CITY SUNSCREEN SERUM BROAD SPECTRUM SPF 30 SUPERGOOP- homosalate, octisalate, avobenzene, octocrylene cream
City Sunscreen serum Broad Spectrum SPF 30 by
Drug Labeling and Warnings
City Sunscreen serum Broad Spectrum SPF 30 by is a Otc medication manufactured, distributed, or labeled by Taylor James, Ltd., Baxter Laboratories Pty. Ltd. . Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
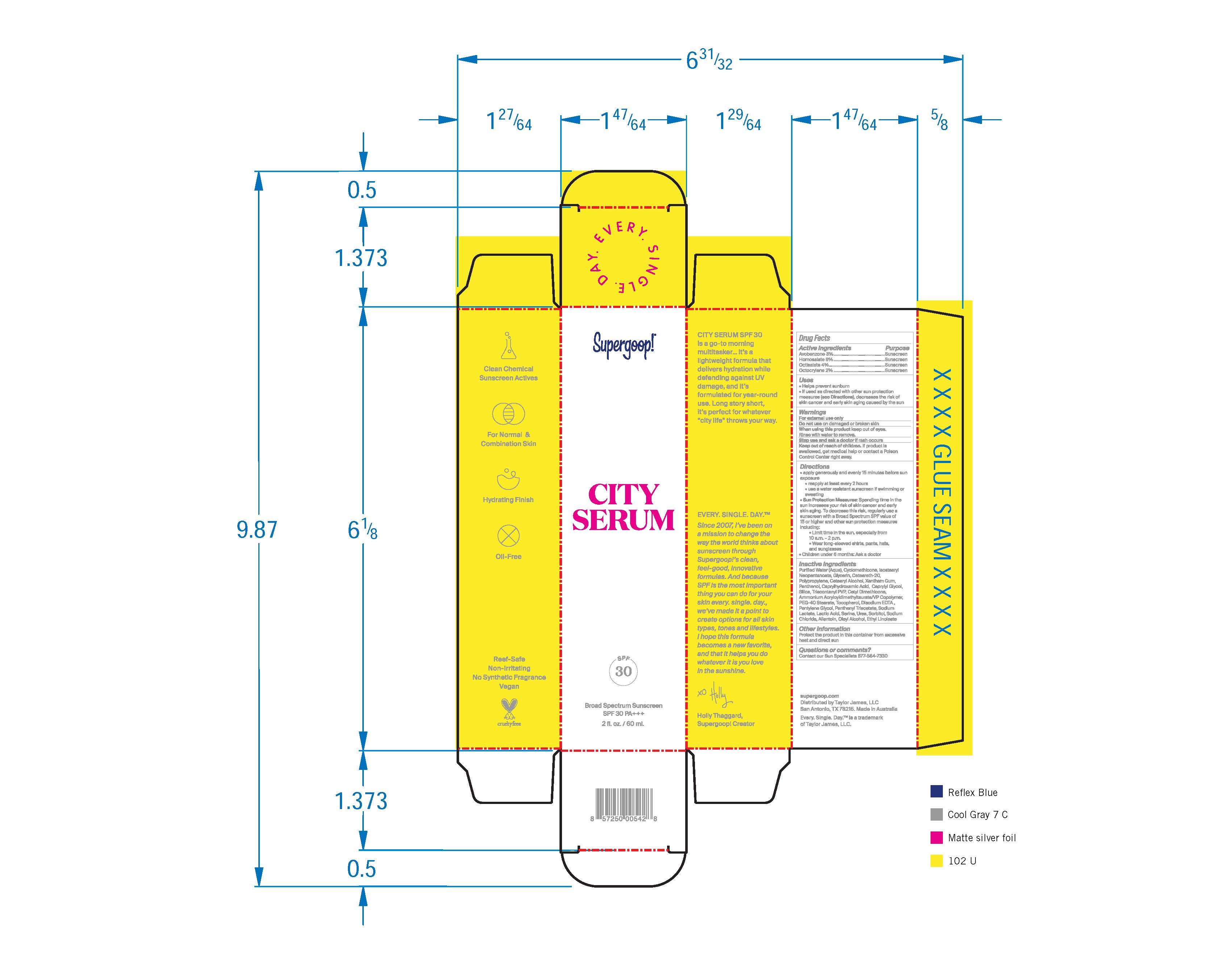
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
Directions: Apply liberally 15 minutes before sun exposure
Use a water resistant sunscreen if swimming or sweating
Reapply at least every 2 hours
children under 6 months: ask a doctor
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreenwith Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
Limit time in the sun,especially from 10 a.m. -2p.m.
Wear long-sleeved shirts, pants, hats, and sunglasses
-
INACTIVE INGREDIENT
Inactive Ingredients Purified water (Aqua), Cyclometicone, Isostearyl Neopentanoate, Glycerin, Ceteareth-20, Polypropylene, Cetearyl Alcohol, Xanthan Gum, d-Panthenol, Octanohydroxamic acid, Caprylyl Glycol, Silica, Triacontanyl PVP, Cetyl Dimethicone, Ammonium Acryloyldimethyltaurate/VP Copolymer, PEG-40 Stearate, Tocopheryl, Disodium EDTA, Pentylene Glycol, Pantheyl Triacetate, Sodium Lactate, Lactic Acid, Serine, Urea, Sorbitol, Sodium Chloride, Allantoin, Oleyl Alcohol, Ethyl Linoleate.
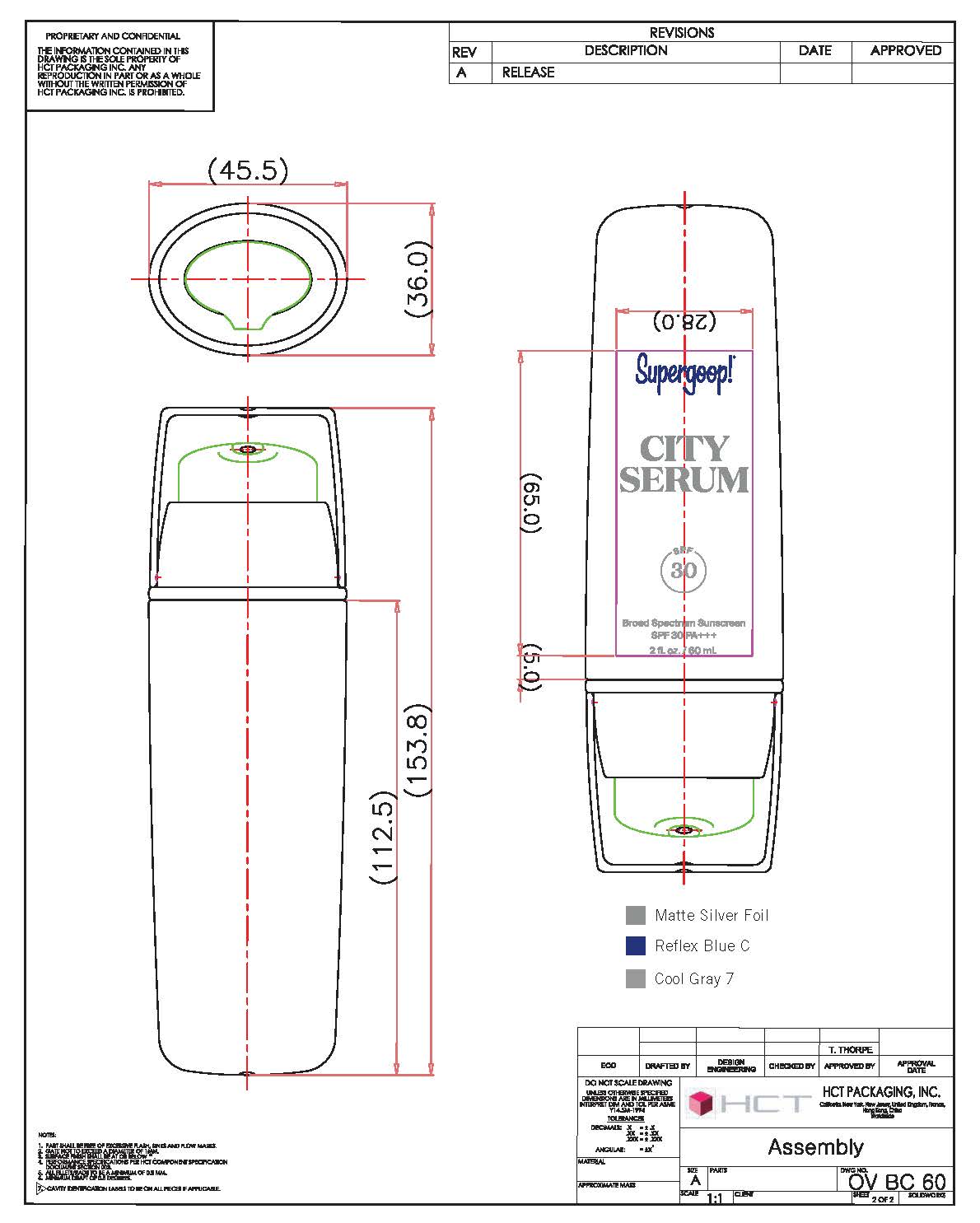
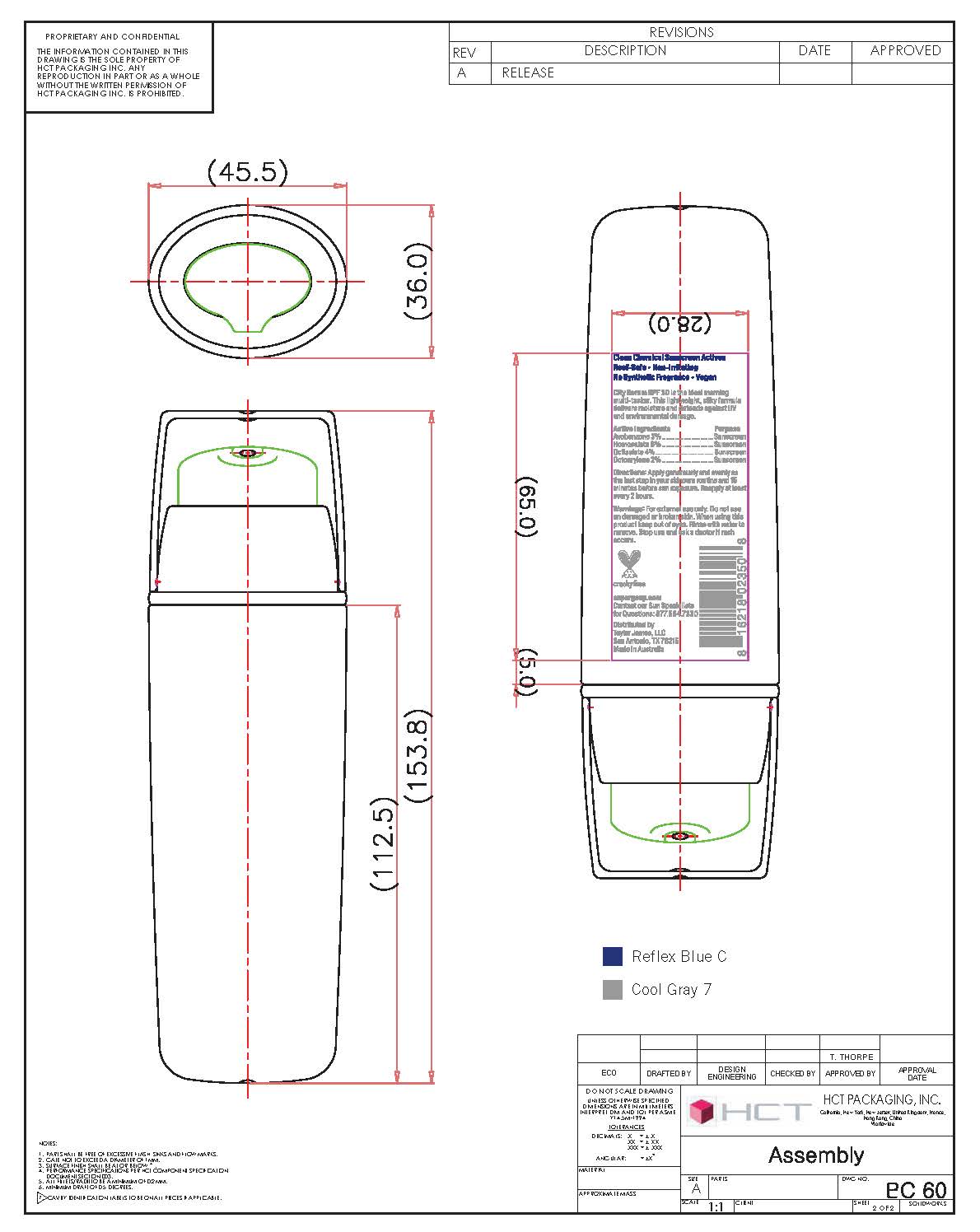
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CITY SUNSCREEN SERUM BROAD SPECTRUM SPF 30 SUPERGOOP
homosalate, octisalate, avobenzene, octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 75936-111 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 8 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 4 g in 100 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 2 g in 100 mL Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE (UNII: NMQ347994Z) GLYCERIN (UNII: PDC6A3C0OX) XANTHAN GUM (UNII: TTV12P4NEE) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLYL GLYCOL (UNII: 00YIU5438U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) POLYOXYL 40 STEARATE (UNII: 13A4J4NH9I) EDETATE DISODIUM (UNII: 7FLD91C86K) PENTYLENE GLYCOL (UNII: 50C1307PZG) SODIUM LACTATE (UNII: TU7HW0W0QT) LACTIC ACID (UNII: 33X04XA5AT) SERINE (UNII: 452VLY9402) UREA (UNII: 8W8T17847W) SORBITOL (UNII: 506T60A25R) SODIUM CHLORIDE (UNII: 451W47IQ8X) ALLANTOIN (UNII: 344S277G0Z) OLEYL ALCOHOL (UNII: 172F2WN8DV) ETHYL LINOLEATE (UNII: MJ2YTT4J8M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 75936-111-01 50 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 12/07/2010 2 NDC: 75936-111-02 20 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 12/07/2010 3 NDC: 75936-111-05 10 mL in 1 TUBE; Type 0: Not a Combination Product 12/07/2010 4 NDC: 75936-111-06 60 mL in 1 TUBE; Type 0: Not a Combination Product 12/07/2010 5 NDC: 75936-111-07 3 mL in 1 PACKET; Type 0: Not a Combination Product 12/07/2010 6 NDC: 75936-111-08 15 mL in 1 TUBE; Type 0: Not a Combination Product 07/19/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 12/07/2010 Labeler - Taylor James, Ltd. (033381850) Establishment Name Address ID/FEI Business Operations Baxter Laboratories Pty. Ltd. 740537709 manufacture(75936-111)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.