LOPERAMIDE HYDROCHLORIDE tablet
Loperamide Hydrochloride by
Drug Labeling and Warnings
Loperamide Hydrochloride by is a Otc medication manufactured, distributed, or labeled by Preferred Pharmaceuticals Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Purpose
- Use
- Warnings
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
adults and children 12 years and over
2 tablets after the first loose stool; 1 tablet after each subsequent loose stool; but no more than 4 tablets in 24 hours
children 9 to 11 years (60 to 95 lbs)
1 tablet after the first loose stool; 1/2 tablet after each subsequent loose stool; but no more than 3 tablets in 24 hours
children 6 to 8 years (48 to 59 lbs)
1 tablet after the first loose stool; 1/2 tablet after each subsequent loose stool; but no more than 2 tablets in 24 hours
children 2 to 5 years (34 to 47 lbs)
ask a doctor
children under 2 years (up to 33 lbs)
do not use
- Other information
- Inactive ingredients
- Questions or comments?
-
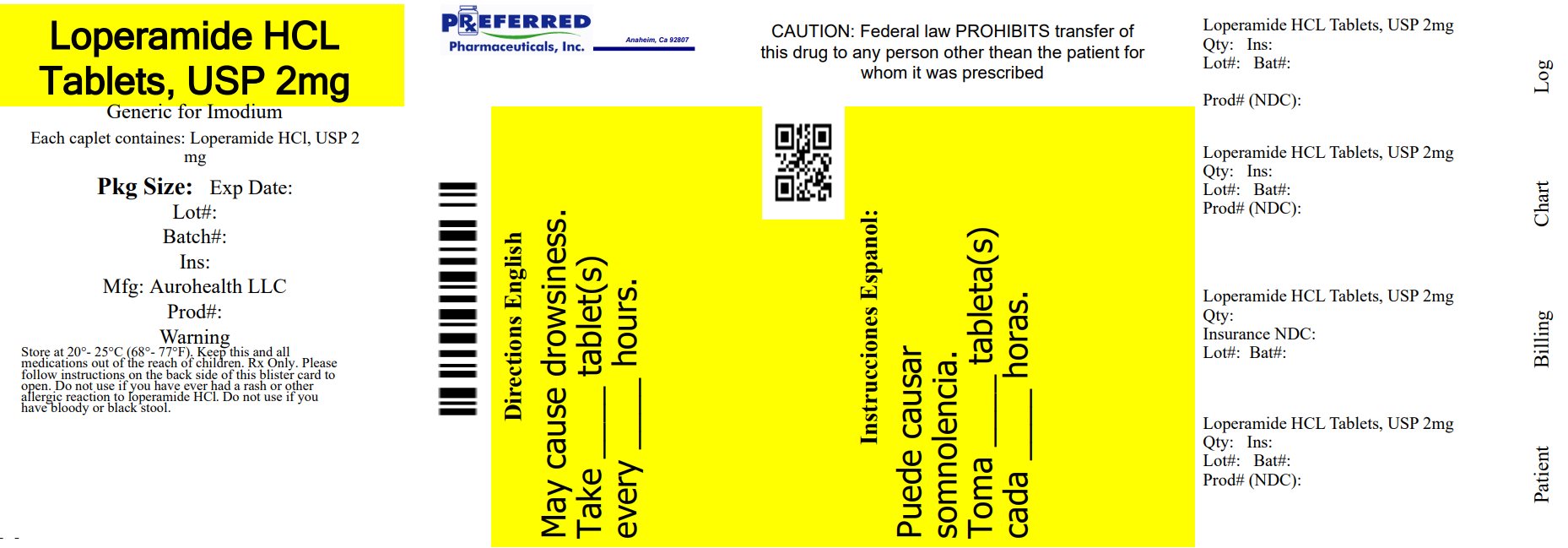
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 2 mg Blister Carton (4 x 6's Tablets)
AUROHEALTH
Relabeled By: Preferred Pharmaceuticals Inc.
NDC: 68788-8393
*Compare to the active
ingredient of Imodium® A-D
Loperamide Hydrochloride
Tablets USP 2 mg
Anti-Diarrheal
Controls the symptoms of diarrhea
24 Tablets
-
INGREDIENTS AND APPEARANCE
LOPERAMIDE HYDROCHLORIDE
loperamide hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68788-8393(NDC:58602-701) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 2 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color GREEN (Light Green) Score 2 pieces Shape CAPSULE (Biconvex) Size 10mm Flavor Imprint Code L;28 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68788-8393-2 4 in 1 CARTON 03/01/2023 1 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206548 03/01/2023 Labeler - Preferred Pharmaceuticals Inc. (791119022) Registrant - Preferred Pharmaceuticals Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals Inc. 791119022 RELABEL(68788-8393)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.