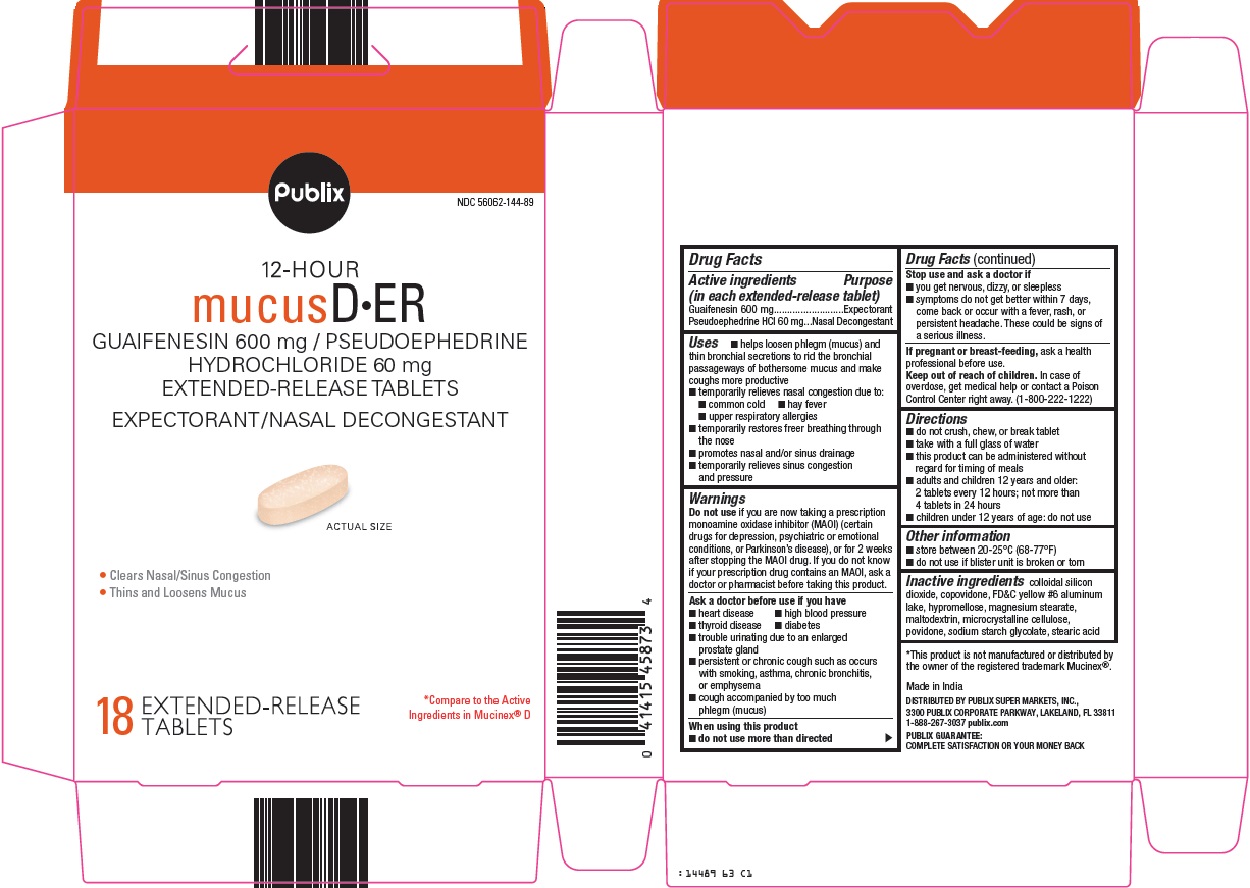
MUCUS D ER- guaifenesin, pseudoephedrine hydrochloride tablet, extended release
Mucus D ER by
Drug Labeling and Warnings
Mucus D ER by is a Otc medication manufactured, distributed, or labeled by Publix Super Markets Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredients (in each extended-release tablet)
- Purpose
-
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves nasal congestion due to:
- common cold
- hay fever
- upper respiratory allergies
- temporarily restores freer breathing through the nose
- promotes nasal and/or sinus drainage
- temporarily relieves sinus congestion and pressure
- Warnings
-
Do not use if you are
now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
MUCUS D ER
guaifenesin, pseudoephedrine hydrochloride tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 56062-144 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 600 mg PSEUDOEPHEDRINE HYDROCHLORIDE (UNII: 6V9V2RYJ8N) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE HYDROCHLORIDE 60 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) COPOVIDONE K25-31 (UNII: D9C330MD8B) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color ORANGE (peach) Score no score Shape OVAL (biconvex) Size 18mm Flavor Imprint Code 600;Watson Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 56062-144-89 18 in 1 CARTON 06/23/2016 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 56062-144-68 36 in 1 CARTON 06/23/2016 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091071 06/23/2016 Labeler - Publix Super Markets Inc (006922009)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.