AWIQLI- insulin icodec-abae injection, solution
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Awiqli® safely and effectively. See full prescribing information for Awiqli.
Awiqli (insulin icodec-abae) injection, for subcutaneous use
Initial U.S. Approval: 2026INDICATIONS AND USAGE
Awiqli is a long-acting human insulin analog indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus (1)
DOSAGE AND ADMINISTRATION
- Individualize dose based on type of diabetes, metabolic needs, blood glucose monitoring results and glycemic control goals. (2.1)
- See Full Prescribing Information for important administration instructions (2.2)
- Inject Awiqli subcutaneously into the thigh, upper arm, or abdomen. (2.2)
- Rotate injection sites to reduce risk of lipodystrophy and localized cutaneous amyloidosis. (2.2)
- See Full Prescribing Information for the recommended starting dosage in insulin naïve patients (2.3) and recommendations for switching patients from daily basal insulin. (2.4)
- Closely monitor glucose when switching to Awiqli. (2.4)
DOSAGE FORMS AND STRENGTHS
Injection: 700 units/mL (U-700) available as a clear and colorless solution:
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Hypoglycemia Due to Medication Errors and Accidental Overdose: Accidental mix-ups between insulin products can occur. Advise patients to always check the product label before each injection to confirm they are using Awiqli and not another insulin or injectable antidiabetic medicine. DO NOT transfer Awiqli from the Awiqli FlexTouch pen into a syringe for administration as overdosage and severe hypoglycemia can result. (5.1)
- Hypoglycemia: May be life-threatening. Increase monitoring with changes to: insulin dosage, co-administered glucose lowering medications, meal pattern, physical activity, and in patients with renal impairment, hepatic impairment or hypoglycemia unawareness. (5.2)
- Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen: Make changes to a patient’s insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) under close medical supervision with increased frequency of blood glucose monitoring. (5.3)
- Hypersensitivity Reactions: Severe, life-threatening, generalized allergy, including anaphylaxis, can occur. Discontinue Awiqli FlexTouch, monitor and treat if indicated. (5.4)
- Hypokalemia: May be life-threatening. Monitor potassium levels in patients at risk for hypokalemia and treat if indicated. (5.5)
- Never share an Awiqli FlexTouch pen between patients, even if the needle is changed. (5.6)
- Fluid Retention and Heart Failure with Concomitant Use of Thiazolidinediones (TZDs): Observe for signs and symptoms of heart failure; consider dosage reduction or discontinuation if heart failure occurs. (5.7)
ADVERSE REACTIONS
Adverse reactions commonly associated with Awiqli are:
- hypoglycemia, hypersensitivity reactions (e.g., urticaria, swelling face and lips), injection site reactions, lipodystrophy, pruritus, rash, edema, and weight gain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Novo Nordisk at 1-844-668-6463 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Drugs that may increase the risk of hypoglycemia: antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analog (e.g., octreotide), sulfonamide antibiotics, GLP-1 receptor agonists, DPP-4 inhibitors, and SGLT-2 inhibitors. (7)
- Drugs that may decrease the blood glucose lowering effect: atypical antipsychotics, corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. (7)
- Drugs that may increase or decrease the blood glucose lowering effect: Alcohol, beta-blockers, clonidine, lithium salts, and pentamidine. (7)
- Drugs that may blunt the signs and symptoms of hypoglycemia: beta-blockers, clonidine, guanethidine, and reserpine. (7)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Instructions
2.2 Important Administration Instructions
2.3 Recommended Dosage in Insulin Naive Patients
2.4 Switching to Awiqli from Daily Basal Insulin Therapy
2.5 Recommendations Regarding Missed Doses
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypoglycemia Due to Medication Errors and Accidental Overdose
5.2 Hypoglycemia
5.3 Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen
5.4 Hypersensitivity Reactions
5.5 Hypokalemia
5.6 Never Share an Awiqli FlexTouch Pen or Needle Between Patients
5.7 Fluid Retention and Congestive Heart Failure with Concomitant Use of a PPAR Gamma Agonist
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 Immunogenicity
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Overview of Clinical Trials
14.2 Insulin Naïve Adults with Type 2 Diabetes Mellitus
14.3 Adults with Type 2 Diabetes Mellitus Previously Treated with Basal Insulin
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Instructions
Awiqli FlexTouch is available as a single-patient-use FlexTouch pen.
- Inject Awiqli subcutaneously once-weekly on any day of the week on the same day each week.
- The Awiqli FlexTouch pen delivers doses in 10 unit increments and can deliver up to 700 units in a single injection.
- Individualize and titrate the dose of Awiqli based on the patient’s metabolic needs, blood glucose monitoring results, and glycemic control goal.
- The potency of insulin analogues, including insulin icodec-abae is expressed in units. One (1) unit of insulin icodec-abae corresponds to 1 international unit of human insulin.
- Dose adjustments may be needed with changes in renal or hepatic function or during illness to minimize the risk of hypoglycemia or hyperglycemia. Due to the long half-life of Awiqli, adjustment of dose is not advised during acute illness nor if patients make short-term changes in their physical activity level or usual diet. In these situations, consider other applicable adjustments, e.g. glucose intake or changes to other glucose lowering medication [see Warnings and Precautions (5.2, 5.3)].
2.2 Important Administration Instructions
- Always check the product label before administration [see Warnings and Precautions (5.1)].
- Inspect visually for particulate matter and discoloration. Only use Awiqli if the solution appears clear and colorless.
- Inject Awiqli subcutaneously into the thigh, upper arm, or abdomen.
- Rotate injection sites within the same region from one injection to the next to reduce the risk of lipodystrophy and localized cutaneous amyloidosis. Do not inject into areas of lipodystrophy or localized cutaneous amyloidosis [see Warnings and Precautions (5.3), Adverse Reactions (6.1)].
- During changes to a patient’s insulin regimen, increase the frequency of blood glucose monitoring [see Warnings and Precautions (5.2, 5.3)].
- Use Awiqli FlexTouch pen with caution in patients with visual impairment that may rely on audible clicks to dial their dose.
- DO NOT administer Awiqli intramuscularly, intravenously or in an insulin infusion pump.
- DO NOT dilute or mix Awiqli with any other insulin or solution.
- DO NOT transfer Awiqli from the Awiqli FlexTouch pen into a syringe for administration [see Warnings and Precautions (5.1)].
2.3 Recommended Dosage in Insulin Naive Patients
The recommended weekly starting dose of Awiqli in insulin naïve patients is 70 units administered subcutaneously once-weekly on the same day each week.
2.4 Switching to Awiqli from Daily Basal Insulin Therapy
- Administer the first dose of Awiqli on the day after the last dose of daily basal insulin.
- Week 1 dosage: The recommended one-time starting dosage of Awiqli FlexTouch is 1.5 times the total daily basal dosage multiplied by 7 rounded to the nearest 10 units.
- Week 2 dosage: The recommended dosage is the previous total daily basal insulin dose multiplied by 7 and then rounded to the nearest 10 units.
- See Table 1 for examples of Awiqli dosage for Week 1 and 2, when switching from daily basal insulin therapy.
- Week 3 dosage and beyond: The recommended dosage of Awiqli can be titrated from the previous dosage based on the patient’s metabolic needs, blood glucose monitoring results, and glycemic control goal.
- When switching from daily basal insulin to once-weekly Awiqli close glucose monitoring is recommended. Doses and timing of concurrent rapid-acting or short-acting insulin products or other concomitant antidiabetic treatment may need to be adjusted [see Warnings and Precautions (5.1)].
Table 1. Example Awiqli Weekly Dosages When Switching from Daily Basal Insulin Therapy
Previous total daily dosage of
basal insulin (units)Week 1 Dosage of Awiqli (units)a
Week 2 Dosage of Awiqli (units)b
10
110
70
11
120
80
12
130
80
13
140
90
14
150
100
15
160
110
16
170
110
17
180
120
18
190
130
19
200
130
20
210
140
21
220
150
22
230
150
23
240
160
24
250
170
25
260
180
26
270
180
27
280
190
28
290
200
29
300
200
30
320
210
31
330
220
32
340
220
33
350
230
34
360
240
35
370
250
36
380
250
37
390
260
38
400
270
39
410
270
40
420
280
41
430
290
42
440
290
43
450
300
44
460
310
45
470
320
46
480
320
47
490
330
48
500
340
49
510
340
50
530
350
100
1,050c
700
a Week 1 dose only: Multiply the previous total daily basal insulin dosage by 7, then multiply by 1.5, and round to the nearest 10 units.
b Week 2 dose: Previous total daily basal insulin dosage multiplied by 7, then rounded to the nearest 10 units.
c When the required dose is larger than 700 units, split the dose into two injections (e.g., a 1,050 unit dose could be administered as a 700 unit injection followed by a 350 unit injection)
2.5 Recommendations Regarding Missed Doses
- If a dose is missed, administer the missed dose as soon as possible within 4 days. Resume the once-weekly dosing schedule, one week from the day the missed dose was administered.
- If more than 4 days have passed, skip the missed dose and administer the next dose on the regularly scheduled day.
- Increase blood glucose monitoring with missed doses.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypoglycemia Due to Medication Errors and Accidental Overdose
Serious hypoglycemia requiring hospitalization has occurred due to accidental mix-ups between Awiqli and other insulin products or once-weekly injectable antidiabetic medicines, incorrect dose selection, and dosing frequency errors.
To avoid dosing errors when switching from daily basal insulin to Awiqli, follow the dosage recommendations in the Dosage and Administration Section (2.2). Administer Awiqli once weekly only.
Advise patients to always check the product label before each injection to confirm they are using Awiqli and not another insulin or injectable antidiabetic medicine. Prior to initiation, train patients and their caregiver(s) on how to select their weekly Awiqli dosage. Advise patients using other injectable medications for glycemic control that the dosage selection of Awiqli differs [see Dosage and Administration (2.2, 2.3, 2.4), Instructions for Use]. Instruct patients to visually verify the dialed units on the dose counter of the Awiqli FlexTouch prefilled pen before each injection to avoid dosing errors. Do not dial the maximum single dose (700 units) of Awiqli unless this is the prescribed dose [see Dosage and Administration (2.4)]. Do not use a syringe to remove Awiqli from the Awiqli FlexTouch disposable insulin prefilled pen.
Monitor patients for signs and symptoms of hypoglycemia, particularly during the first several weeks after initiation or dose escalation of Awiqli. Ensure patients understand how to recognize and manage hypoglycemia [see Warnings andPrecautions (5.2)].
5.2 Hypoglycemia
Hypoglycemia is the most common adverse reaction associated with insulin, including Awiqli [see Adverse Reactions(6.1)]. Severe hypoglycemia can cause seizures, may be life-threatening or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place an individual and others at risk in situations where these abilities are important (e.g., driving or operating other machinery). Awiqli, or any insulin, should not be used during episodes of hypoglycemia [see Contraindications (4)].
Hypoglycemia can happen suddenly and symptoms may differ in each individual and change over time in the same individual. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic nerve disease, in patients using medications that block the sympathetic nervous system (e.g., beta-blockers) [see Drug Interactions (7)], or in patients who experience recurrent hypoglycemia.
The long-acting effect of Awiqli may delay recovery from hypoglycemia compared to shorter-acting insulins.
Risk Factors for Hypoglycemia
The risk of hypoglycemia generally increases with intensity of glycemic control. The risk of hypoglycemia after an injection is related to the duration of action of the insulin [see Clinical Pharmacology (12.2)] and, in general, is highest when the glucose lowering effect of the insulin is maximal. As with all insulins, the glucose lowering effect over time of Awiqli may vary among different individuals or at different times in the same individual and depends on many conditions, including the area of injection as well as the injection site blood supply and temperature.
Other factors which may increase the risk of hypoglycemia include changes in meal pattern (e.g., macronutrient content or timing of meals), changes in level of physical activity, or changes to co-administered medication [see Drug Interactions(7)]. Patients with renal or hepatic impairment may be at higher risk of hypoglycemia [see Use in Specific Populations(8.6, 8.7)].
Risk Mitigation Strategies for Hypoglycemia
Educate patients and caregivers to recognize and manage hypoglycemia. Self-monitoring of blood glucose plays an essential role in the prevention and management of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended.
5.3 Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen
Changes in an insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) may affect glycemic control and predispose to hypoglycemia [see Warnings and Precautions (5.2)] or hyperglycemia. Repeated insulin injections into areas of lipodystrophy or localized cutaneous amyloidosis have been reported to result in hyperglycemia; and a sudden change in the injection site (to an unaffected area) has been reported to result in hypoglycemia [see Adverse Reactions (6)].
Make any changes to a patient’s insulin regimen under close medical supervision with increased frequency of blood glucose monitoring. Advise patients who have repeatedly injected into areas of lipodystrophy or localized cutaneous amyloidosis to change the injection site to unaffected areas and closely monitor for hypoglycemia. Adjustments in concomitant anti-diabetic treatment may be needed [see Dosage and Administration (2.4)].
5.4 Hypersensitivity Reactions
Severe, life-threatening, generalized allergy, including anaphylaxis, can occur with insulins, including Awiqli. If hypersensitivity reactions occur, discontinue Awiqli; treat per standard of care and monitor until symptoms and signs resolve. Awiqli is contraindicated in patients who have had hypersensitivity reactions to insulin icodec-abae or any of the excipients [see Contraindications (4)].
5.5 Hypokalemia
All insulins, including Awiqli, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia. Untreated hypokalemia may cause respiratory paralysis, ventricular arrhythmia, and death. Monitor potassium levels in patients at risk for hypokalemia if indicated (e.g., patients using potassium-lowering medications, patients taking medications sensitive to serum potassium concentrations).
5.6 Never Share an Awiqli FlexTouch Pen or Needle Between Patients
Awiqli FlexTouch disposable prefilled pens should never be shared between patients, even if the needle is changed. Sharing poses a risk for transmission of blood-borne pathogens.
5.7 Fluid Retention and Congestive Heart Failure with Concomitant Use of a PPAR Gamma Agonist
Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)-gamma agonists can cause dose related fluid retention, particularly when used in combination with insulin. Fluid retention may lead to or exacerbate congestive heart failure. Patients treated with insulin, including Awiqli and a PPAR-gamma agonist should be observed for signs and symptoms of congestive heart failure. If congestive heart failure develops, it should be managed according to current standards of care and discontinuation or dose reduction of the PPAR-gamma agonist must be considered.
-
6 ADVERSE REACTIONS
The following adverse reactions are also discussed elsewhere:
- Hypoglycemia Due to Medication Errors and Accidental Overdose [see Warnings and Precautions (5.1)]
- Hypoglycemia [see Warnings and Precautions (5.2)]
- Hypersensitivity reactions [see Warnings and Precautions (5.4)]
- Hypokalemia [see Warnings and Precautions (5.5)]
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Awiqli in patients with type 2 diabetes was evaluated in five clinical trials involving 1,880 adults with type 2 diabetes exposed to Awiqli, with a mean exposure duration of 26 to 52 weeks across the five trials [see Clinical Studies(14)]. The type 2 diabetes population had the following characteristics: mean age was 59 years and 5% were older than 75 years, 59% were male, 71% were White, 3.6% were Black or African American, and 13% were Hispanic or Latino ethnicity. The mean BMI was 30.7 kg/m2. The mean duration of diabetes was 13 years and the mean HbA1c at baseline was 8.6%. At baseline, the mean eGFR was 86.1 mL/min/1.73 m2 and 11% of patients had an eGFR less than 60 mL/min/1.73 m2.
Common Adverse Reactions
Hypoglycemia
Hypoglycemia is the most commonly observed adverse reaction in patients treated with Awiqli [see Warnings andPrecautions (5.2)]. The rates of reported hypoglycemia depend on the definition of hypoglycemia used, diabetes type, insulin dose, intensity of glucose control, background therapies, and other intrinsic and extrinsic patient factors. For these reasons, comparing rates of hypoglycemia in clinical trials for Awiqli with the incidence of hypoglycemia for other products may be misleading and also, may not be representative of hypoglycemia rates that will occur in clinical practice.
In clinical trials [see Clinical Studies (14)], events of severe hypoglycemia (level 3) were defined as an episode associated with severe cognitive impairment requiring external assistance for recovery. Hypoglycemia episodes with a glucose level below 54 mg/dL with or without associated symptoms (level 2 hypoglycemia) were also assessed in patients with type 2 diabetes.
In the clinical trials of patients with type 2 diabetes, percentages of adults randomized to Awiqli who experienced at least one episode of severe or clinically significant (level 2) hypoglycemia in clinical trials are shown in Table 2.
Table 2: Proportion (%) of Patients with Type 2 Diabetes Experiencing at Least One Episode of Severe (Level 3) or Clinically Significant (Level 2) Hypoglycemia in Clinical Trials Type 2 Diabetes Trial A Trial B Trial C Trial D Trial Ec Awiqli + anti-diabetic drugsd
insulin naïve
52 weeks (N=492)Awiqli + anti-diabetic drugse
insulin naïve
26 weeks
(N=293)Awiqli + anti-diabetic drugsd
26 weeks
(N=262)Awiqli ± anti-diabetic drugsd +
insulin aspart
26 weeks
(N=291)Awiqli + anti-diabetic drugsd
insulin naïve
52 weeks
(N=542)Level 3 Hypoglycemiaa
0.2
0
0
1.4
0
Level 2 Hypoglycemiab
9.8
8.9
14.1
50.9
11.8
a Level 3 hypoglycemia is an episode associated with severe cognitive impairment requiring external assistance for recovery.
b Level 2 hypoglycemia is hypoglycemia episode with a self-measured blood glucose level below 54 mg/dL with or without associated symptoms.
c In Trial E, Awiqli arm titration was performed via a digital titration app.
d Excludes sulfonylureas and glinides.
e Sulfonulurea and glinide dosage decreased by 50% at the discretion of the investigator.
Insulin Initiation and Intensification of Glucose Control
Intensification or rapid improvement in glucose control has been associated with a transitory, reversible ophthalmologic refraction disorder, worsening of diabetic retinopathy, and acute painful peripheral neuropathy. However, long-term glycemic control decreases the risk of diabetic retinopathy and neuropathy.
Allergic Reactions
Severe, life-threatening, generalized allergy, including anaphylaxis, generalized skin reactions, angioedema, bronchospasm, hypotension, and shock may occur with any insulin, including Awiqli and may be life threatening. Hypersensitivity (manifested with urticaria, swelling of face and lips swelling) were reported in 0.4% of patients treated with Awiqli.
In the three clinical trials in type 2 patients with antibody samples collected (Trial B, C, and D), hypersensitivity reactions occurred in 0.6% of Awiqli-treated patients with anti-insulin icodec-abae antibodies and in 0.4% of Awiqli-treated patients who did not develop anti-insulin icodec-abae antibodies [see Clinical Pharmacology (12.6)].
Lipodystrophy
Long-term use of insulin, including Awiqli, can cause lipodystrophy at the site of repeated insulin injections. Lipodystrophy includes lipohypertrophy (thickening of adipose tissue) and lipoatrophy (thinning of adipose tissue) and may affect insulin absorption [see Dosage and Administration (2.2)]. In clinical trials, lipodystrophy, lipohypertrophy, or lipoatrophy was reported in 0.0% of patients treated with Awiqli.
Injection Site Reactions
Patients taking Awiqli may experience injection site reactions, including pruritus, bruising, erythema, pain, injection site hypersensitivity, swelling, urticaria and injection site mass. In the clinical trials, injection site reactions occurred in 1.8% of patients treated with Awiqli.
In the three clinical trials in type 2 patients with antibody samples collected (Trial B, C, and D), injection site reactions occurred in 2.3% of Awiqli-treated patients with anti-insulin icodec-abae antibodies and in 2.4% of Awiqli-treated patients who did not develop anti-insulin icodec-abae antibodies [see Clinical Pharmacology (12.6)].
Weight Gain
Weight gain can occur with insulin therapy, including Awiqli, and has been attributed to the anabolic effects of insulin. In the clinical trials after 26 to 52 weeks of treatment, patients with type 2 diabetes treated with Awiqli gained an average weight of 1.4 to 2.8 kg.
Peripheral Edema
Awiqli, may cause sodium retention and edema. In the clinical trials, peripheral edema occurred in 1.2% of patients with type 2 diabetes mellitus treated with Awiqli.
-
7 DRUG INTERACTIONS
Table 3 includes clinically significant drug interactions with Awiqli.
Table 3: Clinically Significant Drug Interactions with Awiqli
Drugs That May Increase the Risk of Hypoglycemia
Drugs:
Antidiabetic agents, angiotensin converting enzyme (ACE) inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analogs (e.g., octreotide), and sulfonamide antibiotics, glucagon-like peptide-1 (GLP-1) receptor agonists, dipeptidyl peptidase-4 (DPP-4) inhibitors, sodium-glucose co-transporter 2 (SGLT-2) inhibitors.
Intervention:
Dose reductions and increased frequency of glucose monitoring may be required when Awiqli is co-administered with these drugs.
Drugs That May Decrease the Blood Glucose Lowering Effect of Awiqli
Drugs:
Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones.
Intervention:
Dose increases and increased frequency of glucose monitoring may be required when Awiqli is co-administered with these drugs.
Drugs That May Increase or Decrease the Blood Glucose Lowering Effect of Awiqli
Drugs:
Alcohol, beta-blockers, clonidine, and lithium salts.Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia.
Intervention:
Dose adjustment and increased frequency of glucose monitoring may be required when Awiqli is co-administered with these drugs.
Drugs That May Blunt Signs and Symptoms of Hypoglycemia
Drugs:
Beta-blockers, clonidine, guanethidine, and reserpine
Intervention:
Increased frequency of glucose monitoring may be required when Awiqli is co-administered with these drugs.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data with Awiqli in pregnant women to inform a drug-associated risk for major birth defects and miscarriage. There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy [seeClinicalConsiderations].
Rats and rabbits were exposed to insulin icodec-abae in animal reproduction studies during organogenesis. No adverse developmental effects were observed in rats or rabbits at exposures approximately equal to human exposure at a dose of 230 U/week [see Data].
The estimated background risk of major birth defects is 6-10% in women with pre-gestational diabetes with an HbA1c >7 and has been reported to be as high as 20-25% in women with an HbA1c >10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-associated Maternal and/or Embryo/fetal Risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, still birth, and macrosomia related morbidity.
Data
Animal Data
Insulin icodec-abae was investigated in studies covering the periods of embryo-fetal development and pre- and post-natal development in rats and the period of embryo-fetal development in rabbits. In these studies, insulin icodec-abae did not cause adverse effects on embryo-fetal development when given subcutaneously at up to 10 U/kg/day in rats and 3 U/kg/day in rabbits, resulting in exposures comparable to human exposure (AUC) at a human subcutaneous dose of 230 U/week. Maternal deaths and abortions were observed in rabbits at human exposures secondary to maternal hypoglycemia.
In a pre- and postnatal developmental study in rats where insulin icodec-abae was given by the subcutaneous route at doses up to 8.3 U/kg/day (from Gestation Day 6 through Lactation Day 20), maternal and pup mortality occurred during the lactation period at exposures approximately equal to human exposures, which were secondary to maternal hypoglycemia.
8.2 Lactation
Risk Summary
There are no data on the presence of insulin icodec-abae in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Awiqli and any potential adverse effects on the breastfed infant from Awiqli or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of Awiqli have not been established in pediatric patients.
8.5 Geriatric Use
In controlled clinical trials [see Clinical Studies (14)] a total of 646 (34.4%) of the 1,880 Awiqli-treated patients with type 2 diabetes were 65 years of age or older and 97 (5.2%) were 75 years of age or older. No overall differences in safety or effectiveness of Awiqli have been observed between patients 65 years of age and older and younger adult patients.
Greater caution should be exercised when Awiqli is administered to geriatric patients since greater sensitivity of some older individuals to the effects of Awiqli cannot be ruled out. The initial dosing, dose increments, and maintenance dosage should be conservative to avoid hypoglycemia. Hypoglycemia may be more difficult to recognize in the elderly. More frequent glucose monitoring is recommended and the insulin dose is to be adjusted on an individual basis.
8.6 Renal Impairment
No clinically relevant difference in the pharmacokinetics of Awiqli was identified in a study comparing healthy subjects and subjects with renal impairment including subjects with end stage renal disease. Additional dose adjustment should not be necessary for patients with renal impairment [see Dosage and Administration (2.3, 2.4), Clinical Pharmacology (12.3)]. However, as with all insulin products, glucose monitoring should be intensified and the Awiqli dosage adjusted on an individual basis in patients with renal impairment.
8.7 Hepatic Impairment
No difference in the pharmacokinetics of Awiqli was identified in a study comparing healthy subjects and subjects with hepatic impairment (mild, moderate, and severe hepatic impairment) [see Clinical Pharmacology (12.3)]. Additional dose adjustment should not be necessary for patients with hepatic impairment [see Dosage and Administration (2.3, 2.4),Clinical Pharmacology (12.3)]. However, as with all insulin products, glucose monitoring should be intensified and the Awiqli dosage adjusted on an individual basis in patients with hepatic impairment.
-
10 OVERDOSAGE
An excess of insulin relative to food intake, energy expenditure, or both may lead to severe and sometimes prolonged and life-threatening hypoglycemia and hypokalemia [see Warnings and Precautions (5.2, 5.5)]. Mild episodes of hypoglycemia usually can be treated with oral glucose. Lowering the dosage of Awiqli, adjustments in meal patterns, or physical activity may be needed. More severe episodes of hypoglycemia with coma, seizure, or neurologic impairment may be treated with a glucagon product for emergency use or concentrated intravenous glucose. After apparent clinical recovery from hypoglycemia, continued observation and additional carbohydrate intake may be necessary to avoid reoccurrence of hypoglycemia. Hypokalemia must be corrected appropriately. Overdosing has been investigated in a clinical pharmacology trial [see Clinical Pharmacology (12.2)].
-
11 DESCRIPTION
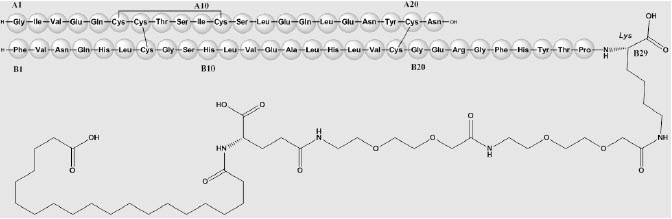
Insulin icodec-abae is a once-weekly basal human insulin analog for subcutaneous injection produced by a process that includes expression of recombinant DNA in Saccharomyces cerevisiae followed by chemical modification. Insulin icodec-abae differs from human insulin in that the amino acid threonine in position B30 has been omitted, Tyr(A14) has been substituted with Glu and Tyr(B16) and Phe(B25) have been substituted with His. The side chain is connected to the peptide backbone via the amino group in the side chain at Lys(B29). Insulin icodec-abae has a molecular formula of C280H435N71O87S6 and a molecular weight of 6380.26 Da. It has the following structure:
Figure 1: Structural Formula of Insulin icodec-abae
Awiqli (insulin icodec-abae) injection is a sterile, clear and colorless solution available as 700 units/mL (U-700) for subcutaneous use.
Each mL contains 700 units of insulin icodec-abae and glycerin (15 mg), metacresol (1.08 mg), phenol (5.65 mg), sodium chloride (1.17 mg), zinc acetate (101 mcg), and Water for Injection.
Awiqli has a pH of approximately 7.4. Hydrochloric acid or sodium hydroxide may be added to adjust pH.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The primary activity of insulin, including Awiqli, is regulation of glucose metabolism. Insulin and its analogs lower blood glucose by stimulating peripheral glucose uptake, especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin also inhibits lipolysis and proteolysis, and enhances protein synthesis. Awiqli binds reversibly to albumin, resulting in a depot in the circulation from which insulin icodec-abae is slowly released. The insulin receptor is activated by insulin icodec-abae leading to a stable glucose-lowering effect over the entire dosing interval of one week. When insulin icodec-abae binds to the human insulin receptor it results in the same pharmacological effects as human insulin.
12.2 Pharmacodynamics
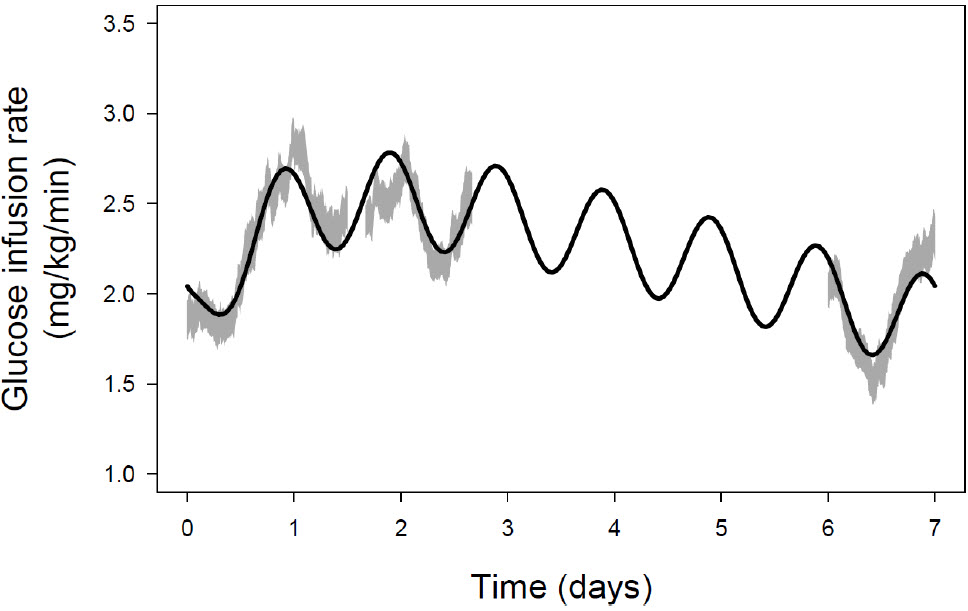
Euglycemic clamps were performed at steady state in a single-center, open-label, one-period, multiple-dose study in 46 patients with Type 2 diabetes from 0 to 36 hours, 40 to 64 hours, and 144 to 168 hours post dose. Individualized insulin icodec-abae doses were used to obtain pre-breakfast self-monitored plasma glucose within 80-126 mg/dL, the mean dose at steady state was 2.9 U/kg/week (range: 1.5-5.6 U/kg/week). Glucose infusion rate profiles for all three clamps are shown together with the model-derived data suggesting the duration of the glucose-lowering effect to cover a full week (Figure 2).
Figure 2: Full-week Glucose Infusion Rate Profile of Insulin icodec-abae at Steady-state in Type 2 Diabetes
12.3 Pharmacokinetics
Absorption
Dose proportionality in total exposure is observed after subcutaneous administration of Awiqli within the therapeutic dose range.
Insulin icodec-abae concentration reached steady state levels after 2 to 3 weeks of Awiqli administration with a one-time 50% additional dose for the first dose [see Dosage and Administration (2.4)] and after 3 to 4 weeks when initiating Awiqli without a one-time additional dose [see Dosage and Administration (2.4)]. In patients with type 2 diabetes, after 8 weekly doses with 2.9 U/kg of Awiqli maximum insulin icodec-abae concentrations of 283 nmol/L were attained at a median of 15 hours (tmax) post-dose.
Distribution
The affinity of Awiqli to serum albumin corresponds to a plasma protein binding of >99% in human plasma.
The results of the in vitro protein binding studies demonstrate that there is no clinically relevant interaction between Awiqli and fatty acids or other protein-bound drugs.
Elimination
The half-life after subcutaneous administration is approximately one week independent of dose. Degradation of insulin icodec-abae is similar to that of human insulin; all metabolites formed are inactive.
Specific Populations
Age, Sex, Race, Ethnicity, and Body Weight
The effect of covariates on the pharmacokinetics of Awiqli was examined in a population PK analysis of several exploratory and confirmatory trials. Age (18 to 86 years), sex, race (68% white, 4% Black, and 25% Asian) and ethnicity (86% not of Hispanic or Latino origin, 13% Hispanic or Latino) did not meaningfully affect the pharmacokinetics and pharmacodynamics of Awiqli.
Body weight had a clear effect on Awiqli exposure which decreased with increasing body weight (40 to 160 kg).
Renal Impairment
The pharmacokinetics of insulin icodec-abae were studied in 58 subjects with normal or impaired renal function/end-stage renal disease following administration of single subcutaneous dose (1.5 U/kg) of Awiqli . Renal function was defined using measured iohexol clearance as follows: ≥90 mL/min (normal), 60-89 mL/min (mild), 30-59 mL/min (moderate) and <30 mL/min (severe). Subjects requiring hemodialysis were classified as having end-stage renal disease (ESRD). In this study, there was no apparent effect of renal function or dialysis on the pharmacokinetics of insulin icodec-abae.
Hepatic Impairment
A single subcutaneous dose of 1.5 U/kg Awiqli was administered in an open-label, single-dose study of 25 subjects with normal or different degree of hepatic impairment (mild, moderate and severe) classified based on Child-Pugh Scores ranging from 0 (healthy volunteers) to 12 (severe hepatic impairment). In this study, there was no apparent impact of the degree of hepatic impairment on the pharmacokinetic parameters of insulin icodec-abae.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADAs in the studies described below with the incidence of ADAs in other studies, including those of Awiqli.
During the 26-week treatment periods with ADA sampling conducted up to 31 weeks in three clinical trials in adults with Type 2 diabetes mellitus [see Clinical Studies (14)], 4/243 (1.6%) (insulin naïve) and 131/548 (23.9%) (insulin experienced) of Awiqli-treated patients were positive for anti-insulin icodec-abae antibodies at baseline.192/243 (79.0%) (insulin naïve) and 389/550 (70.7%) (insulin experienced) were positive for anti-insulin icodec-abae antibodies at least once during the trials. In these trials, 188/243 (77.4%) (insulin naïve) and 370/550 (67.3%) (insulin experienced) of patients were positive for anti-insulin icodec-abae antibodies cross-reacting with human insulin at least once during the trial.
In vitro neutralizing activity of anti-insulin icodec-abae antibodies on insulin receptor action was tested in follow-up (Week 31) samples from one trial in insulin-naïve Type 2 patients. A total of 178 anti-insulin icodec-abae antibody positive samples were tested and in vitro neutralizing activity was detected in 23/178 (12.9%) of the anti-insulin icodec-abae antibody positive follow-up samples.
The patients with anti-insulin icodec-abae antibodies had increased insulin icodec-abae concentrations (increase in geometric mean ratio up to 1.2 compared to patients who did not develop anti-insulin icodec-abae antibodies). There was no identified clinically significant effect of anti-insulin icodec-abae antibodies on safety or effectiveness of Awiqli over the treatment duration of 26 weeks.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Standard 2-year carcinogenicity studies in animals have not been performed to evaluate the carcinogenic potential of insulin icodec-abae. In a 52-week study including human insulin (NPH insulin) as comparator, Sprague-Dawley rats were dosed subcutaneously with insulin icodec-abae at 3.3, 5 (female only), 6.7 and 10 (male only) U/kg/day, resulting in 4 times (males) and 2 times (females) the human exposure (AUC) when compared to a human subcutaneous dose of 230 U/week. Human insulin was dosed at 6.7 U/kg/day. No treatment-related increases in benign or malignant tumors were recorded in female mammary glands from rats dosed with insulin icodec-abae. Further, no treatment related changes in the occurrence of hyperplastic or neoplastic lesions were seen in other tissues in animals dosed with insulin icodec-abae when compared to vehicle or human insulin.
Genotoxicity testing of insulin icodec-abae was not performed.
In a combined fertility and embryo-fetal study in male and female rats, treatment with insulin icodec-abae up to 17 U/kg/day in males and 10 U/kg/day in females (4 times and comparable to human exposure (AUC) at 230 U/week, respectively) prior to mating and in female rats during gestation had no effect on mating performance and fertility.
-
14 CLINICAL STUDIES
14.1 Overview of Clinical Trials
The efficacy of Awiqli administered once-weekly in adult patients with Type 2 diabetes, used in combination with a mealtime insulin or in combination with common oral anti-diabetic agents and/or GLP-1 receptor agonist, was evaluated in three randomized, open-label, treat-to-target, active-controlled trials and one randomized, double-blind, treat-to-target, active-controlled trial (Trials A, B, C, and D). Blinded continuous glucose monitoring (CGM) was utilized in Trials A, C, and D. Across all four trials, insulin icodec-abae was titrated in 20-unit increments, and the fasting plasma glucose (FPG) glycemic target (based on the mean of the 3 most recent FPG values) was 80-130 mg/dL for both treatment arms.
Type 2 insulin naïve patients and basal-only patients treated with Awiqli achieved statistically significant improvement in glycemic control compared to insulin glargine U-100 or insulin degludec U-100. Type 2 patients with basal-bolus regimen achieved similar glycemic control with Awiqli as those achieved with insulin glargine U-100 or insulin degludec U-100.
14.2 Insulin Naïve Adults with Type 2 Diabetes Mellitus
The efficacy of Awiqli was evaluated in a 52-week randomized, open label, active-controlled, parallel-group, multicenter, multinational, treat-to-target trial that enrolled 984 insulin naïve adult patients with type 2 diabetes mellitus inadequately controlled on one or more oral antidiabetic agents (OADs) or GLP-1 receptor agonist [Trial A (NCT04460885)]. Patients were randomized to Awiqli once-weekly or insulin glargine U-100 once-daily according to the approved labeling. Pre-trial non-insulin anti-diabetic medications were continued as background therapy in both treatment arms throughout the entire trial except for sulfonylureas and glinides, which were discontinued at randomization.
The trial population had the following characteristics: mean age was 59 years; mean duration of diabetes was 12 years; 57% were male, 66% were White, 28% were Asian, 3% were Black or African American, and 11% were Hispanic or Latino ethnicity; and 11% of patients had eGFR <60 mL/min/1.73m2. The mean BMI was approximately 30.1 kg/m2.
Treatment with Awiqli once-weekly for 52 weeks resulted in a statistically significant reduction in HbA1c compared to once daily insulin glargine U-100 with an estimated treatment difference of -0.18% [-0.29%; -0.08%]95%CI (see Table 4).
Treatment with Awiqli once-weekly for 52 weeks also resulted in a statistically significantly longer Time in Range (TIR) (4.27% [1.92; 6.62]95CI, ~ 1 hour per day) compared to once daily insulin glargine U-100 treatment during the last 4 weeks of the trial (Weeks 48 to 52) (see Table 4).
Table 4: Results at Week 52 in Trial A Comparing Awiqli to Insulin Glargine U-100 in Insulin Naïve Adult Patients with Type 2 Diabetes Mellitus on OAD(s) or GLP-1 Receptor Agonist
Awiqli + OAD(s)/GLP-1 Receptor Agonist
Insulin glargine + OAD(s)/GLP-1 Receptor Agonist
N
492
492
HbA1c (%)
Baseline
8.5
8.44
End of trial (LSMean)a,b
6.96
7.15
Change from baseline (LSMean)a,b
-1.51
-1.33
Estimated treatment differencea,b [95%CI]
Awiqli – insulin glargine U-100
-0.18 [-0.29; -0.08]c
Proportion Achieving HbA1c < 7% at Trial End (LSMean)d
56.52%
44.57%
FPG (mg/dL)
Baseline
185.31
185.71
End of trial (LSMean)a,b
125.19
125.43
Change from baseline (LSMean)a,b
-60.32
-60.08
TIR (70-180 mg/dL) (%)
Week 48 to 52 (LSMean)e
71.27
67.00
Estimated treatment differencee [95%CI]
Awiqli – insulin glargine U-100
4.27 [1.92; 6.62]f
Weekly basal insulin dose (Units)
End of trial (LSMean)e,g
214.23
222.39
a Estimated using an ANCOVA with treatment, and region as fixed factors and baseline response as covariate.
b Missing values were imputed by the baseline value adding a random term, using multiple imputation.
There were 2.6% of patients in the Awiqli arm and 2.6% in the glargine arm for whom HbA1c data was missing at Week 52.
c p=0.0004 (two-sided) for superiority, adjusted for multiplicity.
d Estimated using logistic regression with treatment, and region as fixed factors and baseline HbA1c as covariate.e Estimated using an ANOVA with treatment, and region as fixed factors. Missing values were imputed using multiple imputation based on patients in the insulin glargine arm who completed their randomized treatment.
f p<0.001 (two-sided) for superiority, adjusted for multiplicity.
g Dose was log-transformed before analysis.
The efficacy of Awiqli was evaluated in a 26-week randomized, double blinded, active-controlled, parallel-group, multicenter, multinational, treat-to-target trial that enrolled 588 adult insulin-naïve patients with type 2 diabetes mellitus inadequately controlled on one or more oral antidiabetic agents (OADs) or GLP-1 receptor agonist [Trial B (NCT04795531)]. Patients were randomized to Awiqli once-weekly or insulin degludec U-100 once daily according to the approved labeling. Pre-trial non-insulin anti-diabetic medications were continued as background therapy in both treatment arms throughout the entire trial except for sulfonylureas and glinides, which were reduced at randomization by approximately 50% at the discretion of the investigator.
The trial population had the following characteristics: mean age was 58 years; mean duration of diabetes was 11 years; 63% were male; 60% were White, 28% were Asian, 3% were Black or African American, and 28% were Hispanic or Latino ethnicity; and 8% of patients had eGFR <60 mL/min/1.73m2. The mean BMI was approximately 29.6 kg/m2.
Treatment with Awiqli once-weekly for 26 weeks resulted in a statistically significant reduction in HbA1c compared to once daily insulin degludec U-100 with an estimated treatment difference of -0.22 [-0.35; -0.09]95%CI. (See Table 5).
Table 5: Results at Week 26 in Trial B Comparing Awiqli to Insulin Degludec U-100 in Insulin Naïve Adult Patients with Type 2 Diabetes Mellitus on OAD(s) or GLP-1 Receptor Agonist
Awiqli + OAD(s)/GLP-1 RA
Insulin degludec + OAD(s)/GLP-1 Receptor Agonist
N
294
294
HbA1c (%)
Baseline
8.55
8.48
End of trial (LS Mean)a,b,*
6.96
7.18
Change from baseline (LS Mean)a,b
-1.56
-1.34
Estimated treatment differencea,b [95%CI]
Awiqli – insulin degludec U-100
-0.22 [-0.35; -0.09]c
Proportion Achieving HbA1c < 7% at Trial End (LS Mean)d
56.45%
40.95%
FPG (mg/dL)
Baseline
186.78
176.20
End of trial (LS Mean)a,b
127.16
127.53
Change from baseline (LS Mean)a,b
-54.28
-53.9
Weekly basal insulin dose (Units)
End of trial (LS Mean)e
204.28
186.52
a Estimated using an ANCOVA with treatment, SU or glinide use (yes/no), and region as fixed factors and baseline response as covariate.
b Missing values were imputed by the baseline value adding a random term, using multiple imputation.
There were 3.7% of patients in the Awiqli arm and 3.1% in the degludec arm for whom HbA1c data was missing at Week 26.
c p=0.0007 (two-sided) for superiority, adjusted for multiplicity.
d Estimated using logistic regression with treatment, SU or glinides use (yes/no) and region as fixed factors and baseline HbA1c as covariate.e Estimated using an ANOVA with treatment, SU or glinide use (yes/no), and region as fixed factors. Dose was log-transformed before analysis. Missing values were imputed using multiple imputation based on patients in the insulin glargine arm who completed their randomized treatment.
14.3 Adults with Type 2 Diabetes Mellitus Previously Treated with Basal Insulin
The efficacy of Awiqli was evaluated in a 26-week randomized, open label, active-controlled, parallel-group, multicenter, multinational, treat-to-target trial in 526 adult patients with Type 2 diabetes mellitus treated with once or twice daily basal insulin with or without OADs [Trial C (NCT04770532)]. Patients were randomized to Awiqli once-weekly or insulin degludec U-100 once daily according to the approved labeling. Pre-trial non-insulin OADs/GLP-1 receptor agonist were continued as background therapy in both treatment arms throughout the entire trial except for sulfonylureas and glinides, which were discontinued at randomization.
The trial population had the following characteristics: mean age was 62 years; mean duration of diabetes was 17 years; 57% were male, 57% were White, 4% were Black or African American, 37% were Asian, and 6% were Hispanic or Latino ethnicity; and 15% of patients had eGFR <60 mL/min/1.73m2. The mean BMI was approximately 29.6 kg/m2.
Treatment with Awiqli once-weekly for 26 weeks resulted in a statistically significant reduction in HbA1c compared with insulin degludec U-100 with estimated treatment difference of -0.19[-0.32; -0.06]95%CI (see Table6).
Estimated time in Range (70-180 mg/dL) during the last 4 weeks of the trial (Week 22 to 26) was 62.34% for Awiqli and 59.93% for insulin degludec U-100.
Table 6: Results at Week 26 in Trial C Comparing Awiqli to Insulin Degludec U-100 in Adult Patients with Type 2 Diabetes Mellitus Previously Treated with Basal Insulin
Awiqli ± OAD(s)/GLP-1 Receptor Agonist
Insulin degludec ± OAD(s)/GLP-1 Receptor Agonist
N
263
263
HbA1c (%)
Baseline
8.17
8.1
End of trial (LS Mean)a,b
7.23
7.43
Change from baseline (LS Mean)a,b
-0.90
-0.71
Estimated treatment difference a,b[95%CI]
Awiqli – insulin degludec U-100
-0.19 [-0.32; -0.06]c
Proportion (%) Achieving HbA1c < 7% at Trial End (LS Mean)d
37.53
25.35
FPG (mg/dL)
Baseline
152.24
150.7
End of trial (LS Mean)a,b
123.01
122.3
Change from baseline (LS Mean)a,b
-28.47
-29.18
Weekly basal insulin dose (Units)
End of trial (LS Mean)e
247.99
262.69
a Estimated using an ANCOVA with treatment, personal CGM device use (yes/no) and region as fixed factors and baseline response as covariate.
b Missing values were imputed by the baseline value adding a random term, using multiple imputation.
There were 2.7% of patients in the Awiqli arm and 3.8% in the degludec arm for whom HbA1c data was missing at Week 26.
c p=0.0032 (two-sided) for superiority, adjusted for multiplicity.
d Estimated using logistic regression with treatment, personal CGM use (yes/no) and region as fixed factors and baseline HbA1c as covariate.e Estimated using an ANOVA with treatment, personal CGM device use (yes/no), and region as fixed factors. Dose was log-transformed before analysis. Missing values were imputed using multiple imputation based on patients in the degludec arm who completed their randomized treatment.
The efficacy of Awiqli was evaluated in a 26-week randomized, open-label, active-controlled, parallel-group, multicenter, multinational, treat-to-target trial in 582 adult patients with type 2 diabetes mellitus inadequately controlled on once-daily basal insulin in combination with mealtime rapid-acting insulin with or without oral antidiabetic agents (OADs) or GLP-1 receptor agonist [Trial D (NCT04880850)]. Patients were randomized to Awiqli once-weekly or insulin glargine U-100 once daily according to the approved labeling both in combination with insulin aspart before each meal. Pre-trial non-insulin OADs or GLP-1 receptor agonist were continued as background therapy in both treatment arms throughout the entire trial except for sulfonylureas and glinides, which were discontinued at randomization.
The trial population had the following characteristics: mean age was 60 years; mean duration of diabetes was 17 years; 52% were male; 64% were White, 32% were Asian, 4% Black or African American, and 18% were Hispanic; and 16% of patients had eGFR <60 mL/min/1.73m2. The mean BMI was approximately 30.3 kg/m2.
At Week 26, the difference in HbA1c reduction from baseline between Awiqli and insulin glargine U-100 was 0.02% with a 95% confidence interval of [-0.11; 0.15] and met the pre-specified non-inferiority margin (0.3%) (see Table7).
Estimated time in Range (70-180 mg/dL) during the last 4 weeks of the trial (Weeks 22 to 26) was 66.75% for the Awiqli arm and 66.46% for insulin glargine U-100.
Table 7: Results at Week 26 in Trial D Comparing Awiqli to Insulin Glargine U-100 in Adult Patients with Type 2 Diabetes Mellitus Receiving Insulin Aspart at Mealtimes with or without OADs or GLP-1 Receptor Agonist
Awiqli + Insulin aspart
± OAD(s)/GLP-1 Receptor Agonist
Insulin glargine + Insulin aspart
± OAD(s)/GLP-1 Receptor Agonist
N
291
291
HbA1c (%)
Baseline
8.29
8.31
End of trial (LS Mean)a,b
7.14
7.12
Change from baseline LS Mean)a, b
-1.16
-1.18
Estimated treatment differencea, b [95%CI]
Awiqli – insulin glargine U-100
0.02 [-0.11; 0.15]
Proportion Achieving HbA1c <7% at Trial End (LS Mean)c
40.69
45.48
FPG (mg/dL)
Baseline
166.59
173.05
End of trial (LS Mean)a,b
138.28
140.76
Change from baseline LS Mean)a,b
-31.54
-29.06
Weekly basal insulin dose (Units)
End of trial (LS Mean)d
321.90
283.46
Weekly bolus insulin dose (Units)
End of trial (LS Mean)d
197.54
260.61
a Estimated using an ANCOVA with treatment, personal CGM device use (yes/no) and region as fixed factors and baseline response as covariate.
b Missing values were imputed by the baseline value adding a random term, using multiple imputation.
There were 5.5% of patients in the Awiqli arm and 9.3% in the glargine arm for whom HbA1c data was missing at Week 26.
c Estimated using logistic regression with treatment, personal CGM use (yes/no) and region as fixed factors and baseline HbA1c as covariate.d Estimated using an ANOVA with treatment, personal CGM device use (yes/no), and region as fixed factors. Dose was log-transformed before analysis. Missing values were imputed using multiple imputation based on patients in the glargine arm who completed their randomized treatment.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Awiqli (insulin icodec-abae) injection is a 700 units/mL (U-700) clear and colorless solution available as follows:
Table 8: Presentations of Awiqli FlexTouch
Awiqli Presentation
NDC number
Total units
Max dose per Injection (units)
Dose Increment
(units)Package Size
3 mL single-patient-use FlexTouch Pen
0169-3121-13
2,100
700
10
1 pen/pack with 13 disposable needles
Trade only
1.5 mL single-patient-use FlexTouch Pen
0169-3105-11
1,050
700
10
1 pen/pack with 13 disposable needles
Trade only
1 mL single-patient-use FlexTouch Pen
0169-3170-97
700
700
10
1 pen/pack with 9 disposable needles
Sample only
Awiqli U-700 FlexTouch pen dials in 10 unit increments.
Dispense in this sealed carton with the enclosed Instructions for Use.
Storage and Handling
Dispense in the original sealed carton with the enclosed Instructions for Use.
Do not store in the freezer or directly adjacent to the refrigerator cooling element. Do not freeze. Do not use Awiqli if it has been frozen.
The storage conditions are summarized in Table 9:
Table 9: Storage Conditions for 3 mL, 1.5 mL, and 1 mL Single-patient-use Awiqli FlexTouch Pen
Not in-use (unopened)
In-use (opened)
Refrigerated
36ºF to 46ºF(2ºC to 8ºC)
Room Temperature
below 86°F (30°C)*Room Temperature
below 86°F (30°C)*Refrigerated
36ºF to 46ºF
(2ºC to 8ºC)
Awiqli FlexTouch
Until expiration date
12 Weeks
- 12 Weeks
12 Weeks
* The total time at room temperature cannot exceed more than 12 Weeks including in-use time.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Hypoglycemia Due to Medication Errors and Accidental Overdose
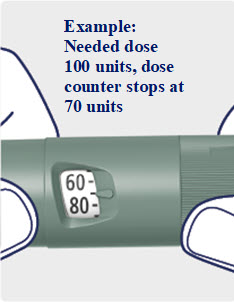
Advise patients to always check the product label before each injection to confirm they are using Awiqli and not another insulin or injectable antidiabetic medicine. Advise patients using other injectable medications for glycemic control that the dosage selection of Awiqli differs. Instruct patients to visually verify the dialed units on the dose counter of the Awiqli FlexTouch prefilled pen before each injection to avoid dosing errors [see Warnings and Precautions (5.1)].
Hypoglycemia
Inform patients that hypoglycemia is the most common adverse reaction with insulin. Inform patients of the symptoms of hypoglycemia (e.g., impaired ability to concentrate and react). This may present a risk in situations where these abilities are especially important, such as driving or operating other machinery. Advise patients who have frequent hypoglycemia or reduced or absent warning signs of hypoglycemia to use caution when driving or operating machinery [see Warnings and Precautions (5.2)].
Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen
Advise patients that changes in insulin regimen can predispose to hyperglycemia or hypoglycemia and that changes in insulin regimen should be made under close medical supervision [see Warnings and Precautions (5.3)].
Hypersensitivity Reactions
Advise patients that hypersensitivity reactions have occurred with Awiqli. Inform patients on the symptoms of hypersensitivity reactions [see Warnings and Precautions (5.4)].
Never Share an Awiqli FlexTouch Pen or Needle Between Patients
Awiqli FlexTouch disposable prefilled pens should never be shared between patients, even if the needle is changed. Sharing poses a risk for transmission of blood-borne pathogens [see Warnings and Precautions (5.6)].
Manufactured by:
Novo Nordisk Inc.
800 Scudders Mill Rd.
Plainsboro, NJ 08536
U.S. License Number 1261
At: Novo Nordisk A/S
Novo Allé
2880 Bagsvaerd
Denmark
www.awiqli.com
For information about Awiqli contact:
Novo Nordisk Inc.
800 Scudders Mill Road
Plainsboro, NJ 08536
1-844-668-6463
Novo Nordisk®, Awiqli®, FlexTouch® are registered trademarks of Novo Nordisk A/S.
© 2026 Novo Nordisk
Patent Information: http://novonordisk-us.com/products/product-patents.html
-
Patient Package Insert
PATIENT INFORMATION
Awiqli® FlexTouch® [Ah–wik-lee]
(insulin icodec-abae)
injection, for subcutaneous use
Do not share your Awiqli FlexTouch pen or needles with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Make sure you use the right type and dose of insulin. Always check the label on your insulin pen before each injection to avoid mix-ups with Awiqli and other insulin products or injectable medicines used to treat diabetes. If you use another injectable medicine to treat your diabetes, pay close attention to how you select your Awiqli dosage. The dosage of Awiqli is different from other injectable medicines used to treat diabetes.
Always make sure that you select the correct dosage of your Awiqli FlexTouch pen as prescribed by your healthcare provider, to avoid dosing errors and accidental overdose. People who are blind or have vision problems should not use this pen without help from a person trained to use the pen.
Do not dial the maximum single dosage (700 units) of your Awiqli FlexTouch Pen, unless prescribed by your healthcare provider.
Do not use a syringe to withdraw Awiqli from your pen.
Talk to your healthcare provider if you have any questions about how to correctly dose Awiqli FlexTouch.
What is Awiqli?
- Awiqli is a long-acting man-made insulin (U-700) that is used to control high blood sugar in adults with type 2 diabetes mellitus.
- It is not known if Awiqli is safe and effective in children and adolescents.
- Awiqli FlexTouch is only available in one concentration (U-700):
- o The Awiqli FlexTouch pen can be injected from 10 units to 700 units in a single injection, in 10 unit increments.
Who should not take Awiqli?
Do not take Awiqli if you:
- are having an episode of low blood sugar (hypoglycemia).
- have an allergy to Awiqli or any of the ingredients in Awiqli. See the end of this Patient Information leaflet for a complete list of ingredients in Awiqli.
Before taking Awiqli, tell your healthcare provider about all your medical conditions including, if you are:
- have liver or kidney problems.
- take other medicines, especially ones called TZDs (thiazolidinediones).
- have heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with Awiqli.
- pregnant, planning to become pregnant, or are breastfeeding.
- taking new prescription or over-the-counter medicines, vitamins, or herbal supplements.
Before you start taking Awiqli, talk to your healthcare provider about low blood sugar and how to manage it.
How should I take Awiqli FlexTouch?
- Read the Instructions for Use that come with your Awiqli FlexTouch Pen.
- Your healthcare provider should show you how to use Awiqli FlexTouch Pen before you use it for the first time.
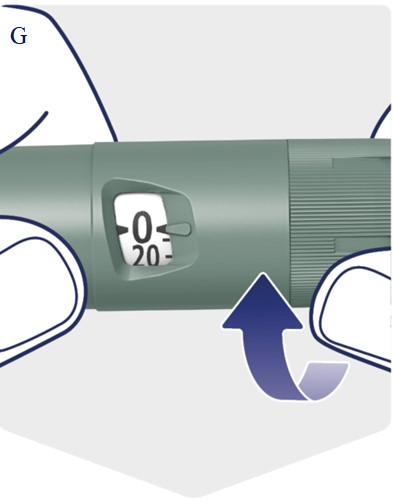
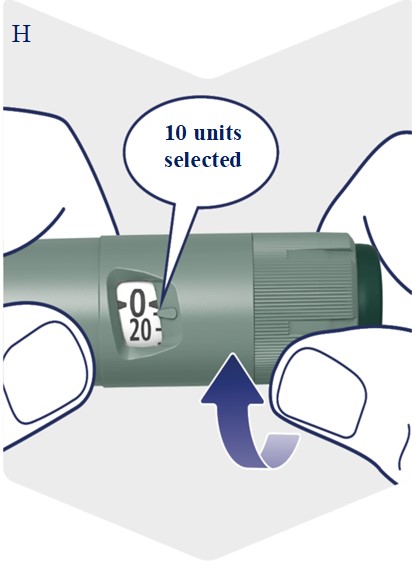
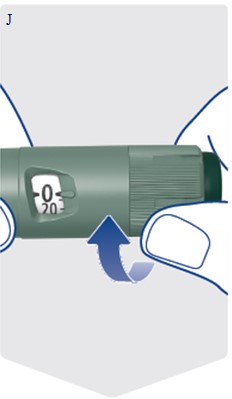
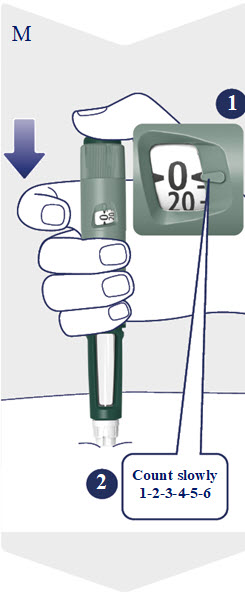
- Take Awiqli exactly as your healthcare provider tells you to. Do not do any conversion of your dose. The dose counter always shows the selected dose in units. Awiqli FlexTouch pens are made to deliver your insulin dose in units.
- Know the type and strength of insulin you take. Do not change the type of insulin you take unless your healthcare provider tells you to. The amount of insulin and the best time for you to take your insulin may need to change if you take different types of insulin.
- Inject Awiqli FlexTouch 1 time each week on any day of the week on the same day each week.
- If you need to change the day of the week, you may do so if your last dose has been at least 4 days.
- If you missed your dose, take your missed dose as soon as possible as long as it has been 4 days or less. Then continue the 1 time each week schedule 1 week from the day your missed dose was taken.
- If more than 4 days has passed, skip the missed dose and take your next Awiqli dose on your regularly scheduled day.
- Check your blood sugar levels. Ask your healthcare provider what your blood sugar should be and when you should check your blood sugar levels.
- Never inject Awiqli FlexTouch into a vein or muscle or use Awiqli in an infusion pump.
- Never use a syringe to remove Awiqli from the FlexTouch pen.
- Do not dilute or mix Awiqli with any other insulin or solution.
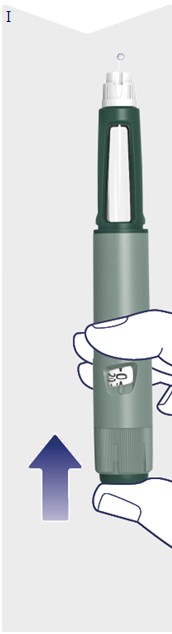
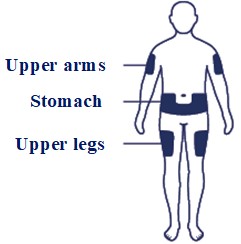
- Awiqli FlexTouch can be injected under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Change (rotate) your injection sites within the area you choose with each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not use the exact same spot for each injection.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
Keep Awiqli and all medicines out of the reach of children.
What should I avoid while taking Awiqli?
While taking Awiqli do not:
- Drive or operate heavy machinery, until you know how Awiqli affects you.
- Drink alcohol or use prescription or over-the-counter medicines that contain alcohol.
What are the possible side effects of Awiqli?
Awiqli may cause serious side effects that can lead to death, including:
- Low blood sugar (hypoglycemia). Signs and symptoms that may indicate low blood sugar include:
- o dizziness or light-headedness
- o blurred vision
- o anxiety, irritability, or mood changes
- o sweating
- o slurred speech
- o hunger
- o confusion
- o shakiness
- o headache
- o fast heartbeat
- Severe allergic reaction (whole body reaction). Stop using Awiqli and get medical help right away, if you have any of these signs or symptoms of a severe allergic reaction:
- o a rash over your whole body, trouble breathing, a fast heartbeat, or sweating.
- Low potassium in your blood (hypokalemia).
- Heart failure. Taking certain diabetes pills called thiazolidinediones or “TZDs” with Awiqli may cause heart failure in some people. This can happen even if you have never had heart failure or heart problems before. If you already have heart failure, it may get worse while you take TZDs with Awiqli. Your healthcare provider should monitor you closely while you are taking TZDs with Awiqli. Tell your healthcare provider if you have any new or worse symptoms of heart failure including shortness of breath, tiredness, swelling of your ankles or feet and sudden weight gain. Treatment with TZDs and Awiqli may need to be adjusted or stopped by your healthcare provider if you have new or worse heart failure.
Your insulin dose may need to change because of:
- change in level of physical activity or exercise
- increased stress
- change in diet
- weight gain or loss
- illness
Common side effects of Awiqli may include:
- hypoglycemia, serious allergic reactions (whole body reactions), reactions at the injection site, skin thickening or pits at the injection site (lipodystrophy), itching, rash, swelling of your hands and feet, and weight gain.
Get emergency medical help if you have:
- trouble breathing, shortness of breath, fast heartbeat, swelling of your face, tongue, or throat, sweating, extreme drowsiness, dizziness, confusion.
These are not all the possible side effects of Awiqli. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Awiqli.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about Awiqli that is written for health professionals. Do not use Awiqli FlexTouch for a condition for which it was not prescribed. Do not give Awiqli to other people, even if they have the same symptoms that you have. It may harm them.
What are the ingredients in Awiqli?
Active Ingredient: insulin icodec-abae
Inactive Ingredients: Glycerin; metacresol; phenol; sodium chloride; zinc acetate and water for injection. Hydrochloric acid or sodium hydroxide may be added to adjust pH.
Manufactured by:
Novo Nordisk Inc.
800 Scudders Mill Road
Plainsboro, NJ 08536
U.S. License No. 1261
At: Novo Nordisk A/S
Novo Allé
2880 Bagsvaerd
Denmark
For more information, go to www.awiqli.com or call 1-844-668-6463.
Patent Information: http://novonordisk-us.com/products/product-patents.html
© 2026 Novo Nordisk
- This Patient Information has been approved by the U.S. Food and Drug Administration.Issued: 03/2026
-
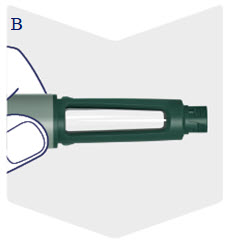
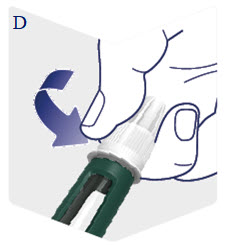
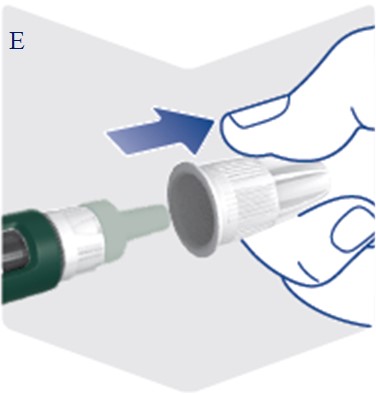
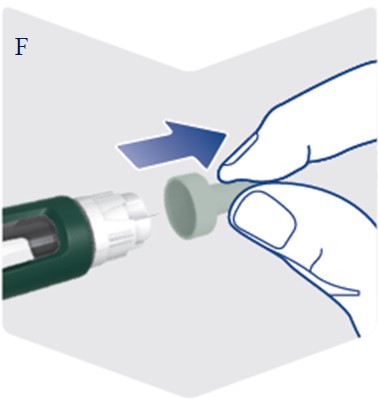
Instructions for Use
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 1 mL Sample Carton
- Awiqli® FlexTouch® NDC 0169- 3170 -97
- (insulin icodec-abae) injection List: 317097
- 700 units/mL (U-700) Use Once Weekly
For Single-Patient-Use Only
- For subcutaneous use Pen delivers doses in 10 unit increments only
- Rx only
Dispense in this sealed carton.
Sample not for resale
Contains: 1 x 1 mL Prefilled Pen, 9 disposable needles
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 1.5 mL Carton
- Awiqli® FlexTouch® NDC 0169- 3105 -11
- (insulin icodec-abae) injection List: 310511
- 700 units/mL (U-700) Use Once Weekly
For Single-Patient-Use Only
- For subcutaneous use Pen delivers doses in 10 unit increments only
- Rx only
Dispense in this sealed carton.
Contains: 1 x 1.5 mL Prefilled Pen, 13 disposable needles
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 3 mL Carton
- Awiqli® FlexTouch® NDC 0169- 3121 -13
- (insulin icodec-abae) injection List: 312113
- 700 units/mL (U-700) Use Once Weekly
For Single-Patient-Use Only
- For subcutaneous use Pen delivers doses in 10 unit increments only
- Rx only
Dispense in this sealed carton.
Contains: 1 x 3 mL Prefilled Pen, 13 disposable needles
-
INGREDIENTS AND APPEARANCE
AWIQLI
insulin icodec-abae injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0169-3170 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Insulin Icodec (UNII: P7YU3ED05N) (Insulin Icodec - UNII:P7YU3ED05N) Insulin Icodec 700 U in 1 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) 15 mg in 1 mL Metacresol (UNII: GGO4Y809LO) 1.08 mg in 1 mL Phenol (UNII: 339NCG44TV) 5.65 mg in 1 mL Zinc Acetate (UNII: FM5526K07A) 101 ug in 1 mL Sodium Chloride (UNII: 451W47IQ8X) 1.17 mg in 1 mL Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0169-3170-97 1 in 1 CARTON 03/26/2026 1 NDC: 0169-3170-90 1 mL in 1 SYRINGE; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761326 03/26/2026 AWIQLI
insulin icodec-abae injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0169-3105 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Insulin Icodec (UNII: P7YU3ED05N) (Insulin Icodec - UNII:P7YU3ED05N) Insulin Icodec 700 U in 1 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) 15 mg in 1 mL Metacresol (UNII: GGO4Y809LO) 1.08 mg in 1 mL Phenol (UNII: 339NCG44TV) 5.65 mg in 1 mL Zinc Acetate (UNII: FM5526K07A) 101 ug in 1 mL Sodium Chloride (UNII: 451W47IQ8X) 1.17 mg in 1 mL Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0169-3105-11 1 in 1 CARTON 03/26/2026 1 NDC: 0169-3105-01 1.5 mL in 1 SYRINGE; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761326 03/26/2026 AWIQLI
insulin icodec-abae injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0169-3121 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Insulin Icodec (UNII: P7YU3ED05N) (Insulin Icodec - UNII:P7YU3ED05N) Insulin Icodec 700 U in 1 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) 15 mg in 1 mL Metacresol (UNII: GGO4Y809LO) 1.08 mg in 1 mL Phenol (UNII: 339NCG44TV) 5.65 mg in 1 mL Zinc Acetate (UNII: FM5526K07A) 101 ug in 1 mL Sodium Chloride (UNII: 451W47IQ8X) 1.17 mg in 1 mL Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0169-3121-13 1 in 1 CARTON 03/26/2026 1 NDC: 0169-3121-01 3 mL in 1 SYRINGE; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761326 03/26/2026 Labeler - Novo Nordisk (622920320)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.