Fluoride by McKesson / Dabur India Limited
Fluoride by
Drug Labeling and Warnings
Fluoride by is a Otc medication manufactured, distributed, or labeled by McKesson, Dabur India Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
FLUORIDE- sodium fluoride paste
McKesson
----------
Warnings
Keep out of reach of children under 6 years of age.
If you accidentally swallow more than used for brushing, seek professional
help or contact a Poison Control Center immediately.
Directions
Adults and children 2 years & older:
Brush teeth thoroughly after meals or at least twice a day or use as directed by a dentist.
Do not swallow.
Instruct children under 6 years of age in good brushing and rinsing habits
(to minimize swallowing, use a pea-sized amount).
Supervise children's brushing until good habits are established.
Children under 2 years: Consult a dentist or doctor.
Inactive ingredients
Sorbitol, Silica, Treated Water, Glycerin, PEG-32, Sodium Lauryl Sulfate, Flavor,
Titanium Dioxide, Cellulose Gum, Carrageenan, Trisodium Phosphate, Sodium Saccharin,
Sodium Benzoate, Tetrasodium Pyrophosphate, FD&C Blue # 1.
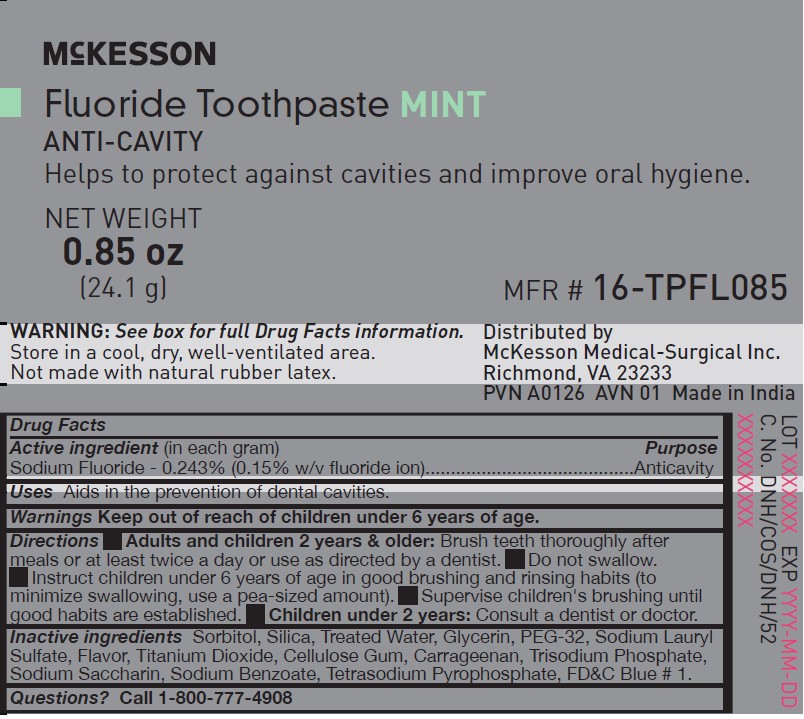
NDC: 68599-9572-1
MFR # 16-TPFL085
NET WEIGHT 0.85 oz (24.1 g)
McKesson
Fluoride Toothpaste
MINT, ANTI-CAVITY
Helps to protect against cavities and improve oral hygiene.
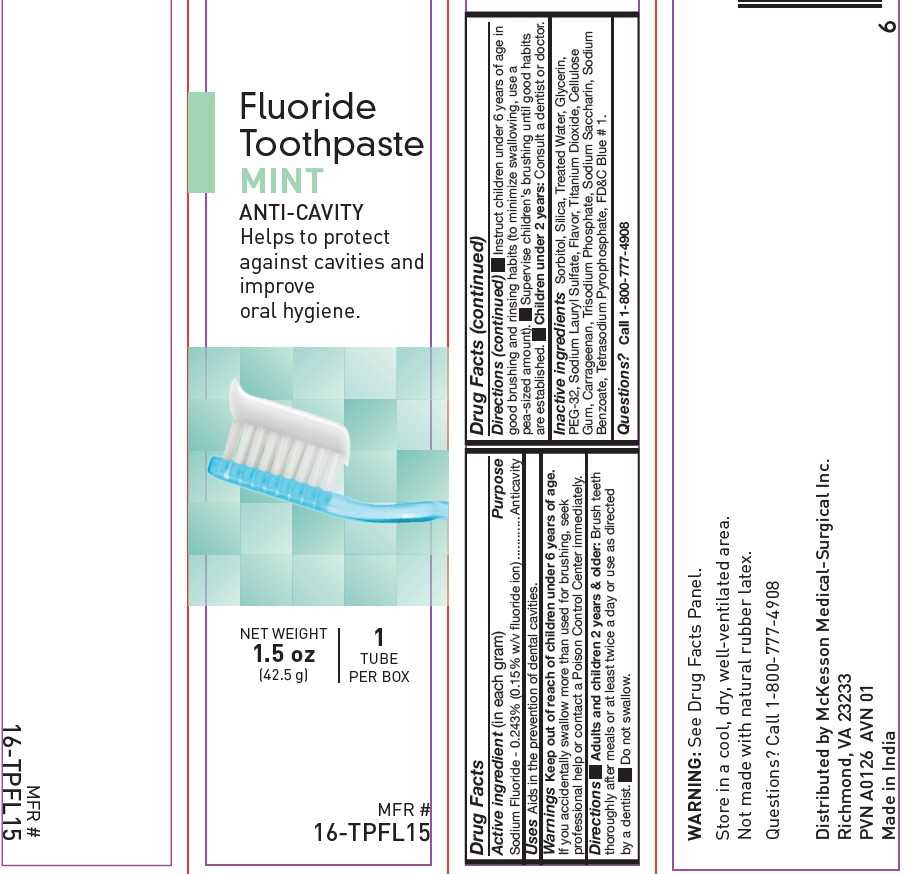
NDC: 68599-9572-2
MFR # 16-TPFL15
NET WEIGHT 1.5 oz (42.5 g)
McKesson
Fluoride Toothpaste
MINT, ANTI-CAVITY
Helps to protect against cavities and improve oral hygiene.
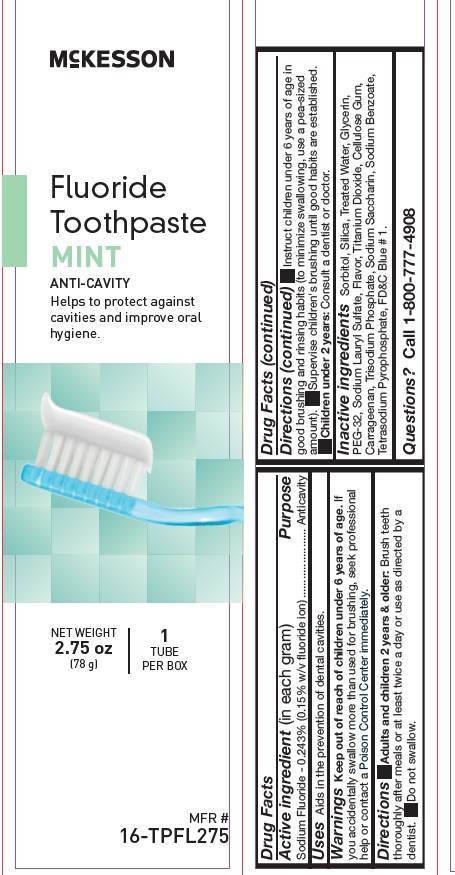
NDC: 68599-9572-3
MFR # 16-TPFL275
NET WEIGHT 2.75 oz (78 g)
McKesson
Fluoride Toothpaste
MINT, ANTI-CAVITY
Helps to protect against cavities and improve oral hygiene.
NDC: 68599-9572-4
MFR # 16-TPFL4
NET WEIGHT 4 oz (113.4 g)
McKesson
Fluoride Toothpaste
MINT, ANTI-CAVITY
Helps to protect against cavities and improve oral hygiene.

| FLUORIDE
sodium fluoride paste |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - McKesson (023904428) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Dabur India Limited | 650599231 | manufacture(68599-9572) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.