Sodium Sulfacetamide 9% and Sulfur 4.5% Suspension
ZMA CLEAR Sodium Sulfacetamide 9% and Sulfur 4.5% by
Drug Labeling and Warnings
ZMA CLEAR Sodium Sulfacetamide 9% and Sulfur 4.5% by is a Prescription medication manufactured, distributed, or labeled by Gabar Health Sciences Corp.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ZMA CLEAR SODIUM SULFACETAMIDE 9% AND SULFUR 4.5%- sulfacetamide sodium and sulfur suspension
Gabar Health Sciences Corp.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Sodium Sulfacetamide 9% and Sulfur 4.5% Suspension
DESCRIPTION
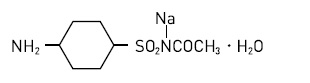
Sodium Sulfacetamide is a sulfonamide with antibacterial with antibacterial activity while sulfur acts as a keratolytic agent. Chemically, Sodium Sulfacetamide is N-[(4-aminophenyl) sulfony]-acetamide, monosodium salt, monohydrate. The structural formula is:
Each mL of Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension contains 90 mg of sodium sulfacetamide and 45 mg of sulfur in a formulation consisting of: aloe vera, fragrance, butylated hydroxytoluene, cetyl alcohol, disodium EDTA, disodium oleamido MEA sulfosuccinate, glyceryl stearate (and) PEG-100 stearate, green tea extract, magnesium aluminum silicate, methylparaben, propylparaben, purified water, sodium cocoyl isethionate, sodium methyl cocoyl tau rate, sodium thiosulfate, stearyl alcohol, and xanthan gum.
CLINICAL PHARMACOLOGY
The most widely accepted mechanism of action of sulfonamides in the Woods-Fildes theory which is based on the fact that sulfonamides act as competitive antagonists to para-aminobenzoic acid (PABA), an essential component for bacterial growth. While absorption through intact skin has not been determined, Sodium Sulfacetamide is readily absorbed from the gastrointestinal tract when taken orally and excreted in the urine, largely unchanged. The biological half-life has variously been reported as 7 to 12.8 hours. The exact mode of action of sulfur in the treatment of acne is unknown, but it has been reported that it inhibits the growth of Propionibacterium acnes and the formation of free fatty acids.
INDICATIONS
Zma Clear™ (Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension) is indicated in the topical control of acne vulgaris, acne rosacea and seborrheic dermatitis.
CONTRAINDICATIONS
Zma Clear™ Sodium Sulfacetamide 9% & Sulfur 4.5% suspension is contraindicated for use by patients having known hypersensitivity to sulfonamides, sulfur, or any other component of this preparation. Zma Clear™ is not to be used by patients with kidney disease.
WARNINGS
Although rare, sensitivity to Sodium Sulfacetamide may occur. Therefore, caution and careful supervision should be observed when prescribing this drug for patients who may be prone to hypersensitivity to topical sulfonamides. Systemic toxic reactions such as agranulocytosis, acute hemolytic anemia, purpura hemorrhagica, drug fever, jaundice, and contact dermatitis indicate hypersensitivity to sulfonamides. Particular caution should be employed if areas of denuded or abraded skin are involved.
FOR EXTERNAL USE ONLY. Keep away from eyes. Keep out of reach of children. Keep container tightly closed.
PRECAUTIONS
General
If irritation develops, use of this product should be discontinued and appropriate therapy instituted. Patients should be very carefully observed for possible local irritation or sensitization during long-term therapy. The object of this therapy is to achieve desquamation without irritation, but Sodium Sulfacetamide and Sulfur can cause reddening and scaling of the epidermis. These side effects are not unusual in the treatment of acne vulgaris, but patients should be cautioned about the possibility.
Information for Patients
Avoid contact with eyes, eyelids, lips and mucous membranes. If accidental contact occurs, rinse with water. If excessive irritation develops, discontinue use and consult your physician.
Carcinogenesis, Mutagenesis and Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential.
Pregnancy
Category C
Animal reproduction studies have not been conducted with Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension. It is also not known whether Sodium Sulfactamide 9% & Sulfur 4.5% Suspension can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension is administered to a nursing woman.
DOSAGE AND ADMINISTRATION
Apply Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension once or twice daily to affected areas or as directed by your physician. Wet skin and liberally apply to areas to be cleansed. Massage gently into skin for 10-20 seconds, working into a lather. Rinse thoroughly and pat dry. If drying occurs, it may be controlled by rinsing off Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension sooner or using less often.
HOW SUPPLIED
Zma Clear™ Sodium Sulfacetamide 9% & Sulfur 4.5% suspension is available in an 8 oz (237 mL) bottle, NDC: 82429-106-08.
STORAGE
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). Brief exposure to temperatures up to 40°C (104°F) may be tolerated provided the mean kinetic temperature does not exceed 25°C (77°F), however such exposure should be minimized. Protect from freezing.
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN
PRINCIPAL DISPLAY PANEL - 8 oz (237 mL) Bottle Label
NDC: 82429-106-08
Rx only
For topical use only
Not ophthalmic use
Sodium Sulfacetamide 9% & Sulfur 4.5% Suspension
Gabar Health Sciences Corp.
Net WT. 8oz. (237 mL)

| ZMA CLEAR SODIUM SULFACETAMIDE 9% AND SULFUR 4.5%
sulfacetamide sodium and sulfur suspension |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Gabar Health Sciences Corp. (118401847) |
| Registrant - Gabar Health Sciences Corp. (118401847) |