LEVETIRACETAM EXTENDED-RELEASE- levetiracetam tablet, film coated, extended release
levetiracetam by
Drug Labeling and Warnings
levetiracetam by is a Prescription medication manufactured, distributed, or labeled by Trupharma, LLC, Pharmadax, Inc, Pharmadax (Foshan) Co.,Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LEVETIRACETAM EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information for LEVETIRACETAM EXTENDED-RELEASE TABLETS.
LEVETIRACETAM extended-release tablet, film coated, extended release for oral use
Initial U.S. Approval: 1999RECENT MAJOR CHANGES
Warnings and Precautions (5.1) [04/2009] Patient Counseling Information (17) [04/2009] INDICATIONS AND USAGE
Levetiracetam extended-release tablets is an antiepileptic drug indicated for adjunctive therapy in the treatment of partial onset seizures in patients >16 years of age with epilepsy ( 1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- Suicidal Behavior and Ideation. ( 5.1)
- Neuropsychiatric Adverse Reactions: Levetiracetam extended-release tablets causes somnolence, dizziness, and behavioral abnormalities. The adverse reactions that may be seen in patients receiving Levetiracetam extended-release tablets are expected to be similar to those seen in patients receiving immediate-release Levetiracetam tablets. ( 5.2)
- In controlled trials of immediate-release Levetiracetam extended-release tablets in patients experiencing partial onset seizures, immediate- release Levetiracetam causes somnolence and fatigue, coordination difficulties, and behavioral abnormalities (e.g., psychotic symptoms, suicidal
ideation, and other abnormalities).( 5.2) - Withdrawal Seizures: Levetiracetam extended-release tablets must be gradually withdrawn. ( 5.3)
ADVERSE REACTIONS
- Most common adverse reactions (difference in incidence rate is ≥5% between levetiracetam extended-release tablet-treated patients and placebo-treated patients and occurred more frequently in levetiracetam extended-release tablet-treated patients) include: somnolence and irritability ( 6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Trupharma, LLC at 1-813-444-6299 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 1/2016
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Adult Patients With Impaired Renal Function
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Suicidal Behavior and Ideation
5.2 Neuropsychiatric Adverse Reactions
5.3 Withdrawal Seizures
5.4 Hematologic Abnormalities
5.5 Hepatic Abnormalities
5.6 Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 General Information
7.2 Phenytoin
7.3 Valproate
7.4 Other Antiepileptic Drugs
7.5 Oral Contraceptives
7.6 Digoxin
7.7 Warfarin
7.8 Probenecid
8 USE IN SPECIFIC POPULATIONS
8.1 PREGNANCY
8.2 LABOR & DELIVERY
8.3 NURSING MOTHERS
8.4 PEDIATRIC USE
8.5 GERIATRIC USE
8.6 Use In Patients With Impaired Renal Function
9 DRUG ABUSE AND DEPENDENCE
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 MECHANISM OF ACTION
12.3 PHARMACOKINETICS
13 NONCLINICAL TOXICOLOGY
13.1 CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
13.2 ANIMAL PHARMACOLOGY & OR TOXICOLOGY
14 CLINICAL STUDIES
16 HOW SUPPLIED/ STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
Treatment should be initiated with a dose of 1000 mg once daily. The daily dosage may be adjusted in increments of 1000 mg every 2 weeks to a maximum recommended daily dose of 3000 mg.
2.1 Adult Patients With Impaired Renal Function
Levetiracetam extended-release tablet dosing must be individualized according to the patients renal function status. Recommended doses and adjustment for dose for adults are shown in Table 1. To use this dosing table, an estimate of the patients creatinine clearance (CLcr) in mL/min is needed. CLcr in mL/min may be estimated from serum creatinine
(mg/dL) determination using the following formula:
Table 1: Dosing Adjustment Regimen For Adult Patients With Impaired Renal Function Group Creatinine
Clearance
(mL/min/1.73m 2)Dosage
(mg)Frequency Normal > 80 1000 to
3000Every 24 h Mild 50 to 80 1000 to
2000Every 24 h Moderate 30 to 50 500 to 1500 Every 24 h Severe < 30 500 to 1000 Every 24 h - 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including levetiracetam extended-release tablets, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed. Table 2 shows absolute and relative risk by indication for all evaluated AEDs.
Table 2: Risk by indication for antiepileptic drugs in the pooled analysis Indication Placebo patients
with Events Per
1000 PatientsDrug patients
with Events
Per 1000
PatientsRelative Risk:
Incidence of Events in
Drug Patients/Incidence
in Placebo PatientsRisk difference:
Additional Drug
Patients with Events
Per 1000 patientsEpilepsy 1.0 3.4 3.5 2.4 Psychiatric 5.7 8.5 1.5 2.9 Other 1.0 1.8 1.9 0.9 Total 2.4 4.3 1.8 1.9 The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing levetiracetam extended-release tablets or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
5.2 Neuropsychiatric Adverse Reactions
Levetiracetam extended-release tablets
In some patients experiencing partial onset seizures, levetiracetam extended-release causes somnolence, dizziness, and behavioral abnormalities.
In the levetiracetam extended-release tablet double-blind, controlled trial in patients experiencing partial onset seizures, 7.8% of levetiracetam extended-release tablet treated patients experienced somnolence compared to 2.5% of placebo-treated patients. Dizziness was reported in 5.2% of levetiracetam extended-release tablet treated patients compared to 2.5% of placebo-treated patients.
A total of 6.5% of levetiracetam extended-release tablet-treated patients experienced non-psychotic behavioral disorders (reported as irritability and aggression) compared to 0% of placebo-treated patients. Irritability was reported in 6.5% of levetiracetam extended-release tablet treated patients. Aggression was reported in 1.3% oflevetiracetam extended-release tablet treated patients.
No patient discontinued treatment or had a dose reduction as a result of these adverse reactions.
The number of patients exposed to levetiracetam extended-release tablets was considerably smaller than the number of patients exposed to immediate-release levetiracetam tablets in controlled trials. Therefore, certain adverse reactions observed in the immediate-release levetiracetam tablets controlled trials may also occur in patients receiving levetiracetam extended-release tablets.
Immediate-Release Levetiracetam Tablets
In controlled trials of immediate-release levetiracetam tablets in patients experiencing partial onset seizures, immediate-release levetiracetam tablets cause the occurrence of central nervous system adverse reactions that can be classified into the following categories: 1) somnolence and fatigue, 2) coordination difficulties, and 3) behavioral abnormalities.
In controlled trials of adult patients with epilepsy experiencing partial onset seizures, 14.8% of immediate-release levetiracetam tablet-treated patients reported somnolence, compared to 8.4% of placebo patients. There was no clear dose response up to 3000 mg/day.
In controlled trials of adult patients with epilepsy experiencing partial onset seizures, 14.7% of treated patients reported asthenia, compared to 9.1% of placebo patients.
A total of 3.4% of immediate-release levetiracetam tablet-treated patients experienced coordination difficulties, (reported as either ataxia, abnormal gait, or incoordination) compared to 1.6% of placebo patients.
Somnolence, asthenia and coordination difficulties occurred most frequently within the first 4 weeks of treatment.
In controlled trials of patients with epilepsy experiencing partial onset seizures, 5 (0.7%) immediate-release levetiracetam tablet-treated patients experienced psychotic symptoms compared to 1 (0.2%) placebo patient.
A total of 13.3% of immediate-release levetiracetam tablet patients experienced other behavioral symptoms (reported as aggression, agitation, anger, anxiety, apathy, depersonalization, depression, emotional lability, hostility, irritability, etc.) compared to 6.2% of placebo patients.
5.3 Withdrawal Seizures
Antiepileptic drugs, including levetiracetam extended-release tablets, should be withdrawn gradually to minimize the potential of increased seizure frequency.
5.4 Hematologic Abnormalities
Although there were no obvious hematologic abnormalities observed in treated patients in the levetiracetam extended-release tablet controlled study, the limited number of patients makes any conclusion tentative. The data from the partial seizure patients in the immediate-release levetiracetam tablets controlled studies should be considered to be relevant for levetiracetam extended-release tablet-treated patients.
In controlled trials of immediate-release levetiracetam tablets in patients experiencing partial onset seizures, minor, but statistically significant, decreases compared to placebo in total mean RBC count (0.03 x 10 6/mm 3), mean hemoglobin (0.09 g/dL), and mean hematocrit (0.38%), were seen in immediate-release levetiracetam-treated patients. A total of 3.2% of treated and 1.8% of placebo patients had at least one possibly significant (≤2.8 x 10 9/L) decreased WBC, and 2.4% of treated and 1.4% of placebo patients had at least one possibly significant (≤1.0 x 10 9/L) decreased neutrophil count. Of the treated patients with a low neutrophil count, all but one rose towards or to baseline with continued treatment. No patient was discontinued secondary to low neutrophil counts.
5.5 Hepatic Abnormalities
There were no meaningful changes in mean liver function tests (LFT) in the levetiracetam extended-release tablets controlled trial. No patients were discontinued from the controlled trial for LFT abnormalities.
There were no meaningful changes in mean liver function tests (LFT) in controlled trials of immediate-release levetiracetam tablets in adult patients; lesser LFT abnormalities were similar in drug and placebo-treated patients in controlled trials (1.4%). No patients were discontinued from controlled trials for LFT abnormalities except for 1 (0.07%) adult epilepsy patient receiving open treatment.
5.6 Laboratory Tests
Although effects on laboratory tests were not clinically significant with levetiracetam extended-release tablet treatment, it is expected that the data from immediate-release levetiracetam tablets controlled studies would be considered relevant for levetiracetam extended-release tablet-treated patients.
Although most laboratory tests are not systematically altered with immediate-release levetiracetam tablets treatment, there have been relatively infrequent abnormalities seen in hematologic parameters and liver function tests.
-
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The prescriber should be aware that the adverse reaction incidence figures in the following table, obtained when levetiracetam extended-release tablets were added to concurrent AED therapy, cannot be used to predict the frequency of adverse experiences in the course of usual medical practice where patient characteristics and other factors may differ from those prevailing during clinical studies. Similarly, the cited frequencies cannot be directly compared with figures obtained from other clinical investigations involving different treatments, uses, or investigators. An inspection of these frequencies, however, does provide the prescriber with one basis to estimate the relative contribution of drug and non-drug factors to the adverse reaction incidences in the population studied.
Levetiracetam Extended-Release Tablets
In the well-controlled clinical study using levetiracetam extended-release tablets in patients with partial onset seizures, the most frequently reported adverse reactions in patients receiving levetiracetam extended-release tablets in combination with other AEDs, not seen at an equivalent frequency among placebo-treated patients, were irritability and somnolence.
Table 3 lists treatment-emergent adverse reactions that occurred in at least 5% of epilepsy patients treated with levetiracetam extended-release tablets participating in the placebo-controlled study and were numerically more common than in patients treated with placebo. In this study, either levetiracetam extended-release tablets or placebo was added to concurrent AED therapy. Adverse reactions were usually mild to moderate in intensity.
Table 3: Incidence (%) Of Treatment-Emergent Adverse Reactions In The Placebo-Controlled, Add-On Study By Body System (Adverse Reactions Occurred In At Least 5% Of levetiracetam extended-release Tablets-Treated Patients And Occurred More Frequently Than Placebo-Treated Patients) Body System/Adverse Reaction Extended-ReleaseLevetiracetamTablets(N=77)% Placebo(N=79)% Gastrointestinal Disorders Nausea 5 3 Infections and Infestations Influenza 8 4 Nasopharyngitis 7 5 Nervous System Disorders Somnolence 8 3 Dizziness 5 3 Psychiatric Disorders Irritability 7 0 Discontinuation Or Dose Reduction In The Levetiracetam extended-release Tablets Well-Controlled Clinical Study
In the well-controlled clinical study using levetiracetam extended-release tablets, 5.2% of patients receiving levetiracetam extended-release tablets and 2.5% receiving placebo discontinued as a result of an adverse event. The adverse reactions that resulted in discontinuation and that occurred more frequently in levetiracetam extended-release tablet-treated patients than in placebo-treated patients were asthenia, epilepsy, mouth ulceration, rash and respiratory failure. Each of these adverse reactions led to discontinuation in an levetiracetam extended-release tablet-treated patient and no placebo-treated patients.
Comparison Of Gender, Age And Race
There are insufficient data for levetiracetam extended-release tablets to support a statement regarding the distribution of adverse experience reports by gender, age and race.
Table 4 lists the adverse reactions seen in the well-controlled studies of immediate-release levetiracetam tablets in adult patients experiencing partial onset seizures. Although the pattern of adverse reactions in the levetiracetam extended-release tablets study seems somewhat different from that seen in partial onset seizure well-controlled studies for immediate-release levetiracetam tablets, this is possibly due to the much smaller number of patients in this study compared to the immediate-release tablet studies. The adverse reactions for levetiracetam extended-release tablets are expected to be similar to those seen with immediate-release levetiracetam tablets.
Immediate-Release Levetiracetam Tablets
In well-controlled clinical studies of immediate-release levetiracetam tablets as adjunctive therapy to other AEDs in adults with partial onset seizures, the most frequently reported adverse reactions, not seen at an equivalent frequency among placebo-treated patients, were somnolence, asthenia, infection and dizziness.
Table 4 lists treatment-emergent adverse reactions that occurred in at least 1% of adult epilepsy patients treated with immediate-release levetiracetam tablets participating in placebo-controlled studies and were numerically more common than in patients treated with placebo. In these studies, either immediate-release levetiracetam tablets or placebo was added to concurrent AED therapy. Adverse reactions were usually mild to moderate in intensity.
Table 4: Incidence (%) Of Treatment-Emergent Adverse Reactions In Placebo-Controlled, Add-On Studies In Adults Experiencing Partial Onset Seizures By Body System (Adverse Reactions Occurred In At Least 1% Of Immediate-release Levetiracetam Tablet-Treated Patients And Occurred More Frequently Than Placebo-Treated Patients) Body System/Adverse Reaction Immediate-ReleaseLevetiracetam Tablets(N=769)% Placebo(N=439)% Body as a Whole Asthenia 15 9 Headache 14 13 Infection 13 8 Pain 7 6 Digestive System Anorexia 3 2 Nervous System Somnolence 15 8 Dizziness 9 4 Depression 4 2 Nervousness 4 2 Ataxia 3 1 Vertigo 3 1 Amnesia 2 1 Anxiety 2 1 Hostility 2 1 Paresthesia 2 1 Emotional Lability 2 0 Respiratory System Pharyngitis 6 4 Rhinitis 4 3 Cough Increased 2 1 Sinusitis 2 1 Special Senses Diplopia 2 1 In addition, the following adverse reactions were seen in other well-controlled studies of immediate-release levetiracetam tablets: balance disorder, disturbance in attention, eczema, hyperkinesia, memory impairment, myalgia, personality disorders, pruritus, and vision blurred.
6.2 Postmarketing Experience
In addition to the adverse reactions listed above for immediate-release levetiracetam tablets [ see Adverse Reactions (6.1)], the following adverse events have been identified during postapproval use of immediate-release levetiracetam tablets. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The listing is alphabetized: abnormal liver function test, hepatic failure, hepatitis, leukopenia, neutropenia, pancreatitis, pancytopenia (with bone marrow suppression identified in some of these cases), thrombocytopenia and weight loss. Alopecia has been reported with immediate-release levetiracetam tablet use; recovery was observed in majority of cases where immediate-release levetiracetam tablets were discontinued.
-
7 DRUG INTERACTIONS
7.1 General Information
In vitro data on metabolic interactions indicate that levetiracetam extended-release tablets are unlikely to produce, or be subject to, pharmacokinetic interactions. Levetiracetam and its major metabolite, at concentrations well above Cmax levels achieved within the therapeutic dose range, are neither inhibitors of nor high affinity substrates for human liver cytochrome P450 isoforms, epoxide hydrolase or UDPglucuronidation enzymes. In addition, levetiracetam does not affect the in vitro glucuronidation of valproic acid.
Levetiracetam circulates largely unbound (<10% bound) to plasma proteins; clinically significant interactions with other drugs through competition for protein binding sites are therefore unlikely.
Potential pharmacokinetic interactions were assessed in clinical pharmacokinetic studies (phenytoin, valproate, oral contraceptive, digoxin, warfarin, probenecid) and through pharmacokinetic screening with immediate-release levetiracetam tablets in the placebo-controlled clinical studies in epilepsy patients. The following are the results of these studies. The potential for drug interactions for levetiracetam extended-release tablets is expected to be essentially the same as that with immediate-release levetiracetam tablets.
7.2 Phenytoin
Immediate-release levetiracetam tablets (3000 mg daily) had no effect on the pharmacokinetic disposition of phenytoin in patients with refractory epilepsy. Pharmacokinetics of levetiracetam were also not affected by phenytoin.
7.3 Valproate
Immediate-release levetiracetam tablets (1500 mg twice daily) did not alter the pharmacokinetics of valproate in healthy volunteers. Valproate 500 mg twice daily did not modify the rate or extent of levetiracetam absorption or its plasma clearance or urinary excretion. There also was no effect on exposure to and the excretion of the primary metabolite, ucb L057.
7.4 Other Antiepileptic Drugs
Potential drug interactions between immediate-release levetiracetam tablets and other AEDs (carbamazepine, gabapentin, lamotrigine, phenobarbital, phenytoin, primidone and valproate) were also assessed by evaluating the serum concentrations of levetiracetam and these AEDs during placebo-controlled clinical studies. These data indicate that levetiracetam does not influence the plasma concentration of other AEDs and that these AEDs do not influence the pharmacokinetics of levetiracetam.
7.5 Oral Contraceptives
Immediate-release levetiracetam tablets (500 mg twice daily) did not influence the pharmacokinetics of an oral contraceptive containing 0.03 mg ethinyl estradiol and 0.15 mg levonorgestrel, or of the luteinizing hormone and progesterone levels, indicating that impairment of contraceptive efficacy is unlikely. Coadministration of this oral contraceptive did not influence the pharmacokinetics of levetiracetam.
7.6 Digoxin
Immediate-release levetiracetam tablets (1000 mg twice daily) did not influence the pharmacokinetics and pharmacodynamics (ECG) of digoxin given as a 0.25 mg dose every day. Coadministration of digoxin did not influence the pharmacokinetics of levetiracetam.
7.7 Warfarin
Immediate-release levetiracetam tablets (1000 mg twice daily) did not influence the pharmacokinetics of R and S warfarin. Prothrombin time was not affected by levetiracetam. Coadministration of warfarin did not affect the pharmacokinetics of levetiracetam.
7.8 Probenecid
Probenecid, a renal tubular secretion blocking agent, administered at a dose of 500 mg four times a day, did not change the pharmacokinetics of levetiracetam 1000 mg twice daily. C ssmax of the metabolite, ucb L057, was approximately doubled in the presence of probenecid while the fraction of drug excreted unchanged in the urine remained the same. Renal clearance of ucb L057 in the presence of probenecid decreased 60%, probably related to competitive inhibition of tubular secretion of ucb L057. The effect of immediate-release levetiracetam tablets on probenecid was not studied.
-
8 USE IN SPECIFIC POPULATIONS
8.1 PREGNANCY
Pregnancy Category C
There are no adequate and well-controlled studies in pregnant women. In animal studies, levetiracetam produced evidence of developmental toxicity, including teratogenic effects, at doses similar to or greater than human therapeutic doses. levetiracetam extended-release tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Oral administration of levetiracetam to female rats throughout pregnancy and lactation led to increased incidences of minor fetal skeletal abnormalities and retarded offspring growth pre- and/or postnatally at doses ≥350 mg/kg/day (approximately equivalent to the maximum recommended human dose of 3000 mg [MRHD] on a mg/m 2 basis) and with increased pup mortality and offspring behavioral alterations at a dose of 1800 mg/kg/day (6 times the MRHD on a mg/m 2 basis). The developmental no effect dose was 70 mg/kg/day (0.2 times the MRHD on a mg/m 2 basis). There was no overt maternal toxicity at the doses used in this study.
Oral administration of levetiracetam to pregnant rabbits during the period of organogenesis resulted in increased embryofetal mortality and increased incidences of minor fetal skeletal abnormalities at doses ≥600 mg/kg/day (approximately 4 times MRHD on a mg/m 2 basis) and in decreased fetal weights and increased incidences of fetal malformations at a dose of 1800 mg/kg/day (12 times the MRHD on a mg/m 2 basis). The developmental no effect dose was 200 mg/kg/day (1.3 times the MRHD on a mg/m 2 basis). Maternal toxicity was also observed at 1800 mg/kg/day.
When levetiracetam was administered orally to pregnant rats during the period of organogenesis, fetal weights were decreased and the incidence of fetal skeletal variations was increased at a dose of 3600 mg/kg/day (12 times the MRHD). 1200 mg/kg/day (4 times the MRHD) was a developmental no effect dose. There was no evidence of maternal toxicity in this study.
Treatment of rats with levetiracetam during the last third of gestation and throughout lactation produced no adverse developmental or maternal effects at oral doses of up to 1800 mg/kg/day (6 times the MRHD on a mg/m 2 basis).
Pregnancy Registry
To provide information regarding the effects of in utero exposure to levetiracetam extended-release tablets, physicians are advised to recommend that pregnant patients taking levetiracetam extended-release tablets enroll in the North American Antiepileptic Drug (NAAED) pregnancy registry. This can be done by calling the toll free number 1-888-233-2334, and must be done by the patients themselves. Information on the registry can also be found at the website http://www.aedpregnancyregistry.org/.
8.2 LABOR & DELIVERY
The effect of levetiracetam extended-release tablets on labor and delivery in humans is unknown.
8.3 NURSING MOTHERS
Levetiracetam is excreted in breast milk. Because of the potential for serious adverse reactions in nursing infants from levetiracetam extended-release tablets, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
8.4 PEDIATRIC USE
Safety and effectiveness of levetiracetam extended-release tablets in patients below the age of 16 years have not been established.
8.5 GERIATRIC USE
There were insufficient numbers of elderly subjects in controlled trials of epilepsy to adequately assess the effectiveness of levetiracetam extended-release tablets in these patients. It is expected that the safety of levetiracetam extended-release tablets in elderly patients 65 and over would be comparable to the safety observed in clinical studies of immediate-release levetiracetam tablets.
Of the total number of subjects in clinical studies of immediate-release levetiracetam, 347 were 65 and over. No overall differences in safety were observed between these subjects and younger subjects. There were insufficient numbers of elderly subjects in controlled trials of epilepsy to adequately assess the effectiveness of immediate-release levetiracetam tablets in these patients.
A study in 16 elderly subjects (age 61-88 years) with oral administration of single dose and multiple twice-daily doses of immediate-release levetiracetam tablets for 10 days showed no pharmacokinetic differences related to age alone.
Levetiracetam is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
8.6 Use In Patients With Impaired Renal Function
The effect of levetiracetam extended-release tablets on renally impaired patients was not assessed in the well-controlled study. However, it is expected that the effect on levetiracetam extended-release tablet-treated patients would be similar to the effect seen in well-controlled studies of immediate-release levetiracetam tablets. Caution should be taken in dosing patients with moderate and severe renal impairment and in patients undergoing hemodialysis. The dosage should be reduced in patients with impaired renal function receiving levetiracetam extended-release tablets [ see Clinical Pharmacology (12.3) and Dosage and Administration (2.1)].
Clearance of immediate-release levetiracetam is decreased in patients with renal impairment and is correlated with creatinine clearance.
- 9 DRUG ABUSE AND DEPENDENCE
-
10 OVERDOSAGE
Signs, Symptoms And Laboratory Findings Of Acute Overdosage In Humans
The signs and symptoms for levetiracetam extended-release tablets overdose are expected to be similar to those seen with immediate-release levetiracetam tablets.
The highest known dose of oral immediate-release levetiracetam tablets received in the clinical development program was 6000 mg/day. Other than drowsiness, there were no adverse reactions in the few known cases of overdose in clinical trials. Cases of somnolence, agitation, aggression, depressed level of consciousness, respiratory depression and coma were observed with immediate-release levetiracetam tablets overdoses in postmarketing use.
Treatment Or Management Of Overdose
There is no specific antidote for overdose with levetiracetam extended-release tablets. If indicated, elimination of unabsorbed drug should be attempted by emesis or gastric lavage; usual precautions should be observed to maintain airway. General supportive care of the patient is indicated including monitoring of vital signs and observation of the patient’s clinical status. A Certified Poison Control Center should be contacted for up to date information on the management of overdose with levetiracetam extended-release tablets.
Hemodialysis
Standard hemodialysis procedures result in significant clearance of levetiracetam (approximately 50% in 4 hours) and should be considered in cases of overdose. Although hemodialysis has not been performed in the few known cases of overdose, it may be indicated by the patient's clinical state or in patients with significant renal impairment.
-
11 DESCRIPTION
Levetiracetam is an antiepileptic drug available as 500 mg and 750 mg (white) extended-release tablets for oral administration.
The chemical name of levetiracetam, a single enantiomer, is (-)-(S)-α-ethyl-2-oxo-1-pyrrolidine acetamide, its molecular formula is C 8H 14N 2O 2 and its molecular weight is 170.21. Levetiracetam is chemically unrelated to existing antiepileptic drugs (AEDs). It has the following structural formula:
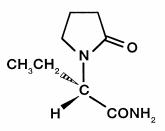
Levetiracetam is a white to off-white crystalline powder with a faint odor and a bitter taste. It is very soluble in water (104.0 g/100 mL). It is freely soluble in chloroform
(65.3 g/100 mL) and in methanol (53.6 g/100 mL), soluble in ethanol (16.5 g/100 mL), sparingly soluble in acetonitrile (5.7 g/100 mL) and practically insoluble in n-hexane. (Solubility limits are expressed as g/100 mL solvent.)
Levetiracetam extended-release tablets contain the labeled amount of levetiracetam. Inactive ingredients: alcohol (anhydrous), colloidal silicon dioxide, ethylcellulose, magnesium stearate, microcrystalline cellulose and talc. The imprinting ink contains ammonium hydroxide, D&C yellow
#10, FD&C blue #1, isopropyl alcohol, n-butyl alcohol, propylene glycol, shellac and titanium dioxide. -
12 CLINICAL PHARMACOLOGY
12.1 MECHANISM OF ACTION
The precise mechanism(s) by which levetiracetam exerts its antiepileptic effect is unknown. The antiepileptic activity of levetiracetam was assessed in a number of animal models of epileptic seizures. Levetiracetam did not inhibit single seizures induced by maximal stimulation with electrical current or different chemoconvulsants and showed only minimal activity in submaximal stimulation and in threshold tests. Protection was observed, however, against secondarily generalized activity from focal seizures induced by pilocarpine and kainic acid, two chemoconvulsants that induce seizures that mimic some features of human complex partial seizures with secondary generalization. Levetiracetam also displayed inhibitory properties in the kindling model in rats, another model of human complex partial seizures, both during kindling development and in the fully kindled state. The predictive value of these animal models for specific types of human epilepsy is uncertain.
In vitro and in vivo recordings of epileptiform activity from the hippocampus have shown that levetiracetam inhibits burst firing without affecting normal neuronal excitability, suggesting that levetiracetam may selectively prevent hypersynchronization of epileptiform burst firing and propagation of seizure activity.
Levetiracetam at concentrations of up to 10 μM did not demonstrate binding affinity for a variety of known receptors, such as those associated with benzodiazepines, GABA (gamma-aminobutyric acid), glycine, NMDA (Nmethyl-D-aspartate), re-uptake sites, and second messenger systems. Furthermore, in vitro studies have failed to find an effect of levetiracetam on neuronal voltage-gated sodium or T-type calcium currents and levetiracetam does not appear to directly facilitate GABAergic neurotransmission. However, in vitro studies have demonstrated that levetiracetam opposes the activity of negative modulators of GABA- and glycine-gated currents and partially inhibits N-type calcium currents in neuronal cells.
A saturable and stereoselective neuronal binding site in rat brain tissue has been described for levetiracetam. Experimental data indicate that this binding site is the synaptic vesicle protein SV2A, thought to be involved in the regulation of vesicle exocytosis. Although the molecular significance of levetiracetam binding to synaptic vesicle protein SV2A is not understood, levetiracetam and related analogs showed a rank order of affinity for SV2A which correlated with the potency of their antiseizure activity in audiogenic seizure-prone mice. These findings suggest that the interaction of levetiracetam with the SV2A protein may contribute to the antiepileptic mechanism of action of the drug.
12.3 PHARMACOKINETICS
Overview
Bioavailability of levetiracetam extended-release tablets is similar to that of the immediate-release levetiracetam tablets . The pharmacokinetics (AUC and Cmax) were shown to be dose proportional after single dose administration of 1000 mg, 2000 mg, and 3000 mg levetiracetam extended-release . Plasma half-life of levetiracetam extended-release is approximately 7 hours.
Levetiracetam is almost completely absorbed after oral administration. The pharmacokinetics of levetiracetam are linear and time-invariant, with low intra- and inter-subject variability. Levetiracetam is not significantly protein-bound (<10% bound) and its volume of distribution is close to the volume of intracellular and extracellular water. Sixty-six percent (66%) of the dose is renally excreted unchanged. The major metabolic pathway of levetiracetam (24% of dose) is an enzymatic hydrolysis of the acetamide group. It is not liver cytochrome P450 dependent. The metabolites have no known pharmacological activity and are renally excreted. Plasma half-life of levetiracetam across studies is approximately 6-8 hours. The half-life is increased in the elderly (primarily due to impaired renal clearance) and in subjects with renal impairment.
Absorption and Distribution
levetiracetam extended-release peak plasma concentrations occur in about 4 hours. The time to peak plasma concentrations is about 3 hours longer with levetiracetam extended-release than with immediate-release tablets.
Single administration of two 500 mg levetiracetam extended-release tablets once daily produced comparable maximal plasma concentrations and area under the plasma concentration versus time as did the administration of one 500 mg immediate-release tablet twice daily in fasting conditions. After multiple dose levetiracetam extended-release tablets intake, extent of exposure (AUC0-24) was similar to extent of exposure after multiple dose immediate-release tablets intake. Cmax and Cmin were lower by 17% and 26% after multiple dose levetiracetam extended-release tablets intake in comparison to multiple dose immediate-release tablets intake. Intake of a high fat, high calorie breakfast before the administration of levetiracetam extended-release tablets resulted in a higher peak concentration, and longer median time to peak. The median time to peak (Tmax) was 2 hours longer in the fed state.
Two 750 mg levetiracetam extended-release tablets were bioequivalent to a single administration of three 500 mg levetiracetam extended-release tablets.
Metabolism
Levetiracetam is not extensively metabolized in humans. The major metabolic pathway is the enzymatic hydrolysis of the acetamide group, which produces the carboxylic acid metabolite, ucb L057 (24% of dose) and is not dependent on any liver cytochrome P450 isoenzymes. The major metabolite is inactive in animal seizure models. Two minor metabolites were identified as the product of hydroxylation of the 2-oxo-pyrrolidine ring (2% of dose) and opening of the 2-oxo-pyrrolidine ring in position 5 (1% of dose). There is no enantiomeric interconversion of levetiracetam or its major metabolite.
Elimination
Levetiracetam plasma half-life in adults is 7 ± 1 hour and is unaffected by either dose or repeated administration. Levetiracetam is eliminated from the systemic circulation by renal excretion as unchanged drug which represents 66% of administered dose. The total body clearance is 0.96 mL/min/kg and the renal clearance is 0.6 mL/min/kg. The mechanism of excretion is glomerular filtration with subsequent partial tubular reabsorption. The metabolite ucb L057 is excreted by glomerular filtration and active tubular secretion with a renal clearance of 4 mL/min/kg. Levetiracetam elimination is correlated to creatinine clearance. Levetiracetam clearance is reduced in patients with impaired renal function [ see Use in Specific Populations (8.6) and Dosage and Administration (2.1)].
Pharmacokinetic Interactions
In vitro data on metabolic interactions indicate that levetiracetam is unlikely to produce, or be subject to, pharmacokinetic interactions. Levetiracetam and its major metabolite, at concentrations well above Cmax levels achieved within the therapeutic dose range, are neither inhibitors of, nor high affinity substrates for, human liver cytochrome P450 isoforms, epoxide hydrolase or UDPglucuronidation enzymes. In addition, levetiracetam does not affect the in vitro glucuronidation of valproic acid. The pharmacokinetics of immediate-release levetiracetam are linear over the dose range of 500-5000 mg. Levetiracetam and its major metabolite are less than 10% bound to plasma proteins; clinically significant interactions with other drugs through competition for protein binding sites are therefore unlikely.
Potential pharmacokinetic interactions of or with immediate-release levetiracetam were assessed in clinical pharmacokinetic studies (phenytoin, valproate, warfarin, digoxin, oral contraceptive, probenecid) and through pharmacokinetic screening in the placebo-controlled clinical studies in epilepsy patients [ see Drug Interactions (7)]. The potential for drug interactions for levetiracetam extended-release is expected to be similar to that with immediate-release levetiracetam.
Special Populations
Elderly
There are insufficient pharmacokinetic data to specifically address the use of levetiracetam extended-release in the elderly population. Pharmacokinetics of immediate-release levetiracetam were evaluated in 16 elderly subjects (age 61-88 years) with creatinine clearance ranging from 30 to 74 mL/min. Following oral administration of twice-daily dosing for 10 days, total body clearance decreased by 38% and the half-life was 2.5 hours longer in the elderly compared to healthy adults. This is most likely due to the decrease in renal function in these subjects.
Pediatric Patients
Safety and effectiveness of levetiracetam extended-release tablets in patients below the age of 16 years have not been established.
Gender
levetiracetam extended-release Cmax was 21-30% higher and AUC was 8-18% higher in women (N=12) compared to men (N=12). However, clearances adjusted for body weight were comparable.
Race
Formal pharmacokinetic studies of the effects of race have not been conducted with extended-release or immediate-release levetiracetam. Cross study comparisons involving Caucasians (N=12) and Asians (N=12), however, show that pharmacokinetics of immediate-release levetiracetam were comparable between the two races.
Renal Impairment
The effect of levetiracetam extended-release tablets on renally impaired patients was not assessed in the well-controlled study. However, it is expected that the effect on levetiracetam extended-release tablet-treated patients would be similar to that seen in well-controlled studies of immediate-release levetiracetam tablets. In patients with end stage renal disease on dialysis, it is recommended that immediate-release levetiracetam tablets be used instead of levetiracetam extended-release tablets.
The disposition of immediate-release levetiracetam was studied in adult subjects with varying degrees of renal function. Total body clearance of levetiracetam is reduced in patients with impaired renal function by 40% in the mild group (CLcr = 50-80 mL/min), 50% in the moderate group (CLcr = 30-50 mL/min) and 60% in the severe renal impairment group (CLcr <30 mL/min). Clearance of levetiracetam is correlated with creatinine clearance.
In anuric (end stage renal disease) patients, the total body clearance decreased 70% compared to normal subjects (CLcr >80mL/min). Approximately 50% of the pool of levetiracetam in the body is removed during a standard 4 hour hemodialysis procedure.
Dosage should be reduced in patients with impaired renal function receiving levetiracetam; immediate-release levetiracetam should be given to patients on dialysis [ see Dosage and Administration (2.1)].
Hepatic Impairment
In subjects with mild (Child-Pugh A) to moderate (Child-Pugh B) hepatic impairment, the pharmacokinetics of levetiracetam were unchanged. In patients with severe hepatic impairment (Child-Pugh C), total body clearance was 50% that of normal subjects, but decreased renal clearance accounted for most of the decrease. No dose adjustment is needed for patients with hepatic impairment.
-
13 NONCLINICAL TOXICOLOGY
13.1 CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
Carcinogenesis
Rats were dosed with levetiracetam in the diet for 104 weeks at doses of 50, 300 and 1800 mg/kg/day. The highest dose corresponds to 6 times the maximum recommended daily human dose (MRHD) of 3000 mg on a mg/m 2 basis and it also provided systemic exposure (AUC) approximately 6 times that achieved in humans receiving the MRHD. There was no evidence of carcinogenicity. A study was conducted in which mice received levetiracetam in the diet for 80 weeks at doses of 60, 240 and 960 mg/kg/day (high dose is equivalent to 2 times the MRHD on a mg/m 2 or exposure basis). Although no evidence for carcinogenicity was seen, the potential for a carcinogenic response has not been fully evaluated in that species because adequate doses have not been studied.
Mutagenesis
Levetiracetam was not mutagenic in the Ames test or in mammalian cells in vitro in the Chinese hamster ovary/HGPRT locus assay. It was not clastogenic in an in vitro analysis of metaphase chromosomes obtained from Chinese hamster ovary cells or in an in vivo mouse micronucleus assay. The hydrolysis product and major human metabolite of levetiracetam (ucb L057) was not mutagenic in the Ames test or the in vitro mouse lymphoma assay.
Impairment Of Fertility
No adverse effects on male or female fertility or reproductive performance were observed in rats at oral doses up to 1800 mg/kg/day (approximately 6 times the maximum recommended human dose on a mg/m 2 or exposure basis).
-
14 CLINICAL STUDIES
The effectiveness of the immediate-release formulation of levetiracetam as adjunctive therapy (added to other antiepileptic drugs) in adults was established in three multicenter, randomized, double-blind, placebo controlled clinical studies in 904 patients who had refractory partial onset seizures with or without secondary generalization for at least two years and had taken two or more classical AEDs.
The effectiveness of levetiracetam extended-release tablets as adjunctive therapy (added to other antiepileptic drugs) was established in one multicenter, randomized, double-blind, placebo-controlled clinical study across 7 countries in patients who had refractory partial onset seizures with or without secondary generalization. Patients enrolled had at least eight partial seizures with or without secondary generalization during the 8-week baseline period and at least two partial seizures in each 4-week interval of the baseline period. Patients were taking a stable dose regimen of at least one and could take a maximum of three AEDs. After a prospective baseline period of 8 weeks, 158 patients were randomized to placebo (N=79) or levetiracetam extended-release tablets (2x500 mg tablets) (N=79) given once daily over a 12-week treatment period.
The primary efficacy endpoint was the percent reduction over placebo in mean weekly frequency of partial onset seizures. The median percent reduction in weekly partial onset seizure frequency from baseline over the treatment period was 46.1% in the levetiracetam extended-release tablets 1000 mg treatment group (N=74) and 33.4% in the placebo group (N=78). The estimated percent reduction over placebo in weekly partial onset seizure frequency over the treatment period was 14.4% (statistically significant).
The relationship between the effectiveness of the same daily dose of levetiracetam extended-release tablets and immediate-release levetiracetam tablets has not been studied and is unknown.
-
16 HOW SUPPLIED/ STORAGE AND HANDLING
16.1 How Supplied
Levetiracetam extended-release tablets 500 mg are white, oblong-shaped, film coated tablets imprinted with “P46” in green on one side. They are supplied in white HDPE bottles containing 300 tablets (NDC: 52817-110-32), and in bottles containing 60 tablets (NDC: 52817-110-60).Levetiracetam extended-release tablets 750 mg are white, oblong-shaped, film coated tablets imprinted with “P47” in green on one side. They are supplied in white HDPE bottles containing 300 tablets (NDC: 52817-111-32), and in bottles containing 60 tablets (NDC: 52817-111-60).
-
17 PATIENT COUNSELING INFORMATION
Patients and caregivers should be informed of the availability of a Medication Guide, and they should be instructed to read the Medication Guide prior to taking levetiracetam extended-release tablets. Medication Guide may also be found in the full prescribing information for levetiracetam extended-release tablets posted on http://www.anchen.com or by calling 1-888-493-0857. Patient should be instructed to take levetiracetam extended-release tablets only as prescribed.
Patients, their caregivers, and families should be counseled that AEDs, including levetiracetam extended-release tablets, may increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
Patients should be advised that levetiracetam extended-release tablets may cause irritability and aggression. In addition, patients should be advised that they may experience changes in behavior that have been seen with other formulations of levetiracetam, which include agitation, anger, anxiety, apathy, depression, hostility, irritability and, in rare cases, psychotic symptoms.
Patients should be instructed to only take levetiracetam extended-release tablets as prescribed and to swallow the tablets whole. They should not be chewed, broken, or crushed. Patients should be advised to notify their physician if they become pregnant or intend to become pregnant during therapy. Patients should be encouraged to enroll in the NAAED Pregnancy Registry if they become pregnant. This registry is collecting information about the safety of antiepileptic drugs during pregnancy. To enroll, patients can call the toll free number 1-888-233-2334.
Patients should be advised that levetiracetam extended-release tablets may cause dizziness and somnolence. Accordingly, patients should be advised not to drive or operate heavy machinery or engage in other hazardous activities until they have gained sufficient experience on levetiracetam extended-release tablets to gauge whether it adversely affects their performance of these activities.
Manufactured for:
TruPharma, LLCC, Tampa, Florida 33609
www.trupharma.com 1-813-444-6299 Rev. 01/2016 -
MEDICATION GUIDE
Levetiracetam Extended-Release Tablets
Read this Medication Guide before you start taking levetiracetam extended-release tablets and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is the most important information I should know about levetiracetam extended-release tablets?
Like other antiepileptic drugs, levetiracetam extended-release tablets may cause suicidal thoughts or actions in a very small number of people, about 1 in 500 people taking it.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
Do not stop levetiracetam extended-release tablets without first talking to a healthcare provider.
- Stopping levetiracetam extended-release tablets suddenly can cause serious problems. Stopping a seizure medicine suddenly can cause seizures that will not stop (status epilepticus).
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
How can I watch for early symptoms of suicidal thoughts and actions?
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
What islevetiracetam extended-release tablets?
Levetiracetam extended-release tablet is a prescription medicine taken by mouth that is used with other medicines to treat partial onset seizures in people 16 years of age and older with epilepsy.
It is not known if levetiracetam extended-release tablets are safe or effective in people under 16 years of age.
Before taking your medicine, make sure you have received the correct medicine. Compare the name above with the name on your bottle and the appearance of your medicine with the description of levetiracetam extended-release tablets provided below. Tell your pharmacist immediately if you think you have been given the wrong medicine.
500 mg levetiracetam extended-release tablets are white, oblong-shaped, film coated tablets marked with "P46" in green on one side.
750 mg levetiracetam extended-release tablets are white, oblong-shaped, film-coated tablets marked with "P47" in green on one side.
What should I tell my healthcare provider before starting levetiracetam extended-release tablets?
Before taking levetiracetam extended-release tablets, tell your healthcare provider about all of your medical conditions, including if you:
- have or have had depression, mood problems or suicidal thoughts or behavior
- have kidney problems
- are pregnant or planning to become pregnant. It is not known if levetiracetam extended-release tablets will harm your unborn baby. You and your healthcare provider will have to decide if you should take levetiracetam extended-release tablets while you are pregnant. If you become pregnant while taking levetiracetam extended-release tablets, talk to your healthcare provider about registering with the North American Antiepileptic Drug Pregnancy Registry. You can enroll in this registry by calling 1-888-233-2334. The purpose of the registry is to collect information about the safety of levetiracetam extended-release tablets and other antiepileptic medicine during pregnancy.
- are breast feeding. Levetiracetam can pass into your milk and may harm your baby. You and your healthcare provider should discuss whether you should take levetiracetam extended-release tablets or breast feed; you should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements. Do not start a new medicine without first talking with your healthcare provider.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine.
How should I take levetiracetam extended-release tablets?
Take levetiracetam extended-release tablets exactly as prescribed.
- Your healthcare provider will tell you how much levetiracetam extended-release tablets to take and when to take it. levetiracetam extended-release tablets are usually taken once a day. Take levetiracetam extended-release tablets at the same time each day.
- Your healthcare provider may change your dose. Do not change your dose without talking to your healthcare provider.
- Take levetiracetam extended-release tablets with or without food.
- Swallow the tablets whole. Do not chew, break, or crush tablets.
- If you miss a dose of levetiracetam extended-release tablets, take it as soon as you remember. If it is almost time for your next dose, just skip the missed dose. Take the next dose at your regular time. Do not take two doses at the same time.
- If you take too much levetiracetam extended-release tablets, call your local Poison Control Center or go to the nearest emergency room right away.
What should I avoid while taking levetiracetam extended-release tablets?
Do not drive, operate machinery or do other dangerous activities until you know how levetiracetam extended-release tablets affect you. levetiracetam extended-release tablets may make you dizzy or sleepy.
What are the possible side effects of levetiracetam extended-release tablets?
- See “What is the most important information I should know about levetiracetam extended-release tablets?”
Levetiracetam extended-release tablets can cause serious side effects.
Call your healthcare provider right away if you have any of these symptoms:
- mood and behavior changes such as aggression, agitation, anger, anxiety, apathy, mood swings, depression, hostility, and irritability. A few people may get psychotic symptoms such as hallucinations (seeing or hearing things that are really not there), delusions (false or strange thoughts or beliefs) and unusual behavior.
- extreme sleepiness, tiredness, and weakness
- problems with muscle coordination (problems walking and moving)
Common side effects seen in people who take levetiracetam extended-release tablets and other formulations of levetiracetam include:
- sleepiness
- weakness
- dizziness
- infection
These side effects can happen at any time but happen more often within the first 4 weeks of treatment.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of levetiracetam extended-release tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may also report side effects to Anchen Pharmaceuticals at 1-888-493-0857 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How should I store levetiracetam extended-release tablets?
Store levetiracetam extended-release tablets at room temperature, 20 to 25ºC (68 to 77ºF) [see USP Controlled Room Temperature] away from heat and light.
Keep levetiracetam extended-release tablets and all medicines out of the reach of children.General information about levetiracetam extended-release tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use levetiracetam extended-release tablets for a condition for which it was not prescribed. Do not give levetiracetam extended-release tablets to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about levetiracetam extended-release tablets. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about levetiracetam extended-release tablets that is written for health professionals.
What are the ingredients of levetiracetam extended-release tablets?
Levetiracetam extended-release tablet active ingredient: levetiracetam
Inactive ingredients: alcohol (anhydrous), colloidal silicon dioxide, ethylcellulose, magnesium stearate, microcrystalline cellulose and talc. The imprinting ink contains ammonium hydroxide, D&C yellow #10, FD&C blue #1, isopropyl alcohol, n-butyl alcohol, propylene glycol, shellac and titanium dioxide. Levetiracetam extended-release does not contain lactose or gluten.
Rx only
This Medication Guide has been approved by the US Food and Drug Administration.
Manufactured for:
TruPharma, LLCC
Tampa, Florida 33609www.trupharma.com
1-813-444-6299Rev. 01/2016
-
Levetiracetam Extended-Release Tablets 750mg
TruPharma
NDC: 52817-111-60
ONCE DAILY DOSING
Levetiracetam
Extended-Release Tablets
750 mg
Dispense accompanying Medication Guide to each patient
60 Tablets
Rx Only

-
Levetiracetam Extended-Release Tablets 500 mg
TruPharma
NDC: 52817-110-60
ONCE DAILY DOSING
Levetiracetam
Extended-Release Tablets
500 mg
Dispense accompanying Medication Guide to each patient
60 Tablets
Rx Only

-
INGREDIENTS AND APPEARANCE
LEVETIRACETAM EXTENDED-RELEASE
levetiracetam tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 52817-110 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVETIRACETAM (UNII: 44YRR34555) (LEVETIRACETAM - UNII:44YRR34555) LEVETIRACETAM 500 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) TALC (UNII: 7SEV7J4R1U) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) AMMONIA (UNII: 5138Q19F1X) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ISOPROPYL ALCOHOL (UNII: ND2M416302) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score no score Shape OVAL (oblong) Size 17mm Flavor Imprint Code BOCA696 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 52817-110-60 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2018 2 NDC: 52817-110-32 300 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA201464 06/01/2018 LEVETIRACETAM EXTENDED-RELEASE
levetiracetam tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 52817-111 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVETIRACETAM (UNII: 44YRR34555) (LEVETIRACETAM - UNII:44YRR34555) LEVETIRACETAM 750 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) TALC (UNII: 7SEV7J4R1U) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) AMMONIA (UNII: 5138Q19F1X) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ISOPROPYL ALCOHOL (UNII: ND2M416302) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score no score Shape OVAL (oblong) Size 20mm Flavor Imprint Code BOCA697 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 52817-111-60 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2018 2 NDC: 52817-111-32 300 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA201464 06/01/2018 Labeler - Trupharma, LLC (078533947) Registrant - Pharmadax, Inc (656535874) Establishment Name Address ID/FEI Business Operations Pharmadax (Foshan) Co.,Ltd 421295980 manufacture(52817-110, 52817-111)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.