IBUPROFEN capsule, liquid filled
IBUPROFEN by
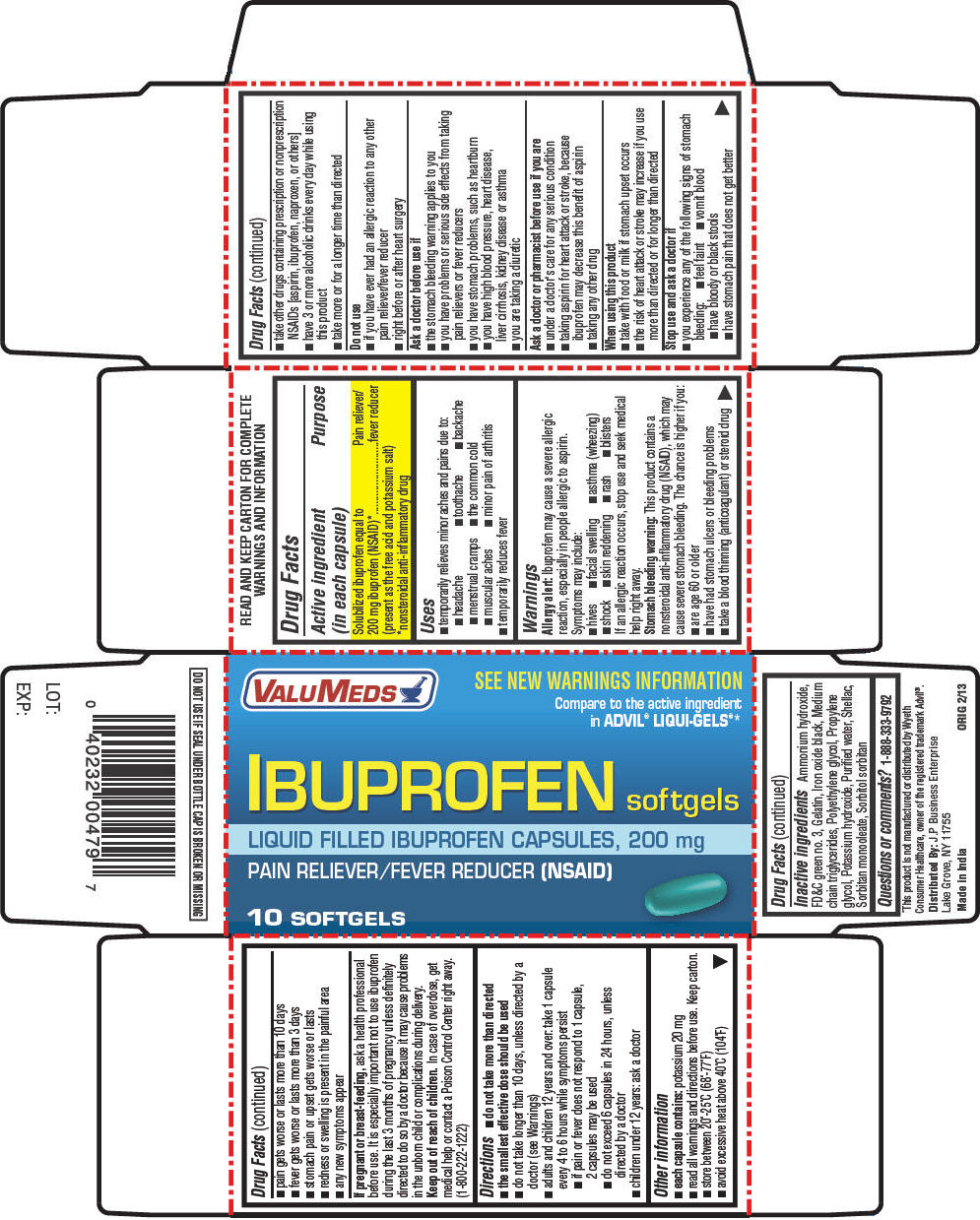
Drug Labeling and Warnings
IBUPROFEN by is a Otc medication manufactured, distributed, or labeled by J.P BUSINESS ENTERPRISE. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each capsule)
- Purpose
- Uses
-
Warnings
Allergy alert
Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
- skin reddening
- rash
- blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning
This product contains a nonsteroidal anti-inflammatory drug (NSAID), which may cause severe stomach bleeding. The chance is higher if you:
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Do not use
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- right before or after heart surgery
Ask a doctor before use if
- the stomach bleeding warning applies to you
- you have problems or serious side effects from taking pain relievers or fever reducers
- you have stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease or asthma
- you are taking a diuretic
Ask a doctor or pharmacist before use if you are
- under a doctor's care for any serious condition
- taking aspirin for heart attack or stroke, because ibuprofen may decrease this benefit of aspirin
- taking any other drug
When using this product
- take with food or milk if stomach upset occurs
- the risk of heart attack or stroke may increase if you use more than directed or for longer than directed
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- stomach pain or upset gets worse or lasts
- redness or swelling is present in the painful area
- any new symptoms appear
-
Directions
- do not take more than directed
- the smallest effective dose should be used
- do not take longer than 10 days, unless directed by a doctor (see Warnings)
- adults and children 12 years and over: take 1 capsule every 4 to 6 hours while symptoms persist
- if pain or fever does not respond to 1 capsule, 2 capsules may be used
- do not exceed 6 capsules in 24 hours, unless directed by a doctor
- children under 12 years: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 10 Softgel Bottle Carton
-
INGREDIENTS AND APPEARANCE
IBUPROFEN
ibuprofen capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 59105-002 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 200 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) GELATIN (UNII: 2G86QN327L) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) FERROSOFERRIC OXIDE (UNII: XM0M87F357) WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) Product Characteristics Color GREEN Score no score Shape OVAL Size 18mm Flavor Imprint Code 133 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59105-002-10 1 in 1 CARTON 1 10 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA079205 08/20/2013 Labeler - J.P BUSINESS ENTERPRISE (078775890)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.