These highlights do not include all the information needed to use VABOMERE safely and effectively. See full prescribing information forVABOMERE. VABOMERE™ (meropenem and vaborbactam) for injection, for intravenous use Initial U.S. Approval: 2017
VABOMERE by
Drug Labeling and Warnings
VABOMERE by is a Prescription medication manufactured, distributed, or labeled by The Medicines Company, Facta Farmaceutici S.p.A.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
VABOMERE- meropenem-vaborbactam injection, powder, for solution
The Medicines Company
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use VABOMERE safely and effectively. See full prescribing information forVABOMERE.
VABOMERE™ (meropenem and vaborbactam) for injection, for intravenous use Initial U.S. Approval: 2017 INDICATIONS AND USAGEVABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. ( 1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2) DOSAGE AND ADMINISTRATION
a As calculated using the Modification of Diet in Renal Disease (MDRD) formula;
b All doses of VABOMERE are administered intravenously over 3 hours;
c Doses adjusted for renal impairment should be administered after a hemodialysis session;
d The total duration of treatment is for up to 14 days.
DOSAGE FORMS AND STRENGTHSVABOMERE 2 grams (meropenem and vaborbactam) for injection, is supplied as a sterile powder for constitution in single‑dose vials containing meropenem 1 gram (equivalent to 1.14 grams of meropenem trihydrate) and vaborbactam 1 gram. ( 3) CONTRAINDICATIONSKnown hypersensitivity to the components of VABOMERE (meropenem and vaborbactam) or anaphylactic reactions to beta‑lactams. ( 4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most frequently reported adverse reactions occurring in ≥3% of patients treated with VABOMERE were headache, phlebitis/infusion site reactions, and diarrhea. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact The Medicines Company at 1-888-977-6326 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION Revised: 8/2017 |
FULL PRESCRIBING INFORMATION
1. INDICATIONS AND USAGE
1.1 Complicated Urinary Tract Infections (cUTI), including Pyelonephritis
VABOMERE™ is indicated for the treatment of patients 18 years of age and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by the following susceptible microorganisms: Escherichia coli, Klebsiella pneumoniae, and Enterobacter cloacae species complex.
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage of VABOMERE is 4 grams (meropenem 2 grams and vaborbactam 2 grams) administered every 8 hours by intravenous (IV) infusion over 3 hours in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) greater than or equal to 50 mL/min/1.73m 2. The duration of treatment is for up to 14 days.
2.2 Dosage Adjustments in Patients with Renal Impairment
Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/1.73m 2. The recommended dosage of VABOMERE in patients with varying degrees of renal function is presented in Table 1. For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly [see Use in Specific Populations (8.6) and Clinical Pharmacology ( 12.3)] .
Meropenem and vaborbactam are removed by hemodialysis [see Clinical Pharmacology ( 12.3)] . For patients maintained on hemodialysis, administer VABOMERE after a hemodialysis session
|
eGFR a (mL/min/ 1.73m2) |
Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) b, c, d | Dosing Interval |
| 30 to 49 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 8 hours |
| 15 to 29 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 12 hours |
| Less than 15 |
VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) | Every 12 hours |
a As calculated using the Modification of Diet in Renal Disease (MDRD) formula as follows: eGFR (mL/min/1.73m 2) = 175 x (serum creatinine) -1.154 x (age) -0.203x (0.742 if female) x (1.212 if African American)
b All doses of VABOMERE are administered intravenously over 3 hours.
c Doses adjusted for renal impairment should be administered after a hemodialysis session.
d The total duration of treatment is for up to 14 days.
2.3 Preparation and Administration of VABOMERE for Intravenous Infusion
Preparation
VABOMERE is supplied as a dry powder in a single-dose vial that must be constituted and further diluted prior to intravenous infusion as outlined below. VABOMERE does not contain preservatives. Aseptic technique must be used for constitution and dilution.
1. To prepare the required dose for intravenous infusion, constitute the appropriate number of vials, as determined from below. Withdraw 20 mL of 0.9% Sodium Chloride Injection, USP, from an infusion bag and constitute each vial of VABOMERE.
2. Mix gently to dissolve. The constituted VABOMERE solution will have an approximate meropenem concentration of 0.05 gram/mL and an approximate vaborbactam concentration of 0.05 gram/mL. The final volume is approximately 21.3 mL. The constituted solution is not for direct injection.
3. The constituted solution must be diluted further, immediately, in a 0.9% Sodium Chloride Injection, USP infusion bag before intravenous infusion. The intravenous infusion of the diluted solution must be completed within 4 hours if stored at room temperature or 22 hours if stored refrigerated at 2°C to 8°C (36°F to 46°F).
4. To dilute the constituted solution, withdraw the full or partial constituted vial contents from each vial and add it back into the infusion bag in accordance with Table 2 below. After dilution, the final infusion concentration of meropenem and vaborbactam will be approximately between 2 mg/mL and 8 mg/mL.
| VABOMERE (meropenem and vaborbactam) Dose | Number of Vials to Constitute for Further Dilution | Volume to Withdraw from Each Constituted Vial for Further Dilution | Volume of Infusion Bag |
| 4 grams (2 grams-2 grams) | 2 vials | Entire contents (approximately 21 mL) | 250-1000 mL |
| 2 grams (1 gram-1 gram) | 1 vial | Entire contents (approximately 21 mL) | 125-500 mL |
| 1 gram (0.5 gram-0.5 gram) |
1 vial | 10.5 mL (discard unused portion) | 70-250 mL |
5. Visually inspect the diluted VABOMERE solution for particulate matter and discoloration prior to administration (the color of the VABOMERE infusion solution for administration ranges from colorless to light yellow). Discard unused portion after use.
3 DOSAGE FORMS AND STRENGTHS
VABOMERE 2 grams (meropenem and vaborbactam) for injection, is supplied as a white to light yellow sterile powder for constitution in single‑dose, clear glass vials containing meropenem 1 gram (equivalent to 1.14 grams meropenem trihydrate) and vaborbactam 1 gram.
4 CONTRAINDICATIONS
VABOMERE is contraindicated in patients with known hypersensitivity to any components of VABOMERE (meropenem and vaborbactam), or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactam antibacterial drugs [see Warnings and Precautions ( 5.1)] .
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Hypersensitivity reactions were reported in patients treated with VABOMERE in the clinical trials [see Adverse Reactions ( 6.1)] . Serious and occasionally fatal hypersensitivity (anaphylactic) reactions and serious skin reactions have been reported in patients receiving therapy with beta-lactam antibacterial drugs. These reactions are more likely to occur in individuals with a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another beta-lactam antibacterial drug. Before initiating therapy with VABOMERE, it is important to inquire about previous hypersensitivity reactions to penicillins, cephalosporins, other beta-lactam antibacterial drugs, and other allergens. If an allergic reaction to VABOMERE occurs, discontinue the drug immediately.
5.2 Seizure Potential
Seizures and other adverse Central Nervous System (CNS) experiences have been reported during treatment with meropenem, which is a component of VABOMERE. These experiences have occurred most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) or with bacterial meningitis and/or compromised renal function [see Adverse Reactions ( 6.1) and Drug Interactions ( 7.1)] .
Close adherence to the recommended dosage regimens is urged, especially in patients with known factors that predispose to convulsive activity. Continue anti-convulsant therapy in patients with known seizure disorders. If focal tremors, myoclonus, or seizures occur, evaluate neurologically, place on anti-convulsant therapy if not already instituted, and reexamine the dosage of VABOMERE to determine whether it should be decreased or discontinued.
5.3 Clostridium difficile-associated Diarrhea
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including VABOMERE, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of
C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin‑producing isolates of
C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.4 Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid
The concomitant use of VABOMERE and valproic acid or divalproex sodium is generally not recommended. Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. Consider administration of antibacterial drugs other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of VABOMERE is necessary, consider supplemental anticonvulsant therapy [see Drug Interactions ( 7.1)] .
5.5 Thrombocytopenia
In patients with renal impairment, thrombocytopenia has been observed in patients treated with meropenem, but no clinical bleeding has been reported [see Dosage and Administration ( 2.2), Adverse Reactions ( 6.1), Use in Specific Populations ( 8.5) and ( 8.6), and Clinical Pharmacology ( 12.3) ].
5.6 Potential for Neuromotor Impairment
Alert patients receiving VABOMERE on an outpatient basis regarding adverse reactions such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until it is reasonably well established that VABOMERE is well tolerated, advise patients not to operate machinery or motorized vehicles [see Adverse Reactions ( 6.1)].
5.7 Development of Drug-Resistant Bacteria
Prescribing VABOMERE in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of drug-resistant bacteria [see Indications and Usage ( 1.2)] .
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Hypersensitivity Reactions [see Warnings and Precautions ( 5.1)]
- Seizure Potential [see Warnings and Precautions ( 5.2)]
- Clostridium difficile-associated Diarrhea [see Warnings and Precautions ( 5.3)]
- Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid [see Warnings and Precautions ( 5.4)]
- Thrombocytopenia [see Warnings and Precautions ( 5.5)]
- Potential for Neuromotor Impairment [see Warnings and Precautions ( 5.6)]
- Development of Drug-Resistant Bacteria [see Warnings and Precautions ( 5.7)]
- Overgrowth of Nonsusceptible Organisms [see Warnings and Precautions ( 5.8)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
VABOMERE was evaluated in a Phase 3 comparator-controlled clinical trial in cUTI, including pyelonephritis, which included 272 patients treated with VABOMERE and 273 patients treated with the comparator piperacillin/tazobactam 4.5 grams (piperacillin 4 g/tazobactam 0.5 g) every 8 hours. After a minimum of 15 doses of IV therapy, patients could be switched to oral levofloxacin (500 mg daily every 24 hours) to complete the treatment course. Mean duration of IV therapy was 8 days in both treatment groups. Mean duration of IV and oral therapy was 10 days; patients with baseline bacteremia could receive up to 14 days of treatment.
The mean age of patients treated with VABOMERE was 53 years (range 18 to 92 years), and 32% of patients were 65 years of age or older. Patients were predominantly female (66.5%) and White (93.4%). Most patients were enrolled in Europe (89.7%).
Serious Adverse Reactions and Adverse Reactions Leading to Discontinuation
Treatment was discontinued due to adverse reactions in 2.9% (8/272) of patients receiving VABOMERE and in 5.1% (14/273) of patients receiving piperacillin/tazobactam. Most common adverse reactions resulting in discontinuation of VABOMERE included hypersensitivity, 1.1% (3/272) and infusion-related reactions, 0.7% (2/272). Death occurred in 2 (0.7%) patients who received VABOMERE and in 2 (0.7%) patients who received piperacillin/tazobactam.
Common Adverse Reactions
The most frequently reported adverse reactions (3% or greater) in patients receiving VABOMERE in the Phase 3 cUTI trial were headache, phlebitis/infusion site reactions, and diarrhea. Table 3 provides adverse reactions occurring in 1% or greater of patients receiving VABOMERE in the Phase 3 cUTI trial.
| Adverse Reactions |
VABOMERE
|
Piperacillin/Tazobactama
|
| Headache | 8.8 | 4.4 |
| Phlebitis/Infusion site reactions b | 4.4 | 0.7 |
| Diarrhea | 3.3 | 4.4 |
| Hypersensitivity | 1.8 | 1.8 |
| Nausea | 1.8 | 1.5 |
| Alanine aminotransferase increased | 1.8 | 0.4 |
| Aspartate aminotransferase increased | 1.5 | 0.7 |
| Pyrexia | 1.5 | 0.7 |
| Hypokalemia | 1.1 | 1.5 |
a Piperacillin/tazobactam 4.5 grams (piperacillin 4 g/tazobactam 0.5 g) IV infused over 30 minutes every 8 hours.
b Infusion site reactions include infusion/injection site phlebitis, infusion site thrombosis, and infusion site erythema.
c Hypersensitivity includes hypersensitivity, drug hypersensitivity, anaphylactic reaction, rash urticaria, and bronchospasm
Adverse Reactions Occurring in Less Than 1% of Patients Receiving VABOMERE in the Phase 3 cUTI trial:
Blood and lymphatic system disorders: leukopenia
General disorders and administration site conditions: chest discomfort
Infections and infestations: pharyngitis, vulvovaginal candidiasis, oral candidiasis
Investigations: creatinine phosphokinase increase
Metabolism and nutrition disorders: decreased appetite, hyperkalemia, hyperglycemia, hypoglycemia
Nervous system disorders: dizziness, tremor, paresthesia, lethargy
Psychiatric disorders: hallucination, insomnia
Renal and urinary disorders: azotemia, renal impairment
Vascular disorders: deep vein thrombosis, hypotension, vascular pain
Other Adverse Reactions Associated with Meropenem
Additionally, adverse reactions reported with meropenem alone that were not reported in VABOMERE-treated patients in the Phase 3 clinical trial are listed below:
Blood and lymphatic system disorders: thrombocytosis, neutropenia, eosinophilia, thrombocytopenia, agranulocytosis, hemolytic anemia
Gastrointestinal disorders: abdominal pain
Hepatobiliary disorders: jaundice
Nervous system disorders: convulsions
Investigations: blood alkaline phosphatase increased, blood lactate dehydrogenase increased, blood bilirubin increased, blood creatinine increased, blood urea increased, blood thromboplastin decreased, prothrombin time decreased, Direct and Indirect Coombs test positive
Skin and subcutaneous tissue disorders: pruritus, toxic epidermal necrolysis, Stevens Johnson syndrome, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome, erythema multiforme
Immune system disorders: angioedema
General disorders and administration site conditions: pain
7 DRUG INTERACTIONS
7.1 Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid’s glucuronide metabolite (VPA‑g) back to valproic acid, thus decreasing the serum concentrations of valproic acid. If administration of VABOMERE is necessary, then supplemental anti‑convulsant therapy should be considered [see Warnings and Precautions ( 5.4)] .
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Fetal malformations were observed in vaborbactam-treated rabbits, therefore advise pregnant women of the potential risks to the fetus. There are insufficient human data to establish whether there is a drug-associated risk of major birth defects or miscarriages with VABOMERE, meropenem, or vaborbactam in pregnant women.
Malformations (supernumerary lung lobes, interventricular septal defect) were observed in offspring from pregnant rabbits administered intravenous vaborbactam during the period of organogenesis at doses approximately equivalent to or above the maximum recommended human dose (MRHD) based on plasma AUC comparison. The clinical relevance of the malformations is uncertain. No similar malformations or fetal toxicity were observed in offspring from pregnant rats administered intravenous vaborbactam during organogenesis or from late pregnancy and through lactation at a dose equivalent to approximately 1.6 times the MRHD based on body surface area comparison [see Data].
No fetal toxicity or malformations were observed in pregnant rats and cynomolgus monkeys administered intravenous meropenem during organogenesis at doses up to 1.6 and 1.2 times the MRHD based on body surface area comparison, respectively. In rats administered intravenous meropenem in late pregnancy and during the lactation period, there were no adverse effects on offspring at doses equivalent to approximately 1.6 times the MRHD based on body surface area comparison [see Data].
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15‑20%, respectively.
Data
Animal Data
Meropenem
Reproductive studies have been performed with meropenem in rats at doses of up to 1000 mg/kg/day and in cynomolgus monkeys at doses of up to 360 mg/kg/day (on the basis of body surface area comparisons, approximately 1.6 times and 1.2 times higher, respectively, than the MRHD of 2 grams every 8 hours). These studies revealed no evidence of harm to the fetus due to meropenem, although there were slight changes in fetal body weight at doses of 250 mg/kg/day (equivalent to approximately 0.4 times the MRHD of 2 grams every 8 hours based on body surface area comparison) and above in rats. In a published study 1, meropenem administered to pregnant rats from Gestation Day 6 to Gestation Day 17, was associated with mild maternal weight loss at all doses, but did not produce malformations or fetal toxicity. The no-observed-adverse-effect-level (NOAEL) for fetal toxicity in this study was considered to be the high dose of 750 mg/kg/day (equivalent to approximately 1.2 times the MRHD based on body surface area comparison).
In a peri-postnatal study in rats described in the published literature 1, intravenous meropenem was administered to dams from Gestation Day 17 until Postpartum Day 21. There were no adverse effects in the dams and no adverse effects in the first generation offspring (including developmental, behavioral, and functional assessments and reproductive parameters) except that female offspring exhibited lowered body weights which continued during gestation and nursing of the second generation offspring. Second generation offspring showed no meropenem-related effects. The NOAEL value was considered to be 1000 mg/kg/day (approximately 1.6 times the MRHD based on body surface area comparisons).
Vaborbactam
In a rat embryo-fetal toxicology study, intravenous administration of vaborbactam during Gestation Days 6‑17 showed no evidence of maternal or embryofetal toxicity at doses up to 1000 mg/kg, which is equivalent to approximately 1.6 times the MRHD based on body surface area comparisons. In the rabbit, intravenous administration of vaborbactam during Gestation Days 7–19 at doses up to 1000 mg/kg/day (approximately 5 times the MRHD based on AUC exposure comparison) was not associated with maternal toxicity or fetal weight loss. A low incidence of malformations occurred in the 300 mg/kg/day mid-dose group (two fetuses from different litters with interventricular septal defects, one fetus with a fused right lung lobe and one fetus with a supernumerary lung lobe), and in the 1000 mg/kg/day high-dose group (two fetuses from different litters with supernumerary lobes). The NOAEL was considered to be 100 mg/kg/day which is equivalent to 0.3 times the MRHD based on plasma AUC exposure comparison and 6-times the MRHD based on maximum plasma concentration (C max) comparison. The clinical relevance of the malformations is uncertain. Vaborbactam C max values may have influenced malformations in the rabbit study, and the recommended 3-hour infusion time for clinical administration of vaborbactam is associated with lower plasma C max values than the 30-minute infusions in rabbits.
In a peri-postnatal study in rats, vaborbactam administered intravenously to pregnant dams from Gestation Day 6 to Lactation Day 20 caused no adverse effects on the dams, or in first and second generation offspring. The NOAEL was considered to be 1000 mg/kg/day (equivalent to approximately 1.6 times the MRHD based on body surface area comparison).
8.2 Lactation
Meropenem has been reported to be excreted in human milk. It is unknown whether vaborbactam is excreted in human milk. No information is available on the effects of meropenem and vaborbactam on the breast-fed child or on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VABOMERE and any potential adverse effects on the breast-fed child from VABOMERE or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of VABOMERE in pediatric patients (younger than 18 years of age) has not been established. Studies of VABOMERE have not been conducted in patients younger than 18 years of age.
8.5 Geriatric Use
Of the 272 patients treated with VABOMERE in the Phase 3 cUTI trial, 48 (18%) patients were 65 years of age and older, while 39 (14%) patients were 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Meropenem, a component of VABOMERE, is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with renal impairment. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Population pharmacokinetic (PK) analysis found no clinically relevant change in pharmacokinetic parameters in elderly patients. No dosage adjustment based on age is required. Dosage adjustment for elderly patients should be based on renal function [see Dosage and Administration ( 2.2) and Clinical Pharmacology ( 12.3)] .
8.6 Renal Impairment
Pharmacokinetic studies conducted with meropenem and vaborbactam in subjects with renal impairment have shown that the plasma exposures of both meropenem and vaborbactam increased with decreasing renal function [see Clinical Pharmacology (12.3)]. Dosage adjustment for VABOMERE is recommended in patients with renal impairment (eGFR less than 50 mL/min/1.73m 2) [see Dosage and Administration ( 2.2)].
For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly. Meropenem and vaborbactam are removed by hemodialysis. Following a single dose of VABOMERE, vaborbactam exposure was substantially greater when VABOMERE was administered after hemodialysis than before hemodialysis [see Clinical Pharmacology ( 12.3)] .
10 OVERDOSAGE
In the event of overdose, discontinue VABOMERE and institute general supportive treatment.
Meropenem and vaborbactam can be removed by hemodialysis. In subjects with end-stage renal disease (ESRD) administered meropenem 1 gram and vaborbactam 1 gram, the mean total recovery in dialysate following a hemodialysis session was 38% and 53% of the administered dose of meropenem and vaborbactam, respectively.
No clinical information is available on the use of hemodialysis to treat VABOMERE overdosage.
11 DESCRIPTION
VABOMERE (meropenem and vaborbactam) for injection is a combination product that contains meropenem, a synthetic penem antibacterial drug and vaborbactam, a cyclic boronic acid beta-lactamase inhibitor, for intravenous administration.
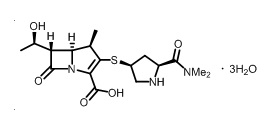
Meropenem, present as a trihydrate, is a white to light yellow crystalline powder, with a molecular weight of 437.52. The chemical name for meropenem trihydrate is (4 R,5 S,6 S)-3-[[(3 S,5 S)-5-(dimethylcarbamoyl)-3-pyrrolidinyl]thio]-6-[(1 R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid, trihydrate. The empirical formula of meropenem trihydrate is C 17H 25N 3O 5S·3H 2O and its chemical structure is:
Figure 1: Structure of Meropenem Trihydrate
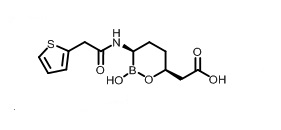
Vaborbactam is a white to off-white powder, with a molecular weight of 297.14. The chemical name for vaborbactam is (3 R,6 S)-2-hydroxy-3-[[2-(2-thienyl)acetyl]amino]-1,2-oxaborinane-6-acetic acid. Its empirical formula is C 12H 16BNO 5S and its chemical structure is:
Figure 2: Structure of Vaborbactam
VABOMERE is supplied as a white to light yellow sterile powder for constitution that contains meropenem trihydrate, vaborbactam, and sodium carbonate. Each 50 mL glass vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate. The total sodium content of the mixture is approximately 0.25 grams (10.9 mEq)/vial.
Each vial is constituted and further diluted with 0.9% Sodium Chloride Injection, USP. Both the constituted solution and the diluted solution for intravenous infusion should be a colorless to light yellow solution [see Dosage and Administration ( 2.3)].
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
Similar to other beta‑lactam antibacterial drugs, the percentage of time of a dosing interval that unbound plasma concentration of meropenem exceeds the meropenem-vaborbactam minimum inhibitory concentration (MIC) against the infecting organism has been shown to best correlate with efficacy in animal and in vitro models of infection. The ratio of the 24-hour unbound plasma vaborbactam AUC to meropenem-vaborbactam MIC is the index that best predicts efficacy of vaborbactam in combination with meropenem in animal and in vitro models of infection.
12.3 Pharmacokinetics
Pharmacokinetic (PK) Parameters
The mean PK parameters of meropenem and vaborbactam in healthy adults with normal renal function after single and multiple 3-hour infusions of VABOMERE 4 grams (meropenem 2 grams and vaborbactam 2 grams) administered every 8 hours are summarized in Table 4.
The PK parameters of meropenem and vaborbactam were similar for single and multiple dose administration of VABOMERE.
| Parameters | Meropenem | Vaborbactam | ||
| Single VABOMERE 4 gram
a Dose
(N=8) | Multiple VABOMERE 4 gram a Doses Administered Every 8 hours for 7 Days (N=8) | Single VABOMERE 4 grama Dose
(N=8) | Multiple VABOMERE 4 gram a Doses Administered Every 8 hours for 7 Days (N=8) | |
| C max (mg/L) | 46.0 (5.7) | 43.4 (8.8) | 50.7 (8.4) | 55.6 (11.0) |
| CL (L/h) | 14.6 (2.7) | 15.1 (2.8) | 12.3 (2.2) | 10.9 (1.8) |
| AUC (mgh/L) b | 142.0 (28.0) | 138.0 (27.7) | 168.0 (32.2) | 196.0 (36.7) |
| T 1/2 (h) | 1.50 (1.0) | 1.22 (0.3) | 1.99 (0.8) | 1.68 (0.4) |
C max = maximum observed concentration; CL = plasma clearance; AUC = area under the concentration time curve; T ½ = half-life.
a Meropenem 2 grams and vaborbactam 2 grams administered as a 3-hour infusion
b AUC 0-inf reported for single-dose administration; AUC 0-8 reported for multiple-dose administration; AUC 0 – 24 is 414 mgh/L for meropenem and 588 mgh/L for vaborbactam.
The maximum plasma concentration (Cmax) and area under the plasma drug concentration time curve (AUC) of meropenem and vaborbactam proportionally increased with dose across the dose range studied (1 gram to 2 grams for meropenem and 0.25 grams to 2 grams for vaborbactam) when administered as a single 3‑hour intravenous infusion. There is no accumulation of meropenem or vaborbactam following multiple intravenous infusions administered every 8 hours for 7 days in subjects with normal renal function.
The mean population PK parameters of meropenem and vaborbactam in 295 patients (including 35 patients with reduced renal function) after 3-hour infusions of VABOMERE 4 grams (meropenem 2 grams and vaborbactam 2 grams) administered every 8 hours (or dose adjusted based on renal function) are summarized in Table 5
| Parameter | Meropenem | Vaborbactam |
| C max (mg/L) | 57.3 (23.0) |
71.3 (28.6) |
| AUC 0-24, Day 1 (mgh/L) | 637 (295) | 821 (369) |
| AUC 0-24, steady-state (mgh/L) |
650 (364 | 835 (508) |
| CL (L/h) | 10.5 (6.4) | 7.95 (4.3) |
| T 1/2 (h) | 2.30 (2.5) |
2.25 (2.1) |
aMeropenem 2 grams and vaborbactam 2 grams administered as a 3-hour infusion..
Distribution
The plasma protein binding of meropenem is approximately 2%. The plasma protein binding of vaborbactam is approximately 33%.
The steady‑state volumes of distribution of meropenem and vaborbactam in patients were 20.2 L and 18.6 L, respectively.
Elimination
The clearance of meropenem in healthy subjects following multiple doses is 15.1 L/h and for vaborbactam is 10.9 L/h. The t 1/2 is 1.22 hours and 1.68 hours for meropenem and vaborbactam, respectively.
Metabolism
A minor pathway of meropenem elimination is hydrolysis of the beta‑lactam ring (meropenem open lactam), which accounts for 22% of a dose eliminated via the urine.
Vaborbactam does not undergo metabolism.
Excretion
Both meropenem and vaborbactam are primarily excreted via the kidneys.
Approximately 40–60% of a meropenem dose is excreted unchanged within 24‑48 hours with a further 22% recovered as the microbiologically inactive hydrolysis product. The mean renal clearance for meropenem was 7.8 L/h. The mean non‑renal clearance for meropenem was 7.3 L/h which comprises both fecal elimination (~2% of dose) and degradation due to hydrolysis.
For vaborbactam, 75 to 95% of the dose was excreted unchanged in the urine over a 24 to 48 hour period. The mean renal clearance for vaborbactam was 8.9 L/h. The mean non‑renal clearance for vaborbactam was 2.0 L/h indicating nearly complete elimination of vaborbactam by the renal route.
Specific Populations
Patients with Renal Impairment
Following a single dose of VABOMERE, pharmacokinetic studies with meropenem and vaborbactam in subjects with renal impairment have shown that meropenem AUC 0-inf ratios to subjects with normal renal function are 1.28, 2.07, and 4.63 for subjects with mild (eGFR of 60 to 89 mL/min/1.73m 2), moderate (eGFR of 30 to 59 mL/min/1.73m 2), and severe (eGFR <30 mL/min/1.73m 2) renal impairment, respectively; vaborbactam AUC 0-inf ratios to subjects with normal renal function are 1.18, 2.31, and 7.8 for subjects with mild, moderate, and severe renal impairment, respectively [see Dosing and Administration ( 2.2)] . Hemodialysis removed 38% of the meropenem dose and 53% of the vaborbactam dose. Vaborbactam exposure was high in subjects with ESRD (eGFR <15 ml/min/1.73 m 2). Vaborbactam exposure was higher when VABOMERE was administered after hemodialysis (AUC 0-inf ratio to subjects with normal renal function of 37.5) than when VABOMERE was administered before hemodialysis (AUC 0-inf ratio to subjects with normal renal function of 10.2) [see Use in Specific Populations ( 8.6) and Dosing and Administration ( 2.2)] .
Patients with Hepatic Impairment
A pharmacokinetic study conducted with an intravenous formulation of meropenem in patients with hepatic impairment has shown no effects of liver disease on the pharmacokinetics of meropenem.
Vaborbactam does not undergo hepatic metabolism. Therefore, the systemic clearance of meropenem and vaborbactam is not expected to be affected by hepatic impairment.
Geriatric Patients
In elderly patients with renal impairment, plasma clearances of meropenem and vaborbactam were reduced, correlating with age‑associated reduction in renal function [see Dosage and Administration ( 2.2) and Use in Specific Populations ( 8.5)] .
Male and Female Patients
Meropenem and vaborbactam C max and AUC were similar between males and females using a population pharmacokinetic analysis.
Racial or Ethnic Groups
No significant difference in mean meropenem or vaborbactam clearance was observed across race groups using a population pharmacokinetic analysis.
Drug Interactions
No drug-drug interaction was observed between meropenem and vaborbactam in clinical studies with healthy subjects.
Based upon the in vitro and in vivo data available to date, there is a low potential for clinically significant drug interactions with vaborbactam.
Vaborbactam at clinically relevant concentrations does not inhibit the cytochrome P450 isoforms CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A4 in vitro human liver microsomes. Vaborbactam showed no potential for in vitro induction of CYP1A2, CYP2B6, and CYP3A4 in human hepatocytes. Studies evaluating the potential for meropenem to interact with CYP450 enzymes or active transport systems have not been conducted. However, carbapenems as a class have not shown the potential for inhibition or induction CYP450 enzymes and clinical experience suggests that such effects are unlikely.
Vaborbactam does not inhibit the following hepatic and renal transporters in vitro at clinically relevant concentrations: P‑gp, BCRP, OAT1, OAT3, OCT1, OCT2, OATP1B1, OATP1B3 or BSEP. Vaborbactam was not a substrate of OAT1, OAT3, OCT2, P‑gp, and BCRP.
Meropenem is a substrate of OAT1 and OAT3 and as such, probenecid competes with meropenem for active tubular secretion and thus inhibits the renal excretion of meropenem. Following administration of probenecid with meropenem, the mean systemic exposure increased 56% and the mean elimination half-life increased 38% [see Drug Interactions ( 7.2)] .
Concomitant administration of meropenem and valproic acid has been associated with reductions in valproic acid concentrations with subsequent loss in seizure control [see Drug Interactions ( 7.1)].
12.4 Microbiology
Mechanism of Action
The meropenem component of VABOMERE is a penem antibacterial drug. The bactericidal action of meropenem results from the inhibition of cell wall synthesis. Meropenem penetrates the cell wall of most gram-positive and gram-negative bacteria to bind penicillin-binding protein (PBP) targets. Meropenem is stable to hydrolysis by most beta‑lactamases, including penicillinases and cephalosporinases produced by gram‑negative and gram‑positive bacteria, with the exception of carbapenem hydrolyzing beta‑lactamases.
The vaborbactam component of VABOMERE is a non‑suicidal beta-lactamase inhibitor that protects meropenem from degradation by certain serine beta-lactamases such as Klebsiella pneumoniae carbapenemase (KPC). Vaborbactam does not have any antibacterial activity. Vaborbactam does not decrease the activity of meropenem against meropenem-susceptible organisms.
Resistance
Mechanisms of beta-lactam resistance may include the production of beta-lactamases, modification of PBPs by gene acquisition or target alteration, up-regulation of efflux pumps, and loss of outer membrane porin. VABOMERE may not have activity against gram‑negative bacteria that have porin mutations combined with overexpression of efflux pumps.
Clinical isolates may produce multiple beta-lactamases, express varying levels of beta-lactamases, or have amino acid sequence variations, and other resistance mechanisms that have not been identified.
Culture and susceptibility information and local epidemiology should be considered in selecting or modifying antibacterial therapy.
VABOMERE demonstrated in vitro activity against Enterobacteriaceae in the presence of some beta-lactamases and extended-spectrum beta-lactamases (ESBLs) of the following groups: KPC, SME, TEM, SHV, CTX‑M, CMY, and ACT. VABOMERE is not active against bacteria that produce metallo‑beta lactamases or oxacillinases with carbapenemase activity.
In the Phase 3 cUTI trial with VABOMERE, some isolates of E. coli, K. pneumoniae, E. cloacae, C. freundii, P. mirabilis, P. stuartii that produced beta‑lactamases, were susceptible to VABOMERE (minimum inhibitory concentration ≤4 mcg /mL). These isolates produced one or more beta‑lactamases of the following enzyme groups: OXA (non‑carbapenemases), KPC, CTX-M, TEM, SHV, CMY, and ACT.
Some beta-lactamases were also produced by an isolate of K. pneumoniae that was not susceptible to VABOMERE (minimum inhibitory concentration ≥32 mcg/mL). This isolate produced beta-lactamases of the following enzyme groups: CTX-M, TEM, SHV, and OXA.
No cross-resistance with other classes of antimicrobials has been identified. Some isolates resistant to carbapenems (including meropenem) and to cephalosporins may be susceptible to VABOMERE.
Interaction with Other Antimicrobials
In vitro synergy studies have not demonstrated antagonism between VABOMERE and levofloxacin, tigecycline, polymyxin, amikacin, vancomycin, azithromycin, daptomycin, or linezolid.
Activity against Meropenem Non-susceptible Bacteria in Animal Infection Models
Vaborbactam restored activity of meropenem in animal models of infection (e.g., mouse thigh infection, urinary tract infection and pulmonary infection) caused by some meropenem non-susceptible KPC-producing Enterobacteriaceae.
Antimicrobial Activity
VABOMERE has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections [see Indications and Usage ( 1.1)] .
Gram-negative bacteria:
- Enterobacter cloacae species complex
- Escherichia coli
- Klebsiella pneumoniae
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following bacteria exhibit an in vitro MIC less than or equal to the susceptible breakpoint for VABOMERE against isolates of a similar genus or organism group. However, the efficacy of VABOMERE in treating clinical infections due to these bacteria has not been established in adequate and well-controlled clinical trials.
Gram-negative bacteria:
- Citrobacter freundii
- Citrobacter koseri
- Enterobacter aerogenes
- Klebsiella oxytoca
- Morganella morganii
- Proteus mirabilis
- Providencia spp.
- Pseudomonas aeruginosa
- Serratia marcescens
Susceptibility Test Methods
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term carcinogenicity studies have not been performed with VABOMERE, meropenem, or vaborbactam.
Mutagenesis
Meropenem
Genetic toxicity studies were performed with meropenem using the bacterial reverse mutation test, the Chinese hamster ovary HGPRT assay, cultured human lymphocytes cytogenic assay, and the mouse micronucleus test. There was no evidence of mutation potential found in any of these tests.
Vaborbactam
Genetic toxicity studies were performed with vaborbactam using the bacterial reverse mutation test, chromosomal aberration test and the mouse micronucleus test. There was no evidence of mutagenic potential found in any of these tests.
Impairment of Fertility
Meropenem
Reproductive studies were performed with meropenem in male and female rats at doses up to 1000 mg/kg/day with no evidence of impaired fertility (approximately equivalent to 1.6 times the MRHD based on body surface area comparison).
In a reproductive study in cynomolgus monkeys at doses of meropenem up to 360 mg/kg/day (on the basis of body surface area comparison, approximately equivalent to 1.2 times the MRHD) no reproductive toxicity was seen.
Vaborbactam
Vaborbactam had no adverse effect on fertility in male and female rats at doses up to 1000 mg/kg/day, which is equivalent to approximately 1.6 times the MRHD based on body surface area comparison.
14 CLINICAL STUDIES
14.1 Complicated Urinary Tract Infections (cUTI), including Pyelonephritis
A total of 545 adults with cUTI, including pyelonephritis were randomized into a double‑blind, double dummy, multi‑center trial comparing VABOMERE (meropenem 2 grams and vaborbactam 2 grams) to piperacillin/tazobactam (piperacillin 4 grams/tazobactam 0.5 grams) intravenously every 8 hours. Switch to an oral antibacterial drug, such as levofloxacin was allowed after a minimum of 15 doses of IV therapy.
The microbiologically modified intent to treat population (m‑MITT) included all randomized patients who received any study drug and had at least 1 baseline uropathogen. Clinical and microbiological response at the end of IV treatment (EOIVT) required both a clinical outcome of cure or improvement and a microbiologic outcome of eradication (all baseline uropathogens at >10 5 CFU/mL are to be reduced to <10 4 CFU/mL). Clinical and microbiological response was also assessed at the Test of Cure (TOC) visit (approximately 7 days after completion of treatment) in the m‑MITT population and required both a clinical outcome of cure and a microbiological outcome of eradication.
Patient demographic and baseline characteristics were balanced between treatment groups in the m‑MITT population. Approximately 93% of patients were Caucasian and 66% were females in both treatment groups. The mean age was 54 years with 32% and 42% patients greater than 65 years of age in VABOMERE and piperacillin/tazobactam treatment groups, respectively. Mean body mass index was approximately 26.5 kg/m 2 in both treatment groups. Concomitant bacteremia was identified in 12 (6%) and 15 (8%) patients at baseline in VABOMERE and piperacillin/tazobactam treatment groups respectively. The proportion of patients with diabetes mellitus at baseline was 17% and 19% in VABOMERE and piperacillin/tazobactam treatment groups, respectively. The majority of patients (approximately 90%) were enrolled from Europe, and approximately 2% of patients were enrolled from North America. Overall, in both treatment groups, 59% of patients had pyelonephritis and 40% had cUTI, with 21% and 19% of patients having a non-removable and removable source of infection, respectively.
Mean duration of IV treatment in both treatment groups was 8 days and mean total treatment duration (IV and oral) was 10 days; patients with baseline bacteremia could receive up to 14 days of therapy. Approximately 10% of patients in each treatment group in the m-MITT population had a levofloxacin-resistant pathogen at baseline and received levofloxacin as the oral switch therapy. This protocol violation may have impacted the assessment of the outcomes at the TOC visit. These patients were not excluded from the analysis presented in Table 8, as the decision to switch to oral levofloxacin was based on post-randomization factors.
VABOMERE demonstrated efficacy with regard to clinical and microbiological response at the EOIVT visit and TOC visits in the m‑MITT population as shown in Table 8.
|
VABOMERE n/N (%) |
Piperacillin/ Tazobactam n/N (%) |
Difference (95% CI) |
|
| Clinical cure or improvement AND microbiological eradication at the End of IV Treatment Visit* | 183/186 (98.4) | 165/175 (94.3) |
4.1% (0.3%, 8.8%) |
| Clinical cure AND microbiological eradication at the Test of Cure visit approximately 7 days after completion of treatment** | 124/162 (76.5) | 2/153 (73.2) |
3.3% (-6.2%, 13.0%) |
CI = confidence interval; EOIVT = End of Intravenous Treatment; TOC = Test of Cure
*End of IV Treatment visit includes patients with organisms resistant to piperacillin/tazobactam at baseline
**Test of Cure visit excludes patients with organisms resistant to piperacillin/tazobactam at baseline
In the m-MITT population, the rate of clinical and microbiological response in VABOMERE- treated patients with concurrent bacteremia at baseline was 10/12 (83.3%).
In a subset of the E. coli and K. pneumoniae isolates, genotypic testing identified certain ESBL groups (e.g., TEM, CTX-M, SHV and OXA) in both treatment groups of the Phase 3 cUTI trial. The rates of clinical and microbiological response were similar in the ESBL-positive and ESBL-negative subset at EOIVT; at TOC, clinical and microbiological response was lower in the ESBL-positive as compared to ESBL-negative subset in both treatment groups.
15 REFERENCES
- Kawamura S, Russell AW, Freeman SJ, and Siddall, RA: Reproductive and Developmental Toxicity of Meropenem in Rats. Chemotherapy, 40:S238-250 (1992).
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard‑Tenth Edition. CLSI document M07‑A10, Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; Twenty‑seventh Informational Supplement, CLSI document M100‑S27, Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2017.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests; Approved Standard‑Twelfth Edition. CLSI document M02-A12, Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
16 HOW SUPPLIED/STORAGE AND HANDLING
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single‑dose, clear glass vials (NDC 65293‑009‑01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 65293‑009‑06).
Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
Store VABOMERE vials at 20°C to 25°C (68°F to 77°F); excursions are permitted to 15°C to 30°C (59°F to 86°F) [see USP, Controlled Room Temperature (CRT)].
17 PATIENT COUNSELING INFORMATION
Serious Allergic Reactions
Advise patients that allergic reactions, including serious allergic reactions, could occur and that serious reactions require immediate treatment. Ask patient about any previous hypersensitivity reactions to VABOMERE (meropenem and vaborbactam), penicillins, cephalosporins, other beta‑lactams, or other allergens [see Warnings and Precautions ( 5.1)] .
Seizures
Patients receiving VABOMERE on an outpatient basis must be alerted of adverse events such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until it is reasonably well established that VABOMERE is well tolerated, patients should not operate machinery or motorized vehicles [see Warnings and Precautions ( 5.2)] .
Potentially Serious Diarrhea
Counsel patients that diarrhea is a common problem caused by antibacterial drugs including VABOMERE, which usually ends when the antibacterial drug is discontinued. Sometimes after starting treatment with antibacterial drugs, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial drug. If this occurs, patients should contact their physician as soon as possible [see Warnings and Precautions ( 5.3)] .
Interaction with Valproic Acid
Counsel patients to inform their physician if they are taking valproic acid or divalproex sodium. Valproic acid concentrations in the blood may drop below the therapeutic range upon co-administration with VABOMERE. If treatment with VABOMERE is necessary and continued, alternative or supplemental anti‑convulsant medication to prevent and/or treat seizures may be needed [see Warnings and Precautions ( 5.4)] .
Antibacterial Resistance
Counsel patients that antibacterial drugs, including VABOMERE, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When VABOMERE is prescribed to treat a bacterial infection, tell patients that although it is common to feel better early in the course of therapy, take the medication exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by VABOMERE or other antibacterial drugs in the future [see Warnings and Precautions ( 5.7)] .
PRINICIPAL DISPLAY PANEL - VABOMERETM - Carton
Rx Only
VABOMERE
TM
(meropenem and vaborbactam) for injection
2 g per vial*
*Meropenem 1 gram (equivalent to 1.14 g meropenem trihydrate) and vaborbactam 1 g
For Intravenous Infusion Only
Single Dose Only
Discard Unused Portion After Use
PN 1502
NDC: 65293-009-06
MUST BE CONSTITUTED THEN DILUTED
See prescribing information for constitution and dilution instructions and complete directions for use.
Storage:
Store at 20°C to 25°C (68°F to 77°F); excursions are permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]
Manufactured by:
Facta Farmaceutici S.p.A.
Nucleo Industriale S. Atto
S. Nicolo a Tordino
64100 Teramo (TE) Italy
Marketed by:
The Medicines Company
Parsippany, NJ 07054
Product of Italy
Contains 6 single dose 2 g vials
PRINCIPAL DISPLAY PANEL - VABOMERE TM - Vial

Rx Only NDC: 65293-009-01
VABOMERE
TM
(meropenem and vaborbactam) for injection
2 g per vial*
*Meropenem 1 gram (equivalent to 1.14 g meropenem trihydrate) and vaborbactam 1 g
For Intravenous Infusion Only
Single Dose Only
Discard Unused Portion After Use
MUST BE CONSTITUTED THEN DILUTED
See prescribing information for constitution and dilution instructions and complete directions for use.
Each vial contains meropenem 1 g, vaborbactam 1 g, and sodium carbonate 0.575 g. The total sodium content of themixture is approximately 0.35 g (10.9 mEq).
Storage: Store at 20°C to 25°C (68°F to 77°F); excursions are permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]
Manufactured by:
Facta Farmaceutici S.p.A.
Nucleo Industriale S. Atto
S. Nicolo a Tordino
64100 Teramo (TE), Italy
Marketed by:
The Medicines Company
Parsippany, NJ 07054
Product of Italy
PN 1501
| VABOMERE
meropenem-vaborbactam injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - The Medicines Company (040861601) |
| Registrant - The Medicines Company (040861601) |
Trademark Results [VABOMERE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 VABOMERE 87394992 5566623 Live/Registered |
The Medicines Company 2017-03-31 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.