These highlights do not include all the information needed to use LYNPARZA safely and effectively. See full prescribing information for LYNPARZA. LYNPARZA® (olaparib) capsules, for oral useInitial U.S. Approval: 2014
Lynparza by
Drug Labeling and Warnings
Lynparza by is a Prescription medication manufactured, distributed, or labeled by AstraZeneca Pharmaceuticals LP, AstraZeneca PLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LYNPARZA- olaparib capsule
AstraZeneca Pharmaceuticals LP
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use LYNPARZA safely and effectively. See full prescribing information for LYNPARZA.
LYNPARZA® (olaparib) capsules, for oral use Initial U.S. Approval: 2014 RECENT MAJOR CHANGESINDICATIONS AND USAGELynparza is a poly (ADP-ribose) polymerase (PARP) inhibitor indicated for the treatment of adult patients with deleterious or suspected deleterious germline BRCA-mutated advanced ovarian cancer who have been treated with three or more prior lines of chemotherapy. Select patients for therapy based on an FDA-approved companion diagnostic for Lynparza. (1.1, 2.2) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSCapsules: 50 mg. (3) CONTRAINDICATIONSNone. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca at 1-800-236-9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSUSE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 9/2018 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Advanced gBRCA-mutated Ovarian Cancer after 3 or More Lines of Chemotherapy
Lynparza is indicated for the treatment of adult patients with deleterious or suspected deleterious germline BRCA-mutated (gBRCAm) advanced ovarian cancer who have been treated with three or more prior lines of chemotherapy. Select patients for therapy based on an FDA-approved companion diagnostic for Lynparza.
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
Lynparza is also available as 100 mg and 150 mg tablets. DO NOT substitute Lynparza capsules (50 mg) with Lynparza tablets (100 mg and 150 mg) on a milligram-to-milligram basis due to differences in the dosing and bioavailability of each formulation [see Clinical Pharmacology (12.3)]. Refer to the full prescribing information for Lynparza tablets for specific tablet dosing.
2.2 Patient Selection
Select patients for the treatment of advanced ovarian cancer with Lynparza based on the presence of deleterious or suspected deleterious germline BRCA-mutations [see Indications and Usage (1) and Clinical Studies (14)]. Information on FDA-approved test for the detection of BRCA-mutations is available at http://www.fda.gov/companiondiagnostics.
2.3 Recommended Dosing
The recommended dose of Lynparza is 400 mg (eight 50 mg capsules) taken orally twice daily with or without food, for a total daily dose of 800 mg.
Continue treatment until disease progression or unacceptable toxicity.
If a patient misses a dose of Lynparza, instruct patients to take their next dose at its scheduled time.
Swallow capsule whole. Do not chew, dissolve, or open capsule. Do not take capsules which appear deformed or show evidence of leakage [see How Supplied/Storage and Handling (16.2)].
2.4 Dose Adjustments for Adverse Reactions
To manage adverse reactions, consider interruption of treatment or dose reduction.
The recommended dose reduction is 200 mg (four 50 mg capsules) taken twice daily, for a total daily dose of 400 mg.
If a further dose reduction is required, then reduce to 100 mg (two 50 mg capsules) taken twice daily, for a total daily dose of 200 mg.
2.5 Dose Modifications for Use with CYP3A Inhibitors
Avoid concomitant use of strong and moderate CYP3A inhibitors and consider alternative agents with less CYP3A inhibition. If the inhibitor cannot be avoided, reduce the Lynparza dose to 150 mg (three 50 mg capsules) taken twice daily for a strong CYP3A inhibitor or 200 mg (four 50 mg capsules) taken twice daily for a moderate CYP3A inhibitor [see Drug Interactions (7.2)].
2.6 Dose Modifications for Patients with Renal Impairment
Patients with mild renal impairment (CLcr 51-80 mL/min as estimated by Cockcroft-Gault equation) do not require an adjustment in Lynparza dosing. In patients with moderate renal impairment (CLcr 31-50 mL/min) the recommended dose reduction is to 300 mg (six 50 mg capsules) twice daily, for a total daily dose of 600 mg. The pharmacokinetics of olaparib have not been evaluated in patients with severe renal impairment or end-stage renal disease (CLcr ≤30 mL/min) [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
3 DOSAGE FORMS AND STRENGTHS
Capsules (50 mg): white, opaque, marked in black ink with “OLAPARIB 50 mg” on the cap and the AstraZeneca logo on the body.
5 WARNINGS AND PRECAUTIONS
5.1 Myelodysplastic Syndrome/Acute Myeloid Leukemia
Overall, the incidence of Myelodysplastic Syndrome/Acute Myeloid Leukemia (MDS/AML) in patients treated with Lynparza monotherapy in clinical trials, including long-term follow up, was <1.5% (21/1680) and the majority of events had a fatal outcome. Of these, 19/21 patients had a documented BRCA mutation, 1 patient had gBRCA wildtype and in 1 patient the BRCA mutation status was unknown. Additional cases of MDS/AML have been documented in patients treated with Lynparza in combination studies. The duration of therapy with Lynparza in patients who developed secondary MDS/cancer-therapy related AML varied from < 6 months to > 2 years. All of these patients had received previous chemotherapy with platinum agents and/or other DNA damaging agents including radiotherapy. Some of these patients also had a history of previous cancer or bone marrow dysplasia.
Do not start Lynparza until patients have recovered from hematological toxicity caused by previous chemotherapy (≤ Grade 1). Monitor complete blood count for cytopenia at baseline and monthly thereafter for clinically significant changes during treatment. For prolonged hematological toxicities, interrupt Lynparza and monitor blood counts weekly until recovery. If the levels have not recovered to Grade 1 or less after 4 weeks, refer the patient to a hematologist for further investigations, including bone marrow analysis and blood sample for cytogenetics. If MDS/AML is confirmed, discontinue Lynparza.
5.2 Pneumonitis
Pneumonitis, including fatal cases, occurred in <1% of patients treated with Lynparza. If patients present with new or worsening respiratory symptoms such as dyspnea, cough and fever, or a radiological abnormality occurs, interrupt Lynparza treatment and promptly assess the source of symptoms. If pneumonitis is confirmed, discontinue Lynparza treatment and treat the patient appropriately.
5.3 Embryo-Fetal Toxicity
Lynparza can cause fetal harm when administered to a pregnant woman based on its mechanism of action and findings in animals. In an animal reproduction study, administration of olaparib to pregnant rats during the period of organogenesis caused teratogenicity and embryo-fetal toxicity at exposures below those in patients receiving the recommended human dose of 400 mg twice daily. Apprise pregnant women of the potential hazard to a fetus. Advise females of reproductive potential to use effective contraception during treatment and for 6 months following the last dose of Lynparza [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
6 ADVERSE REACTIONS
The following adverse reactions are discussed elsewhere in the labeling:
- Myelodysplastic Syndrome/Acute Myeloid Leukemia [see Warnings and Precautions (5.1)]
- Pneumonitis [see Warnings and Precautions (5.2)]
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Treatment of Advanced gBRCAm Ovarian Cancer after 3 or More Lines of Chemotherapy
Pooled data
Treatment with Lynparza capsules 400 mg twice daily as monotherapy, was studied in 223 patients (pooled from 6 studies) with gBRCAm advanced ovarian cancer who had received 3 or more prior lines of chemotherapy.
Adverse reactions led to dose interruption in 40% of patients, dose reduction in 4% of patients, and discontinuation in 7% of patients. There were 8 (4%) patients with adverse reactions leading to death, two were attributed to acute leukemia, and one each was attributed to COPD, cerebrovascular accident, intestinal perforation, pulmonary embolism, sepsis, and suture rupture. The median exposure to Lynparza in these patients was 5.2 months.
Table 1 presents adverse reactions reported in ≥20% of patients and Table 2 presents laboratory abnormalities that occurred in at least 25% of patients from the pooled studies.
|
3 or more lines of prior chemotherapy |
||
|
Adverse Reaction |
Grades 1-4 n=223 % |
Grades 3-4 n=223 % |
|
Blood and Lymphatic disorders |
||
|
Anemia |
34 |
18 |
|
Gastrointestinal disorders |
||
|
Decreased appetite |
22 |
1 |
|
Nausea |
64 |
3 |
|
Vomiting |
43 |
4 |
|
Diarrhea |
31 |
1 |
|
Dyspepsia |
25 |
0 |
|
General disorders |
||
|
Fatigue/asthenia |
66 |
8 |
|
Infections and infestations | ||
|
Nasopharyngitis/URI |
26 |
0 |
|
Musculoskeletal and Connective Tissue disorders | ||
|
Arthralgia/musculoskeletal pain |
21 |
0 |
|
Myalgia |
22 |
0 |
|
|
||
|
Laboratory Parameter* |
3 or more lines of prior chemotherapy |
|
|
Grades 1-4 n=223 % |
Grades 3-4 n=223 % |
|
|
Decrease in hemoglobin |
90 |
15 |
|
Decrease in absolute neutrophil count |
25 |
7 |
|
Decrease in platelets |
30 |
3 |
|
Decrease in lymphocytes |
56 |
17 |
|
Mean corpuscular volume elevation |
57 |
- |
|
Increase in creatinine |
30 |
2 |
The following adverse reactions and laboratory abnormalities have been identified in ≥10 to <20% of the 223 patients receiving Lynparza and not included in the table: cough, constipation, dysgeusia, peripheral edema, back pain, dizziness, headache, urinary tract infection, dyspnea, and rash.
The following adverse reactions and laboratory abnormalities have been identified in ≥1 to <10% of the 223 patients receiving Lynparza and not included in the table: leukopenia, stomatitis, peripheral neuropathy, pyrexia, hypomagnesemia, and venous thrombosis (including pulmonary embolism).
Study 19
The safety of Lynparza capsules as maintenance monotherapy was also evaluated in patients with platinum sensitive ovarian cancer who had received 2 or more previous platinum containing regimens in Study 19, a randomized, placebo-controlled, double-blind, multi-center study in which 264 patients received Lynparza 400 mg twice daily (n=136) or placebo (n=128). At the time of final analysis, the median duration of exposure was 8.7 months in patients who received Lynparza and 4.6 months in patients who received placebo.
Adverse reactions led to dose interruptions in 35% of those receiving Lynparza and 10% of those receiving placebo; dose reductions in 26% of Lynparza patients and 4% of placebo patients; and discontinuation in 6% of Lynparza patients and 2% in placebo patients.
Table 3 summarizes the adverse reactions that occurred in at least 20% of patients who received Lynparza in Study 19. Table 4 presents the laboratory abnormalities that occurred in at least 25% of patients from Study 19.
|
|
||||
|
Adverse Reactions |
Lynparza n=136 |
Placebo n=128 |
||
|
Grades 1-4 % |
Grades 3-4 % |
Grades 1-4 % |
Grades 3-4 % |
|
|
Blood and Lymphatic disorders | ||||
|
Anemia† |
23 |
7 |
7 |
1 |
|
Gastrointestinal disorders |
||||
|
Nausea |
71 |
2 |
36 |
0 |
|
Vomiting |
35 |
2 |
14 |
1 |
|
Diarrhea |
28 |
2 |
25 |
2 |
|
Constipation |
22 |
1 |
12 |
0 |
|
General disorders |
||||
|
Fatigue (including asthenia) |
63 |
9 |
46 |
3 |
|
Infections and infestations | ||||
|
Respiratory tract infection |
22 |
2 |
11 |
0 |
|
Metabolism and nutrition disorders | ||||
|
Decreased appetite |
21 |
0 |
13 |
0 |
|
Nervous system disorder |
||||
|
Headache |
21 |
0 |
13 |
1 |
In addition, the adverse reactions in Study 19 that occurred in <20% of patients receiving Lynparza were dyspepsia, stomatitis, dysgeusia, dizziness, increase in creatinine, neutropenia, thrombocytopenia, leukopenia, lymphopenia, dyspnea, pyrexia and edema.
|
|
||||
|
Laboratory parameter* |
Lynparza n=136 |
Placebo n=129 |
||
|
Grades 1-4 % |
Grades 3-4 % |
Grades 1-4 % |
Grades 3-4 % |
|
|
Decrease in hemoglobin |
82 |
8 |
58 |
1 |
|
Increase in mean corpuscular volume |
82 |
- |
51 |
- |
|
Decrease in leukocytes |
58 |
4 |
37 |
2 |
|
Decrease in lymphocytes |
52 |
10 |
32 |
3 |
|
Decrease in absolute neutrophil count |
47 |
7 |
40 |
2 |
|
Mean corpuscular volume elevation |
85 |
- |
44 |
- |
|
Increase in serum creatinine† |
45 |
0 |
14 |
0 |
|
Decrease in platelets |
36 |
4 |
18 |
0 |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Lynparza. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity (rash/dermatitis)
7 DRUG INTERACTIONS
7.1 Anticancer Agents
Clinical studies of Lynparza in combination with other myelosuppressive anticancer agents, including DNA damaging agents, indicate a potentiation and prolongation of myelosuppressive toxicity.
7.2 Drugs That May Increase Olaparib Plasma Concentrations
Olaparib is primarily metabolized by CYP3A. In patients (n=57), co-administration of itraconazole, a strong CYP3A inhibitor, increased AUC of olaparib by 170%. A moderate CYP3A inhibitor, fluconazole, is predicted to increase the AUC of olaparib by 121%.
Avoid concomitant use of strong CYP3A inhibitors such as itraconazole, telithromycin, clarithromycin, ketoconazole, voriconazole, nefazodone, posaconazole, ritonavir, lopinavir/ritonavir, indinavir, saquinavir, nelfinavir, boceprevir, and telaprevir. Avoid use of moderate CYP3A inhibitors such as amprenavir, aprepitant, atazanavir, ciprofloxacin, crizotinib, darunavir/ritonavir, diltiazem, erythromycin, fluconazole, fosamprenavir, imatinib, and verapamil. If the strong or moderate CYP3A inhibitors must be co-administered, reduce the dose of Lynparza [see Dosage and Administration (2.5)].
Avoid grapefruit, grapefruit juice, Seville oranges and Seville orange juice during Lynparza treatment since they are CYP3A inhibitors [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
7.3 Drugs That May Decrease Olaparib Plasma Concentrations
In patients (n=22), co-administration of rifampicin, a strong CYP3A inducer, decreased AUC of olaparib by 87%. A moderate CYP3A inducer, efavirenz, is predicted to decrease the AUC of olaparib by approximately 50%.
Avoid concomitant use of strong CYP3A inducers such as phenytoin, rifampicin, carbamazepine, and St. John’s Wort. Avoid concomitant use of moderate CYP3A4 inducers such as bosentan, efavirenz, etravirine, modafinil, and nafcillin. If a moderate CYP3A inducer cannot be avoided, be aware of a potential for decreased efficacy of Lynparza [see Clinical Pharmacology (12.3)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on findings in animals and its mechanism of action [see Clinical Pharmacology (12.1)], Lynparza can cause fetal harm when administered to a pregnant woman. There are no available data on Lynparza use in pregnant women to inform the drug associated risk. In an animal reproduction study, the administration of olaparib to pregnant rats during the period of organogenesis caused teratogenicity and embryo-fetal toxicity at exposures below those in patients receiving the recommended human dose of 400 mg twice daily [see Data]. Apprise pregnant women of the potential hazard to the fetus and the potential risk for loss of the pregnancy.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. The estimated background risk in the U.S. general population of major birth defects is 2-4%; and the risk for spontaneous abortion is approximately 15-20% in clinically recognized pregnancies.
Data
Animal Data
In a fertility and early embryonic development study in female rats, olaparib was administered orally for 14 days before mating through to day 6 of pregnancy, which resulted in increased post-implantation loss at a dose level of 15 mg/kg/day (with maternal systemic exposures approximately 7% of the human exposure (AUC0-24h) at the recommended dose).
In an embryo-fetal development study, pregnant rats received oral doses of 0.05 and 0.5 mg/kg/day olaparib during the period of organogenesis. A dose of 0.5 mg/kg/day (with maternal systemic exposures approximately 0.3% of human exposure (AUC0-24h) at the recommended dose) caused embryo-fetal toxicities including increased post-implantation loss and major malformations of the eyes (anophthalmia, microphthalmia), vertebrae/ribs (extra rib or ossification center; fused or absent neural arches, ribs, and sternebrae), skull (fused exoccipital) and diaphragm (hernia). Additional abnormalities or variants included incomplete or absent ossification (vertebrae/sternebrae, ribs, limbs) and other findings in the vertebrae/sternebrae, pelvic girdle, lung, thymus, liver, ureter and umbilical artery. Some findings noted above in the eyes, ribs and ureter were observed at a dose of 0.05 mg/kg/day olaparib at lower incidence.
8.2 Lactation
Risk Summary
No data are available regarding the presence of olaparib in human milk, or on its effects on the breastfed infant or on milk production. Because of the potential for serious adverse reactions in the breastfed infants from Lynparza, advise a lactating woman not to breastfeed during treatment with Lynparza and for one month after receiving the last dose.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Pregnancy testing is recommended for females of reproductive potential prior to initiating treatment with Lynparza.
Contraception
Females
Lynparza can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use highly effective contraception during treatment with Lynparza and for at least 6 months following the last dose.
8.4 Pediatric Use
The safety and efficacy of Lynparza have not been established in pediatric patients.
8.5 Geriatric Use
In clinical studies of Lynparza enrolling 482 patients with advanced solid tumors who received Lynparza tablets 300 mg twice daily as monotherapy, 135 (28%) patients were aged ≥65 years. There appeared to be no major difference in the safety profile of patients treated with olaparib aged <65 years versus ≥65 years, nor within the age categories of 65 to 74 years, 75 to 84 years. No patients were aged ≥85 years.
8.6 Hepatic Impairment
No adjustment to the starting dose is required in patients with mild or moderate hepatic impairment. A 15% increase and an 8% increase in mean exposure (AUC), respectively, were observed in patients with mild and moderate hepatic impairment (Child-Pugh classification A and B), compared to patients with normal hepatic function. There are no data in patients with severe hepatic impairment (Child-Pugh classification C) [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No adjustment to the starting dose is required in patients with mild renal impairment, but patients should be monitored closely for toxicity. A 24% increase in mean exposure (AUC) was observed in patients with mild renal impairment (CLcr=51-80 mL/min) compared to patients with normal renal function (CLcr >80 mL/min). A 44% increase in AUC was observed in patients with moderate renal impairment (CLcr=31-50 mL/min) compared to patients with normal renal function (CLcr >80 mL/min). For patients with moderate renal impairment, reduce the dose of Lynparza capsules to 300 mg twice daily [see Dosage and Administration (2.6)]. There are no data in patients with severe renal impairment or end-stage disease (CLcr ≤30 mL/min) [see Clinical Pharmacology (12.3)].
10 OVERDOSAGE
There is no specific treatment in the event of Lynparza overdose, and symptoms of overdose are not established. In the event of an overdose, physicians should follow general supportive measures and should treat symptomatically.
11 DESCRIPTION
Olaparib is an inhibitor of the mammalian polyadenosine 5’-diphosphoribose polymerase (PARP) enzyme.
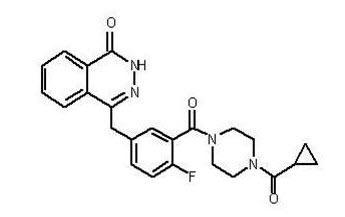
The chemical name is 4-[(3-{[4-(cyclopropylcarbonyl)piperazin-1-yl]carbonyl}-4-fluorophenyl)methyl]phthalazin-1(2H)-one and it has the following chemical structure:
The empirical molecular formula for Lynparza is C24H23FN4O3 and the relative molecular mass is 434.46.
Olaparib is a crystalline solid, is non-chiral and shows pH-independent low solubility of approximately 0.1 mg/mL across the physiological pH range.
Lynparza is available in 50 mg capsules for oral administration. Each capsule contains olaparib as the active ingredient and the following inactive ingredients:
- Capsule content: lauroyl polyoxylglycerides
- Capsule shell: hypromellose, titanium dioxide, gellan gum, potassium acetate
- Capsule printing ink: shellac, ferrosoferric oxide
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Lynparza is an inhibitor of poly (ADP-ribose) polymerase (PARP) enzymes, including PARP1, PARP2, and PARP3. PARP enzymes are involved in normal cellular functions, such as DNA transcription and DNA repair. Olaparib has been shown to inhibit growth of select tumor cell lines in vitro and decrease tumor growth in mouse xenograft models of human cancer both as monotherapy or following platinum-based chemotherapy. Increased cytotoxicity and anti-tumor activity following treatment with olaparib were noted in cell lines and mouse tumor models with deficiencies in BRCA and non-BRCA proteins involved in the homologous recombination repair (HRR) of DNA damage and correlated with platinum response. In vitro studies have shown that olaparib-induced cytotoxicity may involve inhibition of PARP enzymatic activity and increased formation of PARP-DNA complex, resulting in disruption of cellular homeostasis and cell death.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of olaparib on cardiac repolarization was assessed in 119 patients following a single dose of 300 mg tablets and in 109 patients following multiple dosing of 300 mg tablets twice daily. No clinically relevant effect of olaparib on QT interval was observed.
12.3 Pharmacokinetics
Lynparza is available as a tablet and capsule formulation. The oral bioavailability of the tablet formulation is higher than the capsule formulation. Population pharmacokinetic analyses have shown that the steady state exposure (AUC) following 300 mg tablet twice daily was 77% higher compared to that following 400 mg capsule twice daily. The olaparib geometric mean AUC and Cmax following a single 400 mg capsule dose were 37.2 μg*h/mL (N=48) and 4.02 μg/mL (N=48), respectively, and the steady state geometric mean AUC and Cmax following 400 mg capsule twice daily were 43.5 μg*h/mL (N=48) and 6.18 μg/mL (N=48), respectively. Olaparib showed time-dependent PK that the steady state clearance decreased by 15% after multiple dosing.
Absorption
Following oral administration of olaparib via the capsule formulation, absorption is rapid with peak plasma concentrations typically achieved between 1 to 3 hours after dosing. On multiple dosing there is no marked accumulation (accumulation ratio of 1.4 – 1.5 for twice daily dosing), with steady state exposures achieved within 3 to 4 days.
Limited data suggest that the systemic exposure (AUC) of olaparib increases less than proportionally with dose over the dose range of 100 to 400 mg, but the PK data were variable across trials.
Co-administration with a high fat meal slowed the rate (Tmax delayed by 2 hours) of absorption, but did not significantly alter the extent of olaparib absorption (mean AUC increased by approximately 20%).
Distribution
Olaparib had a mean (± standard deviation) apparent volume of distribution at steady state of 167 ± 196 L after a single 400 mg dose of olaparib. The in vitro protein binding of olaparib is approximately 82%.
Metabolism
In vitro, CYP3A4/5 were shown to be the enzymes primarily responsible for the metabolism of olaparib.
Following oral dosing of 14C-olaparib to female patients, unchanged olaparib accounted for the majority of the circulating radioactivity in plasma (70%). It was extensively metabolized with unchanged drug accounting for 15% and 6% of radioactivity in urine and feces, respectively. The majority of the metabolism is attributable to oxidation reactions with a number of the components produced undergoing subsequent glucuronide or sulfate conjugation.
Excretion
A mean (± standard deviation) terminal plasma half-life of 11.9 ± 4.8 hours and apparent plasma clearance of 8.6 ± 7.1 L/h were observed after a single 400 mg dose of olaparib.
Following a single dose of 14C-olaparib, 86% of the dosed radioactivity was recovered within a 7-day collection period, 44% via the urine and 42% via the feces. The majority of the material was excreted as metabolites.
Drug Interactions
Based on the data from a drug-interaction trial (n=57), the AUC and Cmax of olaparib increased by 2.7- and 1.4-fold, respectively, when olaparib was administered in combination with itraconazole, a strong CYP3A inhibitor. Simulations suggested that a moderate CYP3A inhibitor (fluconazole) may increase the AUC and Cmax of olaparib by 2.2- and 1.2-fold, respectively.
Based on the data from a drug-interaction trial (n=22), the AUC and Cmax of olaparib decreased by 87% and 71%, respectively, when olaparib was administered in combination with rifampicin, a strong CYP3A inducer. Simulations suggested that a moderate CYP3A inducer (efavirenz) may decrease the AUC and Cmax of olaparib by approximately 50% and 30%, respectively.
In vitro studies have shown that olaparib is both an inhibitor and inducer of CYP3A and an inducer of CYP2B6. Olaparib is predicted to be a weak CYP3A substrate in humans. In vitro studies also indicated that olaparib is an inhibitor of UGT1A1, BCRP, OATP1B1, OCT1, OCT2, OAT3, MATE1 and MATE2K. The clinical relevance of these findings is unknown. In vitro, olaparib is a substrate of, and inhibits, the efflux transporter P-gp. The potential for olaparib to induce P-gp has not been evaluated.
Pharmacokinetics in Specific Populations
Hepatic Impairment
In a hepatic impairment trial, the mean AUC increased by 15% and the mean Cmax increased by 13% when olaparib was dosed in patients with mild hepatic impairment (Child-Pugh classification A; n=10) and the mean AUC increased by 8% and the mean Cmax decreased by 13% when olaparib was dosed in patients with moderate hepatic impairment, compared to patients with normal hepatic function (n=13). Hepatic impairment had no effect on the protein binding of olaparib and, therefore, total plasma exposure was representative of free drug. There are no data in patients with severe hepatic impairment (Child-Pugh classification C).
Renal Impairment
In a renal impairment trial, the mean AUC increased by 24% and the Cmax increased by 15%, when olaparib was dosed in patients with mild renal impairment (CLcr=51-80 mL/min defined by the Cockcroft-Gault equation; n=13) and by 44% and 26%, respectively, when olaparib was dosed in patients with moderate renal impairment (CLcr=31-50 mL/min; n=13), compared to those with normal renal function (CLcr ≥81 mL/min; n=12). There was no evidence of a relationship between the extent of plasma protein binding of olaparib and creatinine clearance. There are no data in patients with severe renal impairment or end-stage renal disease (CLcr≤30 mL/min).
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with olaparib.
Olaparib was clastogenic in an in vitro chromosomal aberration assay in mammalian Chinese hamster (CHO) cells and in an in vivo rat bone marrow micronucleus assay. This clastogenicity is consistent with genomic instability resulting from the primary pharmacology of olaparib and indicates potential for genotoxicity in humans. Olaparib was not mutagenic in a bacterial reverse mutation (Ames) test.
In a fertility study, female rats received oral olaparib at doses of 0.05, 0.5, and 15 mg/kg/day for at least 14 days before mating through the first week of pregnancy. There were no adverse effects on mating and fertility rates at doses up to 15 mg/kg/day (maternal systemic exposures approximately 11% of the human exposure (AUC0-24h) at the recommended dose).
In a male fertility study, olaparib had no effect on mating and fertility in rats at oral doses up to 40 mg/kg/day following at least 70 days of olaparib treatment (with systemic exposures of approximately 7% of the human exposure (AUC0-24h) at the recommended dose).
14 CLINICAL STUDIES
Advanced gBRCA-mutated Ovarian Cancer Treated with 3 or More Prior Lines of Chemotherapy
The efficacy of Lynparza was investigated in a single-arm study in patients with deleterious or suspected deleterious gBRCAm advanced cancers. A total of 137 patients with measurable, gBRCAm ovarian cancer treated with three or more prior lines of chemotherapy were enrolled. All patients received Lynparza capsules at a dose of 400 mg twice daily as monotherapy until disease progression or intolerable toxicity. Objective response rate (ORR) and duration of response (DOR) were assessed by the investigator according to RECIST v1.0.
The median age of the patients was 58 years, the majority were White (94%) and 93% had an ECOG PS of 0 or 1. Deleterious or suspected deleterious, gBRCAm status was verified retrospectively in 97% (59/61) of the patients for whom blood samples were available by the BRACAnalysis CDx™.
Efficacy results from Study 1 are summarized in Table 5.
Table 5 Overall Response and Duration of Response in Patients with gBRCA-mutated Advanced Ovarian Cancer Who Received 3 or More Lines of Chemotherapy
|
n=137 |
|
|
Objective Response Rate (95% CI) |
34% (26, 42) |
|
Complete Response |
2% |
|
Partial Response |
32% |
|
Median DOR in months (95% CI) |
7.9 (5.6, 9.6) |
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Lynparza 50 mg is a white, opaque, hard capsule, marked in black ink with: “OLAPARIB 50 mg” on the cap and AstraZeneca logo on the body; available in:
Bottles of 112 capsules NDC: 0310-0657-58
16.2 Storage
Refrigerate at 2ºC to 8ºC (36ºF to 46ºF). Protect from freezing.
If needed, Lynparza may be kept at room temperature not to exceed 30ºC (86ºF) for no more than 3 months. Lynparza should not be exposed to temperatures greater than 40ºC (104ºF). Do not take Lynparza if it is suspected of having been exposed to temperatures greater than 40ºC (104ºF) or has been frozen.
Refer to full prescribing information for storage conditions for Lynparza tablets.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
- Administration Instructions: Inform patients on how to take Lynparza [see Dosage and Administration (2.3)]. Lynparza should be taken twice daily with or without food. Instruct patients that if they miss a dose of Lynparza, not to take an extra dose to make up for the one that they missed. They should take their next normal dose at the usual time. Each capsule should be swallowed whole. Do not chew, dissolve, or open the capsule. Inform patients to avoid grapefruit, grapefruit juice, Seville oranges, and Seville orange juice while taking Lynparza [see Drug Interactions (7.3)].
- Inform patients not to substitute Lynparza capsules (50 mg) with Lynparza tablets (100 mg and 150 mg) on a milligram-to-milligram basis due to differences in the dosing and bioavailability of each formulation [see Dosage and Administration (2.1)].
- MDS/AML: Advise patients to contact their healthcare provider if they experience weakness, feeling tired, fever, weight loss, frequent infections, bruising, bleeding easily, breathlessness, blood in urine or stool, and/or laboratory findings of low blood cell counts, or a need for blood transfusions. This may be a sign of hematological toxicity or a more serious uncommon bone marrow problem called ‘myelodysplastic syndrome’ (MDS) or ‘acute myeloid leukemia’ (AML) which have been reported in patients treated with Lynparza [see Warnings and Precautions (5.1)].
- Pneumonitis: Advise patients to contact their healthcare provider if they experience any new or worsening respiratory symptoms including shortness of breath, fever, cough, or wheezing [see Warnings and Precautions (5.2)].
- Embryo-Fetal Toxicity: Advise females to inform their healthcare provider if they are pregnant or become pregnant. Inform female patients of the risk to a fetus and potential loss of the pregnancy [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with Lynparza and for 6 months after receiving the last dose [see Warnings and Precautions (5.3) and Use in Specific Populations (8.1, 8.3)].
- Lactation: Advise patients not to breastfeed while taking Lynparza and for one month after receiving the last dose [see Use in Special Populations (8.2)].
- Nausea/Vomiting: Advise patients that mild or moderate nausea and/or vomiting is very common in patients receiving Lynparza and that they should contact their healthcare provider who will advise on available antiemetic treatment options.
|
Medication Guide Lynparza® (Lin-par-zah) (olaparib) capsules |
|
|
What is the most important information I should know about Lynparza? Lynparza may cause serious side effects, including: Bone marrow problems called Myelodysplastic Syndrome (MDS) or Acute Myeloid Leukemia (AML). Some people who have ovarian cancer or who have received previous treatment with chemotherapy, radiotherapy or certain other medicines for their cancer have developed MDS or AML during treatment with Lynparza. MDS or AML may lead to death. If you develop MDS or AML, your healthcare provider will stop treatment with Lynparza. Symptoms of low blood cell counts are common during treatment with Lynparza, but can be a sign of serious bone marrow problems, including MDS or AML. Symptoms may include: |
|
|
|
|
Your healthcare provider will do blood tests to check your blood cell counts:
|
|
|
What is Lynparza? Lynparza is a prescription medicine used alone to treat women who have a certain type of abnormal inherited BRCA gene, advanced ovarian cancer, and:
Your healthcare provider will perform a test to make sure that Lynparza is right for you. It is not known if Lynparza is safe and effective in children. |
|
|
What should I tell my healthcare provider before taking Lynparza? Before you take Lynparza, tell your healthcare provider about all of your medical conditions including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking Lynparza and certain other medicines may affect how Lynparza works and may cause side effects. |
|
|
How should I take Lynparza?
|
|
|
What should I avoid while taking Lynparza?
|
|
|
What are the possible side effects of Lynparza? Lynparza may cause serious side effects. See “What is the most important information I should know about Lynparza?” The most common side effects of Lynparza are: |
|
|
|
|
|
|
These are not all the possible side effects of Lynparza. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store Lynparza?
3 months.
Keep Lynparza and all medicines out of the reach of children. |
|
|
General information about the safe and effective use of Lynparza Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Lynparza for a condition for which it was not prescribed. Do not give Lynparza to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about Lynparza that is written for health professionals. |
|
|
What are the ingredients in Lynparza? Active ingredient: olaparib |
|
|
Inactive ingredients: Capsule contains: lauroyl polyoxylglycerides Capsule shell contains: hypromellose, titanium dioxide, gellan gum, potassium acetate Capsule printing ink contains: shellac, ferrosoferric oxide Lynparza is a trademark of the AstraZeneca group of companies. © AstraZeneca 2018 Distributed by: AstraZeneca Pharmaceuticals LP Wilmington, DE 19850 For more information, call 1-800-236-9933 or go to www.Lynparza.com. |
|
|
|
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 50 mg
NDC: 0310-0657-58
Lynparza™
(olaparib) capsules
50 mg
112 capsules
Oral use
Rx only
AstraZeneca
| LYNPARZA
olaparib capsule |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - AstraZeneca Pharmaceuticals LP (054743190) |
| Registrant - AstraZeneca PLC (230790719) |