Sumi-Plantar Pain Relief Patch
Plantar Pain Relief Patch by
Drug Labeling and Warnings
Plantar Pain Relief Patch by is a Otc medication manufactured, distributed, or labeled by Guangzhou Sumi Technology Innovation Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
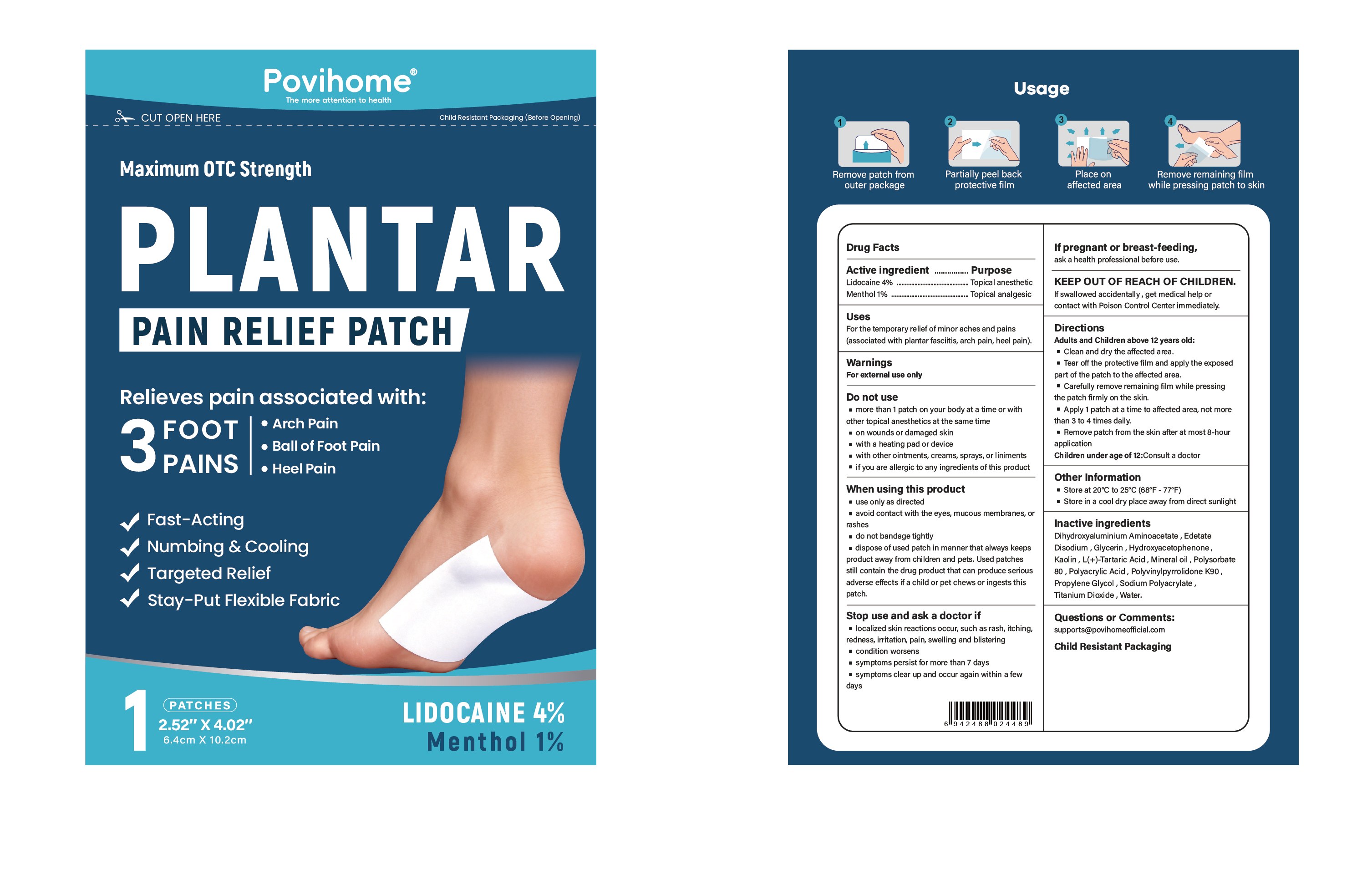
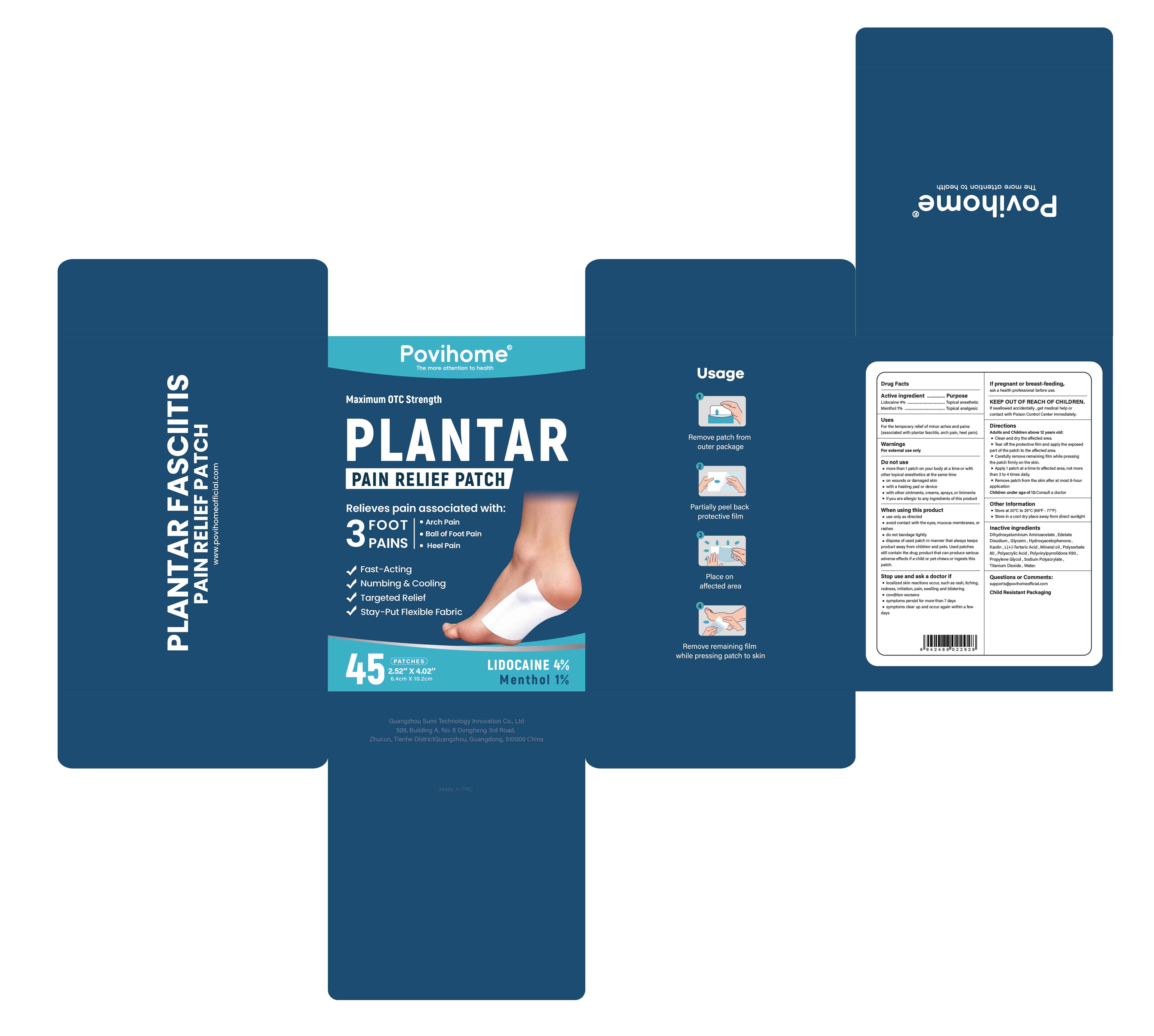
PLANTAR PAIN RELIEF PATCH- lidocaine and menthol patch
Guangzhou Sumi Technology Innovation Co., Ltd
----------
Sumi-Plantar Pain Relief Patch
Active Ingredient
Lidocaine 4% ...... Purpose: Topical anesthetic
Menthol 1% ...... Purpose: Topical analgesic
Uses
For the temporary relief of minor aches and pains (associated with plantar fasciitis, arch pain, heel pain).
Do not use:
- more than 1 patch on your body at a time or with other topical anesthetics at the same time
- on wounds or damaged skin
- with a heating pad or device
- with other ointments, creams, sprays, or liniments
- If you are allergic to any ingredients of this product
When using this product:
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- do not bandage tightly
- dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Stop use and ask a doctor if:
- localized skin reactions occur, such as rash, itching, redness, irritation, pain, swelling and blistering
- condition worsen
- symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center immediately.
Directions
Adult and Children above12 years old:
- Clean and dry the affected area
- Tear off the protective film and apply the exposed part of the patch to the affected area.
- Carefully remove remaining film while pressing the patch firmly on the skin.
- Apply 1 patch at a time to affected area, not morethan 3 to 4 times daily.
- Remove patch from the skin after at most 8-hour application
Children under age of 12:Consult a doctor
Other Information
· Store at 20°C to 25°C(68°F - 77°F)
· Store in a cool dry place away from direct sunlight
| PLANTAR PAIN RELIEF PATCH
lidocaine and menthol patch |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Guangzhou Sumi Technology Innovation Co., Ltd (518555082) |
| Registrant - Guangzhou Sumi Technology Innovation Co., Ltd (518555082) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.