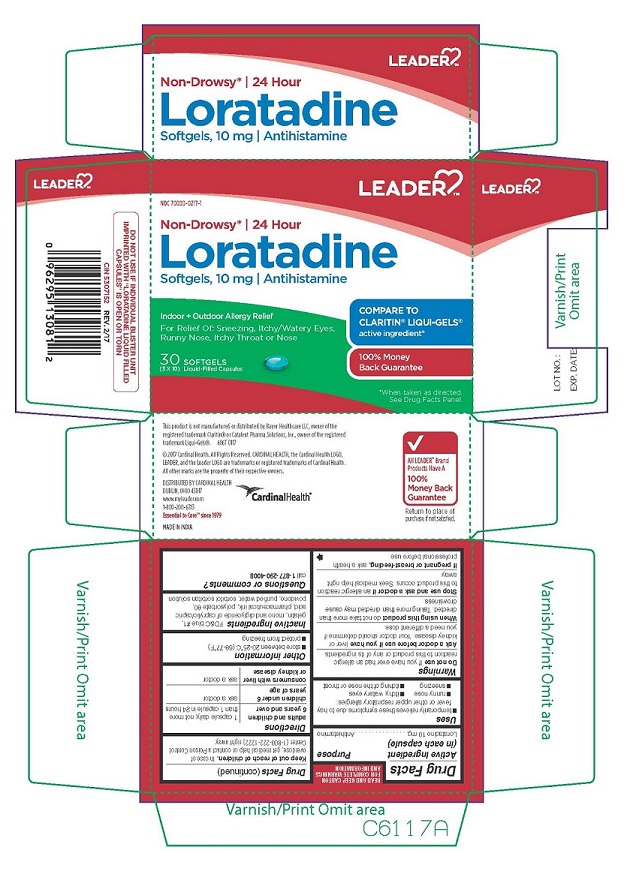
LORATADINE by Cardinal Health / TIME CAP LABORATORIES, INC / MARKSANS PHARMA LIMITED LEADER 686T
LORATADINE by
Drug Labeling and Warnings
LORATADINE by is a Otc medication manufactured, distributed, or labeled by Cardinal Health, TIME CAP LABORATORIES, INC, MARKSANS PHARMA LIMITED. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LORATADINE- loratadine capsule, liquid filled
Cardinal Health
----------
LEADER 686T
|
DO NOT USE IF YOU HAVE EVER HAD AN ALLERGIC REACTION TO THIS PRODUCT OR ANY OF ITS INGREDIENTS. WHEN USING THIS PRODUCT DO NOT TAKE MORE THAN DIRECTED. TAKING MORE THAN DIRECTED MAY CAUSE DROWSINESS. STOP USE AND ASK A DOCTOR IF AN ALLERGIC REACTION TO THIS PRODUCT OCCURS. SEEK MEDICAL HELP RIGHT AWAY. IF PREGNANT OR BREAST FEEDING, ASK A HEALTH PROFESSIONAL BEFORE USE. |
|
TEMPORARILY RELIEVES THESE SYMPTOMS DUE TO HAY FEVER OR OTHR UPPER RESPIRATORY ALLERGIES: RUNNY NOSE ITCHY, WATERY EYES SNEEZING ITCHING OF THE NOSE OR THROAT |
|
ADULTS AND CHILDREN 6 YEARS AND OVER: 1 CAPSULE DAILY; NOT MORE THAN 1 CAPSULE IN 24 HOURS. CHILDREN UNDER 6 YEARS OF AGE: ASK A DOCTOR. CONSUMERS WITH LIVER OR KIDNEY DISEASE: ASK A DOCTOR. |
| LORATADINE
loratadine capsule, liquid filled |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Cardinal Health (097537435) |
| Registrant - TIME CAP LABORATORIES, INC (037052099) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| MARKSANS PHARMA LIMITED | 925822975 | manufacture(70000-0217) | |