Sky Regular Strength Gas Relief Chewable Tablets
Gas relief by
Drug Labeling and Warnings
Gas relief by is a Otc medication manufactured, distributed, or labeled by McKesson Corporation dba SKY Packaging, Guardian Drug Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
GAS RELIEF - simethicone tablet, chewable
McKesson Corporation dba SKY Packaging
----------
Sky Regular Strength Gas Relief Chewable Tablets
KEEP OUT OF REACH OF CHILDREN
In case of overdose, get medical help or contact a Poison Control Center immediately.
DIRECTIONS
- chew 2 to 4 tablets thoroughly as needed after meals and at bedtime
- do not exceed 6 tablets per day unless directed by a physician
OTHER INFORMATION
- do not use if inner seal is broken or missing
- store at room temperature
- avoid high humidity and excessive heat, above 40oC (104oF)
INACTIVE INGREDIENT
compressible sugar, dextrose, flavor, magnesium stearate, maltodextrin, microcrystalline cellulose, sorbitol.
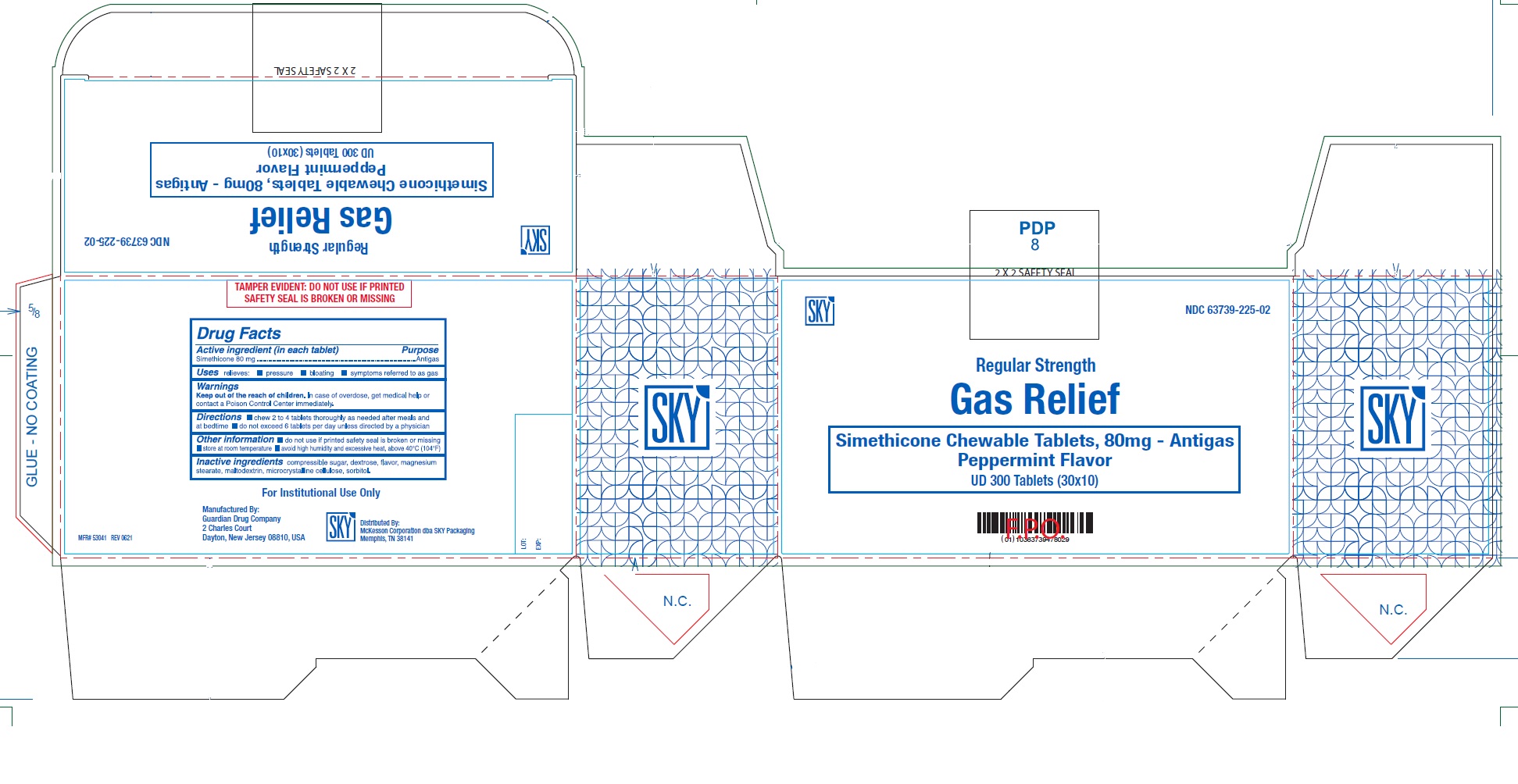
PRINCIPAL DISPLAY PANEL
NDC: 63739-225-02
SKY
Regular Strength
Gas Relief
Simethicone Chewable Tablets, 80 mg - Antigas
Peppermint Flavor
UD 300 Tablets (30x10)
For Institutional Use Only
| GAS RELIEF
simethicone tablet, chewable |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - McKesson Corporation dba SKY Packaging (140529962) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Guardian Drug Company | 119210276 | MANUFACTURE(63739-225) | |
Revised: 12/2025
Document Id: f034d882-f5fd-4656-9628-12f48b6ae632
Set id: 619f1270-59bb-4214-896c-230bd7f6d6d7
Version: 3
Effective Time: 20251201
Trademark Results [Gas relief]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 GAS RELIEF 85443589 not registered Dead/Abandoned |
Traditional Medicinals 2011-10-10 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.