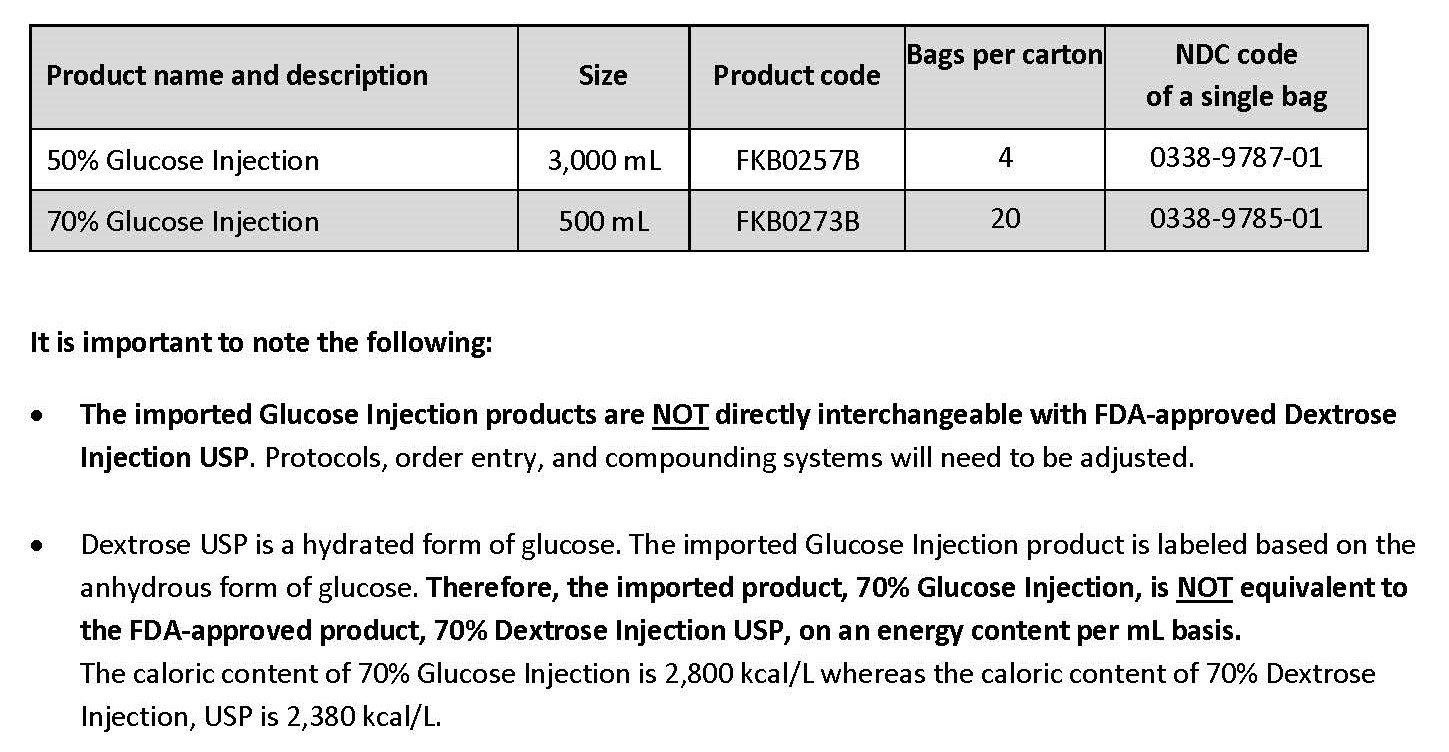
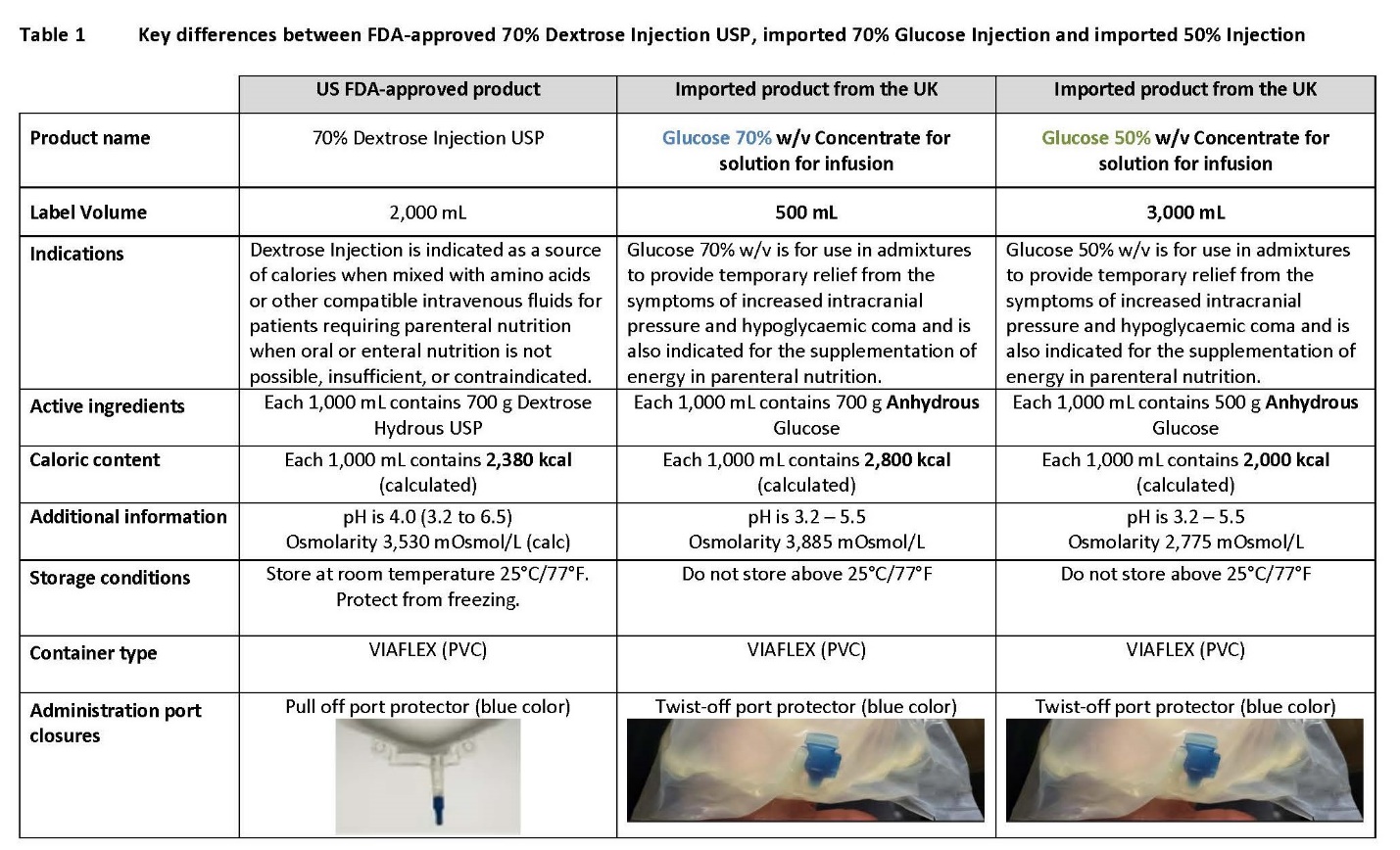
50% and 70% glucose injection
Glucose by
Drug Labeling and Warnings
Glucose by is a Prescription medication manufactured, distributed, or labeled by Baxter Healthcare Company, Baxter Healthcare Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
GLUCOSE- dextrose anhydrous injection, solution
Baxter Healthcare Company
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
50% and 70% glucose injection
Health Care Professional Letter
Baxter has worked proactively to prepare the Training Materials for Baxter Medical Information page (https://meded.baxter.com/hurricane-helene-clinical-resources/baxter-resources-for-products-authorized-for-temporary-importation)
Please refer to the UK prescribing information as follows for any pharmaceutical calculations of a final product that uses concentrated Glucose injection:
- 70% Glucose Injection (click https://mhraproducts4853.blob.core.windows.net/docs/6be6cb79cc56be57f99cfd218cfbd1b34bca8047)) – Local product name in the UK: Glucose 70% w/v Concentrate for solution for infusion
- 50% Glucose Injection (click https://mhraproducts4853.blob.core.windows.net/docs/6d79a27acece3f48020b83e2b3716a2353b6f8b3) – Local product name in the UK: Glucose 50% w/v Concentrate for solution for infusion
Please refer to the FDA-approved prescribing information for the 70% Dextrose Injection USP:
- 70% Dextrose Injection USP (click /DailyMed/0332f34a-7038-47cc-ba59-d55cad6b73ca)
Reporting Adverse Events or Product (Quality Issues)
To report adverse events associated with these imported products, please call Baxter at 1-866-888-2472, or fax: 1-800-759-1801. Adverse events or quality problems experienced with the use of these imported products may also be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax:
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178 (1-800-332-0178).
To report product quality issues associated with these imported products, please contact Baxter Product Surveillance through Baxter Product Feedback Portal (https://productfeedback.baxter.com/).
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
Container Label
Code B0257 3000 ml
Baxter Logo
Glucose 50% w/v
Concentrate for solution for infusion
VIAFLEX container Hypertonic
Free from bacterial endotoxins
Formula per 1000 ml
Anhydrous Glucose
Water for Injections
HCI for pH adjustment
Megajoules (approx.)
500 g
8.4 (2000 kcal)
50%
Dilute before use – bulk source container
Not for direct intravenous infusion
For use under medical supervision
For intravenous use following dilution under aseptic conditions
Check compatibility with other admixture components before use
Keep out of the sight and reach of children
Do not store above 25°C
Do not use unless solution is clear and container is undamaged
Single use only Do not store partially used containers
Discard any unused portion, waste materials and all associated
devices
Do not administer simultaneously with blood or, before
or after, using the same transfusion equipment
Discontinue infusion if adverse reaction occurs
Baxter Healthcare Ltd
Caxton Way Thetford
Norfolk IP24 3SE UK
LOT EXP
POM
PL 00116/0271
TH-35-01-934
Carton Label
Baxter Logo
Code FKB0257B
Glucose 50% w/v
Concentrate for
solution for infusion
Hypertonic
Do not store above 25°C
Keep out of sight and reach of children
Baxter Healthcare Ltd
Caxton Way, Thetford, Norfolk, IP24 3SE, UK
POM
LOT: EXP:
4 x 3000 ml
(≈15.5 kg)
50%
PL 00116/0271
TH-25-02-116
Baxter Logo
Code FKB0257B
Glucose 50% w/v
Concentrate for
solution for infusion
Hypertonic
POM
4 x 3000 ml
(≈15.5 kg)
50%
LOT: EXP:
Container Label
1 –
-
2-
-
3-
-
4-
Code B0273 500 ml
Baxter Logo
Glucose 70% w/v
Concentrate for solution for infusion
VIAFLEX container Hypertonic
Free from bacterial endotoxins
Formula per 1000 ml
Anhydrous Glucose
Water for Injections
HCI for pH adjustment
Megajoules (approx.)
700 g
11.8 (2800 kcal)
70%
Dilute before use – bulk source container
Not for direct intravenous infusion
For use under medical supervision
For intravenous use following dilution under aseptic conditions
Check compatibility with other admixture components before use
Keep out of the sight and reach of children
Do not store above 25°C
Do not use unless solution is clear and container is undamaged
Single use only Do not store partially used containers
Discard any unused portion, waste materials and all associated devices
Do not administer simultaneously with blood or, before or after, using
the same transfusion equipment
Discontinue infusion if adverse reaction occurs
Baxter Healthcare Ltd
Caxton Way Thetford
Norfolk IP24 3SE UK
Lot Expiry
POM
PL 00116/0272
TH-35-01-931
-1
-
-2
-
-3
-
-4
Carton Label
Baxter Logo
Code FKB0273B
Glucose 70% w/v
Concentrate for
solution for infusion
Hypertonic
Do not store above 25°C
Keep out of sight and reach of children
Baxter Healthcare Ltd
Caxton Way, Thetford, Norfolk, IP24 3SE, UK
LOT: EXP:
20 x 500 ml
(≈15.0 kg)
70%
POM
PL 00116/0272
TH-25-02-117
Baxter Logo
Code FKB0273B
Glucose 70% w/v
Concentrate for
solution for infusion
Hypertonic
20 x 500 ml
(≈15.0 kg)
70%
POM
PL 00116/0272
LOT: EXP:
| GLUCOSE
dextrose anhydrous injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GLUCOSE
dextrose anhydrous injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Baxter Healthcare Company (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Healthcare Ltd | 221478644 | ANALYSIS(0338-9787, 0338-9785) , LABEL(0338-9787, 0338-9785) , MANUFACTURE(0338-9787, 0338-9785) , PACK(0338-9787, 0338-9785) , STERILIZE(0338-9787, 0338-9785) | |
Trademark Results [Glucose]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 GLUCOSE 78764828 not registered Dead/Abandoned |
Glucose Media, Inc. 2005-12-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.