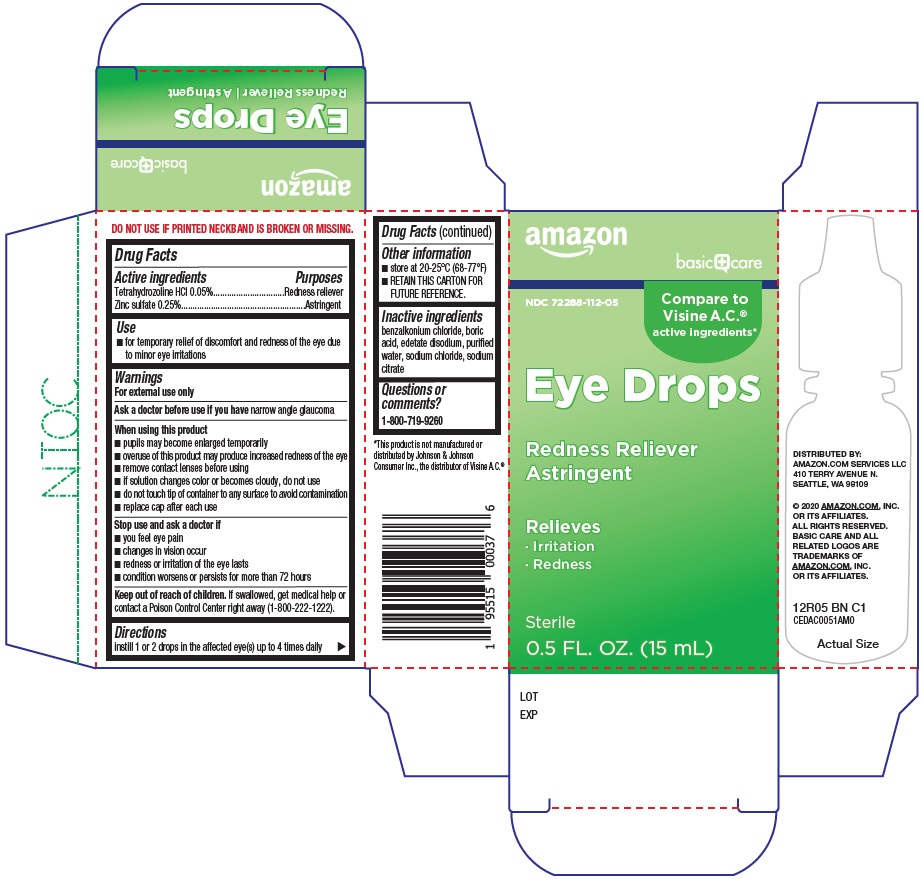
Amazon Eye Drops Drug Facts
Basic Care Eye by
Drug Labeling and Warnings
Basic Care Eye by is a Otc medication manufactured, distributed, or labeled by Amazon.com Services LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BASIC CARE EYE- tetrahydrozoline hcl, zinc sulfate solution/ drops
Amazon.com Services LLC
----------
Amazon Eye Drops Drug Facts
Warnings
For external use only
When using this product
- pupils may become enlarged temporarily
- overuse of this product may produce increased redness of the eye
- remove contact lenses before using
- if solution changes color or becomes cloudy, do not use
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
| BASIC CARE EYE
tetrahydrozoline hcl, zinc sulfate solution/ drops |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Amazon.com Services LLC (128990418) |
Revised: 11/2024
Document Id: 8103e86e-889e-40ea-8b1c-23d60489a3b7
Set id: 51340b77-68d1-4131-9342-b22c5fa2d55c
Version: 3
Effective Time: 20241114