Nexalife by Trivia Pharmaceuticals, LLC Nexalife
Nexalife by
Drug Labeling and Warnings
Nexalife by is a Other medication manufactured, distributed, or labeled by Trivia Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
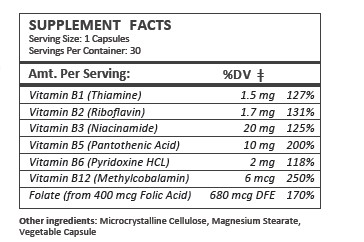
NEXALIFE- b-complex vitamin capsule
Trivia Pharmaceuticals, LLC
----------
Nexalife
Description:
Nexalife B-Complex Capsules are supplied as clear capsules dispensed in plastic bottles of 30 ct.
84781-0016-30
Reserved for Professional Recommendation
This listed product is not a National Drug Code, but instead has merely been formatted to comply with standard industry practice for pharmacy and insurance computer systems.
All prescriptions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product. This product may be administered only under a physician’s supervision. There are no implied or explicit claims on therapeutic equivalence.
Manufactured for:
Trivia Pharmaceuticals
Gilbert, AZ
Warnings and Precautions:
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.
As with any supplement, if you are pregnant, nursing, taking medication, or have a medical condition, ask a health professional before taking this product.
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
Dosage and Administration:
Usual adult dose is 1 capsule by mouth daily once daily, or as prescribed by a licensed medical practitioner.
Nexalife capsules is an orally administered prescription vitamin to provide significant amounts of B Vitamins and folate to supplement the diet, and to help assure that nutritional deficiencies of these vitamines will not develop.
How Supplied:
CONTRAINDICATIONS
This product is contraindicated in patients with known hypersensitivity to any of the ingredients.
PRECAUTIONS
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.
ADVERSE REACTIONS
Allergic sensitizations have been reported following oral administration of folic acid. Consult your physician immediately if adverse side effects occur.
KEEP OUT OF THE REACH OF CHILDREN.
| NEXALIFE
b-complex vitamin capsule |
|||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||
| Supplement Facts | ||
| Serving Size : | Serving per Container : | |
| Amount Per Serving | % Daily Value | |
|---|---|---|
| color | ||
| shape | ||
| size (solid drugs) | 25 mm | |
| scoring | 1 | |
| Labeler - Trivia Pharmaceuticals, LLC (119353862) |
Trademark Results [Nexalife]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NEXALIFE 98674788 not registered Live/Pending |
Guangzhou Xiya Creative Culture Co., Ltd. 2024-07-30 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.