DEXTROSE injection, solution
DEXTROSE by
Drug Labeling and Warnings
DEXTROSE by is a Prescription medication manufactured, distributed, or labeled by HF Acquisition Co. LLC. DBA HealthFirst. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
TABLE OF CONTENTS
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
2.2 Recommended Dosage
2.3 Instructions for Use
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hyperglycemia and Hyperosmolar Hyperglycemic State
5.2 Hypersensitivity Reactions
5.3 Vein Damage and Thrombosis
5.4 Hyponatremia
5.5 Electrolyte Imbalance and Fluid Overload
5.6 Refeeding Syndrome
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Other Products that Affect Glycemic Control, Vasopressin or Fluid and/or Electrolyte Balance
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
* Sections or subsections omitted from the full prescribing information are not listed.
- 1. INDICATIONS AND USAGE
-
DOSAGE & ADMINISTRATION
2.1 Important Administration Instructions
10% Dextrose Injection is intended for intravenous use.
Peripheral administration of 5% dextrose is generally acceptable, however, consider central vein when administering more than 5% dextrose or with an osmolarity of at least 900 mOsm/L or when there is peripheral vein irritation, phlebitis, and/or associated pain [see Warnings and Precautions (5.3)].
Do not administer 10% Dextrose Injection simultaneously with blood products through the same administration set because of the possibility of pseudoagglutination or hemolysis.
To prevent air embolism, use a non-vented infusion set or close the vent on a vented set, avoid multiple connections, do not connect flexible containers in series, fully evacuate residual gas in the container prior to administration, do not pressurize the flexible container to increase flow rates, and if administration is controlled by a pumping device, turn off pump before the container runs dry.
Prior to infusion, visually inspect the diluted dextrose solution for particulate matter. The solution should be clear and there should be no precipitates. Do not administer unless solution is clear and container is undamaged.
Use of a final filter is recommended during administration of parenteral solutions, where possible.2.2 Recommended Dosage
The choice of dextrose concentration, rate and volume depends on the age, weight, clinical and metabolic conditions of the patient and concomitant therapy. Electrolyte supplementation may be indicated according to the clinical needs of the patient.
The administration rate should be governed, especially for premature infants with low birth weight, during the first few days of therapy, by the patient's tolerance to dextrose.
Increase the infusion rate gradually as indicated by frequent monitoring of blood glucose concentrations [see Warnings and Precautions (5.1), Use in Specific Populations (8.4)].
2.3 Instructions for Use
Check flexible container solution composition, lot number, and expiry date. Prior to administration, visually inspect for particulate matter and discoloration.
The intact port caps provides visual tamper evidence. Do not use if a port cap is prematurely removed. Do not remove solution container from its overwrap until immediately before use. Use sterile equipment and aseptic technique.
To Open
Place the solution container on a clean, flat surface. Using the pre-cut corner tabs, peel open the overwrap and remove solution container.
Check the solution container for leaks by squeezing firmly. If leaks are found, or if the seal is not intact, discard the solution.
Do not use if the solution is cloudy or a precipitate is present.To Add Medication
Identify WHITE Additive Port with arrow pointing toward container.
Immediately before injecting additives, break off WHITE Additive Port Cap with the arrow pointing toward container.
Hold base of WHITE Additive Port horizontally.
Insert needle (18 to 23 gauge) horizontally through the center of WHITE Additive Port's septum and inject additives.
Mix container contents thoroughly. For high density medication such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.Preparation for Administration
Immediately before inserting the infusion set, break off BLUE Infusion Port Cap with the arrow pointing away from container.
Use a non-vented infusion set or close the air-inlet on a vented set.
Close the roller clamp of the infusion set.
Hold the base of BLUE Infusion Port.
Insert spike through BLUE Infusion Port by rotating wrist slightly until the spike is inserted.
Suspend solution container from hanger hole
For Single Use Only. Discard unused portion.NOTE: See full directions accompanying administration set.
WARNING: Do not use flexible container in series connections.
- DOSAGE FORMS & STRENGTHS
- CONTRAINDICATIONS
-
WARNINGS AND PRECAUTIONS
- Hyperglycemia or Hyperosmolar Hyperglycemic State: Monitor blood glucose and administer insulin as needed. (5.1)
- Hypersensitivity Reactions: Monitor for signs and symptoms and discontinue infusion if reactions occur. (5.2)
- Vein Damage and Thrombosis: Consider central vein when administering more than 5% dextrose or with an osmolarity of at least 900 mOsm/L or when there is peripheral vein irritation, phlebitis, and/or associated pain. (2.2, 5.3)
- Hyponatremia: Avoid in patients with or at risk for hyponatremia. If use cannot be avoided, monitor serum sodium concentrations. (5.4)
- Electrolyte Imbalance and Fluid Overload: Avoid in patients with or at risk for fluid and/or solute overloading. If use cannot be avoided, monitor daily fluid balance, electrolyte concentrations, and acidbase balance, as needed and especially during prolonged use. (5.5)
- Refeeding Syndrome: Monitor severely undernourished patients and slowly increase nutrient intake. (5.6)
-
ADVERSE REACTIONS
The most common adverse reactions are, hyperglycemia, hypersensitivity reactions, hyponatremia, infection both systemic and at the injection site, vein thrombosis or phlebitis, and electrolyte imbalance. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551- 7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DRUG INTERACTIONS
Other Products that Affect Glycemic Control, Vasopressin or Fluid and/or Electrolyte Balance: Monitor blood glucose concentrations, fluid balance, serum electrolyte concentrations and acid-base balance. (7.1)
Pediatric Use: Increased risk of hypoglycemia/hyperglycemia; monitor serum glucose concentrations. (8.4)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2021
-
USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Appropriate administration of 10% Dextrose Injection during pregnancy is not expected to cause adverse developmental outcomes, including congenital malformations. Animal reproduction studies have not been conducted with injectable dextrose solutions.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of dextrose in human milk, the effects on a breastfed infant, or the effects on milk production. The lack of clinical data during lactation precludes a clear determination of the risk of 10% Dextrose Injection to an infant during lactation; therefore, the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for 10% Dextrose Injection and any potential adverse effects on the breastfed infant from 10% Dextrose Injection or from the underlying maternal condition.
8.4 Pediatric Use
The safety profile of 10% Dextrose Injection in pediatric patients is similar to adults.
Neonates, especially premature infants with low birth weight, are at increased risk of developing hypo- or hyperglycemia and therefore need close monitoring during treatment with intravenous glucose infusions to ensure adequate glycemic control in order to avoid potential long-term adverse effects.
Closely monitor plasma electrolyte concentrations in pediatric patients who may have impaired ability to regulate fluids and electrolytes. In very low birth weight infants, excessive or rapid administration of 10% Dextrose Injection may result in increased serum osmolality and risk of intracerebral hemorrhage.
Children (including neonates and older children) are at increased risk of developing hyponatremia as well as for developing hyponatremic encephalopathy [see Warnings and Precautions (5.4)].
8.5 Geriatric Use
Clinical studies of 10% Dextrose Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Elderly patients are at increased risk of developing hyponatremia as well as for developing hyponatremic encephalopathy [see Warnings and Precautions (5.4)]. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Dextrose is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
-
OVERDOSAGE
An increased infusion rate of 10% Dextrose Injection or administration of dextrose solutions can cause hyperglycemia, hyperosmolality, and adverse effects on water and electrolyte balance [see Warnings and Precautions (5.1, 5.5)].
Severe hyperglycemia and severe dilutional hyponatremia, and their complications, can be fatal. Discontinue infusion, reduce dose and institute appropriate corrective measures such as administration of exogenous insulin.
Discontinue infusion and institute appropriate corrective measures in the event of overhydration or solute overload during therapy, with particular attention to CNS, respiratory and cardiovascular systems.
If over-exposure occurs, call your Poison Control Center at 1-800-222-1222 for current information on the management of poisoning or overdosage.
-
DESCRIPTION
10% Dextrose Injection, USP is a sterile, non-pyrogenic solution for fluid replenishment and caloric supply in single dose containers for intravenous administration.
The solution contains no bacteriostatic, antimicrobial agent or added buffer and is intended only for use as a single-dose injection. The pH range is 4.0 (3.2 to 6.5)
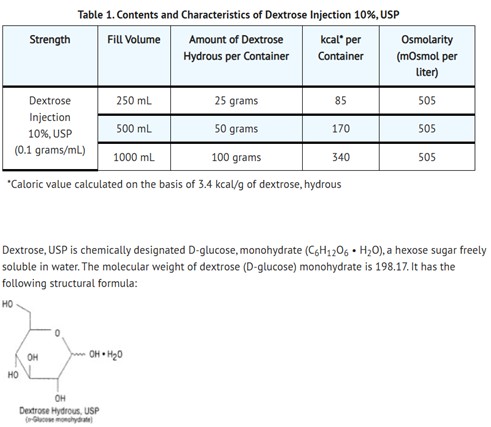
Water for Injection, USP is chemically designated H2O.
Dextrose is derived from corn.
The flexible container is fabricated from a specially formulated non-plasticized, film containing polypropylene and thermoplastic elastomers (freeflex® bag). The amount of water that can permeate from the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the flexible container can leach out certain of the container's chemical components in very small amounts within the expiration period. The suitability of the container material has been confirmed by tests in animals according to USP biological tests for plastic containers.
The flexible container is a closed system, and air is prefilled in the container to facilitate drainage. The container does not require entry of external air during administration.
The container has two ports: one is the administration outlet port for attachment of an intravenous administration set and the other port has a medication site for addition of supplemental medication ([see Instructions for Use (2.3)]). The primary function of the overwrap is to protect the container from the physical environment.
- CLINICAL PHARMACOLOGY
-
HOW SUPPLIED
10% Dextrose Injection, USP is supplied in single dose flexible plastic containers as follows:
Product Number Unit of Sale Strength Each
NDC: 51662-1680-1 250mL BAG
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat.
STORE AT: 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]; brief exposure up to 40°C/104°F does not adversely affect the product. Keep from freezing.
The container closure is not made with natural rubber latex. Non-PVC, Non-DEHP, Sterile.
-
88436-1 - Section Title Not Found In Database
Inform patients, caregivers, or home healthcare providers of the following risks of 10% Dextrose Injection:
Hyperglycemia and hyperosmolar hyperglycemic state [see Warnings and Precautions (5.1)]
Hypersensitivity reactions [see Warnings and Precautions (5.2)]
Vein damage and thrombosis [see Warnings and Precautions (5.3)]
Hyponatremia [see Warnings and Precautions (5.4)]
Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.5)]
Refeeding syndrome [see Warnings and Precautions (5.6)]Manufactured for:
Lake Zurich, IL 60047
Made in Germany
www.fresenius-kabi.com/us
451516BDistributed By:
HF Acquisition Co LLC dba HealthFirst
11629 49th PL W, Mukilteo, WA 98275
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DEXTROSE
dextrose injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 51662-1680(NDC:63323-824) Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 100 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51662-1680-1 250 mL in 1 BAG; Type 0: Not a Combination Product 03/29/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209448 03/29/2019 Labeler - HF Acquisition Co. LLC. DBA HealthFirst (045657305) Registrant - HF Acquisition Co. LLC. DBA HealthFirst (045657305) Establishment Name Address ID/FEI Business Operations HF Acquisition Co. LLC. DBA HealthFirst 045657305 relabel(51662-1680)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
