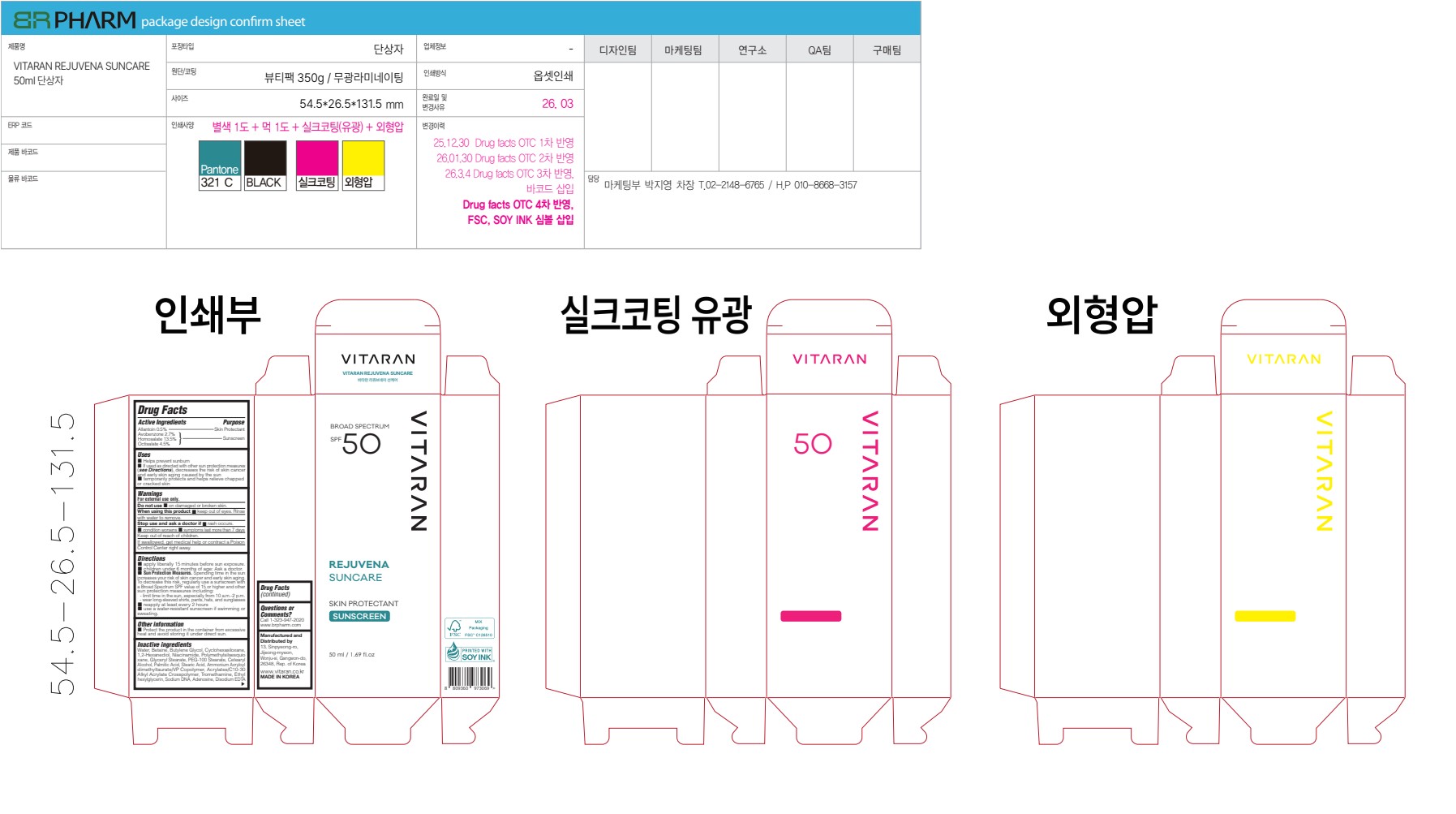
VITARAN REJUVENA SUNCARE- allantoin, avobenzone, homosalate, octisalate cream
VITARAN REJUVENA SUNCARE by
Drug Labeling and Warnings
VITARAN REJUVENA SUNCARE by is a Otc medication manufactured, distributed, or labeled by BR PHARM CO. , Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
■ apply liberally 15 minutes before sun exposure.
■ children under 6 months of age: Ask a doctor.
■ Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m-2 p.m
- wear long-sleeved shirts, pants, hats, and sunglasses
■ reapply at least every 2 hours.
■ use a water-resistant sunscreen if swimming or sweating - OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Water
Betaine
Butylene Glycol
Cyclohexasiloxane
1,2-Hexanediol
Niacinamide
Polymethylsilsesquioxane
Glyceryl Stearate
PEG-100 Stearate
Cetearyl Alcohol
Palmitic Acid
Stearic Acid
Ammonium Acryloyldimethyltaurate/VP Copolymer
Acrylates/C10-30 Alkyl Acrylate Crosspolymer
Tromethamine
Ethylhexylglycerin
Sodium DNA
Adenosine
Disodium EDTA - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
VITARAN REJUVENA SUNCARE
allantoin, avobenzone, homosalate, octisalate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 87435-1000 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 13.5 g in 100 g ALLANTOIN (UNII: 344S277G0Z) (ALLANTOIN - UNII:344S277G0Z) ALLANTOIN 0.5 g in 100 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 4.5 g in 100 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 2.7 g in 100 g Inactive Ingredients Ingredient Name Strength AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) BETAINE (UNII: 3SCV180C9W) TROMETHAMINE (UNII: 023C2WHX2V) WATER (UNII: 059QF0KO0R) PALMITIC ACID (UNII: 2V16EO95H1) STEARIC ACID (UNII: 4ELV7Z65AP) RAINBOW TROUT TESTIS DNA SODIUM (UNII: 4F8CQ9LQW9) EDETATE DISODIUM (UNII: 7FLD91C86K) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) CYCLOHEXASILOXANE (UNII: XHK3U310BA) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) GLYCERYL STEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) CETEARYL ALCOHOL (UNII: 2DMT128M1S) ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER (60000 MPA.S AT 0.5%) (UNII: YY2HMJ9NZF) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ADENOSINE (UNII: K72T3FS567) NIACINAMIDE (UNII: 25X51I8RD4) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 87435-1000-2 1 in 1 BOX 03/11/2026 1 NDC: 87435-1000-1 50 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 03/11/2026 Labeler - BR PHARM CO. , Ltd. (557782379) Registrant - BR PHARM CO. , Ltd. (557782379) Establishment Name Address ID/FEI Business Operations BR PHARM CO. , Ltd. 557782379 manufacture(87435-1000)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.