MOTION SICKNESS RELIEF- dimenhydrinate tablet
Motion Sickness Relief by
Drug Labeling and Warnings
Motion Sickness Relief by is a Otc medication manufactured, distributed, or labeled by FRED'S, INC., LNK International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem such as emphysema or chronic bronchitis
-
Directions
- to prevent motion sickness, the first dose should be taken one-half to one hour before starting activity
adults and children 12 years and over 1 to 2 tablets every 4-6 hours; do not exceed 8 tablets in 24 hours, or as directed by a doctor children 6 to under 12 years ½ to 1 tablet every 6-8 hours; do not exceed 3 tablets in 24 hours, or as directed by a doctor children 2 to under 6 years ½ tablet every 6-8 hours; do not exceed 1½ tablets in 24 hours, or as directed by a doctor
- to prevent motion sickness, the first dose should be taken one-half to one hour before starting activity
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal display panel
fred's
PharmacyNDC: 55315-198-02
*Compare to active ingredient in
Dramamine® Original FormulaORIGINAL FORMULA
Motion Sickness Relief
Dimenhydrinate 50 mg AntiemeticPrevents Nausea, Vomiting
& Dizziness for Children & Adults12 TABLETS
Actual Size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING*This product is not manufactured or distributed by Medtech Products Inc., owner
of the registered trademark Dramamine® Original Formula.50844 ORG051819802
DISTRIBUTED BY: fred's, Inc.
4300 NEW GETWELL RD., MEMPHIS, TN 38118
www.fredsinc.com100%
satisfaction
guaranteed
Questions or comments:
1-855-331-FRED(3733)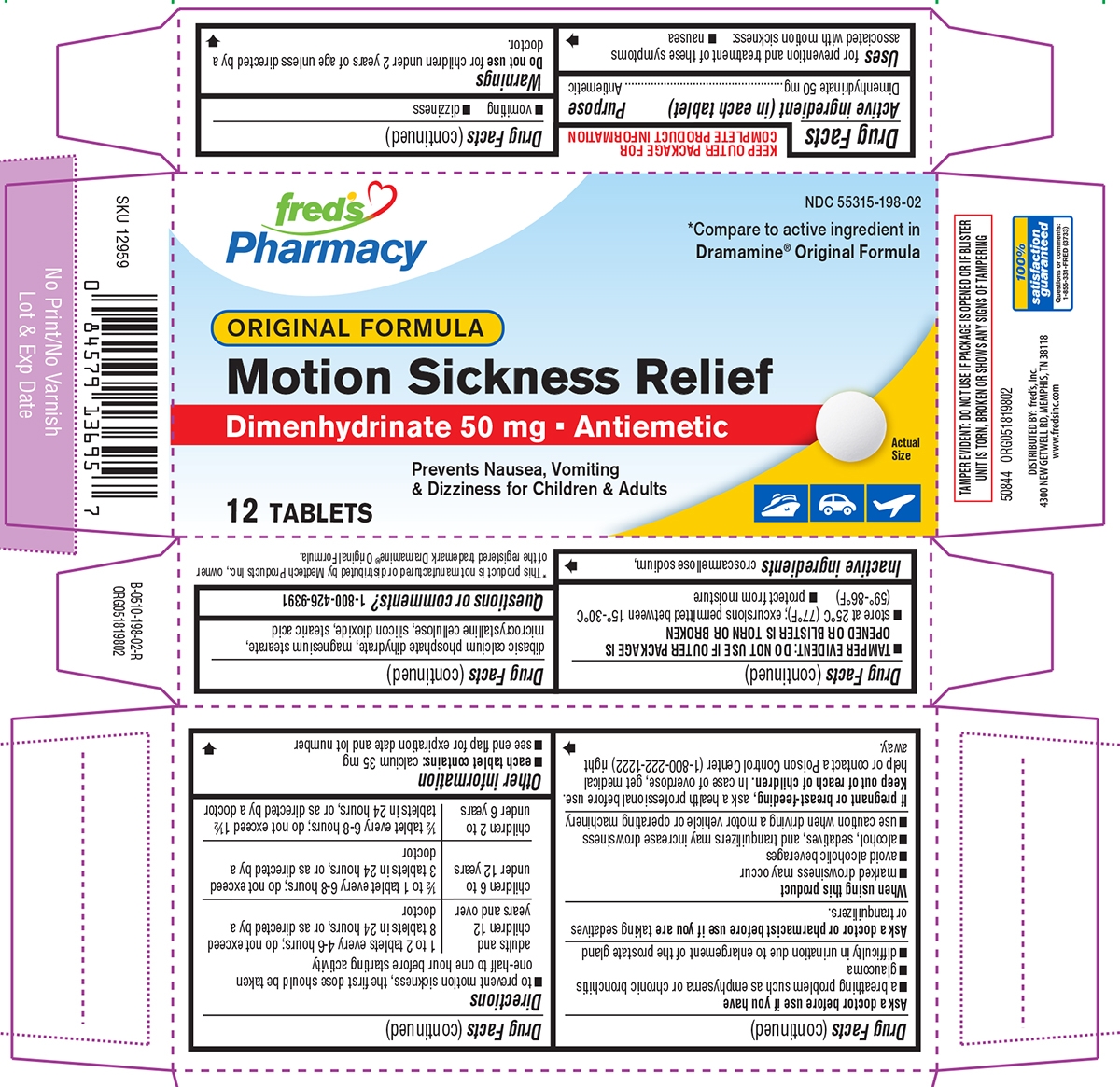
Freds Pharmacy 44-198
-
INGREDIENTS AND APPEARANCE
MOTION SICKNESS RELIEF
dimenhydrinate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 55315-198 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMENHYDRINATE (UNII: JB937PER5C) (DIPHENHYDRAMINE - UNII:8GTS82S83M, CHLORTHEOPHYLLINE - UNII:GE2UA340FM) DIMENHYDRINATE 50 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 9mm Flavor Imprint Code 44;198 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 55315-198-02 2 in 1 CARTON 05/01/2019 1 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part336 05/01/2019 Labeler - FRED'S, INC. (005866116) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 MANUFACTURE(55315-198) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 PACK(55315-198) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 PACK(55315-198) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 PACK(55315-198) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 PACK(55315-198)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.