LAVERDIA- verdinexor tablet, coated
Laverdia by
Drug Labeling and Warnings
Laverdia by is a Animal medication manufactured, distributed, or labeled by Dechra Veterinary Products, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
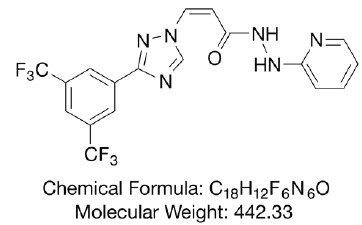
- DESCRIPTION
- INDICATION:
-
DOSAGE AND ADMINISTRATION:
Always provide the Client Information Sheet to the dog owner with each prescription.
Dosing Instructions:
1. Feed the dog immediately before giving LAVERDIA.
2. Wear protective disposable chemotherapy resistant gloves when handling LAVERDIA (see USER SAFETY WARNINGS).
3. Use an appropriate combination of tablets to administer the dose:
a) Administer LAVERDIA at an initial dose of 1.25 mg/kg administered orally twice per week (e.g., Monday and Thursday or Tuesday and Friday) with at least 72 hours between doses.
b) If tolerated after 2 weeks, increase the dose of LAVERDIA to 1.5 mg/kg twice per week with at least 72 hours between doses.
c) Dose reductions of 0.25 mg/kg to a minimum dose of 1 mg/kg twice per week with at least 72 hours between doses or dose interruptions may be considered if the dog has adverse reactions (see ANIMAL SAFETY WARNINGS, PRECAUTIONS, and ADVERSE REACTIONS).
4. Do not split or crush tablets.
-
CONTRAINDICATIONS:
Do not use in dogs that are pregnant, lactating or intended for breeding. LAVERDIA is a possible teratogen and can affect female and male fertility. Laboratory studies in the rat have shown reduced fertility, embryotoxicity, teratogenicity and maternal toxicity. Administration of LAVERDIA caused degeneration/atrophy and vacuolation in the seminiferous tubules and oligospermia in the epididymides in male dogs in the margin of safety study (see TARGET ANIMAL SAFETY).
-
WARNINGS:
USER SAFETY WARNINGS:
NOT FOR USE IN HUMANS. KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN. CHILDREN SHOULD NOT COME INTO CONTACT WITH LAVERDIA. Children should not come in contact with the feces, urine, vomit, or saliva of treated dogs.
Pregnant women, women who may become pregnant, and nursing women should not handle or administer LAVERDIA or come in contact with the feces, urine, vomit, or saliva from LAVERDIA-treated dogs. Based on animal studies, LAVERDIA can cause birth defects and affect female fertility.
LAVERDIA can affect male fertility based on animal studies and studies in humans.
Wear protective disposable chemotherapy resistant gloves when handling LAVERDIA to avoid exposure to drug.
Wear protective disposable chemotherapy resistant gloves to prevent direct contact with moistened, broken, or crushed LAVERDIA tablets.
Wear protective disposable chemotherapy resistant gloves to prevent contact with feces, urine, vomit, and saliva during treatment and for 3 days after the dog has received the last treatment. Place all waste material in a plastic bag and seal before general disposal. Wash hands immediately and thoroughly with soap and water if contact with the feces, urine, vomit, or saliva from LAVERDIA-treated dogs occurs.
Any items that come in contact with feces, urine, vomit, or saliva should not be washed with other laundry during treatment and for 3 days after the last treatment with LAVERDIA.
Wear protective disposable chemotherapy resistant gloves when handling the dog’s toys, food bowl, and water bowl. Wash food and water bowls separately from other items during treatment and for 3 days after the dog has received the last treatment.
If LAVERDIA is accidentally ingested, or if there is significant contact with feces, urine, vomit, or saliva of dogs during treatment or within 3 days after the last treatment without proper precautions, seek medical advice immediately.
It is important to show the treating physician a copy of the package insert, label, or client information sheet.
Special instructions for handling and administering the product
It is recommended that LAVERDIA be administered under the supervision of, or in consultation with, a veterinarian experienced in the use of cancer therapeutic agents.
Use standard measures for the safe handling of all chemotherapeutic drugs. Refer to Occupational Safety and Health Administration (OSHA) for appropriate guidelines, recommendations, and regulations for handling antineoplastic agents.
Do not eat, drink or smoke while handling the product.
Do not store near food, in or near a food preparation area, or with medications intended for use in humans.
Skin contact
In case of contact with skin, wash the affected area immediately and thoroughly with soap and water.
Accidental eye exposure
Rinse eyes with large amounts of tap water (use eyewash station if present) for 10 minutes while holding back the eyelid.
Remove contact lenses.
Seek medical advice immediately and show the package insert or label to the physician.
Accidental oral exposure or ingestion
Seek medical advice immediately and show the package insert or label to the physician.
ANIMAL SAFETY WARNINGS:
LAVERDIA can cause severe anorexia. Dogs should be carefully monitored for inappetence, vomiting, diarrhea, dehydration and weight loss, and supportive care should be provided as clinically indicated (see ADVERSE REACTIONS).
In the studies used to support approval, appetite stimulants, anti-nausea medication, and corticosteroids (prednisone) were found to reduce the incidence of anorexia and gastrointestinal adverse reactions associated with LAVERDIA.
Keep LAVERDIA in a secure location out of reach of dogs, cats, and other animals to prevent accidental ingestion or overdose.
-
PRECAUTIONS:
Safe use of LAVERDIA has not been evaluated in dogs with concurrent serious infections; in dogs with concurrent renal, cardiovascular, or hepatic disease; in dogs with concurrent diabetes mellitus; in dogs with clinically relevant hypercalcemia; or in dogs with concurrent malignancy.
LAVERDIA can cause hematologic and serum chemistry abnormalities. Dogs should be frequently monitored for evidence of hematologic and serum chemistry abnormalities when initiating and maintaining treatment with LAVERDIA (see ADVERSE REACTIONS and TARGET ANIMAL SAFETY).
The safety and effectiveness of LAVERDIA has not been evaluated in conjunction with other chemotherapeutic agents or other treatment modalities for lymphoma.
The effect of concomitant medications on the metabolism of LAVERDIA has not been evaluated.
The safe use of LAVERDIA has not been evaluated in dogs younger than 7 months of age.
The primary metabolism of LAVERDIA in vitro and in vivo is thought to be inactivation by glutathione (GSH) conjugation. Therefore, administration of LAVERDIA with drugs which undergo substantial GSH conjugation (e.g., acetaminophen) should be minimized.
-
ADVERSE REACTIONS:
A randomized, masked, controlled, multicenter clinical field study evaluated the effectiveness and safety of LAVERDIA for the treatment of lymphoma. One hundred and twenty-seven dogs received at least one dose of LAVERDIA. Thirty-three dogs received control tablets.
The Veterinary Cooperative Oncology Group - common terminology criteria for adverse events (VCOG-CTCAE) definitions1 were used to grade the adverse reactions observed:
Grade 1 (mild), Grade 2 (moderate), Grade 3 (severe), Grade 4 (life-threatening), and Grade 5 (death or euthanasia). Most adverse reactions during the study were Grade 1 or 2.
The adverse reactions observed in the study and number of dogs experiencing each adverse reaction are summarized in Table 1 below.
Table 1. Adverse Reactions Reported During the Field Study
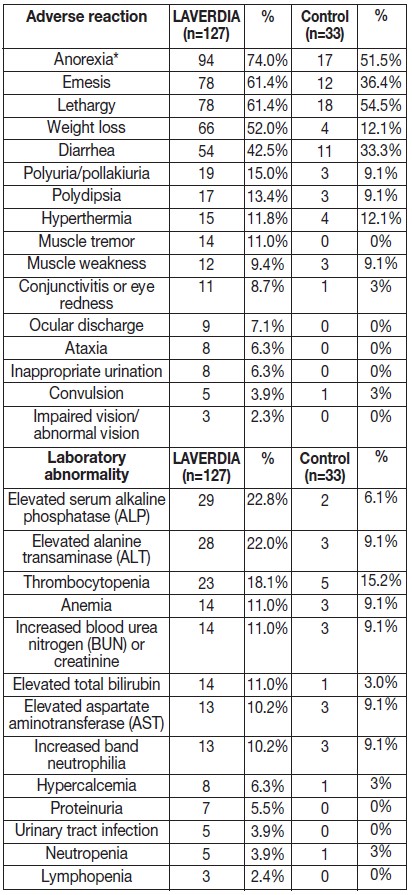
* Of the dogs who had anorexia, 53/94 (56%) in the LAVERDIA group and 5/17 (29%) in the control group received a concomitant medication added to improve appetite.
During the study, additional reported adverse reactions for hematologic and serum chemistry abnormalities included neutrophilia, leukocytosis, leukopenia, hypokalemia, hypochloremia.
Serious Adverse Reactions:
Serious adverse events (SAEs) were reported in 22% (28 of 127) dogs in the LAVERDIA group and 15.2% (5 of the 33) dogs in the control group. Twenty-two dogs in the LAVERDIA group and 4 dogs in the control group were euthanized or died. SAEs reported in other dogs in the LAVERDIA group included thrombocytopenia (one dog), pleural effusion (two dogs), elevated total bilirubin (one dog), neutropenia (one dog), and ventricular arrythmias/swollen face/collapse (one dog). In the control group, one dog had necrosis of the skin on tail secondary to self-trauma.
Conditional Approval Experience:
LAVERDIA was conditionally approved and marketed from 2021-2025 under the proprietary name LAVERDIA-CA1. The following adverse events
were voluntarily reported for LAVERDIA-CA1 during the conditional approval: anemia, polyuria/pollakiuria, leukocytosis, neutrophilia, muscle weakness, and lymphopenia.CONTACT INFORMATION:
To report suspected adverse events, for technical assistance, or to obtain a copy of the Safety Data Sheet (SDS) contact Dechra Veterinary Products at 1-866-933-2472.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or at www.fda.gov/reportanimalae
-
INFORMATION FOR DOG OWNERS:
Always provide the Client Information Sheet with each prescription and review it with the dog owner or person responsible for care of the dog. Advise dog owners about possible adverse reactions, when to contact a veterinarian, how to handle and administer the product, and how to clean up any feces, urine, vomit, or saliva from dogs treated with LAVERDIA. The Client Information Sheet also contains warnings for humans and what to do in case of accidental human exposure to LAVERDIA.
-
CLINICAL PHARMACOLOGY:
Mechanism of action:
Verdinexor is a reversible, selective inhibitor of nuclear export (SINE) that specifically blocks chromosome region maintenance 1 (CRM1), also known as exportin 1 (XPO1). Verdinexor inhibits the export of tumor suppressor proteins (TSP) and growth regulatory proteins (GRP) from the nucleus where they carry out their normal functions; it is selectively cytotoxic for cells with genomic damage (i.e. for tumor cells).
Pharmacokinetics:
Following oral administration of a non-final formulation of verdinexor three times per week for 13 weeks to fed healthy young adult Beagle dogs, overall mean exposure in terms of area under the plasma concentration time curve from time zero to the last quantifiable plasma concentration (AUClast) and maximum plasma concentration (Cmax) showed an increase from 1.25 mg/kg to 1.5 mg/kg; however, exposure at 1.75 mg/kg was either similar or lower than exposure for the 1.5 mg/kg dose group on both evaluation days. The increase in exposure as assessed by dose normalized AUClast and dose normalized Cmax was approximately dose proportional from 1.25 mg/kg to 1.5 mg/kg and less than dose proportional from 1.5 mg/kg to 1.75 mg/kg in males on both evaluation days. In females, the increase in AUClast and Cmax was greater than dose proportional from 1.25 mg/kg to 1.5 mg/kg and there was minimal increase in exposure from 1.5 mg/kg to 1.75 mg/kg on both evaluation days.
Inter-animal variability was relatively high across all dose groups (%CV ranging from 16% to 81% for AUClast); therefore, pharmacokinetic differences or trends should be interpreted cautiously.
Following oral administration of a non-final formulation of verdinexor to fed and fasted dogs, there was a significant food effect on the pharmacokinetics of verdinexor with a 3-fold and 5-fold increase in AUC and Cmax, respectively, when verdinexor was administered orally in the presence of food. Time to reach Cmax (Tmax) was markedly more variable (1-18 hour) when dogs were dosed under fasted conditions as compared to when verdinexor was administered under fed conditions (1.1-2.5 hour).
Table 2. Arithmetic mean (± standard deviation) of verdinexor pharmacokinetic parameters following the first administration of a non-final formulation of verdinexor (maximum proposed label dose 1.5 mg/kg body weight) in fed male and female healthy young adult Beagle Dogs
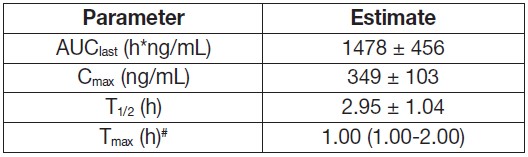
AUClast = area under the curve from the time of dosing to the last quantifiable plasma concentration
Cmax = maximum plasma concentration
T1/2 = half-life
Tmax = time to maximum plasma concentration
#Median (range) -
EFFECTIVENESS:
The effectiveness and safety of LAVERDIA for the treatment of lymphoma was evaluated in a randomized, controlled, masked, multicenter clinical field study. LAVERDIA was compared to control (identical tablets without verdinexor) using time to progression (TTP) as the primary endpoint. Progression was monitored at Days 7, 14, 28, 42, and 56. Progression was defined as the time from Day 0 to progressive disease (PD) for target and non-target lesions using assessment by a modified response evaluation criteria for peripheral nodal lymphoma in dogs2. The modification accommodated dogs that had an initial response to treatment and demonstrated clinical benefit despite lymph node (the target lesion) enlargement.
One hundred and sixty (160) dogs were enrolled and randomly assigned to treatment with either LAVERDIA (n=127) or control (n=33). LAVERDIA was administered as described in the DOSAGE AND ADMINISTRATION section with food or immediately after a meal. The population included dogs with B-cell (80.6% of dogs) or T-cell lymphoma (18.3%) that were naïve to chemotherapy (60% of dogs) or relapsed after chemotherapy (40%), aged 3 to 14 years old, weighing between 9.3 and 72.7 kg, with the most common breeds being mixed breeds, Labrador retriever, and German Shepherd. Use of concomitant appetite stimulants was allowed. If no response to appetite stimulants occurred, prednisone administered at 0.5-1 mg/kg per day was allowed.
Effectiveness was evaluated in 134 dogs (106 dogs in the LAVERDIA group and 28 in control). Including the administration of prednisone as a covariate in the statistical model, the TTP of dogs treated with LAVERDIA was statistically significantly longer (p-value = 0.011) than that of dogs treated with control. Time to progression was 37 days in the LAVERDIA group (with a 95% confidence interval of 29 to 57 days) and 23 days in the control group (with a 95% confidence interval of 14 to 30 days).
Thirty-one (29.2%) dogs in the LAVERDIA group completed the 56-day study while 2 (7.1%) dogs in the control group completed the study. The primary reason that dogs did not complete the study was due to progressive disease, adverse events, and/or death.
Concomitant medications were used primarily for gastrointestinal adverse events or sedation for diagnostics during the study. The most commonly used concomitant medications included maropitant, prednisone, capromorelin, butorphanol, metronidazole, ondansetron, gabapentin, dexmedetomidine, trazadone, atipamezole, isotonic solutions, amoxicillin and clavulanic acid, and heartworm, flea, and tick preventives.
TARGET ANIMAL SAFETY:
In a 13-week margin of safety study, 32 healthy Beagle dogs (4/sex/group), approximately 7 months old at study initiation, were administered
LAVERDIA at either 0, 1.0, 1.5, or 1.75 mg/kg of body weight 3 times a week (Monday, Wednesday, and Friday). Dogs in the control group were sham-dosed. Dogs were fed prior to dosing. All dogs survived to study termination.Dose-dependent LAVERDIA-related clinical findings included vomiting, inappetence, decreased body condition, decreased body weight, loss of skin elasticity, lacrimation, slight depression, and slight decrease of forelimb strength.
Non-dose-dependent LAVERDIA-related findings included abnormal feces (soft, watery, or mucoid feces), excessive shedding, and sparse hair.
Dogs in the 1.0 mg/kg group and dogs in the 1.5 and 1.75 mg/kg groups had lower body weight values, starting on study days 28 and 21 respectively, that continued to the end of the study compared to control dogs.
Dose-dependent LAVERDIA-related clinical pathology findings included decreases in chloride and increases in fibrinogen. Non-dose-dependent LAVERDIA-related clinical pathology findings included decreases in lymphocytes, eosinophils, and monocytes, and increases in albumin and blood urea nitrogen.
Dose-dependent organ weight findings in the dogs administered LAVERDIA included lower testes, thymus, and thyroid/parathyroid gland weights. Dose-dependent histopathological findings in the dogs administered LAVERDIA included lesions in the testes and epididymides (moderate to marked seminiferous tubules degeneration/atrophy, minimal to moderate vacuolation, and minimal Leydig cell hypertrophy in the testes; and severe oligospermia/germ cell debris in the epididymides) and in the thymus (minimal to mild cortical lymphoid depletion).
- HOW SUPPLIED:
- DISPOSAL:
- STORAGE INFORMATION:
-
REFERENCES:
1. Veterinary Cooperative Oncology Group- Common Terminology Criteria for Adverse Events (VCOG-CTCAE v2) following investigational therapy in dogs and cats. Vet Compar Oncol. 2021, Vol.19(2), p.311-352.
2. Response evaluation criteria for peripheral nodal lymphoma in dogs (v1.0) - a Veterinary Cooperative Oncology Group (VCOG) consensus document. Vail DM, Michels GM, Khanna C, Selting KA, London CA; Veterinary Cooperative Oncology Group. Vet Compar Oncol. 2010; 8(1), p. 28-37.
-
SPL UNCLASSIFIED SECTION
Laverdia is a registered trademark of Dechra Limited. Dechra is a registered trademark of Dechra Pharmaceuticals Limited.
Manufactured for:
Dechra Veterinary Products, LLC
7015 College Boulevard, Suite 525, Overland Park, KS 66211Product inquiries should be directed to Dechra Veterinary Products, (866) 933-2472.
Approved by FDA under NADA # 141-614
Issued: January 2026

1 8 9 4 - 0 3 P I N - 7 0 0 1 - 0 9
-
Client Information Sheet
LAVERDIA®
(verdinexor tablets)The client information sheet contains important information about LAVERDIA (verdinexor tablets). You should read this information before you start giving your dog LAVERDIA and review it each time the prescription is refilled as there may be new information. This sheet is provided only as a summary and does not take the place of instructions from your veterinarian. Talk with your veterinarian if you do not understand any of this information or if you want to know more about LAVERDIA.
What is LAVERDIA?
LAVERDIA is an FDA approved drug to treat lymphoma in dogs. The active ingredient in LAVERDIA is verdinexor, a substance that works by preventing tumor suppressing proteins from leaving the nucleus of cells, resulting in disruption of cancer cell survival and eventual cancer cell death.What do I need to tell my veterinarian before my dog is treated with LAVERDIA?
Tell your veterinarian:
About other medications your dog is taking, including prescription drugs, over the counter drugs, heartworm preventatives, flea and tick medications, and vitamins and supplements, including herbal supplements.
About your dog’s previous or current medical conditions, including any infection.
If your dog is pregnant, nursing puppies, or you intend to breed him/her.How is my dog given LAVERDIA?
ALWAYS WEAR GLOVES when handling LAVERDIA and its packaging. See the Handling Instructions section (on the reverse side of this sheet) to learn more about safely administering LAVERDIA to your dog.
LAVERDIA is given by oral administration (by mouth) to dogs.
Feed your dog a small meal immediately before giving LAVERDIA.
LAVERDIA may be hidden inside a treat but be certain your dog swallows the entire tablet(s). It is not recommended that tablets be placed directly in a meal/food bowl as it may be difficult to determine if they have been consumed if the entire meal is not consumed by your dog.
Follow your veterinarian’s instructions for how much and how often to give LAVERDIA. Your dog may need to take a combination of different tablet sizes to receive the correct dose of LAVERDIA. Give all tablets at the same time. Do not change this dose or regimen without consulting with your veterinarian.
Give each dose of LAVERDIA at least 72 hours apart (for example, give on Monday and Thursday or Tuesday and Friday).
If your dog misses a dose of LAVERDIA, do not make up the dose. Instead, simply give the dose on the next regularly scheduled day. Be sure to tell your veterinarian about any missed doses or any difficulty with giving the tablets.How will LAVERDIA affect my dog’s cancer?
LAVERDIA is intended to treat your dog’s cancer. Frequent check-ups by your veterinarian are necessary to determine whether your dog is responding to treatment, and to decide whether your dog should continue to receive LAVERDIA.What are some possible side effects of LAVERDIA?
LAVERDIA may cause side effects, even at the prescribed dose. Serious side effects can occur, with or without warning.The most frequent side effects are:
Decreased/Loss of appetite
Vomiting
Lethargy
Weight Loss
DiarrheaThere are other side effects that may occur. For more
information about side effects ask your veterinarian.Stop LAVERDIA immediately and contact your veterinarian if you notice any of the following changes in your dog:
Refusal to eat for more than 1 day
Repeated vomiting or diarrhea
Lethargy
If your dog experiences other changes that concern youThis client information sheet gives the most important information about LAVERDIA. For more information about LAVERDIA, talk to your veterinarian.
To report a suspected adverse reaction (side effect) call Dechra Veterinary Products at 866-933-2472.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or at www.fda.gov/reportanimalae
See reverse side for HANDLING INSTRUCTIONS
HANDLING INSTRUCTIONS:
What do I need to know to handle LAVERDIA (verdinexor tablets) safely?
ALWAYS WEAR GLOVES when handling LAVERDIA and its packaging. Because LAVERDIA is an anti-cancer drug, extra care must be taken when handling the tablets, giving the drug to your dog, and cleaning up after your dog.
LAVERDIA is not for use in humans.
Do not eat, drink or smoke while handling the product.
Keep LAVERDIA in a secure storage area:
o Out of the reach of children. Children should not come in contact with LAVERDIA.
o Out of reach of dogs, cats, and other animals to prevent accidental ingestion or overdose.
Do not store near food or near a food preparation area, or with medications intended for use in humans.
Pregnant women, women who may become pregnant, and nursing women should not handle or administer LAVERDIA or come in contact with the feces, urine, vomit, or saliva from LAVERDIA-treated dogs.
LAVERDIA may harm an unborn baby. For pregnant and nursing women, accidental ingestion of LAVERDIA may have adverse effects on pregnancy or the nursing baby.
LAVERDIA may affect female and male fertility.How to minimize exposure to the active ingredient when handling LAVERDIA?
ALWAYS WEAR GLOVES when handling LAVERDIA and its packaging. The following handling procedures will help to minimize exposure to the active ingredient in LAVERDIA for you and other members of your household:
Anyone who administers LAVERDIA to your dog should wear protective chemotherapy resistant gloves when handling LAVERDIA. Check with your veterinarian to ensure you have the appropriate gloves.
Minimize the number of people handling LAVERDIA.
When you or others are handling LAVERDIA tablets:
o Do not split or crush the tablets because this will disrupt the protective film coating.
o LAVERDIA tablets should be administered to your dog immediately after they are removed from the bottle.
o Protective disposable chemotherapy resistant gloves should be worn if handling broken or moistened tablets. If your dog spits out the LAVERDIA tablet, the tablet will be moistened and should be handled with protective disposable chemotherapy resistant gloves.
o If the LAVERDIA tablet is “hidden” in a treat, make sure that your dog has eaten the entire dose. This will minimize the potential for exposure to children or other household members to LAVERDIA.
Return any unused LAVERDIA tablets to your veterinarian.What should I do in case of accidental contact when handling LAVERDIA?
In case of contact with skin, wash the affected area immediately and thoroughly with soap and water.
In the case of accidental eye exposure:
o Rinse the eyes with large amounts of tap water (use eyewash station if present) for 10 minutes while holding back the eyelid.
o Remove contact lenses.
o Seek medical advice immediately and show the package insert, label, or client information sheet to the physician.If LAVERDIA is accidentally ingested, seek medical advice immediately. If there is significant contact with feces, urine, vomit, or saliva from your dog during treatment, or within 3 days after the last treatment, without proper precautions, seek medical advice immediately. It is important to show the treating physician a copy of the package insert, label, or client information sheet.
How do I safely clean up after my dog during treatment with LAVERDIA?Because LAVERDIA is a cancer treatment drug, extra care must be taken when cleaning up after your dog during treatment and for 3 days after the last treatment with LAVERDIA.
Avoid direct contact with feces, urine, vomit, or saliva during treatment and for 3 days after the dog has completed treatment with LAVERDIA.
Any skin that comes in contact with feces, urine, vomit, or saliva should be washed immediately with soap and water.
When cleaning up feces, urine, vomit, or saliva you should wear protective disposable chemotherapy resistant gloves and collect the contaminated material with disposable absorptive material (such as paper towels) and place them into a plastic bag. Carefully remove the gloves and place them in the bag and tie or fasten it securely. Wash your hands thoroughly afterwards.
You should not wash any items soiled with feces, urine, vomit, or saliva from your dog with other laundry during treatment and for 3 days after the dog has completed treatment.
Do not let your dog urinate or defecate in areas where people may come in direct contact with the urine or feces.
Children should not come in contact with the feces, urine, vomit, or saliva of treated dogs.
If there is significant contact with feces, urine, vomit, or saliva from your dog during treatment, or within 3 days after the last treatment, without proper precautions, seek medical advice immediately. It is important to show the treating physician a copy of the package insert, label, or client information sheet.Because LAVERDIA may be present in your dog's saliva during treatment and for 3 days after the last treatment, wear protective disposable chemotherapy resistant gloves when handling the dog’s toys, food bowl, and water bowl. Wash food and water bowls separately from other items.
LAVERDIA® is a trademark of Dechra Pharmaceuticals Limited. Dechra is a registered trademark of Dechra Pharmaceuticals Limited.
Manufactured for:
Dechra Veterinary Products, LLC
7015 College Boulevard, Suite 525 Overland Park, KS 66211
(866) 933-2472Approved by FDA under NADA # 141-614
Issued 01/2026

PPT-7002-04
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 2.5 mg Carton
2.5 mg
NDC: 17033-021-01
LAVERDIA®
(verdinexor tablets)2.5 mg per tablet
Antineoplastic
For Oral Use in Dogs OnlyIndicated for the treatment of lymphoma in dogs
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under NADA # 141-614
NET CONTENTS
16 tablets
Dechra

PRINCIPAL DISPLAY PANEL - 10 mg Carton
10 mg
NDC: 17033-022-01
LAVERDIA®
(verdinexor tablets)10 mg per tablet
Antineoplastic
For Oral Use in Dogs OnlyIndicated for the treatment of lymphoma in dogs
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under NADA # 141-614
NET CONTENTS
16 tablets
Dechra

PRINCIPAL DISPLAY PANEL - 50 mg Carton
50 mg
NDC: 17033-024-01
LAVERDIA®
(verdinexor tablets)50 mg per tablet
Antineoplastic
For Oral Use in Dogs OnlyIndicated for the treatment of lymphoma in dogs
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under NADA # 141-614
NET CONTENTS
16 tablets
Dechra

-
INGREDIENTS AND APPEARANCE
LAVERDIA
verdinexor tablet, coatedProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 17033-021 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VERDINEXOR (UNII: 85Q03215IW) (VERDINEXOR - UNII:85Q03215IW) VERDINEXOR 2.5 mg Product Characteristics Color orange Score no score Shape OVAL Size 7mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17033-021-01 1 in 1 CARTON 1 16 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141614 03/01/2026 LAVERDIA
verdinexor tablet, coatedProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 17033-022 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VERDINEXOR (UNII: 85Q03215IW) (VERDINEXOR - UNII:85Q03215IW) VERDINEXOR 10 mg Product Characteristics Color blue Score no score Shape OVAL Size 9mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17033-022-01 1 in 1 CARTON 1 16 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141614 03/01/2026 LAVERDIA
verdinexor tablet, coatedProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 17033-024 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VERDINEXOR (UNII: 85Q03215IW) (VERDINEXOR - UNII:85Q03215IW) VERDINEXOR 50 mg Product Characteristics Color green Score no score Shape OVAL Size 15mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17033-024-01 1 in 1 CARTON 1 16 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141614 03/01/2026 Labeler - Dechra Veterinary Products, LLC (362142734)
Trademark Results [Laverdia]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 LAVERDIA 88290878 not registered Live/Pending |
Anivive Lifesciences, Inc. 2019-02-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.