DESMODA- desmopressin acetate oral solution solution
DESMODA by
Drug Labeling and Warnings
DESMODA by is a Prescription medication manufactured, distributed, or labeled by Eton Pharmaceuticals, Inc., PolyPeptide Laboratories AB, Tulex Pharmaceuticals, Inc., Eurofins BioPharma Product Testing, Denmark A/S, Mikrolab Stockholm AB, New Jersey Laboratories, SGS North America Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DESMODA safely and effectively. See full prescribing information for DESMODA.
DESMODA TM (desmopressin acetate) oral solution
Initial U.S. Approval: 1978INDICATIONS AND USAGE
DESMODA is a vasopressin analog indicated for the management of central diabetes insipidus as antidiuretic replacement therapy for adults and pediatric patients.
Limitations of Use
DESMODA is not indicated for the treatment of nephrogenic diabetes insipidus. (1)DOSAGE AND ADMINISTRATION
Instruct patients about appropriate fluid restriction during DESMODA treatment due to risk of hyponatremia. (2.1)
Recommended starting dosage for adults and pediatric patients is 0.05 mg twice. (2.1)
Titrate the daily dosage as needed to obtain an adequate antidiuretic response. (2.1)
Take DESMODA on an empty stomach, at least 1 hour before or 2 hours after food. (2.1)
Monitor response by measuring urine volume and osmolarity. (2.1)
For patients with central diabetes insipidus switching to DESMODA from desmopressin acetate nasal spray, wait twelve hours after the last dose. Monitor patients closely during the initial dose titration period. (2.2) (2)DOSAGE FORMS AND STRENGTHS
Oral solution: 0.05 mg desmopressin acetate in 1 mL (0.05 mg/mL) (3) (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
Hyponatremia: Excessive fluid intake when urine output is limited by the antidiuretic effect of desmopressin may lead to water intoxication with hyponatremia. Fluid restriction is recommended. (5.1)
Fluid Retention: Desmopressin acetate may cause fluid retention. Use with caution in patients with heart failure or uncontrolled hypertension. DESMODA is not recommended in patients at risk for increased intracranial pressure or those with a history of urinary retention. (5.2)
Hypersensitivity: Severe reactions have occurred. Monitor for reactions during administration and interrupt if reaction occurs. (5.3) (5)ADVERSE REACTIONS
DRUG INTERACTIONS
Drugs that may Increase Risk of Hyponatremia: Requires more frequent monitoring of serum sodium. (7.1)
Other Vasoconstrictors: Concomitant use may elevate blood pressure and require a reduction in DESMODA dosage. (7.2) (7)USE IN SPECIFIC POPULATIONS
Pediatric Use: Use requires careful fluid restriction to prevent hyponatremia with water intoxication. (5.1, 8.4)
Geriatric Use: Carefully select dose and monitor renal function; restrict fluid intake to prevent hyponatremia with water intoxication. (5.1, 8.5)
Renal Impairment: The risk of adverse reactions may be greater in patients with renal impairment, compared to patients with normal renal function. (8.6) (8)(8)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. (8)
Revised: 02/2026 (8)See 17 for Medication Guide.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage for Central Diabetes Insipidus
Instruct patients about appropriate fluid restriction during DESMODA treatment due to risk of hyponatremia [see Warnings and Precautions (5.1)].
Individualize the dosage of DESMODA for each patient and adjust the dosage according to the diurnal pattern of response. Estimate patient response by adequate duration of sleep and water turnover.
Recommended starting dosage of DESMODA for adults and pediatric patients is 0.05 mg orally twice daily.
Titrate the daily dosage as needed to obtain an adequate antidiuretic response.
Administer DESMODA on an empty stomach, at least 1 hour prior to or 2 hours after food.
Administer DESMODA using an oral syringe provided by the pharmacy.
Monitor response by measuring urine volume and osmolarity. Monitoring measurements of plasma osmolarity may also be useful.
2.2 Switching Patients with Central Diabetes Insipidus to DESMODA From Desmopressin Acetate Nasal Spray
For patients with central diabetes insipidus switching to DESMODA from desmopressin acetate nasal spray, wait twelve hours after the last dose.
Monitor patients closely during the initial dose titration period. - 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
DESMODA is contraindicated in patients with:
Hyponatremia or a history of hyponatremia [see Warnings and Precautions (5.1)].
Hypersensitivity to desmopressin acetate or any of the inactive ingredients of DESMODA.Reactions have included anaphylaxis [see Warnings and Precautions (5.3)].
Moderate to severe renal impairment (defined as creatinine clearance (CLcr) less than 50 mL/min)[see Use in Specific Populations (8.6)]. -
5 WARNINGS AND PRECAUTIONS
5.1 Hyponatremia
Excessive fluid intake when urine output is limited by the antidiuretic effect of desmopressin may lead to water intoxication with hyponatremia. Cases of hyponatremia have been reported from postmarketing experience in patients treated with desmopressin acetate. Unless properly diagnosed and treated hyponatremia can be fatal.
Observe all patients receiving DESMODA for the following signs or symptoms associated with hyponatremia: headache, nausea/vomiting, decreased serum sodium, weight gain, restlessness, fatigue, lethargy, disorientation, depressed reflexes, loss of appetite, irritability, muscle weakness, muscle spasms or cramps and abnormal mental status such as hallucinations, decreased consciousness and confusion. Severe symptoms due to an extreme decrease in serum sodium and plasma osmolality may include one or a combination of the following: seizure, coma and/or respiratory arrest.
In order to decrease the risk of water intoxication with hyponatremia, fluid restriction is recommended. Careful fluid intake is particularly important in pediatric and geriatric patients because these patients are at greater risk of developing hyponatremia [see Use in Specific Populations (8.4, 8.5)]. More frequent monitoring of serum sodium levels is recommended in the following patients: those with conditions associated with fluid and electrolyte imbalance, such as cystic fibrosis, heart failure, renal disorders, habitual or psychogenic polydipsia or those taking concomitant drugs that may cause hyponatremia [see Drug Interactions (7.1)].
Temporarily stop treatment with DESMODA during acute intercurrent illness characterized by fluid and/or electrolyte imbalance (e.g., systemic infections, fever, recurrent vomiting or diarrhea) or under conditions of extremely hot weather, vigorous exercise or other conditions associated with increased water intake.
5.2 Fluid Retention
Desmopressin acetate can cause fluid retention, which can worsen underlying conditions that are susceptible to volume status. Closely monitor patients with heart failure or uncontrolled hypertension. DESMODA is not recommended in patients at risk for increased intracranial pressure or those with a history of urinary retention.
5.3 Hypersensitivity Reactions
Hypersensitivity reactions including anaphylaxis have been reported with intravenous and nasal administration of desmopressin acetate, including cases of fatal anaphylaxis with intravenous desmopressin acetate [see Adverse Reactions (6)]. DESMODA is contraindicated in patients with a history of hypersensitivity to desmopressin acetate or any of the inactive ingredients of DESMODA [see Contraindications (4)]. If signs or symptoms of hypersensitivity reactions occur, discontinue DESMODA and initiate appropriate supportive care.
5.4 Risk of Benzyl Alcohol Toxicity in Neonates
DESMODA contains 2.74 mg/mL of benzoic acid, a metabolite of benzyl alcohol. Serious adverse reactions of metabolic acidosis, including fatalities, have been reported in low-birth-weight neonates (less than 2,500 grams) and preterm neonates (gestational age less than 34 weeks) who received benzyl alcohol-containing drugs intravenously. The relationship between systemic levels of benzoic acid and toxicity is not well-characterized [see Use in Specific Populations (8.4)]. Use DESMODA with caution in low-birth-weight neonates or preterm neonates and monitor for signs and symptoms of metabolic acidosis. -
6 ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in the labeling:
Hyponatremia [see Warnings and Precautions (5.1)]Fluid Retention [see Warnings and Precautions (5.2)]
Hypersensitivity [see Warnings and Precautions (5.3)]
Risk of Benzyl Alcohol Toxicity in Neonates [see Warnings and Precautions (5.4)]
The following adverse reactions associated with the use of desmopressin acetate were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac disorders: hypertension
Gastrointestinal disorders: nausea, vomiting, diarrhea, constipation, abdominal cramps
General disorders: headache, malaise, drug ineffective, edema, fatigue
Hepatobiliary disorders: transient increases in AST (1.5 times the upper limit of normal)
Immune system disorders: severe allergic reactions, anaphylaxis has occurred with other formulations of desmopressin
Metabolism and nutrition: dehydration, hyponatremia, water intoxication with hyponatremia
Nervous system disorders: hyponatremic convulsions, hyponatremic convulsions associated with concomitant use of the following medications: oxybutynin and imipramine [see Drug Interactions (7.1)], asthenia, coma, disturbance in attention, psychomotor hyperactivity, dizziness, somnolence, depressed level of consciousness, falls.
Psychiatric disorders: confusional state, abnormal behavior, emotional disorder, depression, hallucination, insomnia
Renal and urinary disorders: bladder and urethral symptoms, including urinary retention, urine flow decreased, dysuria
Skin and subcutaneous tissue disorders: rash, dermatitis allergic, sweating, flushing, urticaria
Respiratory, thoracic and mediastinal disorders: dyspnea, epistaxis
In long-term clinical studies in which patients with diabetes insipidus were followed for periods up to 44 months of desmopressin acetate oral therapy, transient increases in AST (SGOT) no higher than 1.5 times the upper limit of normal were occasionally observed. Elevated AST (SGOT) returned to the normal range despite the continued use of desmopressin acetate oral therapy. -
7 DRUG INTERACTIONS
7.1 Other Drugs that may Increase Risk of Hyponatremia
Concomitant administration of DESMODA with other drugs that may increase the risk of water intoxication with hyponatremia, (e.g., tricyclic antidepressants, selective serotonin re-uptake inhibitors, chlorpromazine, opiate analgesics, thiazide diuretics, NSAIDs, lamotrigine, sulfonylureas, particularly chlorpropamide, oxybutynin and carbamazepine). Monitor serum sodium more frequently in patients taking DESMODA concomitantly with these drugs and when doses of these drugs are increased [see Warnings and Precautions (5.1), Adverse Reactions (6)].
7.2 Other Vasoconstrictors
Desmopressin acetate can elevate blood pressure. Use of large doses of DESMODA with other vasoconstrictors may require a reduction of the DESMODA dosage [see Adverse Reactions (6)]. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data on the use of desmopressin acetate during pregnancy over decades of use have not identified a drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. There are risks associated with untreated central diabetes insipidus during pregnancy (see Clinical Considerations). In vitro studies with human placenta demonstrate poor placental transfer of desmopressin. No adverse developmental outcomes were observed in animal reproduction studies with administration of desmopressin during organogenesis to pregnant rats and rabbits at doses approximately <1 to 38 times, respectively, the maximum recommended human dose based on body surface areas (mg/m2) (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.Clinical Considerations
Disease Associated Maternal and Embryo-Fetal Risk
There are risks associated with untreated central diabetes insipidus during pregnancy such as dehydration, preterm birth and oligohydramnios. Pregnant patients with diabetes insipidus should be monitored for electrolyte imbalance, maternal hypotension, and low amniotic fluid levels.
Data
Animal Data
In a developmental toxicity study in rats, desmopressin acetate was administered intravenously at doses of 9.68, 48.4, or 241 mcg/kg/day during the period of organogenesis (gestations days 7 to 17). Laparohysterectomy for fetal examinations were conducted on gestation day 20 for twenty females in each group; the remaining 10 females were allowed to litter in order to determine any postnatal effects that might be attributable to pre-natal treatment. No effects were seen on maternal and fetal survival, growth and morphology or post-natal offspring survival, growth, development, behavior and reproductive performance up to 241 mcg/kg/day (4 times a 0.6 mg dose received by a 60 kg patient based on body surface area).
In an embryo-fetal development study and a pre- and postnatal development study in rabbits, desmopressin acetate was administered subcutaneously at doses of 2, 20 or 200 mcg/kg/day (embryo-fetal development) and 0.1, 1 or 10 mcg/kg/day (pre- and postnatal development) during the period of organogenesis (gestation days 6 to 18). No effects on maternal and fetal survival or morphology were observed in both studies at doses of up to 200 mcg/kg/day (6.5 times a 0.6 mg dose received by a 60 kg patient based on body surface area) nor were there effects in the pre- and postnatal development study on parturition, postnatal survival, growth, development or behavior, up to the highest dose tested of 10 mcg/kg/day (0.3 times a 0.6 mg dose received by a 60 kg patient, based on body surface area).8.2 Lactation
Risk Summary
Breastfeeding is not expected to result in clinically relevant exposure of the infant to desmopressin following maternal administration. Desmopressin is transferred into human breastmilk at negligible amounts (see Data). There is no information on the effects of desmopressin on the breastfed infant or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for DESMODA and any potential adverse effects on the breastfed infant from DESMODA or from the underlying maternal condition.
Data
A trial was conducted in six healthy lactating women, at greater than 4 months postpartum, to evaluate intranasal administration of 300 mcg single dose of desmopressin acetate. Samples of maternal plasma and breastmilk were obtained at 0, 30, 60, 120, 240, 360 and 480 min after the drug administration. At 8 hours after dose intake, the levels in the milk ranged between 4.16 and 101 pg/ml, and the plasma levels ranged between 40 and 242 pg/ml. The total amount of desmopressin present in the milk over the 8 hours ranged between 491 pg and 16 ng, which corresponds to 0.0001 to 0.005% of the administered dose to the breastfeeding mother.8.4 Pediatric Use
Desmopressin acetate is indicated for the management of central diabetes insipidus as antidiuretic replacement therapy in pediatric patients. Use in pediatric patients requires careful fluid intake restriction to prevent possible water intoxication with hyponatremia. In younger pediatric patients, the dose must be individually adjusted to prevent an excessive decrease in plasma osmolality leading to hyponatremia and possible convulsions. Fluid restriction should be discussed with the patient and/or guardian [see Warnings and Precautions (5.2)].
DESMODA contains benzoic acid, a metabolite of benzyl alcohol. Serious adverse reactions, including fatal reactions, of new onset or worsening metabolic acidosis that progressed to neurotoxicity, and in some cases gasping syndrome, have been reported in low-birth weight neonates and preterm neonates who received benzyl alcohol-containing drugs intravenously (DESMODA is not approved for intravenous use [see Dosage and Administration (2.1)]). Gasping syndrome is a life-threatening condition in neonates caused by benzyl alcohol toxicity that is characterized by new onset or worsening metabolic acidosis with gradual neurological deterioration, seizures, intracranial hemorrhage, hematologic abnormalities, skin breakdown, hepatic and kidney failure, hypotension, bradycardia, and gasping respirations followed by death. In reported cases, benzyl alcohol in amounts of 99 to 234 mg/kg/day produced blood BA levels of 6.6 to 14.9 mg/dL, but the minimum amount of benzyl alcohol at which gasping syndrome may occur in neonates is not known [see Warnings and Precautions (5.4)]. The rate at which benzyl alcohol is metabolized to benzoic acid cannot be reliably predicted, and the relationship between systemic levels of benzoic acid and toxicity is not well-characterized.
DESMODA contains 2.74 mg of benzoic acid per mL [see Warnings and Precautions (5.4)].8.5 Geriatric Use
Because geriatric patients are more likely to have decreased renal function, care should be taken in dose selection and monitoring renal function is recommended [see Clinical Pharmacology (12.3)].
Use of DESMODA in geriatric patients requires careful fluid intake restriction to prevent possible water intoxication with hyponatremia [see Warnings and Precautions (5.1)].8.6 Renal Impairment
Desmopressin acetate is substantially excreted by the kidney, and the risk of adverse events may be greater in patients with renal impairment than patients with normal renal function. Desmopressin acetate is contraindicated in patients with estimated CLcr by Cockcroft Gault equation less than 50 mL/min [see Contraindications (4), Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Signs of overdose may include confusion, drowsiness, continuing headache, problems with passing urine and rapid weight gain due to fluid retention [see Warnings and Precautions (5.1)]. In case of overdosage, reduce the dosage, decrease the frequency of administration, or discontinue the DESMODA. There is no known specific antidote for desmopressin acetate. The patient should be observed and treated with appropriate symptomatic therapy.
-
11 DESCRIPTION
DESMODA oral solution contains active moiety desmopressin, an analog of natural pituitary hormone 8-arginine vasopressin (antidiuretic hormone), in the form of acetate salt. The chemical name of desmopressin acetate is 1-desamino-8-D-arginine vasopressin acetate.
Desmopressin acetate has a molecular weight of 1129.30 g/mol, an empirical formula of C 46H 64N 14O 12S 2·C 2H 4O 2, and structural formula: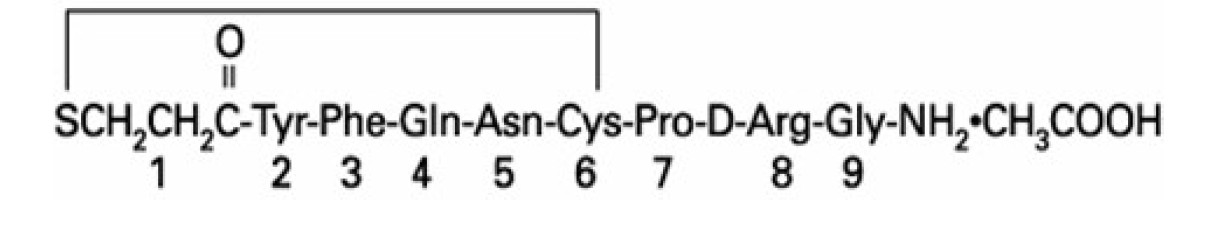
Desmopressin acetate is white amorphous powder and is soluble in water.
DESMODA oral solution: Each mL contains 0.05 mg of desmopressin acetate (equivalent to 0.047 mg of desmopressin free base) as active ingredient. The inactive ingredients include benzoic acid and sodium benzoate as buffering agents and antimicrobial agents, and purified water. The pH of the oral solution is 4.8 to 5.2. -
12 CLINICAL PHARMACOLOGY
-
12.1 Mechanism of Action
Desmopressin acetate’s primary pharmacodynamic action involves its binding to vasopressin 2 (V2) receptors. V2 receptors are primarily located in the distal convoluted tubule and collecting ducts of the nephron in the kidneys. These receptors play a crucial role in regulating water reabsorption and urine concentration. By influencing the permeability of the renal tubules, V2 receptors help maintain the body’s water balance and overall homeostasis.
12.2 Pharmacodynamics
Dose response studies in patients with diabetes insipidus following desmopressin acetate tablets 0.025 mg to 0.4 mg produced clinically significant antidiuretic effects. In most patients, doses of 0.1 mg to 0.2 mg produced optimal antidiuretic effects lasting up to eight hours. With doses of 0.4 mg, antidiuretic effects were observed for up to 12 hours; measurements beyond 12 hours were not recorded.
Following administration of desmopressin acetate tablets, the onset of antidiuretic effect occurs at around 1 hour, and it reaches a maximum at about 4 to 7 hours based on the measurement of increased urine osmolality.12.3 Pharmacokinetics
Following a single oral administration of DESMODA in healthy subjects at 0.6 mg, mean maximum concentration (SD) is 82 (33) pg/mL, and mean total systemic exposure (SD) is 314 (138) (hrs∙pg/mL).
Absorption
Desmopressin absolute oral bioavailability is 0.16%. Desmopressin area under the plasma drug concentration time curve (AUC) is reduced by 95% following administration of the oral solution compared to the intranasal formulation.
Effect of Food
DESMODA Cmax was decreased 72% and the AUC was decreased 64% following administration of 0.6 mg DESMODA with a high-fat meal (1000 calories, 50% fat).
Distribution
DESMODA mean apparent (oral) volume of distribution (SD) is 11717 (5086) L.
Elimination
Desmopressin is mainly excreted in the urine. Desmopressin estimated elimination half-life is 3.6 hours. DESMODA mean apparent (oral) clearance (SD) is 2264 (940) L/hr.Specific Populations
Renal Impairment
A pharmacokinetic study was conducted in subjects with normal renal function and patients with mild, moderate, and severe renal impairment (n=24, 6 subjects each group) with a single dose of 2 mcg desmopressin acetate intravenous injection. The geometric mean terminal half-life was 2.8 hours in subjects with normal renal function, and 4, 6.6, and 8.7 hours in patients with mild, moderate, and severe renal impairment, respectively. In patients with mild, moderate and severe renal impairment, mean desmopressin AUC was 1.5-fold, 2.4-fold and 3.6-fold higher, respectively, compared to that of subjects with normal renal function. - 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
The effectiveness of DESMODA was supported by dose response studies of desmopressin acetate tablets in patients with diabetes insipidus. These studies demonstrated that oral doses of 0.025 mg to 0.4 mg of desmopressin acetate tablets produced clinically significant antidiuretic effects. In patients treated with desmopressin acetate tablets, doses of 0.1 mg to 0.2 mg produced optimal antidiuretic effects lasting up to eight hours [see Clinical Pharmacology (12.2)].
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
DESMODA (desmopressin acetate) oral solution is a clear, colorless to slightly yellow colored solution supplied in an amber glass bottle:Strength Volume in Bottle NDC 0.05 mg/mL 145 mL 71863-123-14 Storage and Handling
Store at 2°C to 25°C (36°F to 77°F). Excursions permitted to 30°C (86°F) [see USP refrigerated and controlled room temperature]. Do not freeze.
Discard any unused DESMODA oral solution after 120 days after first opening the bottle. -
17 PATIENT COUNSELING INFORMATION
Advise patients and/or caregivers to read the FDA-approved patient labeling (Medication Guide).
Administration
Instruct caregivers to use an oral dosing syringe to correctly measure the prescribed amount of medication. Inform caregivers that oral dosing syringes may be obtained from their pharmacy [see Dosage and Administration (2.1)].
Hyponatremia
Advise patients and/or caregivers that DESMODA is a potent antidiuretic, which when administered may lead to water intoxication and /or hyponatremia. Discuss recommendations for fluid restrictions [see Warnings and Precautions (5.1)].
Manufactured for:
Eton Pharmaceuticals, Inc. Deer Park, IL 60010
DESMODA TM is a trademark of Eton Pharmaceuticals, Inc.
U.S. Patent No. 12,214,010; U.S. Patent No. 12,343,372
©2026 Eton Pharmaceuticals, Inc. All rights reserved.
PL-79-1.0 -
Medication Guide
DESMODA
TM (des-MOH-duh)
(desmopressin acetate)
oral solution
What is the most important information I should know about DESMODA?
DESMODA may cause serious side effects, including:
Low levels of salt (sodium) in the blood (hyponatremia). Hyponatremia has happened in people taking desmopressin acetate, an ingredient in DESMODA. Drinking excessive fluids during treatment with DESMODA may lead to water toxicity with hyponatremia. Hyponatremia can lead to death if not treated. Talk to your healthcare provider about fluid limits during treatment with DESMODA.
Do not drink more than you need to satisfy your thirst. You can have serious side effects such as seizures, coma, and death from drinking too much fluid.
Children and the elderly are at higher risk for these problems and should follow their healthcare provider’s limits on drinking fluids.
Tell your healthcare provider if you have any of the following signs or symptoms:
headache restlessness loss of appetite nausea or vomiting tiredness irritability decreased sodium levels in the blood lack of energy muscle weakness weight gain disorientation muscle spasms or cramps abnormal mental status (hallucinations, decreased consciousness, or confusion) weakened reflexes
Your healthcare provider will change the dose of DESMODA depending on your response. Tell your healthcare provider if you cannot swallow medicines by mouth.
People who have taken desmopressin acetate nasal spray previously should start taking the oral solution twelve hours after the last nasal spray dose. Your healthcare provider should monitor you closely during this period.
Your healthcare provider may monitor your sodium levels during your treatment with DESMODA.
See “What are the possible side effects of DESMODA?” for more information about side effects.
What is DESMODA?
DESMODA is a prescription medicine that contains the medicine desmopressin acetate. Desmopressin acetate is an antidiuretic, also known as vasopressin. DESMODA (desmopressin acetate) is a man-made (synthetic) prescription drug used to replace the body’s antidiuretic hormone to manage central diabetes insipidus in adults and children.
DESMODA is not effective in the treatment of diabetes insipidus related to kidney disease (nephrogenic diabetes insipidus).
Who should not take DESMODA?
Do not take DESMODA if you:
have or have had low levels of sodium in the blood.
are allergic to desmopressin acetate or any of the ingredients in DESMODA. See the end of this Medication Guide for a complete list of ingredients in DESMODA.
have moderate to severe kidney problems.
Before you take DESMODA, tell your healthcare provider about all of your medical conditions, including if you:
have heart failure or blood circulation problems.
are thirsty much of the time and drink excessive amounts of fluids (polydipsia).
have or have had low levels of sodium in your blood. have high blood pressure that is not controlled.
have increased pressure in your brain (increased intracranial pressure).
have had problems with emptying your bladder (urinary retention).
are pregnant or plan to become pregnant. It is not known if DESMODA will harm your unborn baby.
are breastfeeding or plan to breastfeed. DESMODA can pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby if you take DESMODA.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Some medicines, food and drink can affect the way that DESMODA works and may mean that your healthcare provider needs to change the prescribed dose of DESMODA.
How should I take DESMODA?
Take DESMODA exactly as your healthcare provider tells you to take it.
Take DESMODA by mouth on an empty stomach, at least 1 hour before or 2 hours after food.
Take DESMODA using the oral syringe provided by the pharmacy.
Do not stop taking DESMODA without talking to your healthcare provider.
If you take too much DESMODA, call your healthcare provider right away or go to the nearest emergency room.
What are the possible side effects of DESMODA?
DESMODA may cause serious side effects, including:
See “What is the most important information I should know about DESMODA?”
fluid retention. Desmopressin acetate, an ingredient in DESMODA, taken as an injection can cause you to retain fluid in the body. This can make existing health problems worse that are affected by changes in your body’s fluid levels.
allergic reactions. Allergic reactions, including a severe reaction known as anaphylaxis, have happened in people who received desmopressin acetate through a vein (intravenous) or through the nose (nasal). Tell your healthcare provider right away or get emergency medical help if you get any of the following symptoms: swelling of the face, lips or tongue feeling dizzy or faint trouble breathing or swallowing rash or hives
risk of benzyl alcohol toxicity in newborn babies. Serious side effects that can lead to death have happened in low-birth weight and preterm newborns who received medicines through their vein that contained benzyl alcohol. DESMODA contains benzoic acid, a breakdown product of benzyl alcohol.
The most common side effects of DESMODA include: headache dizziness nausea stomach pain high blood pressure diarrhea
These are not all the possible side effects of DESMODA.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store DESMODA?
Store DESMODA in a refrigerator between 36°F to 46°F (2°C to 8°C) or at room temperature up to 77°F (25°C).
Do not freeze DESMODA.
After the bottle has been opened, use DESMODA within 120 days. Throw DESMODA away if it has not been used within the 120 days.
Keep DESMODA and all medicines out of the reach of children.
General information about the safe and effective use of DESMODA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use DESMODA for a condition for which it was not prescribed. Do not give DESMODA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DESMODA that is written for health professionals.
What are the ingredients in DESMODA?
Active ingredient: desmopressin acetate
Inactive ingredients: benzoic acid, sodium benzoate, and purified water.
DESMODA is manufactured for Eton Pharmaceuticals, Inc. by Tulex Pharmaceuticals, Inc., 5 Cedarbrook Dr., Cranbury, NJ 08512, USA.
DESMODA TM is a trademark of Eton Pharmaceuticals, Inc.
DESMODA is covered by the following: U.S. Patent No. 12,214,010; U.S. Patent No. 12,343,372
For more information, go to www.desmoda.com or call 1-833-343-2500.
This Medication Guide has been approved by the U.S. Food and Drug Administration. Issued: 02/2026
PL-76-1.0
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DESMODA
desmopressin acetate oral solution solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71863-123 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DESMOPRESSIN ACETATE (UNII: XB13HYU18U) (DESMOPRESSIN - UNII:ENR1LLB0FP) DESMOPRESSIN ACETATE 0.05 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZOIC ACID (UNII: 8SKN0B0MIM) SODIUM BENZOATE (UNII: OJ245FE5EU) WATER (UNII: 059QF0KO0R) Product Characteristics Color white (white amorphous powder and is soluble in water) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71863-123-14 145 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/02/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219873 03/02/2026 Labeler - Eton Pharmaceuticals, Inc. (080870465) Registrant - Eton Pharmaceuticals, Inc. (080870465) Establishment Name Address ID/FEI Business Operations PolyPeptide Laboratories AB 618054084 analysis(71863-123) , api manufacture(71863-123) Establishment Name Address ID/FEI Business Operations Tulex Pharmaceuticals, Inc. 080119240 analysis(71863-123) , manufacture(71863-123) Establishment Name Address ID/FEI Business Operations Eurofins BioPharma Product Testing, Denmark A/S 311900950 analysis(71863-123) Establishment Name Address ID/FEI Business Operations Mikrolab Stockholm AB 350032022 analysis(71863-123) Establishment Name Address ID/FEI Business Operations New Jersey Laboratories 063155238 analysis(71863-123) Establishment Name Address ID/FEI Business Operations SGS North America Inc. 049859261 analysis(71863-123)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
