males treated with NUZOLVENCE. A reduced number of live embryos and increased number of embryonic losses were observed in untreated female rats mated with male rats administered zoliflodacin at exposures approximately 3.9-times the clinical exposure at the MRHD for 4 weeks.
Advise males with female partners of reproductive potential to use effective contraception for at least 3 months after administration of NUZOLVENCE. [see Use in Specific Populations (8.3) and Nonclinical Toxicology (13.1)].
5.3 Testicular Toxicity and Risks to Male Fertility
Based on findings from animal studies, NUZOLVENCE may cause testicular toxicity and impair male fertility. In repeat-dose toxicity studies, rats and dogs administered zoliflodacin for durations of 2 days to 4 weeks at AUC exposures 3- to 11-times the MRHD experienced minimal to moderate decreased spermatogenesis and testicular histopathological changes. In rats, loss of male rat fertility was no longer observed 4 weeks after the last dose; however, testicular histopathological changes in rats and dogs were only partially reversible after 2 to 3 months [see Nonclinical Toxicology (13.1)]. An evaluation of spermatogenesis has not been conducted in humans.
Advise males that NUZOLVENCE may cause testicular toxicity and impair male fertility [see Use in Specific Populations (8.3) and Nonclinical Toxicology (13.1)].
5.4 Hypersensitivity Reactions
Hypersensitivity reactions, including rash and pruritus, have been reported in patients receiving NUZOLVENCE [see Adverse Reactions (6.1)]. NUZOLVENCE is contraindicated in patients with a known history of hypersensitivity to NUZOLVENCE [see Contraindications (4)]. Before therapy with NUZOLVENCE is instituted, carefully inquire about previous hypersensitivity reactions to NUZOLVENCE.
If an allergic reaction to NUZOLVENCE occurs, institute appropriate supportive measures.
5.5 Clostridioides difficile Infection
Clostridioides difficile infection (CDI) has been reported with use of nearly all antibacterial agents and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDI. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDI must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDI has been reported to occur over two months after the administration of antibacterial agents.
If CDI is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.6 Development of Drug-Resistant Bacteria
Prescribing NUZOLVENCE in the absence of a proven or strongly suspected bacterial infection or prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria [see Indications and Usage (1.2)].
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in the Warnings and Precautions section of the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 782 patients received a 3 g dose of zoliflodacin across all phases of clinical trials.
The safety of NUZOLVENCE was evaluated in a phase 3, randomized, open-label, active-controlled, multicenter, multinational trial (NCT03959527) (Trial 1). In total, 927 patients with suspected uncomplicated gonorrhea due to N. gonorrhoeae were randomized (2:1) and treated with either a single oral 3 g dose of NUZOLVENCE (N=619) or a combination of a single 500 mg intramuscular dose of ceftriaxone and a single 1 g oral dose of azithromycin (N=308) [see Clinical Studies (14)]. Patients were eligible for enrollment if they were ≥12 years old and ≥35 kg. The majority (98%) of patients were adults (≥18 years); 14 patients were 15 to 18 years old: 12/619 (1.9%) in the NUZOLVENCE arm and 2/308 (0.6%) in the ceftriaxone and azithromycin arm. South Africa had the highest proportion of enrolled patients (46%), followed by Thailand (29%), the United States (17%), and the European Union (8%). The majority of patients treated with NUZOLVENCE were male (88%). Patients identified as Black or African American (56%), Asian (31%), White (11%), American Indian or Alaska Native (1%), or Other (1%). A total of 3% of patients treated with NUZOLVENCE identified as Hispanic or Latino and 22% were living with Human Immunodeficiency Virus (HIV).
Serious Adverse Reactions and Adverse Reactions Leading to Discontinuation:
There were no serious adverse reactions or adverse reactions leading to treatment discontinuation or death.
Common Adverse Reactions:
Table 2 lists adverse reactions occurring in ≥2% of patients receiving NUZOLVENCE in Trial 1.
Table 2: Adverse Reactions in ≥2% of Patients with Suspected Uncomplicated Gonorrhea Infection Treated with NUZOLVENCE in Trial 1* (Safety Population) Adverse Reaction NUZOLVENCE
N = 619
n (%)Ceftriaxone and Azithromycin
N = 308
n (%)N, total number of patients in treatment arm; n, number of patients meeting criteria. - * Trial 1 was not designed to evaluate meaningful comparisons of the incidence of adverse reactions in the NUZOLVENCE and the ceftriaxone and azithromycin treatment groups. The safety population includes patients with urogenital gonorrhea as well as those with uncomplicated gonorrhea infections at other body sites not covered under the approved indication.
- † Headache includes tension headache and headache.
Headache† 61 (10) 15 (5) Dizziness 21 (3) 5 (2) Nausea 16 (3) 12 (4) Diarrhea 15 (2) 22 (7) Headache: In Trial 1, headache was reported in 61/619 (10%) of patients receiving NUZOLVENCE; headache severity was mild in 8% of patients and moderate in 2%.
Laboratory Abnormalities
Laboratory abnormalities that occurred at a frequency of 2% or greater in Trial 1 are provided in Table 3.
Table 3: Laboratory Abnormalities in ≥2% of Patients with Suspected Uncomplicated Gonorrhea Infection Treated with NUZOLVENCE, with Normal Baseline Values in Trial 1* (Safety Population) Laboratory
ParametersNUZOLVENCE
N = 609
n (%)Ceftriaxone and Azithromycin
N = 304
n (%)N, total number of patients in treatment arm with normal baseline values; n, number of patients meeting criteria. - * Trial 1 was not designed to evaluate meaningful comparisons of the incidence of laboratory changes in the NUZOLVENCE and the ceftriaxone and azithromycin treatment groups. The safety population includes patients with urogenital gonorrhea as well as those with uncomplicated gonorrhea infections at other body sites not covered under the approved indication.
- † Neutropenia is defined as neutrophil count less than 1500 cells/µL.
- ‡ Leukopenia is defined as white blood cell count less than 3500 cells/µL.
Neutropenia† 71 (12) 43 (14) <1500 to 1000 cells/µL 56 (9) 31 (10) <1000 to 500 cells/µL 14 (2) 12 (4) <500 cells/µL 1 (0.2) 0 Leukopenia‡ 54 (9) 33 (11) <3500 to 3000 cells/µL 34 (6) 20 (7) <3000 to 1000 cells/µL 20 (3) 13 (4) <1000 cells/µL 0 0 Neutropenia: In Trial 1, 41/609 (7%) and 30/609 (5%) patients developed neutropenia from 4 to 8 days and from 27 to 33 days following NUZOLVENCE administration, respectively. Of these 71 patients, 25% were living with HIV and 8% had unknown HIV status. No patients required treatment for neutropenia.
Leukopenia: In Trial 1, 54/609 (9%) of patients developed leukopenia following NUZOLVENCE administration; 16% of these patients were living with HIV and 11% had unknown HIV status. No patients required treatment for leukopenia.
Other Adverse Reactions Associated with NUZOLVENCE in Trial 1
Adverse reactions occurring in less than 2% of patients receiving NUZOLVENCE in Trial 1 (Safety Population), are presented below:
Blood and lymphatic system disorders: Thrombocytopenia, monocyte count decreased, hemoglobin decreased
Cardiac disorders: Palpitations
Gastrointestinal disorders: Vomiting, abdominal pain, constipation, abdominal distension, flatulence
General disorders: Asthenia, fatigue, malaise, pyrexia, chills, feeling hot, night sweats
Hepatobiliary disorders: Alanine aminotransferase increased, blood bilirubin increased
Infections and infestations: Tinea infections, candidal infections
Musculoskeletal and connective tissue disorders: Musculoskeletal pain
Nervous system disorders: Somnolence, hypersomnia, hypoesthesia
Psychiatric disorders: Insomnia
Renal and urinary disorders: Hematuria, blood creatinine increased, glomerular filtration rate decreased
Respiratory, thoracic, and mediastinal disorders: Cough
Skin and subcutaneous tissue disorders: Rash, pruritus, alopecia, eyelid swelling
Vascular disorders: Hot flush
-
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on NUZOLVENCE
Moderate and Strong CYP3A4 Inducers
Concomitant use of moderate or strong inducers of CYP3A4 with NUZOLVENCE is contraindicated [see Contraindications (4)]. Zoliflodacin is a CYP3A4 substrate. Moderate and strong CYP3A4 inducers are predicted to result in decreased plasma concentrations of zoliflodacin and may reduce NUZOLVENCE efficacy [see Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on findings from animal studies, NUZOLVENCE may cause fetal malformations or increased embryo-fetal loss when administered to a pregnant female. In pregnant mice, repeat oral administration of zoliflodacin during organogenesis was associated with fetal malformations (exencephaly) and increased embryo-fetal loss at AUC exposures 1.6-fold the MRHD and decreased fetal weights at 2.9-fold the MRHD. In pregnant rats, zoliflodacin administration resulting in AUC exposures 8.5-fold the MRHD increased embryo-fetal loss, and exposures 3.2-fold the MRHD decreased fetal weights. There was no effect on embryo-fetal survival at exposures 5.5-fold the MRHD to zoliflodacin in pregnant rats. When zoliflodacin was administered to rats throughout pregnancy, parturition, and lactation, no effects on pup survival, birth weight, or growth were observed at maternal exposures up to 2.4-fold the MRHD (see Data).
There are no human data on NUZOLVENCE use in pregnancy to evaluate the drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes.
Advise pregnant females about the potential risk to the fetus with maternal exposure to NUZOLVENCE [see Warnings and Precautions (5.1) and Use in Specific Populations (8.3)].
A postmarketing descriptive pregnancy safety study is available for NUZOLVENCE. If exposure occurs during pregnancy, pregnant females or their healthcare providers should report the pregnancy to Entasis Therapeutics at 1-800-651-3861.
The background risk of major birth defects and miscarriage in the indicated population is unknown. All pregnancies have a background risk of birth defects, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Animal Data
In pregnant mice, repeat oral administration of zoliflodacin at 250, 500, and 1000 mg/kg/day during organogenesis (gestation days [GD] 5-16) was associated with fetal exencephaly at and above doses of 500 mg/kg/day (AUC exposures equal to or greater than 1.6-fold the MRHD). Increased implant losses occurred at 500 mg/kg/day, and decreased fetal weights occurred at 1000 mg/kg/day (AUC exposures 2.9-fold the MRHD). No adverse embryo-fetal effects were observed at 250 mg/kg/day (AUC exposure 0.6-fold the MRHD).
In female rats, repeat oral administration of zoliflodacin at 200, 500, or 1000 mg/kg/day from at least 2 weeks prior to mating through organogenesis (GD 16), decreased pregnancy rates and reduced embryo-fetal survival occurred at 1000 mg/kg/day (AUC exposures 8.5-fold the MRHD), without fetal malformations. No effect on embryo-fetal survival occurred at 500 mg/kg/day (AUC exposures 5.5-fold the MRHD). Across all dose levels, decreased fetal weights and delayed skeletal ossification were observed.
In pregnant rats, repeat oral administration of zoliflodacin from GD 6 through lactation Day 20 at doses up to 200 mg/kg/day (maternal AUC exposures 2.4-fold the MRHD at the end of gestation), resulted in no maternal toxicity or adverse effects on prenatal or postnatal offspring survival or growth. In offspring evaluated at 8-9 weeks, increased motor activity occurred in males and females at 200 mg/kg/day and in females at 100 mg/kg/day (maternal AUC exposures 1.4-fold MRHD at the end of gestation). At both dose levels, maternal AUC exposures were below clinical exposures at the end of lactation. Results of learning and memory assessments were indeterminate due to limitations of the study design.
8.2 Lactation
Risk Summary
There are no data on the presence of zoliflodacin in either human or animal milk, effects on the breastfed infant, or effects on milk production. If NUZOLVENCE is present in breast milk, intestinal flora alteration in the breastfed infant could occur. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for NUZOLVENCE and any potential adverse effects on the breastfed infant from NUZOLVENCE or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Based on animal studies, NUZOLVENCE may cause fetal malformations when administered to a pregnant female at clinically relevant doses. Additionally, based on data from an animal study, the risk of early pregnancy loss may be increased in partners of males treated with NUZOLVENCE [see Use in Specific Populations (8.1) and Nonclinical Toxicology (13.1)].
Pregnancy Testing
Obtain a pregnancy test in females of reproductive potential prior to initiating treatment with NUZOLVENCE [see Dosage and Administration (2.1), Warnings and Precautions (5.1), and Use in Specific Populations (8.1)].
Contraception
Males
Advise males with female partners of reproductive potential to use effective contraception for at least 3 months after their single-dose treatment of NUZOLVENCE [see Warnings and Precautions (5.2) and Nonclinical Toxicology (13.1)].
Infertility
Males
Based on data from repeat-dose animal toxicity and fertility studies, NUZOLVENCE may cause testicular toxicity and impair male fertility [see Warnings and Precautions (5.3) and Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of NUZOLVENCE for the treatment of uncomplicated urogenital gonorrhea have been established in pediatric patients 12 years of age and older, weighing at least 35 kg [see Indications and Usage (1.1]. Use of NUZOLVENCE in this pediatric population is supported by clinical, microbiological and safety data from an adequate and well-controlled trial (Trial 1) in adults and pediatric patients with uncomplicated urogenital gonorrhea and additional pharmacokinetic data in adult patients. In Trial 1, 12 patients aged 16 to <18 years, weighing 46 to 76.2 kg, received a single 3 g dose of NUZOLVENCE [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
The safety and effectiveness of NUZOLVENCE in pediatric patients younger than 12 years of age or weighing less than 35 kg have not been established.
-
11 DESCRIPTION
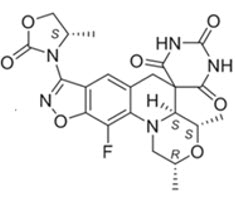
NUZOLVENCE for oral suspension contains zoliflodacin, an oral spiropyrimidinetrione bacterial type II topoisomerase inhibitor. The chemical name of zoliflodacin is (2R,4S,4aS)-11-Fluoro-1,2,4,4a-tetrahydro-2,4-dimethyl-8-[(4S)-4-methyl-2-oxo-3-oxazolidinyl]spiro[isoxazolo[4,5-g][1,4]oxazino[4,3-a]quinoline-5(6H),5'(2'H)-pyrimidine]-2',4',6'(1'H,3'H)-trione. Its molecular formula is C22H22FN5O7 and molecular mass is 487.4 g/mol. Its chemical structure is depicted below.
Each unit dose packet of NUZOLVENCE for oral suspension contains 3 g of zoliflodacin and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, talc, and xanthan gum.
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
The ratio of the unbound plasma zoliflodacin area under the concentration-time curve from time of dosing extrapolated to infinity to the zoliflodacin MIC (fAUC0-inf/MIC) is the best predictor of efficacy based on in vitro models of infection.
Cardiac Electrophysiology
A thorough QTc study of single oral doses of zoliflodacin 2 g and 4 g (not approved doses of NUZOLVENCE) was conducted in 72 healthy subjects aged 18 to 45 years. A concentration-dependent increase in QTc interval was observed in the thorough QTc study. Based on the observed relationship, clinically significant QTc interval prolongation is not expected at the maximum recommended single dose of NUZOLVENCE.
12.3 Pharmacokinetics
Zoliflodacin, as single doses, generally displayed dose-proportional increases in exposure up to 800 mg (0.27 times the recommended dosage). Increases above 800 mg led to slightly less than dose-proportional increases in exposure up to 4 g (1.3 times the recommended dosage).
The pharmacokinetic properties of zoliflodacin in healthy subjects are displayed in Table 4.
Table 4: Pharmacokinetic Properties of Zoliflodacin in Healthy Subjects Abbreviations: AUC = area under the concentration-time curve; CL/F=apparent total body clearance; Cmax = maximum drug concentration; CYP = Cytochrome P450; %GCV = % geometric coefficient of variation; T½=elimination half-life; Tmax=the time to Cmax; Vz/F=apparent volume of distribution. - * Based on studies with a moderate-fat, moderate-calorie meal consisting of approximately 462 kcal (39% fat, 51% carbohydrates, and 10% protein) or a high-fat, high-calorie meal consisting of approximately 884 kcal (estimated to contain approximately 55% fat, 29% carbohydrates, 16% protein).
- † Non-CYP-mediated clearance has not been fully characterized.
Absorption Tmax (h), median (minimum to maximum) Fasted: 2.5 (1.0 to 4.0); Fed: 4.0 (3.0 to 5.5) Food effect* At the 3 g dose, Cmax was increased by approximately 1.5-fold and AUC was increased by approximately 1.5 to 2-fold when given with a moderate or high fat meal vs fasted conditions. Distribution % bound to human plasma proteins 83% Blood-to-plasma ratio 0.69 Vz/F (L), geometric mean (%GCV) Fasted: 177 (26.6); Fed: 98.7 (24.1) Metabolism Metabolic pathways The primary clearance mechanism is metabolism through both CYP-mediated and non-CYP-mediated pathways.† CYP-mediated metabolism is predominantly via CYP 3A4/5 enzymes, with lesser contributions from CYP1A2, CYP2C9, CYP2C8, and CYP2C19. Elimination Major route of elimination Fecal T1/2(h), geometric mean (%GCV) Fasted: 6.4 (20.4); Fed 5.5 (14.0) CL/F (L/h), geometric mean (%GCV) Fasted: 19.1 (28.8); Fed: 12.5 (27.8) Excretion % drug-related material in feces 79.6% % of dose excreted unchanged in feces 1.5% % drug-related material in urine 18.2% % of dose excreted unchanged in urine 2.5% The predicted zoliflodacin exposure parameters in adult patients with uncomplicated urogenital gonorrhea are presented in Table 5.
Table 5: Zoliflodacin Exposures in Patients with Uncomplicated Urogenital Gonorrhea Pharmacokinetic Parameter* Geometric Mean (%GCV) Abbreviations: AUC0-inf = area under the concentration-time curve from time zero to infinity; Cmax = maximum drug concentration. - * Data presented as geometric mean (%CV) based on post hoc parameters from 24 patients enrolled in the Trial 1 (weight range 48.2 to 112.3 kg) who received zoliflodacin 3 g after a low/moderate fat meal.
Cmax (mcg/mL) 28.5 (21.6%) AUC0-inf (h*mcg/mL) 353 (24.1%) Effect of Food
Following administration of the 3 g dose of NUZOLVENCE with a moderate-to-high-fat meal, zoliflodacin Cmax was increased by approximately 1.5-fold and the AUC0-inf was increased by approximately 1.5- to 2-fold. With a high-fat, high-calorie meal (consisting of approximately 884 kcal, 55% fat, 29% carbohydrates, 16% protein), the fed vs. fasted ratios of adjusted geometric means (90% CI) AUC0-inf and Cmax were 2.01 (1.85, 2.18) and 1.52 (1.39-1.67), respectively. With a moderate-fat, moderate-calorie meal (consisting of approximately 462 kcal, [39% fat, 51% carbohydrates, and 10% protein), the fed vs. fasted ratios of adjusted geometric means (90%CI) AUC0-inf and Cmax were 1.53 (1.45, 1.61) and 1.49 (1.38, 1.61), respectively.
Specific Populations
No clinically significant differences in the pharmacokinetics of zoliflodacin were observed based on age (18 to 55 years old), sex, and race (White 61%, Black 28%, Asian 8%).
Body Weight
There is an inverse relationship between body weight and zoliflodacin exposure. Simulations were conducted under moderate-fat/moderate-calorie conditions similar to Trial 1 (e.g., meals with 400-500 calories, < 50% fat) to inform dosing conditions. Considering both the weight and food effects, patients weighing 35 kg to less than 50 kg should take NUZOLVENCE on an empty stomach and patients weighing 50 kg or more should take NUZOLVENCE with food [see Dosage and Administration (2.3)]. There are no clinical data in patients weighing less than 35 kg.
Pediatric Patients
There are no pharmacokinetic data in pediatric patients. Using modeling and simulation, the recommended dosage with administration based on weight is expected to result in comparable plasma exposures of zoliflodacin in pediatric patients 12 years of age and older and weighing at least 35 kg as observed in healthy adults [see Use in Specific Populations (8.4)].
Drug Interaction Studies
Clinical Studies and Model Informed Approaches
In a clinical drug-drug interaction study in 18 healthy subjects, the strong CYP3A4 inhibitor (and P-gp inhibitor) itraconazole, administered as a 400 mg loading dose followed by 200 mg once daily for 6 days, increased zoliflodacin systemic exposure (AUC0-inf) by approximately 1.4-fold with minimal changes in peak concentrations (Cmax) of zoliflodacin, when zoliflodacin was administered under fasted conditions. Co-administration with a strong CYP3A4 inhibitor is unlikely to translate to a clinically significant safety risk relative to NUZOLVENCE administered alone.
Co-administration of zoliflodacin with CYP3A4 inducers, rifampin and efavirenz, is predicted to reduce the geometric mean zoliflodacin exposure (AUC0-inf) by approximately 56 and 41%, respectively [see Contraindications (4) and Drug Interactions (7.1)].
In Vitro Studies
Clearance of zoliflodacin via metabolism by CYP- and non-CYP-mediated pathways is the major clearance mechanism for zoliflodacin.
CYP phenotyping studies indicated that CYP-mediated metabolism of zoliflodacin is via CYP3A4 (68%), with lesser contributions from CYP1A2 (14%), CYP2C9 (10%), CYP2C8 (5%), and CYP2C19 (2.7%). Enzymes for the non-CYP metabolism have not been identified.
Zoliflodacin inhibited CYP2C8, CYP2C9, and CYP2C19 at IC50 values that were approximately 14- to 28-fold higher than the anticipated unbound therapeutic plasma concentrations (total concentrations 2.5 to 5-fold higher). Zoliflodacin was not an inducer of CYP1A2, CYP2B6, and CYP3A4 in inducible human hepatocyte-like HepaRG cells at clinically relevant concentrations.
Zoliflodacin is a substrate for P-gp and BCRP and possible substrate for OATP1B1/3.
Zoliflodacin is not an inhibitor for OCT2, MATE1, MATE2K at clinically relevant concentrations. Zoliflodacin is an inhibitor of P-gp, BCRP, OAT1, OAT3, OATP1B1, and OATP1B3.
12.4 Microbiology
Mechanism of Action
Zoliflodacin is a spiropyrimidinetrione inhibitor of the bacterial type II topoisomerases (DNA gyrase and topoisomerase IV), which are required for DNA synthesis. Zoliflodacin binds within the cleaved DNA–gyrase complex, blocking re-ligation, and interacts with conserved amino acids in the gyrase B subunit.
Resistance
Resistance to spiropyrimidinetriones is associated with mutations in the bacterial type II topoisomerases (DNA gyrase and DNA topoisomerase IV) and efflux pumps. Zoliflodacin primarily targets DNA gyrase B in N. gonorrhoeae. Additionally, zoliflodacin shows reduced in vitro activity due to the up-regulation of efflux pumps such as MtrCDE, MacAB, and NorM. Resistance to zoliflodacin due to spontaneous mutations occurs at mutation frequencies of <5.2 × 10-9 to <1.7 × 10-8 at 8 times the MIC.
Cross-resistance has not been identified with other classes of antimicrobials, including penicillins, cephalosporins, aminoglycosides, macrolides, fluoroquinolones and tetracyclines.
Interaction with Other Antimicrobials
In vitro studies showed no antagonism between zoliflodacin and other antimicrobial drugs including ceftriaxone, cefixime, ciprofloxacin, spectinomycin, gentamicin, and tetracycline.
Antimicrobial Activity
Zoliflodacin has been shown to be active against the following microorganisms both in vitro and in clinical infections [see Indications and Usage (1)]:
Gram-negative bacteria
- Neisseria gonorrhoeae
Susceptibility Test Methods
For specific information regarding susceptibility test interpretive criteria, and associated test methods and quality control standards recognized by FDA for this drug, please see https://www.fda.gov/STIC.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies have not been conducted with zoliflodacin.
Mutagenesis
Zoliflodacin was not genotoxic in the mouse lymphoma assay (MLA) and rat in vivo micronucleus assay. In the Ames bacterial reverse mutation assay, zoliflodacin was mutagenic in the TA102 strain, consistent with its pharmacologic action as a bacterial topoisomerase II inhibitor.
Impairment of Fertility
Male Fertility
In male rats, once daily oral administration of zoliflodacin for 4 weeks resulted in dose-related effects on fertility. At 1000 mg/kg/day (exposures 8-fold the MRHD based on AUC), males were completely infertile. At 500 mg/kg/day (exposures 4-fold the MRHD) fertility was reduced by 31%, with decreased pregnancy rates and increased pre- and post-implantation loss. After a 29-day recovery period, full recovery of fertility was observed; however, minimal to mild testicular tubular degeneration persisted after a 57-days recovery period. No zoliflodacin-related effects on fertility or testicular histopathology were observed at 200 mg/kg/day (exposures 2.4-fold the MRHD).
In a non-GLP study in rats, minimal exfoliation of germinal epithelial cells/immature sperm was observed in the testes at 1000 mg/kg/day and in the epididymides at 500 and 1000 mg/kg/day following 14 days of dosing (exposures greater than or equal to 2.8-fold the MRHD). Similar minimal epididymal findings occurred after 2-days of dosing at 1000 and 2000 mg/kg/day (exposures at and above 4-fold the MRHD).
After 4 weeks of daily zoliflodacin administration in rats, minimal to mild testicular degeneration and cellular debris in the epididymides were observed at or above 500 mg/kg/day (exposures 7.2-fold the MRHD). The microscopic findings, at a dose equivalent to exposures 10.7-fold the MRHD, were partially reversed after a 3-month recovery period. No adverse effects were reported at 200 mg/kg/day (exposures 3.3-fold the MRHD).
In dogs, 4 weeks of daily zoliflodacin administration produced mild to moderate testicular degeneration and moderate epididymides cellular debris at and above 200 mg/kg/day (exposures 7.5-fold the MRHD). No adverse effects were reported at 100 mg/kg/day (exposures 2.3-fold the MRHD). Findings were not present after a 3-month recovery period, although minimal to mild decreased spermatogenesis persisted in 2 of 3 dogs administered 500 mg/kg/day (exposures 8.1-fold the MRHD).
Female Fertility
In female rats, oral administration of zoliflodacin at 1000 mg/kg/day (exposures 12.7-fold the MRHD, extrapolated from nonpregnant rats) for 2 weeks prior to mating reduced pregnancy rates. No effects on pregnancy rates or embryo-fetal survival were observed at 500 mg/kg/day (7.9-fold the MRHD).
-
14 CLINICAL STUDIES
A total of 930 patients with suspected uncomplicated gonorrhea due to Neisseria gonorrhoeae were randomized in an open-label, active-controlled, multicenter, multinational trial (NCT03959527; Trial 1). Patients were randomized 2:1 to receive a single, oral, 3 g dose of NUZOLVENCE or a combination of a single intramuscular 500 mg dose of ceftriaxone and a single 1 g oral dose of azithromycin. Patients were eligible for enrollment if they were ≥12 years old and ≥35 kg. Patients with confirmed or suspected complicated or disseminated gonorrhea were excluded from the study.
The primary analysis population was the microbiologic intent-to-treat (micro-ITT) urogenital population, which included patients who had N. gonorrhoeae isolated at baseline from the urethra or cervix and who were not infected with a strain that showed resistance to both ceftriaxone and azithromycin at baseline. The micro-ITT urogenital population consisted of 744 patients (506 for NUZOLVENCE and 238 for ceftriaxone and azithromycin).
The demographic and baseline characteristics in the micro-ITT urogenital population were comparable between treatment groups. In total, 91% were male; 55% Black, 31% Asian, 13% White, and 4% Hispanic or Latino; the mean age was 30 years (range: 16 to 73) and the mean weight was 70 kg (range: 37 to 139 kg). The primary efficacy endpoint was microbiological cure as determined by confirmed bacterial eradication of N. gonorrhoeae at the urogenital body site at the test of cure (TOC) visit (Day 4 to 8) and was assessed in the micro-ITT urogenital population.
Table 6 shows the primary endpoint of microbiological cure rates at the urogenital site at TOC in the micro-ITT urogenital population. Trial 1 demonstrated non-inferiority of NUZOLVENCE to the combination of ceftriaxone and azithromycin.
Table 6: Microbiological Cure Rates at the Urogenital Site at TOC, Micro-ITT Urogenital Population NUZOLVENCE
n/N (%)Ceftriaxone and Azithromycin
n/N (%)Difference
(95% CI)*TOC = Test of Cure; micro-ITT = microbiological Intent to Treat; CI=confidence interval; n = number of patients in a subcategory; N = number of patients in the specified population. - * Calculated with the Newcombe score method for the NUZOLVENCE – (ceftriaxone and azithromycin) treatment difference
- † Microbiological cure is defined as negative or indeterminate Neisseria gonorrhoeae (NG) culture at the urogenital site at the TOC visit. There were no indeterminate NG cultures at the urogenital site at TOC.
- ‡ Nonassessable outcomes were due to missed TOC visits, unobtainable specimens, or TOC visits out of window.
Microbiological Cure† 460/506 (90.9) 229/238 (96.2) -5.3 (-8.7, -1.4) Failure 46/506 (9.1) 9/238 (3.8) Bacterial Persistence by Culture 15/506 (3.0) 0 Nonassessable‡ 31/506 (6.1) 9/238 (3.8) -
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
NUZOLVENCE (zoliflodacin) for oral suspension is supplied as a kit.
Table 7: NUZOLVENCE Kit Configuration Kit Configuration Strength NDC Code Carton containing: - 1 unit-dose packet of white to off-white granules containing zoliflodacin
- One 120 mL mixing container with lid
- Instructions for Use
- Medication Guide
3 g 68547-915-10 -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved labeling (Medication Guide and Instructions for Use).
Embryo-Fetal Toxicity: Potential Risk for Pregnant Females
- Advise pregnant females about the potential risk to the fetus with maternal exposure to NUZOLVENCE [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1, 8.3)].
- Advise females who take NUZOLVENCE during pregnancy that a pregnancy safety study is available to monitor pregnancy outcomes and encourage them to report their pregnancy to Entasis Therapeutics at 1-800-651-3861 [see Use in Specific Populations (8.1)].
Embryo-Fetal Toxicity Related to Males with Female Partners of Reproductive Potential
Advise males who receive NUZOLVENCE and have female partners of reproductive potential to use effective contraception for at least 3 months after receiving NUZOLVENCE [see Warnings and Precautions (5.2), Use in Specific Populations (8.3), and Nonclinical Toxicology (13.1)].
Testicular Toxicity and Risks to Male Fertility
Advise males that NUZOLVENCE may cause testicular toxicity and impair male fertility [see Warnings and Precautions (5.3), Use in Specific Populations (8.3), and Nonclinical Toxicology (13.1)].
Important Preparation Instructions Prior to Administration
Advise patients that NUZOLVENCE are granules for oral suspension and must be mixed with water before administration. Instruct patients not to take the granules in the dry form [see Dosage and Administration (2.2)].
Important Administration Instructions Regarding the Ingestion of Food
- Advise patients weighing 35 kg (77 pounds) to less than 50 kg (110 pounds) to take NUZOLVENCE on an empty stomach, 1 hour before or 2 hours after food [see Dosage and Administration (2.3)].
- Advise patients weighing 50 kg (110 pounds) or more to take NUZOLVENCE with food [see Dosage and Administration (2.3)].
Diarrhea
Advise the patient, their families, or caregivers that diarrhea is a common problem caused by antibacterial drugs, including NUZOLVENCE. Sometimes, frequent watery or bloody diarrhea may occur and may be a sign of a more serious intestinal infection. If severe watery or bloody diarrhea develops, tell them to contact their healthcare provider [see Warnings and Precautions (5.5)].
Antibacterial Resistance
Patients should be counseled that antibacterial drugs including NUZOLVENCE should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When NUZOLVENCE is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by NUZOLVENCE or other antibacterial drugs in the future [see Warnings and Precautions (5.6)].
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
MEDICATION GUIDE
NUZOLVENCE® (nu-ZOL-vence)
(zoliflodacin)
for oral suspensionThis Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 12/2025 Read this Medication Guide and Instructions for Use before you take NUZOLVENCE. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment. What is the most important information I should know about NUZOLVENCE?
NUZOLVENCE may cause serious side effects, including:-
fetal harm. It is not known if NUZOLVENCE can harm your unborn baby.
- Females: Your healthcare provider should have you take a pregnancy test before you receive NUZOLVENCE. If you take NUZOLVENCE while you are pregnant, your baby may be at risk for serious birth defects.
- Males: Males with female partners who can become pregnant should use effective contraception for at least 90 days (3 months) after treatment with NUZOLVENCE.
- harm to testicles. NUZOLVENCE may be harmful to testicles and lower sperm count in males and cause fertility problems. NUZOLVENCE may harm your sperm.
- allergic reactions. Allergic reactions, including rash and itching, have been reported in people receiving NUZOLVENCE.
- diarrhea caused by Clostridioides difficile infection. Antibiotics can sometimes cause a type of severe diarrhea called Clostridioides difficile infection (CDI). CDI is a severe infection of the intestines (bowels) that can be serious or life-threatening. CDI can happen up to 2 months after finishing treatment with NUZOLVENCE. Call your healthcare provider right away if you develop frequent, watery, or bloody stools, or stomach cramps that are severe, do not go away, or come back, after taking NUZOLVENCE.
What is NUZOLVENCE? - NUZOLVENCE is a prescription antibacterial medicine used to treat uncomplicated urogenital gonorrhea in adults and children 12 years of age and older, who weigh at least 77 pounds (35 kg).
- It is not known if NUZOLVENCE is safe and effective in children younger than 12 years of age or who weigh less than 77 pounds (35 kg).
Do not take NUZOLVENCE if you: - have a history of being allergic to NUZOLVENCE. See the end of this Medication Guide for a complete list of ingredients in NUZOLVENCE.
- are taking certain medicines called CYP3A4 inducers which increase the enzyme CYP3A4 in your liver. These medicines can make NUZOLVENCE less effective. Ask your healthcare provider or pharmacist if you are not sure if any of your medicines are CYP3A4 inducers.
Before taking NUZOLVENCE, tell your healthcare provider about all of your medical conditions, including if you: - are pregnant or plan to become pregnant. It is not known if NUZOLVENCE can harm your unborn baby. See "What is the most important information I should know about NUZOLVENCE?"
- Tell your healthcare provider if you become pregnant or think you are pregnant during treatment with NUZOLVENCE.
- Pregnancy Safety Study: There is a pregnancy safety study for women who are exposed to NUZOLVENCE during pregnancy. The purpose of this registry is to check the health of you and your baby. If you are pregnant while being treated, you or your healthcare providers should report the pregnancy to Entasis Therapeutics at 1-800-651-3861.
- are breastfeeding or plan to breastfeed. It is not known if NUZOLVENCE passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby after taking NUZOLVENCE.
How should I take NUZOLVENCE?
Read the Instructions for Use leaflet for information about the right way to take NUZOLVENCE.- Always mix NUZOLVENCE with water before taking.
- Do not mix NUZOLVENCE with other liquids or sprinkle on food.
- Drink NUZOLVENCE within 15 minutes after mixing. If NUZOLVENCE is not taken within 15 minutes, do not take and call your healthcare provider for a new dose of NUZOLVENCE.
- Take NUZOLVENCE exactly as your healthcare provider tells you.
- If you do not or cannot take the full dose, call your healthcare provider right away.
- Based on your weight, take NUZOLVENCE as follows:
- If you weigh between 77 pounds (35 kg) and less than 110 pounds (50 kg): Take NUZOLVENCE on an empty stomach, 1 hour before eating or 2 hours after eating.
- If you weigh greater than or equal to 110 pounds (50 kg): Take NUZOLVENCE with food.
What are the possible side effects of NUZOLVENCE?
NUZOLVENCE may cause serious side effects, including:- See "What is the most important information I should know about NUZOLVENCE?"
The most common side effects of NUZOLVENCE include:- low white blood cell count
- headache
- dizziness
- nausea
- diarrhea
How should I store NUZOLVENCE? - Store NUZOLVENCE at room temperature between 68 °F to 77 °F (20 °C to 25 °C) in the original packaging.
- Do not freeze.
General information about the safe and effective use of NUZOLVENCE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use NUZOLVENCE for a condition for which it was not prescribed. Do not give NUZOLVENCE to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about NUZOLVENCE that is written for health professionals.What are the ingredients in NUZOLVENCE?
Active ingredient: zoliflodacin
Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, talc, and xanthan gum.
Distributed by:
La Jolla Pharmaceutical Company, Waltham, MA 02451
Product of India
NUZOLVENCE® is a registered trademark of Entasis Therapeutics, Inc.
©2025, Entasis Therapeutics, Inc. All rights reserved. -
fetal harm. It is not known if NUZOLVENCE can harm your unborn baby.
-
INSTRUCTIONS FOR USENUZOLVENCE® (nu-ZOL-vence) (zoliflodacin)for oral suspension
This Instructions for Use contains information on how to take NUZOLVENCE. Read this Instructions for Use before you take NUZOLVENCE.
This information does not take the place of talking to your healthcare provider about your medical condition or treatment. Talk to your healthcare provider or pharmacist if you have any questions on how to prepare or take the prescribed dose of NUZOLVENCE.
NUZOLVENCE kit contents:

Note: you will also need: - drinking water
- a pair of scissors
Important Information You Need to Know Before Taking NUZOLVENCE
- Do not use if the packet or mixing container appears damaged.
- Always mix NUZOLVENCE with water before taking (see Instructions below).
- Do not mix NUZOLVENCE with other liquids or sprinkle on food.
- Take NUZOLVENCE exactly as your healthcare provider tells you.
- If you do not or cannot take the full dose, call your healthcare provider.
Based on your weight, take NUZOLVENCE as follows:
Dose of NUZOLVENCE Body Weight How to Take NUZOLVENCE Relative to the Ingestion of Food 3 g administered as a single, oral dose 77 pounds (35 kg) to less than 110 pounds (50 kg) Take on an empty stomach, 1 hour before or 2 hours after food Greater than or equal to 110 pounds (50 kg) Take with food Storing NUZOLVENCE
- Store NUZOLVENCE in the original package.
- Store NUZOLVENCE at room temperature between 68 °F to 77 °F (20 °C to 25 °C), see prescribing information.
- Do not freeze.
- Keep NUZOLVENCE and all medicines out of the reach of children.
- 1.
Inspect the kit contents and make sure that:
- the expiration date has not passed, and
- the contents are not damaged.
- Do not use if the expiration date on the packet has passed.
- Do not use if the packet or mixing container appear damaged.
- 2.
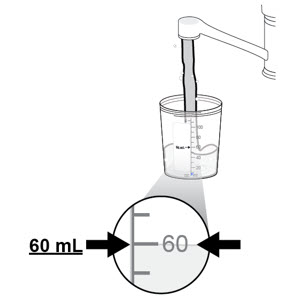
a. Remove the lid from the mixing container.
b. Pour drinking water into the mixing container up to the 60 mL line.- Only use water.
- Do not mix NUZOLVENCE with other liquids or sprinkle on food.
- 3. a. Shake the packet to move the medicine to the bottom of the packet.
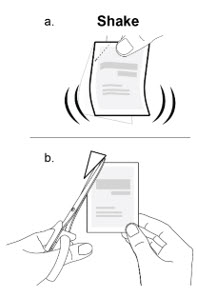
- b. Cut the packet carefully along the diagonal line using scissors.
- 4.
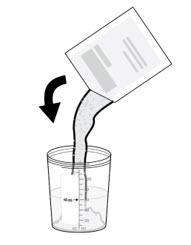
Add the entire packet of NUZOLVENCE into the mixing container with water.
If you spill any medicine, call your healthcare provider for a new dose of NUZOLVENCE.
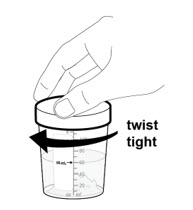
- 5. Immediately secure the lid onto the mixing container by twisting it to the right (clockwise) until it feels tight.
- 6.
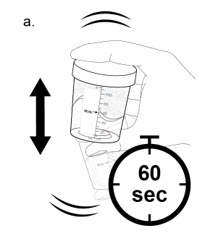
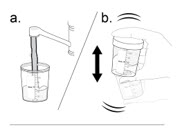
- a.
Holding the lid closed, shake the mixing container vigorously for at least 60 seconds to mix the medicine. The mixture should look cloudy.
- Do not stir or mix the medicine in any other way.
- If you spill any medicine, call your healthcare provider for a new dose of NUZOLVENCE.
- b. Check that the medicine does not have any clumps. Continue shaking until all clumps are gone.
- a.
Holding the lid closed, shake the mixing container vigorously for at least 60 seconds to mix the medicine. The mixture should look cloudy.
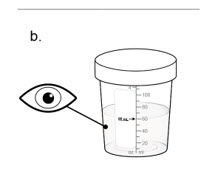
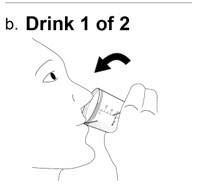
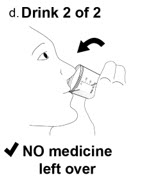
Taking NUZOLVENCE - 7.
Drink 1 of 2:
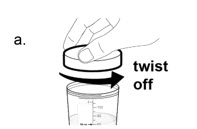
a. Remove the lid from the mixing container by twisting it counter-clockwise.
b. Drink all of the mixture from the mixing container.
Drink the medicine within 15 minutes after you prepare it. If it has been more than 15 minutes, a new dose of NUZVOLENCE must be prepared.
- 8.
Drink 2 of 2:
Some medicine will remain in the container. To ensure you get the full dose:- a. Add another 60 mL of water to the same mixing container.
- b. Repeat Steps 5 and 6.
- c. Remove the lid from the mixing container by twisting it counter-clockwise.
- d. Drink all of the mixture from the mixing container.
c.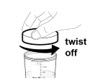
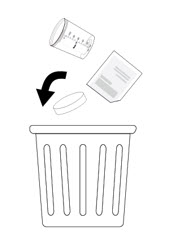
Disposing of NUZOLVENCE - 9. Throw away the empty NUZOLVENCE packet, mixing container, and lid in the trash.
Additional Information
For additional information, call (800) 651-3861
Distributed by:
La Jolla Pharmaceutical Company
Waltham, MA 02451NUZOLVENCE® is a registered trademark of Entasis Therapeutics, Inc.
©2025, Entasis Therapeutics, Inc. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Approved: 12/2025 -
PRINCIPAL DISPLAY PANEL - 3 g Packet Carton
NDC: 68547-915-10
Rx ONLYNUZOLVENCE®
(zoliflodacin) for Oral Suspension3g
UNIT-DOSE KIT
Each carton contains:
- 1 packet containing 3 g of zoliflodacin
- 1 mixing container, 120 mL, with lid
- Instructions for Use
- Medication Guide
PHARMACIST: Dispense
with enclosed
Medication Guide
to each patient.ENTASIS
THERAPEUTICS
-
INGREDIENTS AND APPEARANCE
NUZOLVENCE
zoliflodacin for suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68547-915 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength zoliflodacin (UNII: FWL2263R77) (zoliflodacin - UNII:FWL2263R77) zoliflodacin 3 g Inactive Ingredients Ingredient Name Strength silicon dioxide (UNII: ETJ7Z6XBU4) croscarmellose sodium (UNII: M28OL1HH48) magnesium stearate (UNII: 70097M6I30) mannitol (UNII: 3OWL53L36A) microcrystalline cellulose (UNII: OP1R32D61U) talc (UNII: 7SEV7J4R1U) xanthan gum (UNII: TTV12P4NEE) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68547-915-10 1 in 1 CARTON 04/01/2026 1 1 in 1 PACKET; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219491 04/01/2026 Labeler - La Jolla Pharmaceutical Company (613541192)
Trademark Results [NUZOLVENCE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NUZOLVENCE 98561615 not registered Live/Pending |
Entasis Therapeutics (Ireland) Limited 2024-05-21 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.