JUXTAPID- lomitapide mesylate capsule
Juxtapid by
Drug Labeling and Warnings
Juxtapid by is a Prescription medication manufactured, distributed, or labeled by Chiesi USA, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use JUXTAPID safely and effectively. See full prescribing information for JUXTAPID.
JUXTAPID® (lomitapide) capsules, for oral use
Initial U.S. Approval: 2012WARNING: RISK OF HEPATOTOXICITY
See full prescribing information for complete boxed warning.
JUXTAPID can cause elevations in transaminases (5.1).
- Measure alanine and aspartate aminotransferases (ALT, AST), alkaline phosphatase, and total bilirubin before initiating treatment and then ALT and AST regularly as recommended (2.5, 5.1).
- During treatment, adjust the dose of JUXTAPID if the ALT or AST is ≥3 times the upper limit of normal (ULN) (2.5, 5.1).
- Discontinue JUXTAPID for clinically significant liver toxicity (2.5, 5.1).
JUXTAPID increases hepatic fat (hepatic steatosis) with or without concomitant increases in transaminases (5.1).
- Hepatic steatosis associated with JUXTAPID may be a risk factor for progressive liver disease, including steatohepatitis and cirrhosis (5.1).
Because of the risk of hepatotoxicity, JUXTAPID is available only through a restricted program called the JUXTAPID REMS Program (5.2). Prescribe JUXTAPID only to patients with a clinical or laboratory diagnosis consistent with homozygous familial hypercholesterolemia (HoFH). The safety and effectiveness of JUXTAPID have not been established in patients with hypercholesterolemia who do not have HoFH (1).
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
JUXTAPID is a microsomal triglyceride transfer protein inhibitor indicated as an adjunct to a low-fat diet and exercise and other low-density lipoprotein cholesterol (LDL-C) therapies, to reduce LDL-C in adult and pediatric patients aged 2 years and older with HoFH. (1).
DOSAGE AND ADMINISTRATION
- Before treatment, measure ALT, AST, alkaline phosphatase, and total bilirubin; obtain a negative pregnancy test in females of reproductive potential; initiate a low-fat diet supplying <20% of energy from fat or less than 30 grams of fat, whichever is less. (2.1).
- The recommended initiation dosage is (2.2):
- 2 mg for patients aged 2 to 15 years.
- 5 mg for patients aged 16 years and older.
- Follow the titration schedule presented in Table 1 according to the patient's age. Select the dosage based on the recommended target LDL-C, safety, and tolerability (2.2).
- For pediatric patients, if a patient crosses over into the next age category, escalate the dose of JUXTAPID up to the maximum recommended dose applicable for the new age group (2.2).
- Measure transaminases prior to any dosage increase. If transaminases are abnormal, reduce or withhold dosing of JUXTAPID and monitor as recommended (5.1).
-
Table 1: Recommended JUXTAPID Dosage and Titration Schedule Age group (years) JUXTAPID Dose 2 mg 5 mg 10 mg 20 mg 40 mg 60 mg W:Weeks; ---: Not a recommended dosage; 1Maximum recommended dosage. 2 to 10 W 0 to 8 W 9 to 12 W 13 to 16 W 17 and beyond1 --- --- 11 to 15 W 0 to 4 W 5 to 8 W 9 to 12 W 13 to 16 17 and beyond1 --- 16 to 17 --- W 0 to 4 W 5 to 8 W 9 to 12 13 and beyond1 --- 18 and older --- W 0 to 2 W 3 to 6 W 7 to 10 W 11 to 14 15 and beyond1 - Due to reduced absorption of fat-soluble vitamins/fatty acids: Take daily vitamin E [400 international units (IU) for patients aged 9 years and older, or 200 IU for patients aged 2 to 8 years old] and linoleic acid (200 mg), alpha-linolenic acid (210 mg), eicosapentaenoic acid (110 mg), and docosahexaenoic acid (80 mg) supplements (2.2).
- Take orally once daily, whole, with water and without food, at least 2 hours after evening meal. If unable to swallow intact capsule, patients may sprinkle capsule contents onto a tablespoon of apple sauce or mashed banana (2.3).
- Refer to the Full PI for dosage modifications based on elevated transaminases (2.5); in patients with end-stage renal disease on dialysis require dose adjustment (2.6), and in patients with baseline mild hepatic impairment require dose adjustment (2.7).
DOSAGE FORMS AND STRENGTHS
Capsules: 2 mg, 5 mg, 10 mg, 20 mg, and 30 mg (3).
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. Discontinue JUXTAPID if pregnancy detected (5.3).
- Gastrointestinal adverse reactions occur in 93% of adult and 72% of pediatric patients and could affect absorption of concomitant oral medications (5.5).
ADVERSE REACTIONS
Most common adverse reactions in adult patients (incidence ≥10%) are diarrhea, nausea, dyspepsia, vomiting, and abdominal pain (6.1). Most common adverse reactions in pediatric patients aged 5 to 17 years old (incidence ≥15%) are abdominal pain, alanine aminotransferase increased, aspartate aminotransferase increased, diarrhea, and vomiting (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Chiesi Farmaceutici S.p.A. at 1-888-661-9260 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- CYP3A4 inhibitors increase exposure to lomitapide. Strong and moderate CYP3A4 inhibitors are contraindicated with JUXTAPID. Patients must avoid grapefruit juice (7.1).
- When administered with weak CYP3A4 inhibitors, the dose of JUXTAPID should be decreased by half. Follow the titration recommendations provided in the Full PI (7.2).
- Warfarin: Lomitapide increases plasma concentrations of warfarin. Monitor international normalized ratio (INR) regularly, especially with JUXTAPID dose adjustment (7.3).
- Simvastatin and lovastatin exposure increase with JUXTAPID. Limit dose when co-administered with JUXTAPID due to myopathy risk (7.4).
- P-glycoprotein (P-gp) Substrates: Consider dose reduction of P-gp substrate because of possible increased absorption with JUXTAPID (7.5).
- Bile Acid Sequestrants: Separate JUXTAPID dosing by at least 4 hours (7.6).
USE IN SPECIFIC POPULATIONS
- Lactation: Breastfeeding not recommended (8.2).
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 2/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISK OF HEPATOTOXICITY
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Prior to Initiation
2.2 Recommended Dosage
2.3 Administration
2.4 Dosage Modifications for Cytochrome P450 3A4 Inhibitors
2.5 Dosage Modification Based on Elevated Transaminases
2.6 Recommended Dosage in Patients with Renal Impairment
2.7 Recommended Dosage in Patients with Baseline Hepatic Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Hepatotoxicity
5.2 JUXTAPID REMS Program
5.3 Embryo-Fetal Toxicity
5.4 Reduced Absorption of Fat-Soluble Vitamins and Serum Fatty Acids
5.5 Gastrointestinal Adverse Reactions
5.6 Concomitant Use of CYP3A4 Inhibitors
5.7 Risk of Myopathy with Concomitant Use of Simvastatin or Lovastatin
5.8 Risk of Supratherapeutic or Subtherapeutic Anticoagulation with Warfarin
5.9 Risk of Malabsorption with Rare Hereditary Disorders of Galactose Intolerance
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Moderate and Strong CYP3A4 Inhibitors
7.2 Weak CYP3A4 Inhibitors
7.3 Warfarin
7.4 Simvastatin and Lovastatin
7.5 P-glycoprotein Substrates
7.6 Bile Acid Sequestrants
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED / STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISK OF HEPATOTOXICITY
JUXTAPID can cause elevations in transaminases. In the adult clinical trial, 10 (34%) of the 29 patients treated with JUXTAPID had at least one elevation in alanine aminotransferase (ALT) or aspartate aminotransferase (AST) ≥3 times the upper limit of normal (ULN). There were no concomitant clinically meaningful elevations of total bilirubin, international normalized ratio (INR), or alkaline phosphatase. In the pediatric clinical trial (5 to 17 years of age), 6 (14%) of the 43 patients experienced elevations in ALT and/or AST ≥ 3 times ULN. No concomitant clinically meaningful elevations in total bilirubin or alkaline phosphatase were observed [see Warnings and Precautions (5.1)].
JUXTAPID also increases hepatic fat, with or without concomitant increases in transaminases. The median absolute increase in hepatic fat in adult patients was 6% after both 26 and 78 weeks of treatment, from 1% at baseline, measured by magnetic resonance spectroscopy (MRS). The median absolute increase in hepatic fat in pediatric patients aged 5 to 17 years was 4% after 24 weeks and 104 weeks of treatment, from 3% at baseline, measured by nuclear magnetic resonance (NMR). Hepatic steatosis associated with JUXTAPID treatment may be a risk factor for progressive liver disease, including steatohepatitis and cirrhosis [see Warnings and Precautions (5.1)].
Measure ALT, AST, alkaline phosphatase, and total bilirubin before initiating treatment and then ALT and AST regularly as recommended. During treatment, adjust the dose of JUXTAPID if the ALT or AST are ≥3 times ULN. Discontinue JUXTAPID for clinically significant liver toxicity [see Dosage and Administration (2.7) and Warnings and Precautions (5.1)].
Because of the risk of hepatotoxicity, JUXTAPID is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the JUXTAPID REMS Program [see Warnings and Precautions (5.2)]. Prescribe JUXTAPID only to patients with a clinical or laboratory diagnosis consistent with HoFH. The safety and effectiveness of JUXTAPID have not been established in patients with hypercholesterolemia who do not have HoFH [see Indications and Usage (1)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Prior to Initiation
Before beginning treatment with JUXTAPID:
- Liver function tests: Measure transaminases (ALT, AST), alkaline phosphatase, and total bilirubin [see Warnings and Precautions (5.1)];
- Pregnancy testing: Obtain a negative pregnancy test in females of reproductive potential prior to initiating treatment with JUXTAPID [see Contraindications (4), Warnings and Precautions (5.3), and Use in Specific Populations (8.1, 8.3)];
- Dietary counseling: Initiate a low-fat diet supplying <20% of energy from fat or less than 30 grams of fat, whichever is less [see Warnings and Precautions (5.5)].
2.2 Recommended Dosage
Initiation Dosage
The recommended initiation dosage is
- 2 mg for patients aged 2 to 15 years.
- 5 mg for patients aged 16 years and older.
Dosage Titration
- Follow the titration schedule presented in Table 1 according to the patient's age. Select the dosage based on target LDL-C level recommended in current HoFH treatment guidance, safety, and tolerability.
- For pediatric patients, if a patient crosses over into the next age group, escalate the dosage of JUXTAPID to dosage recommended for the new age group.
- Measure transaminases prior to any dosage increase. If transaminases are abnormal, reduce or withhold dosing of JUXTAPID and monitor as recommended [see Warnings and Precautions (5.1)].
Table 1: Recommended JUXTAPID Dosage and Titration Schedule Age Group JUXTAPID Dosage and Titration Schedule 2 mg 5 mg 10 mg 20 mg 40 mg 60 mg - * Maximum recommended dosage. Select the dosage based on target LDL-C level recommended in current HoFH treatment guidance, safety and tolerability.
2 to 10 years Weeks 0 to 8 Weeks 9 to 12 Weeks 13 to 16 Weeks 17 and beyond* Not recommended Not recommended 11 to 15 years Weeks 0 to 4 Weeks 5 to 8 Weeks 9 to 12 Weeks 13 to 16 Weeks 17 and beyond* Not recommended 16 to l7 years Not recommended Weeks 0 to 4 Weeks 5 to 8 Weeks 9 to 12 Weeks 13 and beyond* Not recommended 18 years and older Not recommended Weeks 0 to 2 Weeks 3 to 6 Weeks 7 to 10 Weeks 11 to 14 Week 15 and beyond* Maximum Recommended Dosage
The maximum recommended dosage is:
- 20 mg for patients aged 2 to 10 years.
- 40 mg for patients aged 11 to 17 years.
- 60 mg for patients aged 18 years and older.
Nutritional Supplementation
To reduce the risk of developing a fat-soluble nutrient deficiency due to JUXTAPID's mechanism of action in the small intestine [see Warnings and Precautions (5.4)], administer JUXTAPID with daily nutritional supplements. See Table 2 for nutritional supplementation recommendations according to age.
Table 2: Nutritional Supplementation Recommendations for Adult and Pediatric Patients Aged 2 Years and Older Receiving JUXTAPID Age Group Vitamin E Essential Fatty Acids 2 to 8 years 200 International Units (IU)
(134 mg d-alpha-tocopherol or 90 mg dl-alpha-tocopherol)- Linoleic acid: 200 mg
- Alpha-linolenic acid (ALA): 210 mg
- Eicosapentaenoic acid (EPA): 110 mg
- Docosahexaenoic acid (DHA): 80 mg
9 years and older 400 IU
(268 mg d-alpha-tocopherol or 180 mg dl-alpha-tocopherol)2.3 Administration
- Take JUXTAPID orally once daily with a glass of water, without food, at least 2 hours after the evening meal. Administration with food may increase the risk of gastrointestinal adverse reactions [see Warnings and Precautions (5.5)].
-
Swallow JUXTAPID capsules whole:
- If a patient is unable to swallow the intact capsule(s), open the capsule(s) and sprinkle the contents on 1 tablespoon of apple sauce or mashed banana.
- For younger patients who require a smaller spoon for administration, sprinkle the capsule contents onto 1 tablespoon of apple sauce or mashed banana, and administer the entire amount using the smaller spoon.
- Administer JUXTAPID at least 4 hours before or 4 hours after administration of a bile acid sequestrant [see Drug Interactions (7.6)].
2.4 Dosage Modifications for Cytochrome P450 3A4 Inhibitors
Moderate or Strong Cytochrome P450 3A4 Inhibitors
JUXTAPID is contraindicated with concomitant use of moderate and strong cytochrome P450 3A4 (CYP3A4) inhibitors [see Contraindications (4) and Drug Interactions (7.1)].
Weak CYP3A4 Inhibitors (With or Without Oral Contraceptives)
-
JUXTAPID Initiation in Patients Treated with a Stable Dosage of a Weak CYP3A4 Inhibitor:
- Follow the recommended dosage and titration schedule described in Table 1. Consider target LDL-C level, safety, and tolerability when selecting a maintenance dosage.
- Refer to the maximum recommended dose of JUXTAPID described in Table 3.
Weak CYP3A4 Inhibitor Initiation in Patients Treated with a Stable Dosage of JUXTAPID:
- Decrease the dosage of JUXTAPID by half.
- The dosage of JUXTAPID may be further titrated according to target LDL-C level, safety, and tolerability, not to exceed the maximum recommended dose of JUXTAPID described in Table 3.
Concomitant Oral contraceptive use Only
-
JUXTAPID Initiation in Patients Treated with a Stable Dosage of an Oral Contraceptive:
- Follow the recommended dosage and titration schedule described in Table 1.
- Consider target LDL-C level, safety, and tolerability when selecting a maintenance dosage, not to exceed the maximum recommended dose of JUXTAPID described in Table 3.
-
Oral contraceptive Initiation in Patients Treated with a Stable Dosage of JUXTAPID:
- Decrease the dose of JUXTAPID by one-third.
- The dosage of JUXTAPID may be further titrated according to target LDL-C level, safety, and tolerability, not to exceed the maximum recommended dose of JUXTAPID described in Table 3.
Table 3: Recommended Maximum JUXTAPID Dosage with Concomitant Weak CYP3A4 inhibitor or an Oral Contraceptive Age group Maximum JUXTAPID Dosage Weak CYP3A4 inhibitor Oral Contraceptive* - * Oral contraceptives are not indicated before menarche.
2 to 10 years 10 mg 15 mg 11 to 17 years 20 mg 30 mg 18 years and older 30 mg 40 mg 2.5 Dosage Modification Based on Elevated Transaminases
Table 4 summarizes recommendations for dosage adjustment and monitoring for patients who develop elevated transaminases during treatment with JUXTAPID [see Warnings and Precautions (5.1)].
Table 4: Dosage Adjustment and Monitoring for Patients with Elevated Transaminases ALT OR AST TREATMENT AND MONITORING RECOMMENDATIONS* - * Recommendations based on an age and gender appropriate ULN.
≥3 times and <5 times ULN - Confirm elevation with a repeat measurement within one week.
- If confirmed, reduce the dosage to the last tolerated dosage, and obtain additional liver-related tests if not already measured (such as alkaline phosphatase, total bilirubin, and INR).
- Repeat tests weekly and withhold dosing if there are signs of abnormal liver function (increase in bilirubin or INR), if transaminase levels rise above 5 times the ULN, or if transaminase levels do not fall below 3 times ULN within approximately 4 weeks. In these cases of persistent or worsening abnormalities, also investigate to identify the probable cause.
- If resuming JUXTAPID after transaminases resolve to <3 times ULN, consider reducing the dose to the last tolerated dosage, and monitor liver-related tests more frequently.
≥5 times ULN - Withhold dosing, obtain additional liver-related tests if not already measured (such as alkaline phosphatase, total bilirubin, and INR), and investigate to identify the probable cause.
- If resuming JUXTAPID after transaminases resolve to <3 times ULN, reduce the dose to the last tolerated dosage, and monitor liver-related tests more frequently.
If transaminase elevations are accompanied by clinical symptoms of liver injury (such as nausea, vomiting, abdominal pain, fever, jaundice, lethargy, flu-like symptoms), increases in bilirubin ≥2 times ULN, or active liver disease, discontinue treatment with JUXTAPID and investigate to identify the probable cause [see Warnings and Precautions (5.1)].
2.6 Recommended Dosage in Patients with Renal Impairment
The recommended dosage of JUXTAPID in patients with end-stage renal disease (eGFR <15 mL/min/1.73m2) receiving hemodialysis is described in Table 5. JUXTAPID has not been studied in patients with mild, moderate, or severe renal impairment, including those with end stage renal disease not receiving dialysis [see Use in Specific Populations (8.6)].
Table 5: Dosage Adjustment for Patients with End Stage Renal Disease Receiving Hemodialysis Age GROUP MAXIMUM RECOMMENDED DAILY DOSAGE 2 to 10 years 15 mg 11 to 17 years 30 mg 18 years and older 40 mg 2.7 Recommended Dosage in Patients with Baseline Hepatic Impairment
JUXTAPID is contraindicated in patients with moderate or severe hepatic impairment (based on Child-Pugh category B or C) and patients with active liver disease, including unexplained persistent elevations of serum transaminases [see Warnings and Precautions (5.6)]. Table 6 summarizes the dosage adjustment for patients with baseline mild hepatic impairment (Child-Pugh A) [see Use in Specific Populations (8.7)].
Table 6: Dosage Adjustment for Patients with Baseline Mild Hepatic Impairment AGE GROUP MAXIMUM RECOMMENDED DAILY DOSAGE 2 to 10 years 15 mg 11 to 17 years 30 mg 18 years and older 40 mg -
3 DOSAGE FORMS AND STRENGTHS
Capsules:
- 2 mg: Grey/grey hard gelatin capsule printed with black ink "A733" and "2 mg"
- 5 mg: Orange/orange hard gelatin capsule printed with black ink "A733" and "5 mg"
- 10 mg: Orange/white hard gelatin capsule printed with black ink "A733" and "10 mg"
- 20 mg: White/white hard gelatin capsule printed with black ink "A733" and "20 mg"
- 30 mg: Orange/yellow hard gelatin capsule printed with black ink "A733" and "30 mg"
-
4 CONTRAINDICATIONS
JUXTAPID is contraindicated in the following conditions:
- Pregnancy [see Warnings and Precautions (5.3) and Use in Specific Populations (8.1)].
- Concomitant administration of JUXTAPID with moderate or strong CYP3A4 inhibitors, as this can increase JUXTAPID exposure [see Warnings and Precautions (5.6), Drug Interactions (7.1), and Clinical Pharmacology (12.3)].
- Patients with moderate or severe hepatic impairment (based on Child-Pugh category B or C) and patients with active liver disease, including unexplained persistent elevations of serum transaminases [see Warnings and Precautions (5.1) and Use in Specific Populations (8.7)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Hepatotoxicity
JUXTAPID can cause elevations in transaminases and hepatic steatosis in adults and pediatric patients, as described below [see Warnings and Precautions (5.2)]. JUXTAPID may induce steatohepatitis, which can progress to cirrhosis over several years. Clinical trials of JUXTAPID for HoFH would have been unlikely to detect this adverse outcome given their size and duration [see Clinical Studies (14)].
Elevation of Transaminases
Elevations in transaminases (ALT and/or AST) are associated with JUXTAPID.
In the 78-week adult clinical trial, 10 (34%) of the 29 patients with HoFH had at least one elevation in ALT or AST ≥3 times ULN, and 4 (14%) of the patients had at least one elevation in ALT or AST ≥5 times ULN. There were no concomitant or subsequent clinically meaningful elevations in bilirubin, INR, or alkaline phosphatase [see Adverse Reactions (6.1)]. No patients discontinued prematurely because of elevated transaminases. Among the 19 patients who subsequently enrolled in the adult HoFH extension trial, one discontinued because of increased transaminases that persisted despite several dose reductions, and one temporarily discontinued because of markedly elevated transaminases (ALT 24 times ULN, AST 13 times ULN) that had several possible causes, including a drug-drug interaction between JUXTAPID and the strong CYP3A4 inhibitor clarithromycin [see Drug Interactions (7.1)].
In the 104-week open-label trial in pediatric patients aged 5 to 17 years with HoFH exposed to JUXTAPID, 6 (14%) of the 43 patients with HoFH had at least one elevation in ALT and/or AST ≥3 times ULN, including 2 (5%) patients who had at least one elevation in ALT ≥5 times ULN. No patients discontinued treatment because of increased transaminases, although some patients required dose interruptions or reductions for management of liver transaminase elevations.
Monitoring of Transaminases
Before initiating JUXTAPID and during treatment, monitor transaminases as recommended in Table 7.
Table 7: Recommendations for Monitoring Transaminases TIME RECOMMENDATIONS Before initiating treatment - Measure ALT, AST, alkaline phosphatase, and total bilirubin.
- If abnormal, consider initiating JUXTAPID only after an appropriate work-up and the baseline abnormalities have been explained or resolved.
- JUXTAPID is contraindicated in patients with moderate or severe hepatic impairment, or active liver disease, including unexplained persistent elevations of serum transaminases [see Contraindications (4)].
During the first year - Measure liver-related tests (ALT and AST, at a minimum) prior to each increase in dose or monthly, whichever occurs first.
After the first year - Measure liver-related tests (ALT and AST, at a minimum) at least every 3 months and before any increase in dose.
At any time during treatment - If transaminases are >1 and <3 times ULN, no dose modification is required. Continue routine monitoring of liver-related tests (once monthly during the first year of treatment and every 3 months thereafter).
- If transaminases are ≥3 times ULN, reduce or withhold dosing of JUXTAPID and monitor as recommended [see Dosage and Administration (2.5)].
- Discontinue JUXTAPID for persistent or clinically significant elevations.
- If transaminase elevations are accompanied by clinical symptoms of liver injury (such as nausea, vomiting, abdominal pain, fever, jaundice, lethargy, flu-like symptoms), increases in bilirubin ≥2 times ULN, or active liver disease, discontinue treatment with JUXTAPID and identify the probable cause.
Hepatic Steatosis
JUXTAPID increases hepatic fat, with or without concomitant increases in transaminases. Hepatic steatosis is a risk factor for progressive liver disease, including steatohepatitis and cirrhosis. The long-term consequences of hepatic steatosis associated with JUXTAPID treatment are unknown. During the HoFH clinical trial conducted in adults, the median absolute increase in hepatic fat was 6% after both 26 weeks and 78 weeks of treatment, from 1% at baseline, measured by magnetic resonance spectroscopy (MRS) [see Adverse Reactions (6.1)]. Clinical data suggest that hepatic fat accumulation is reversible after stopping treatment with JUXTAPID, but whether histological sequelae remain is unknown, especially after long-term use; protocol scheduled liver biopsies were not performed in the adult clinical trial.
In a phase 3 trial in pediatric patients, two patients (both aged 5 to 10 years) developed mild hepatic steatosis (as assessed by ultrasound), which resolved without specific medical interventions, other than the protocol specified follow-up ultrasounds and laboratory monitoring. Overall, the median absolute increase in hepatic fat was 4% after 24 weeks and 104 weeks, from 3% at baseline, measured by NMR. As with data from the adult patients, clinical pediatric data suggest that hepatic fat accumulation is reversible after stopping treatment with JUXTAPID, but whether histological sequelae remain is unknown, especially after long term use.
Alcohol may increase levels of hepatic fat and induce or exacerbate liver injury. Drinking more than one alcoholic drink per day is not recommended for patients taking JUXTAPID.
Exercise caution when using JUXTAPID with other medications known to have potential for hepatotoxicity, such as isotretinoin, amiodarone, acetaminophen (>4 g/day for ≥3 days/week), methotrexate, tetracyclines, and tamoxifen. The effect of concomitant administration of JUXTAPID with other hepatotoxic medications is unknown. More frequent monitoring of liver-related tests may be warranted.
JUXTAPID has not been studied concomitantly with other LDL-lowering agents that can also increase hepatic fat. Therefore, the combined use of such agents is not recommended.
5.2 JUXTAPID REMS Program
Because of the risk of hepatotoxicity associated with JUXTAPID therapy, JUXTAPID is available through a restricted program under the REMS. Under the JUXTAPID REMS, only certified healthcare providers and pharmacies may prescribe and distribute JUXTAPID. Further information is available at www.JUXTAPIDREMSProgram.com or by telephone at 1-85-JUXTAPID (1-855-898-2743).
5.3 Embryo-Fetal Toxicity
Based on findings from animal studies, JUXTAPID use is contraindicated in pregnancy since it may cause fetal harm [see Contraindications (4), Use in Specific Populations (8.1, 8.3)]. In animal reproduction studies in rats and ferrets, embryonic death and fetal malformations were observed at clinically relevant exposures. Females of reproductive potential should have a negative pregnancy test before starting JUXTAPID. Advise females of reproductive potential to use effective contraception during therapy with JUXTAPID and for two weeks after the final dose. If pregnancy is detected, discontinue JUXTAPID.
5.4 Reduced Absorption of Fat-Soluble Vitamins and Serum Fatty Acids
Given its mechanism of action in the small intestine, JUXTAPID may reduce the absorption of fat-soluble nutrients. In clinical trials of adult and pediatric patients with HoFH, patients were provided daily dietary supplements of vitamin E, linoleic acid, ALA, EPA, and DHA.
In the adult clinical trial, the median levels of serum vitamin E, ALA, linoleic acid, EPA, DHA, and arachidonic acid (AA) decreased from baseline to Week 26 but remained above the lower limit of the reference range. Adverse clinical consequences of these reductions were not observed with JUXTAPID treatment of up to 78 weeks.
In the pediatric clinical trial, overall, mean serum vitamin E levels decreased from baseline to Week 104 as expected but were still within or above the upper limit of the reference range. Mean values of linoleic acid, ALA, EPA, AA, and DHA all remained within the normal range or above the upper limit of the reference range during the 104-week trial. Eicosatrienoic acid was below lower limit of normal throughout the trial and increased to a mean normal value by Week 104.
Patients treated with JUXTAPID should take daily nutritional supplements that contain the dosages of vitamin E and essential fatty acids recommended in Dosage and Administration (2.2). Patients with chronic bowel or pancreatic diseases that predispose to malabsorption may be at increased risk for deficiencies in these nutrients with use of JUXTAPID.
5.5 Gastrointestinal Adverse Reactions
Gastrointestinal adverse reactions were reported by 27 (93%) of 29 patients in the adult clinical trial. Diarrhea occurred in 79% of patients, nausea in 65%, dyspepsia in 38%, and vomiting in 34%. Other reactions reported by at least 20% of patients include abdominal pain, abdominal discomfort, abdominal distension, constipation, and flatulence [see Adverse Reactions (6)].
Gastrointestinal adverse reactions of severe intensity were reported by 6 (21%) of 29 patients in the adult clinical trial, with the most common being diarrhea (4 patients, 14%); vomiting (3 patients, 10%); and abdominal pain, distension, and/or discomfort (2 patients, 7%). Gastrointestinal reactions contributed to the reasons for early discontinuation from this trial for 4 (14%) patients.
Gastrointestinal adverse reactions were reported by 31 (72%) of the 43 patients in the pediatric clinical trial. Diarrhea occurred in 22 (51%) patients and was more frequently reported by patients 11 to 17 years of age compared to patients 5 to 10 years of age (57% and 45%, respectively). Abdominal pain occurred in 19 (44%) patients and was reported with similar incidences in patients 5 to 10 years of age and 11 to 17 years of age. Vomiting occurred in 12 (28%) patients and was more frequently reported by patients 5 to 10 years of age compared to patients 11 to 17 years of age (50% and 9% of patients, respectively). Gastrointestinal reactions contributed to the reasons for early discontinuation from the trial for 2 (5%) patients.
There have been post-marketing reports of severe diarrhea in adults treated with JUXTAPID, including patients being hospitalized because of diarrhea-related complications such as volume depletion. Monitor patients who are more susceptible to complications from diarrhea, such as older patients and patients taking drugs that can lead to volume depletion or hypotension. Instruct patients to stop JUXTAPID and contact their healthcare provider if severe diarrhea occurs or if they experience symptoms of volume depletion, such as lightheadedness, decreased urine output, or tiredness. In such cases, consider reducing the dose or suspending use of JUXTAPID.
Absorption of concomitant oral medications may be affected in patients who develop diarrhea or vomiting.
To reduce the risk of gastrointestinal adverse reactions, instruct patients or their caregiver(s) to adhere to a low-fat diet supplying <20% of energy from fat or less than 30 grams of fat, whichever is less. Increase the dosage of JUXTAPID gradually. Individualize the maximum daily fat goal based on caloric needs due to age, growth, activity level, and tolerability. Monitor growth and weight loss in pediatric patients who are below the 10th percentile for height, weight, or BMI [see Dosage and Administration (2.1) and (2.2)].
5.6 Concomitant Use of CYP3A4 Inhibitors
CYP3A4 inhibitors increase the exposure of lomitapide, with strong inhibitors increasing exposure approximately 27-fold. Concomitant use of moderate or strong CYP3A4 inhibitors with JUXTAPID is contraindicated [see Drug Interactions (7.1)]. In the JUXTAPID clinical trials, one adult patient with HoFH developed markedly elevated transaminases (ALT 24 times ULN, AST 13 times ULN) within days of initiating the strong CYP3A4 inhibitor clarithromycin. If treatment with moderate or strong CYP3A4 inhibitors is unavoidable, JUXTAPID should be stopped during the course of treatment.
Avoid food or drinks containing grapefruit during JUXTAPID treatment.
Weak CYP3A4 inhibitors can increase the exposure of lomitapide approximately 2-fold; therefore, when JUXTAPID is administered with weak CYP3A4 inhibitors, the dose of JUXTAPID should be decreased by half. Careful titration may then be considered based on LDL-C response and safety/tolerability to half the maximum recommended dosage except when co-administered with oral contraceptives only, in which case the maximum recommended JUXTAPID dose is approximately two thirds the maximum recommended dose (Table 3) [see Dosage and Administration (2.4) and Drug Interactions (7.2)].
5.7 Risk of Myopathy with Concomitant Use of Simvastatin or Lovastatin
The risk of myopathy, including rhabdomyolysis, with simvastatin and lovastatin monotherapy is dose related. Lomitapide approximately doubles the exposure to simvastatin; therefore, it is recommended to reduce the dose of simvastatin by 50% when initiating JUXTAPID [see Clinical Pharmacology (12.3)]. While taking JUXTAPID, limit simvastatin dosage to 20 mg daily (or 40 mg daily for patients who have previously tolerated simvastatin 80 mg daily for at least one year without evidence of muscle toxicity). Refer to the simvastatin prescribing information for additional dosing recommendations.
Interaction between lovastatin and lomitapide has not been studied. However, the metabolizing enzymes and transporters responsible for the disposition of lovastatin and simvastatin are similar, suggesting that JUXTAPID may increase the exposure of lovastatin; therefore, reducing the dose of lovastatin should be considered when initiating JUXTAPID.
5.8 Risk of Supratherapeutic or Subtherapeutic Anticoagulation with Warfarin
JUXTAPID increases the plasma concentrations of warfarin. Increases in the dose of JUXTAPID may lead to supratherapeutic anticoagulation and decreases in the dose of JUXTAPID may lead to subtherapeutic anticoagulation. Difficulty controlling INR contributed to early discontinuation from the adult clinical trial for one of five patients taking concomitant warfarin. Patients taking warfarin should undergo regular monitoring of the INR, especially after any changes in JUXTAPID dosage. The dose of warfarin should be adjusted as clinically indicated [see Drug Interactions (7.3)].
-
6 ADVERSE REACTIONS
The following important adverse reactions have been observed and are discussed in detail in other sections of the label:
- Risk of hepatotoxicity [see Warnings and Precautions (5.1)]
- Reduced absorption of fat-soluble vitamins, and serum fatty acids [see Warnings and Precautions (5.4)]
- Gastrointestinal adverse reactions [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults with HoFH
One single-arm, open-label, 78-week trial has been conducted in 29 adult patients with HoFH, 23 of whom completed at least one year of treatment. The initial dosage of JUXTAPID was 5 mg daily, with titration up to 60 mg daily during an 18-week period based on safety and tolerability. In this trial, the mean age was 31 years (range, 18 to 55 years), 16 (55%) patients were male, 25 (86%) patients were White, 2 (7%) were Asian, 1 (3%) was Black or African American, and 1 (3%) was multi-racial [see Clinical Studies (14)].
Five (17%) of the 29 patients discontinued treatment due to an adverse reaction. The adverse reactions that contributed to treatment discontinuations included diarrhea (2 patients; 7%) and abdominal pain, nausea, gastroenteritis, weight loss, headache, and difficulty controlling INR on warfarin (1 patient each; 3%).
The most common adverse reactions were gastrointestinal, reported by 27 (93%) of 29 patients. Adverse reactions reported by ≥8 (28%) patients in the clinical trial included diarrhea, nausea, vomiting, dyspepsia, and abdominal pain. Other common adverse reactions, reported by 5 to 7 (17 to 24%) patients, included weight loss, abdominal discomfort, abdominal distension, constipation, flatulence, increased ALT, chest pain, influenza, nasopharyngitis, and fatigue.
The adverse reactions reported in at least 10% of adult patients are presented in Table 8.
Table 8: Adverse Reactions Reported in ≥10% of Patients in the Adult Clinical Trial ADVERSE REACTION N (%) Diarrhea 23 (79) Nausea 19 (65) Dyspepsia 11 (38) Abdominal pain 10 (34) Vomiting 10 (34) Chest pain 7 (24) Decreased weight 7 (24) Abdominal discomfort 6 (21) Abdominal distension 6 (21) Constipation 6 (21) Flatulence 6 (21) Influenza 6 (21) Fatigue 5 (17) Increased ALT 5 (17) Nasopharyngitis 5 (17) Back pain 4 (14) Gastroenteritis 4 (14) Pharyngolaryngeal pain 4 (14) Angina pectoris 3 (10) Defecation urgency 3 (10) Dizziness 3 (10) Fever 3 (10) Gastroesophageal reflux disease 3 (10) Headache 3 (10) Nasal congestion 3 (10) Palpitations 3 (10) Rectal tenesmus 3 (10) Adverse reactions of severe intensity were reported by 8 (28%) of 29 patients, with the most common being diarrhea (4 patients, 14%), vomiting (3 patients, 10%), increased ALT or hepatotoxicity (3 patients, 10%), and abdominal pain, distension, and/or discomfort (2 patients, 7%).
Pediatric Patients with HoFH Aged 5 to 17 years
A single-arm, open label, multinational, 104-week trial was conducted in 43 pediatric patients with HoFH aged 5 to 17 years. Thirty-nine of the patients completed the trial. The dose of JUXTAPID was escalated from an age-dependent starting dose to a maximum tolerated dose (MTD) as applicable to the pediatric age group and based on acceptable safety and tolerability criteria, in addition to LDL-C goals [see Dosage and Administration (2) and Clinical Studies (14)].
Table 9: Adverse Reactions Reported in ≥10% of Pediatric Patients Aged 5 to 17 Years ADVERSE REACTION N (%) - * Grouped terms composed of several similar terms
Elevated transaminases* 23 (53) Abdominal pain* 23 (53) Diarrhea 22 (51) Vomiting 12 (28) Decreased appetite 6 (14) Nausea 5 (12) Transaminase Elevations
During the adult clinical trial, 10 (34%) of 29 patients had at least one elevation in ALT and/or AST ≥3 times ULN (see Table 10). No clinically meaningful elevations in total bilirubin or alkaline phosphatase were observed. Transaminases typically fell within one to four weeks of reducing the dose or withholding JUXTAPID.
Among the 19 adult patients who enrolled in an extension trial following the adult clinical trial, one discontinued because of increased transaminases that persisted despite several dose reductions, and one temporarily discontinued because of markedly elevated transaminases (ALT 24 times ULN, AST 13 times ULN) that had several possible causes, including a drug-drug interaction between JUXTAPID and the strong CYP3A4 inhibitor clarithromycin [see Drug Interactions (7.1)].
In the pediatric trial, 6 patients experienced elevations in ALT and/or AST ≥3 times ULN (see Table 10). No discontinuations occurred due to increased transaminases in the trial, although some patients required interrupting JUXTAPID or reducing the dose.
Table 10: Patient Incidence of Transaminase Elevations During the Clinical Trials ADULTS*
N (%)PEDIATRIC PATIENTS AGED 5 TO 17 YEARS†
N (%)- * Upper limits of normal (ULN) ranged from 33 to 41 international units/L for ALT and 36 to 43 international units/L for AST.
- † ULN ranged from 21 to 55 international units/L for ALT and 24 to 60 international units/L for AST, based on age and gender.
Total Patients 29 43 Maximum ALT ≥3 to <5 × ULN 6 (21) 3 (7) ≥5 to <10 × ULN 3 (10) 2 (5) ≥10 to <20 × ULN 1 (3) 0 ≥20 × ULN 0 0 Maximum AST ≥3 to <5 × ULN 5 (17) 3 (7) ≥5 to <10 × ULN 1 (3) 0 ≥10 to <20 × ULN 0 0 ≥20 × ULN 0 0 Hepatic Steatosis
Hepatic fat was prospectively measured using magnetic resonance spectroscopy (MRS) in all eligible patients during the adult clinical trial. After 26 weeks, the median absolute increase in hepatic fat from baseline was 6%, and the mean absolute increase was 8% (range, 0% to 30%). After 78 weeks, the median absolute increase in hepatic fat from baseline was 6%, and the mean absolute increase was 7% (range, 0% to 18%). Among the 23 patients with evaluable data on at least one occasion during the trial, 18 (78%) exhibited an increase in hepatic fat >5% and 3 (13%) exhibited an increase >20%. Data from individuals who had repeat measurements after stopping JUXTAPID show that hepatic fat accumulation is reversible, but whether histological sequelae remain is unknown.
In the pediatric clinical trial, the median absolute increase in hepatic fat was 4% after 24 weeks and 104 weeks, from 3% at baseline, measured by NMR. Among the 19 patients with hepatic fat measured by NMR on at least one occasion during the trial, 8 (42%) patients exhibited an increase in hepatic fat to >10% including 1 (5%) patient with an increase to >20%. Data from pediatric patients with follow-up measurements after Week 104 suggest that hepatic fat accumulation is reversable after stopping treatment with JUXTAPID, but whether histological sequelae remain is unknown, especially after long term use.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of JUXTAPID. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to JUXTAPID exposure.
Musculoskeletal: Myalgia
Skin reactions: Alopecia
-
7 DRUG INTERACTIONS
7.1 Moderate and Strong CYP3A4 Inhibitors
A strong CYP3A4 inhibitor has been shown to increase lomitapide exposure approximately 27-fold [see Clinical Pharmacology (12.3)]. Concomitant use of strong CYP3A4 inhibitors with JUXTAPID is contraindicated. Concomitant use of moderate CYP3A4 inhibitors has not been studied, but concomitant use with JUXTAPID is contraindicated since lomitapide exposure will likely increase significantly in the presence of these inhibitors.
Avoid food or drinks containing grapefruit during JUXTAPID treatment [see Contraindications (4), Warnings and Precautions (5.6), and Clinical Pharmacology (12.3)].
7.2 Weak CYP3A4 Inhibitors
Weak CYP3A4 inhibitors can increase lomitapide exposure approximately 2-fold [see Clinical Pharmacology (12.3)]. When administered with weak CYP3A4 inhibitors, the dose of JUXTAPID should be decreased by half. Careful titration of JUXTAPID may then be considered based on LDL-C response and safety/tolerability to half the maximum recommended dosage except when co-administered with oral contraceptives only, in which case the maximum recommended JUXTAPID dosage is approximately two thirds of the maximum recommended dosage (Table 3) [see Dosage and Administration (2.4), Warnings and Precautions (5.6), and Clinical Pharmacology (12.3)].
7.3 Warfarin
Lomitapide increases plasma concentrations of both R(+)-warfarin and S(-)-warfarin by approximately 30% and increases the INR 22%. Patients taking warfarin should undergo regular monitoring of INR, particularly after any changes in JUXTAPID dosage. The dose of warfarin should be adjusted as clinically indicated [see Warnings and Precautions (5.8)].
7.4 Simvastatin and Lovastatin
The risk of myopathy, including rhabdomyolysis, with simvastatin and lovastatin monotherapy is dose-related. Lomitapide approximately doubles the exposure of simvastatin; therefore, the recommended dose of simvastatin should be reduced by 50% when initiating JUXTAPID [see Clinical Pharmacology (12.3)]. While taking JUXTAPID, limit simvastatin dosage to 20 mg daily (or 40 mg daily for patients who have previously tolerated simvastatin 80 mg daily for at least one year without evidence of muscle toxicity). Refer to the simvastatin prescribing information for simvastatin dosing recommendations.
Interaction between lovastatin and lomitapide has not been studied. However, the metabolizing enzymes and transporters responsible for the disposition of lovastatin and simvastatin are similar, suggesting that JUXTAPID may increase the exposure of lovastatin; therefore, reducing the dose of lovastatin should be considered when initiating JUXTAPID.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure
There is a registry that monitors pregnancy outcomes in women exposed to JUXTAPID during pregnancy. For additional information visit www.JUXTAPID.com or call the Lomitapide Observational Worldwide Exposure Registry (LOWER) at 1-877-902-4099. Healthcare professionals are encouraged to call the LOWER at 1-877-902-4099 to enroll patients who become pregnant during JUXTAPID treatment.
Risk Summary
Based on findings from animal studies, JUXTAPID use is contraindicated in pregnancy since it may cause fetal harm [see Contraindications (4), Warnings and Precautions (5.3)]. Available human data are insufficient to draw conclusions about any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes. However, in animal reproduction studies, lomitapide was teratogenic in rats at clinically relevant exposures and in ferrets at exposures estimated to be less than human therapeutic exposure at 60 mg when administered during organogenesis, based on AUC comparisons. Embryo-fetal lethality was observed in rabbits at 6-times the maximum recommended human dose (MRHD) of 60 mg based on body surface area. If pregnancy is detected, discontinue JUXTAPID.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Oral gavage doses of 0.04, 0.4, or 4 mg/kg/day lomitapide given to pregnant rats from gestation day 6 through organogenesis were associated with fetal malformations at ≥2-times human exposure at the MRHD (60 mg) based on plasma AUC comparisons. Fetal malformations included umbilical hernia, gastroschisis, imperforate anus, alterations in heart shape and size, limb malrotations, skeletal malformations of the tail, and delayed ossification of cranial, vertebral and pelvic bones.
Oral gavage doses of 1.6, 4, 10, or 25 mg/kg/day lomitapide given to pregnant ferrets from gestation day 12 through organogenesis were associated with both maternal toxicity and fetal malformations at exposures that ranged from less than the human exposure at the MRHD to 5-times the human exposure at the MRHD. Fetal malformations included umbilical hernia, medially rotated or short limbs, absent or fused digits on paws, cleft palate, open eye lids, low-set ears, and kinked tail.
Oral gavage doses of 0.1, 1, or 10 mg/kg/day lomitapide given to pregnant rabbits from gestation day 6 through organogenesis were not associated with adverse effects at systemic exposures up to 3-times the MRHD of 60 mg based on body surface area comparison. Treatment at doses of ≥20 mg/kg/day, ≥6-times the MRHD, resulted in embryo-fetal lethality.
Pregnant female rats given oral gavage doses of 0.1, 0.3, or 1 mg/kg/day lomitapide from gestation day 7 through termination of nursing on lactation day 20 were associated with malformations at systemic exposures equivalent to human exposure at the MRHD of 60 mg based on AUC. Increased pup mortality occurred at 4-times the MRHD.
8.2 Lactation
Risk Summary
There are no data on the presence of lomitapide in human or animal milk, effects on the breastfed infant or on milk production. Because of the potential for serious adverse reactions, including hepatotoxicity, advise patients that breastfeeding is not recommended during treatment with JUXTAPID.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Females of reproductive potential should have a negative pregnancy test before starting JUXTAPID.
Contraception
Based on animal studies, JUXTAPID may cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with JUXTAPID and for two weeks after the final dose.
The use of JUXTAPID may result in reduced efficacy of oral contraceptives if vomiting or diarrhea occurs. Advise patients using oral contraceptives and who experience vomiting or diarrhea to use an effective alternative contraceptive method until 7 days after resolution of symptoms [see Drug Interactions (7.2)].
8.4 Pediatric Use
The safety and effectiveness of JUXTAPID as an adjunct to other LDL-C lowering therapies for the treatment of HoFH have been established in pediatric patients aged 2 years and older. Use of JUXTAPID for this indication is supported by evidence from an open-label trial in adults; an open-label trial of 43 pediatric patients with HoFH aged 5 to 17 years old; and additional pharmacokinetic modeling and simulation data for pediatric patients aged 2 years and older. Adverse reactions reported in pediatric patients aged 5 to 17 years were similar to those reported in adults [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
The safety and effectiveness of JUXTAPID have not been established in pediatric patients younger than 2 years old.
8.5 Geriatric Use
Clinical trials of JUXTAPID did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger patients. In general, dosing for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
Patients with end-stage renal disease (eGFR <15 mL/min/1.73m2) receiving hemodialysis, lomitapide exposure increased approximately 50% compared with healthy volunteers [see Clinical Pharmacology (12.3)]. The maximum recommended dosage of JUXTAPID in patients with end-stage renal disease receiving hemodialysis is lower than in those with normal renal function [see Dosage and Administration (2.6)]. Effects of mild, moderate, and severe renal impairment, including those with end-stage renal disease not yet receiving dialysis, on lomitapide exposure have not been studied. However, it is possible that patients with renal impairment who are not yet receiving dialysis may experience increases in lomitapide exposure exceeding 50% [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
JUXTAPID is contraindicated in patients with moderate (Child-Pugh B) or severe (Child-Pugh C) hepatic impairment since the lomitapide exposure increased 164% compared with healthy volunteers [see Contraindications (4) and Clinical Pharmacology (12.3)]. In adult patients with mild hepatic impairment (Child-Pugh A), lomitapide exposure increased approximately 50% compared with healthy volunteers [see Clinical Pharmacology (12.3)]. The maximum recommended dosage of JUXTAPID in patients with mild hepatic impairment is lower than in those with normal hepatic function [see Dosage and Administration (2.7)].
-
10 OVERDOSAGE
There is no specific treatment in the event of overdose of JUXTAPID. In the event of overdose, the patient should be treated symptomatically, and supportive measures instituted as required. Liver-related tests should be monitored. Hemodialysis is unlikely to be beneficial given that lomitapide is highly protein-bound.
-
11 DESCRIPTION
JUXTAPID capsules contain lomitapide mesylate, a synthetic lipid-lowering agent for oral administration.
The chemical name of lomitapide mesylate is N-(2,2,2-trifluoroethyl)-9-[4-[4-[[[4'a-(trifluoromethyl)[1,1'-biphenyl]-2-yl]carbonyl]amino]-1-piperidinyl]butyl]-9H-fluorene-9-carboxamide, methanesulfonate salt. Its structural formula is:
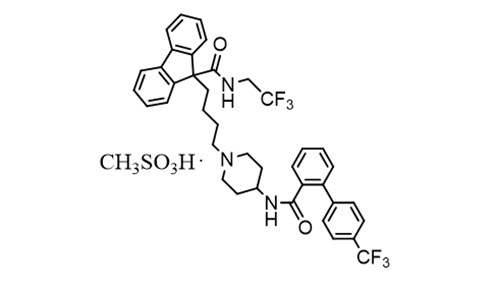
The empirical formula for lomitapide mesylate is C39H37F6N3O2 ∙ CH4O3S and its molecular weight is 789.8.
Lomitapide mesylate is a white to off-white powder that is slightly soluble in aqueous solutions of pH 2 to 5. Lomitapide mesylate is freely soluble in acetone, ethanol, and methanol; soluble in 2-butanol, methylene chloride, and acetonitrile; sparingly soluble in 1-octanol and 2-propanol; slightly soluble in ethyl acetate; and insoluble in heptane.
Each JUXTAPID capsule contains lomitapide mesylate equivalent to 2, 5, 10, 20, or 30 mg lomitapide free base and the following inactive ingredients: pregelatinized starch, sodium starch glycolate, microcrystalline cellulose, lactose monohydrate, silicon dioxide and magnesium stearate. The capsule shells of all strengths contain gelatin and titanium dioxide; the 2 mg capsules contain black iron oxide; the 5 mg, 10 mg and 30 mg capsules also contain red iron oxide; and the 30 mg capsules also contain yellow iron oxide. The imprinting ink contains shellac, black iron oxide, and propylene glycol.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
JUXTAPID directly binds and inhibits microsomal triglyceride transfer protein (MTP), which resides in the lumen of the endoplasmic reticulum, thereby preventing the assembly of apo B-containing lipoproteins in enterocytes and hepatocytes. This inhibits the synthesis of chylomicrons and VLDL. The inhibition of the synthesis of VLDL leads to reduced levels of plasma LDL-C.
12.2 Pharmacodynamics
Effects on QT Interval
At a concentration 23 times the Cmax of the maximum recommended dose, lomitapide does not prolong QTc to any clinically relevant extent.
12.3 Pharmacokinetics
Lomitapide pharmacokinetics is approximately dose-proportional for oral single doses from 10 to 100 mg. Following the maximum recommended dose of 60 mg daily in patients ≥18 years old, the steady state exposure (i.e., AUCtau with tau=24 hours) and Cmax were predicted to be 116.34 ng/mL∙h (with 90% prediction interval of 41.7 to 362.6 ng/mL∙h) and 5.74 ng/mL (2.00 to 17.9 ng/mL) respectively (see Table 11).
Absorption
The absolute bioavailability of lomitapide is approximately 7%. Upon oral administration of a single 60-mg dose of JUXTAPID, the lomitapide tmax is around 6 hours in healthy volunteers.
Distribution
The mean lomitapide volume of distribution at steady state is 985 to 1292 liters. Lomitapide is 99.8% plasma-protein bound.
Elimination
Metabolism
Lomitapide is metabolized extensively by the liver. The metabolic pathways include oxidation, oxidative N-dealkylation, glucuronide conjugation, and piperidine ring opening. Cytochrome P450 (CYP) 3A4 metabolizes lomitapide to its major metabolites, M1 and M3, as detected in plasma. The oxidative N-dealkylation pathway breaks the lomitapide molecule into M1 and M3. M1 is the moiety that retains the piperidine ring, whereas M3 retains the rest of the lomitapide molecule in vitro. CYPs 1A2, 2B6, 2C8, and 2C19 may metabolize lomitapide to a small extent to M1. M1 and M3 do not inhibit activity of microsomal triglyceride transfer protein in vitro.
Excretion
In a mass-balance study, a mean of 59.5% and 33.4% of the dose was excreted in the urine and feces, respectively. In another mass-balance study, a mean of 52.9% and 35.1% of the dose was excreted in the urine and feces, respectively. Lomitapide was not detectable in urine samples. M1 is the major urinary metabolite. Lomitapide is the major component in the feces. The mean lomitapide terminal half-life is 39.7 hours.
Specific Populations
Hepatic Impairment
A single-dose, open-label study was conducted to evaluate the pharmacokinetics of 60 mg lomitapide in healthy adult volunteers with normal hepatic function compared with adult patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment. In patients with moderate hepatic impairment, lomitapide AUC and Cmax were 164% and 361% higher, respectively, compared with healthy volunteers. In patients with mild hepatic impairment, lomitapide AUC and Cmax were 47% and 4% higher, respectively, compared with healthy volunteers. Lomitapide has not been studied in patients with severe hepatic impairment (Child-Pugh score 10 to 15) [see Dosage and Administration (2.7), Contraindications (4), Warnings and Precautions (5.1), and Use in Specific Populations (8.7)].
Renal Impairment
A single-dose, open-label study in adults was conducted to evaluate the pharmacokinetics of 60 mg lomitapide in patients with end-stage renal disease receiving hemodialysis compared with healthy adult volunteers with normal renal function. Healthy volunteers had estimated creatinine clearance >80 mL/min by the Cockcroft-Gault equation. Compared with healthy volunteers, lomitapide AUC0-inf and Cmax were 40% and 50% higher, respectively, in patients with end-stage renal disease receiving hemodialysis. Effects of mild, moderate, and severe renal impairment as well as end-stage renal disease not yet on dialysis on lomitapide exposure have not been studied [see Dosage and Administration (2.6) and Use in Specific Populations (8.6)].
Pediatric Patients
Predicted lomitapide exposures under the maximum dosage levels (i.e., 20 and 40 mg) for pediatric patients in each age group are presented in Table 11.
Table 11: Predicted AUC0-tau and Cmax at Steady-State Age Group Maximum Recommended Dosage Median AUC0-tau (ng/mL∙h) 5th, 95th Percentile AUC0-tau (ng/mL∙h) Median Cmax
(ng/mL)5th, 95th Percentile
Cmax (ng/mL)AUC0-tau=Area under the curve over the dosing interval of 24 hours (tau=24 h); Cmax= Maximum observed plasma concentration - * Simulated adult data for reference.
2 to 4 years 20 mg 213.62 71.62, 637.34 11.11 3.66, 33.76 5 to 10 years 20 mg 142.57 47.52, 455.67 7.33 2.31, 23.70 11 to 15 years 40 mg 240.48 82.96, 783.84 12.16 4.18, 39.09 16 to 17 years 40 mg 201.11 67.58, 646.16 9.95 3.33, 32.53 18 years and older* 60 mg 116.34 41.73, 362.64 5.74 2.00, 17.90 Drug Interactions
In vitro Assessment of Drug Interactions
Lomitapide does not induce CYPs 1A2, 3A4, or 2B6. Lomitapide inhibits CYP3A4. Lomitapide does not inhibit CYPs 1A2, 2B6, 2C9, 2C19, 2D6, or 2E1. M1 and M3 do not induce CYPs 1A2, 3A4, or 2B6. M1 and M3 do not inhibit CYPs 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, or 3A4. Lomitapide is not a P-gp substrate. Lomitapide inhibits P-gp but does not inhibit breast cancer resistance protein (BCRP).
Effects of other Drugs on Lomitapide
Table 12 summarizes the effect of co-administered drugs on lomitapide AUC and Cmax.
Table 12: Effect of Co-administered Drugs on Lomitapide Systemic Exposure CO-ADMINISTERED DRUG DOSING OF CO-ADMINISTERED DRUG DOSING OF LOMITAPIDE RATIO OF LOMITAPIDE EXPOSURE WITH/WITHOUT CO-ADMINISTERED DRUG
NO EFFECT = 1AUC Cmax BID = twice daily; QD = once daily;↑ = increase Contraindicated with lomitapide [see Contraindications (4) and Warnings and Precautions (5.6)] Ketoconazole 200 mg BID for 9 days 60 mg single dose ↑ 27 ↑ 15 Adjustment necessary when co-administered with lomitapide [see Dosage and Administration (2.4) and Warnings and Precautions (5.6)] AUC Cmax Atorvastatin 80 mg QD 20 mg single dose ↑2 ↑2.1 Ethinyl Estradiol (EE) / norgestimate 0.035 mg EE/ 0.25 mg norgestimate QD 20 mg single dose ↑1.3 ↑1.4 Effect of Lomitapide on other Drugs
Table 13 summarizes the effects of lomitapide on the AUC and Cmax of co-administered drugs.
Table 13: Effect of Lomitapide on the Systemic Exposure of Co-administered Drugs CO-ADMINISTERED DRUG DOSING OF CO-ADMINISTERED DRUG DOSING OF LOMITAPIDE CHANGE OF CO-ADMINISTERED DRUG EXPOSURE WITH / WITHOUT LOMITAPIDE AUC Cmax Dosage adjustment necessary when co-administered with lomitapide - * Limit simvastatin dosage to 20 mg daily (or 40 mg daily for patients who have previously tolerated simvastatin 80 mg daily for at least one year without evidence of muscle toxicity). Refer to the simvastatin prescribing information for additional dosing recommendations.
- † Patients taking warfarin should undergo regular monitoring of the INR, especially after any changes in lomitapide dosage. QD = once daily; INR = international normalized ratio; ↑ = increase; ↓ = decrease
Simvastatin* 40 mg single dose 60 mg QD × 7 days Simvastatin
Simvastatin acid↑ 99%
↑ 71%↑
102%
↑ 57%↑ 62% 20 mg single dose 10 mg QD × 7 days Simvastatin
Simvastatin acid↑ 39% ↑65%
↑ 35%Warfarin† 10 mg single dose 60 mg QD × 12 days R(+) warfarin
S(-) warfarin
INR↑ 28%
↑ 30%
↑ 7%↑ 14%
↑ 15%
↑ 22%No dosing adjustments required for the following: Atorvastatin 20 mg single dose 60 mg QD × 7 days Atorvastatin acid ↑ 52%
↑ 11%↑63%
↑19%20 mg single dose 10 mg QD × 7 days Atorvastatin acid Rosuvastatin 20 mg single dose
20 mg single dose60 mg QD × 7 days
10 mg QD × 7 daysRosuvastatin
Rosuvastatin↑ 32%
↑ 2%↑ 4%
↑ 6%Fenofibrate, micronized 145 mg single dose 10 mg QD × 7 days Fenofibric acid ↓ 10% ↓29% Ezetimibe 10 mg single dose 10 mg QD × 7 days Total
ezetimibe↑ 6% ↑ 3% Extended release niacin 1000 mg single dose 10 mg QD × 7 days Nicotinic acid
Nicotinuric acid↑ 10%
↓ 21%↑ 11%
↓ 15%Ethinyl estradiol 0.035 mg QD × 28 days 50 mg QD × 8 days Ethinyl estradiol ↓ 8% ↓ 8% Norgestimate 0.25 mg QD × 28 days 50 mg QD × 8 days 17-Deacetyl norgestimate ↑ 6% ↑ 2% -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year dietary carcinogenicity study in mice, lomitapide was administered at doses of 0.3, 1.5, 7.5, 15, or 45 mg/kg/day. There were statistically significant increases in the incidences of liver adenomas and carcinomas in males at doses ≥1.5 mg/kg/day (≥2-times the MRHD at 60 mg based on AUC) and in females at ≥7.5 mg/kg/day (≥10-times the human exposure at 60 mg based on AUC). Incidences of small intestinal carcinomas in males and combined adenomas and carcinomas in females were significantly increased at doses ≥15 mg/kg/day (≥23-times the human exposure at 60 mg based on AUC).
In a 2-year carcinogenicity study in rats, lomitapide was administered by oral gavage for up to 99 weeks at doses of 0.25, 1.7, or 7.5 mg/kg/day in males and 0.03, 0.35, or 2.0 mg/kg/day in females. While the design of the study was suboptimal, there were no statistically significant drug-related increases in tumor incidences at exposures up to 6-times (males) and 8-times (females) higher than human exposure at the MRHD based on AUC.
Lomitapide did not exhibit genotoxic potential in a battery of studies, including the in vitro Bacterial Reverse Mutation (Ames) assay, an in vitro cytogenetics assay using primary human lymphocytes, and an oral micronucleus study in rats.
Lomitapide had no effect on fertility in rats at doses up to 5 mg/kg/day at systemic exposures estimated to be 4-times (females) and 5-times (males) higher than in humans at 60 mg based on AUC.
-
14 CLINICAL STUDIES
Adults with HoFH
The safety and effectiveness of JUXTAPID as an adjunct to a low-fat diet and other lipid-lowering treatments, including LDL apheresis where available, were evaluated in a multinational, single-arm, open-label, 78-week trial involving 29 adults with HoFH. A diagnosis of HoFH was defined by the presence of at least one of the following clinical criteria: (1) documented functional mutation(s) in both LDL receptor alleles or alleles known to affect LDL receptor functionality, or (2) skin fibroblast LDL receptor activity <20% normal, or (3) untreated TC >500 mg/dL and TG <300 mg/dL and both parents with documented untreated TC >250 mg/dL.
Among the 29 patients enrolled, the mean age was 31 years (range, 18 to 55 years), 16 (55%) were male, and the majority (86%) were White. The mean body mass index (BMI) was 25.8 kg/m2, with four patients meeting BMI criteria for obesity; one patient had type 2 diabetes. Concomitant lipid-lowering treatments at baseline included one or more of the following: statins (93%), ezetimibe (76%), nicotinic acid (10%), bile acid sequestrant (3%), and fibrate (3%); 18 (62%) were receiving apheresis.
After a six-week run-in period to stabilize lipid-lowering treatments, including the establishment of an LDL apheresis schedule if applicable, JUXTAPID was initiated at 5 mg daily and titrated to daily doses of 10 mg, 20 mg, 40 mg, and 60 mg at weeks 2, 6, 10, and 14, respectively, based on tolerability and acceptable levels of transaminases. Patients were instructed to maintain a low-fat diet (<20% calories from fat) and to take dietary supplements that provided approximately 400 international units vitamin E, 210 mg ALA, 200 mg linoleic acid, 110 mg EPA, and 80 mg (DHA per day [see Dosage and Administration (2.2), Warnings and Precautions (5.4)]. After efficacy was assessed at Week 26, patients remained on JUXTAPID for an additional 52 weeks to assess long-term safety. During this safety phase, the dose of JUXTAPID was not increased above each patient's maximum tolerated dose established during the efficacy phase, but changes to concomitant lipid-lowering treatments were allowed.
Twenty-three (79%) patients completed the efficacy endpoint at Week 26, all of whom went on to complete 78 weeks of treatment. Adverse events contributed to premature discontinuation for five patients [see Adverse Reactions (6.1)]. The maximum tolerated doses during the efficacy period were 5 mg (10%), 10 mg (7%), 20 mg (21%), 40 mg (24%), and 60 mg (34%).
The primary efficacy endpoint was percent change in LDL-C from baseline to Week 26. At Week 26, the mean and median percent changes in LDL-C from baseline were -40% (paired t-test p<0.001) and -50%, respectively, based on the intent-to-treat population with last observation carried forward (LOCF) for patients who discontinued prematurely. The mean percent change in LDL-C from baseline through Week 26 is shown in Figure 1 for the 23 patients who completed the efficacy period.
Figure 1: Mean Percent Change in LDL-C from Baseline (Week 26 Completers) Error bars represent 95% confidence intervals of the mean. 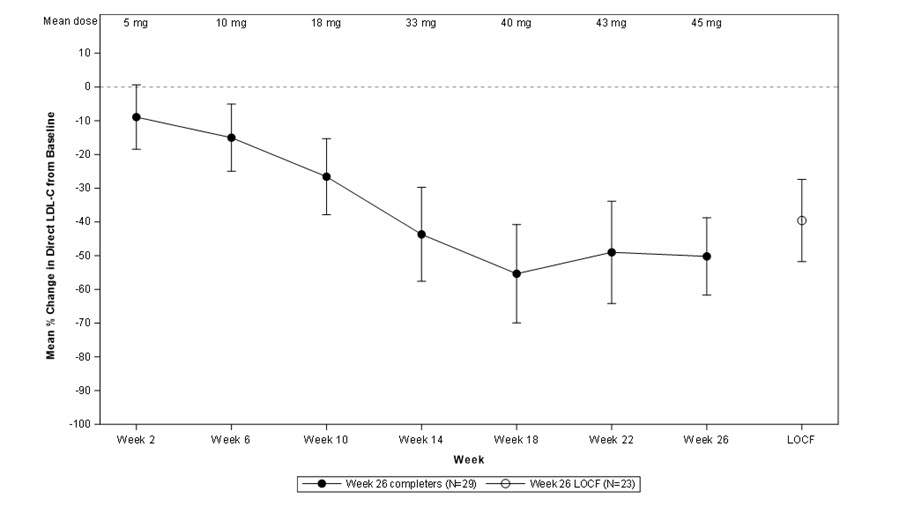
Changes in lipids and lipoproteins through the efficacy endpoint at Week 26 are presented in Table 14.
Table 14: Absolute Values and Percent Changes from Baseline in Lipids and Lipoproteins PARAMETER BASELINE WEEK 26/LOCF
(N=29)Mean (SD) Mean (SD) Mean % Change - * Statistically significant compared with baseline based on the pre-specified gatekeeping method for controlling Type I error among the primary and key secondary endpoints.
- † Median values with interquartile range and median % change presented for TG.
LDL-C, direct (mg/dL) 336 (114) 190 (104) -40 * TC (mg/dL) 430 (135) 258 (118) -36 * apo B (mg/dL) 259 (80) 148 (74) -39 * Non-HDL-C (mg/dL) 386 (132) 217 (113) -40 VLDL-C (mg/dL) 21 (10) 13 (9) -29 TG (mg/dL)† 92 [72, 128] 57 [36, 78] -45 * HDL-C (mg/dL) 44 (11) 41 (13) -7 After Week 26, during the safety phase of the trial, adjustments to concomitant lipid-lowering treatments were allowed. For the study population overall, average reductions in LDL-C, TC, apo B, and non-HDL-C were sustained during chronic therapy.
Pediatric Patients with HoFH
A single-arm, open-label, 104-week trial evaluated the efficacy of JUXTAPID when co-administered with a low-fat diet in pediatric patients aged 5 to 17 years of age with HoFH on stable lipid lowering therapy (LLT), including low density lipoprotein (LDL) apheresis, when applicable. A diagnosis of HoFH was defined by any of the following criteria: (1) genetic confirmation of 2 mutant alleles at the LDLR, apo B, PCSK9, or LDLR adapter protein 1 (LDLRAP1) gene loci, or (2) an untreated LDL-C >500 mg/dL or treated LDL-C ≥300 mg/dL together with either a cutaneous or tendon xanthoma before age 10 years or untreated LDL-C levels consistent with heterozygous familial hypercholesterolemia in both parents.
A total of 43 patients (19 males, 44% and 24 females, 56%) between 5 and 17 years of age (mean age of 11 years) were treated in this trial. The majority (98%) of patients were White (one [2%] patient was Black or African American and one [2%] patient identified as Hispanic/Latino ethnicity). The mean BMI was 19 kg/m2. Concomitant LLT at baseline included one or more of the following: statins (91%), ezetimibe (74%), and evolocumab (12%); 19 (44%) patients were receiving apheresis through Week 24.
The trial consisted of a 6-week run-in period, followed by a 24-week efficacy phase, and then an 80-week safety phase. The dose of JUXTAPID was escalated from an age-dependent starting dose to a maximum tolerated dose as applicable to the pediatric age group (see Table 2). Patients were instructed to maintain a low-fat diet (<20% calories from fat or <30g fat, whichever was the lesser amount) and to take dietary supplements that provided vitamin E (200 IU for patients for 5 to 8 years of age, 400 IU for patients 9 years of age and older) and approximately 200 mg linoleic acid, 210 mg ALA, 110 mg EPA, and 80 mg DHA [see Dosage and Administration 2.2, Warnings and Precautions (5.4)]. After Week 24, patients entered the 80-week safety phase and remained on JUXTAPID to determine the effects of longer-term treatment. At the discretion of the investigator, patients were allowed to change their background LLT and/or increase the JUXTAPID dose (in exceptional cases only) during the safety phase. There was no significant change in LLTs, however apheresis was reduced or discontinued in 47% of patients receiving apheresis at Week 24 (refer to the subgroup analysis below which showed that mean LDL-C levels in the LDL apheresis group showed a gradual increase over time when apheresis was reduced or discontinued).
Forty-six (46) patients were enrolled, of whom 43 completed the run-in period, 41 completed Week 24, and 39 completed the safety phase. In the efficacy phase, 95% of patients aged 5 to 10 years received the MTD of 20 mg, 76% of patients aged 11 to 15 years received the MTD of 40 mg, and 50% of patients aged 16 to 17 years received the MTD of 60 mg but had to down-titrate soon after reaching this dose.
The primary endpoint was percent change in LDL-C from baseline to Week 24. At Week 24, the mean percent change in LDL-C from baseline was -49% (95% CI: -59%, -38%) (see Figure 2 which presents mean percentage change from baseline of observed LDL-C values).
Figure 2: Observed Mean Percent Change in LDL-C from Baseline in Pediatric Patients Aged 5 to 17 years
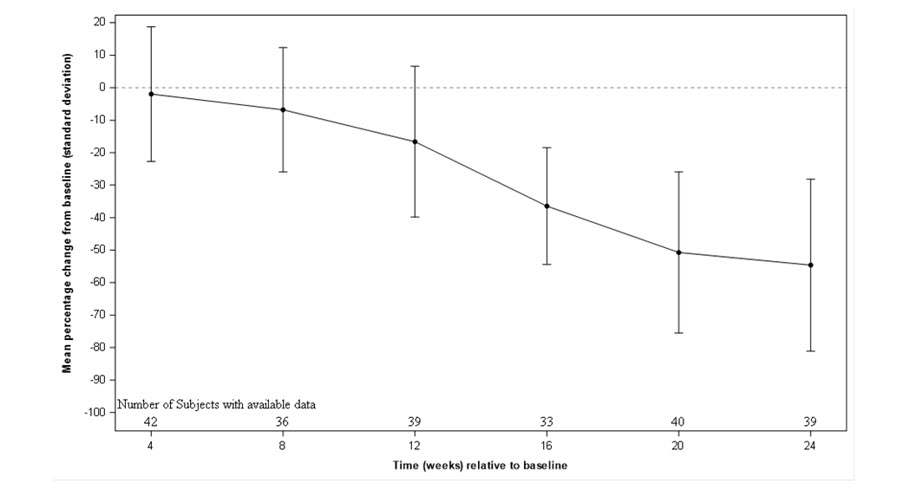
Subgroup analysis was carried out on patients aged 5 to 10 years (N=20) and 11 to 17 years (N=23). Mean decreases from baseline in LDL-C at Week 24 were 52% and 46%, respectively. Subgroup analysis also demonstrated mean decreases from baseline in LDL-C at Week 24 of approximately 36% in patients receiving LDL apheresis at baseline and 62% in patients not receiving LDL apheresis at baseline. During the long-term treatment (over 2 years), mean LDL-C levels in the LDL apheresis group showed a gradual increase over time.
The key secondary endpoint was the mean percent change in lipid parameters (non-HDL-C, total cholesterol, VLDL-C, apo B, and triglycerides) from baseline to Week 24. At Week 24, there were mean percent decreases from baseline in each of the lipid parameters assessed (summarized in Table 15).
Table 15: Absolute Values and Percent Changes from Baseline to Week 24 in Lipids and Lipoproteins in Pediatric Patients Aged 5 to 17 Years PARAMETER (UNITS) BASELINE WEEK 24 (N = 43) MEAN
(SD)MEAN
(95% CI)% CHANGE
(95% CI)LDL-C = Low Density Lipoprotein-C; SD = standard deviation
Missing values at Week 24 were imputed using a return to baseline multiple imputation approach.LDL-C, direct (mg/dL) 436
(189)195
(160, 230)-49
(-59%, -38%)Total Cholesterol (TC) (mg/dL) 486
(188)237
(200, 275)-45
(-55%, -36%)Apolipoprotein B (apo B) (mg/dL) 317
(133)146
(121, 171)-48
(-58%, -37%)Triglycerides (TG) (mg/dL) 92
(39)45
(37, 53)-46
(-57%, -35%)Non-high-density lipoprotein cholesterol (Non-HDL-C) (mg/dL) 454
(192)204
(167, 241)-49
(-59%, -39%)Very-low-density lipoprotein cholesterol (VLDL-C) (mg/dL) 18
(8)9
(7, 11)-46
(-57%, -36%)Average reductions in LDL-C, TC, apo B, TG, non-HDL-C, and VLDL-C were sustained during long-term treatment (over 2 years) particularly in patients not receiving LDL apheresis at baseline.
-
16 HOW SUPPLIED / STORAGE AND HANDLING
How Supplied
JUXTAPID strengths are available as follows:
Capsule Strength Description Package Configuration NDC Number 2 mg Grey/grey hard gelatin capsule printed with black ink "A733" and "2 mg" Bottles of 28 capsules 10122-402-28 5 mg Orange/orange hard gelatin capsule printed with black ink "A733" and "5 mg" Bottles of 28 capsules 10122-405-28 10 mg Orange/white hard gelatin capsule printed with black ink "A733" and "10 mg" Bottles of 28 capsules 10122-410-28 20 mg White/white hard gelatin capsule printed with black ink "A733" and "20 mg" Bottles of 28 capsules 10122-420-28 30 mg Orange/yellow hard gelatin capsule printed with black ink "A733" and "30 mg" Bottles of 28 capsules 10122-430-28 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (between 59°F and 86°F). Brief exposure to temperatures up to 40°C (104°F) may be tolerated provided the mean kinetic temperature does not exceed 25°C (77°F); however, such exposure should be minimized. Keep container tightly closed and protect from moisture.
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved labeling (Medication Guide)
Patients or their caregiver(s) should be informed that a registry for patients taking JUXTAPID has been established to monitor and evaluate the long-term effects of JUXTAPID. Patients or their caregiver(s) are encouraged to participate in the registry and should be informed that their participation is voluntary. For information regarding the registry program visit www.JUXTAPID.com or call 1-877-902-4099.
Advise patients of the following:
Risk of Hepatotoxicity [see Warnings and Precautions (5.1)]
- JUXTAPID can cause both elevations in transaminases and hepatic steatosis. Discuss with patients or their caregiver(s) about the importance of monitoring of liver-related tests before taking JUXTAPID, prior to each dose escalation, and periodically thereafter.
- Advise patients of the potential for increased risk of liver injury if alcohol is consumed while taking JUXTAPID. It is recommended that patients taking JUXTAPID limit consumption to not more than one alcoholic drink per day.
- JUXTAPID is commonly associated with nausea, vomiting, and abdominal pain. Advise patients or their caregiver(s) to promptly report these symptoms if they increase in severity, persist, or change in the character, as they might reflect liver injury. Patients or their caregiver(s) should also report any other symptoms of possible liver injury, including fever, jaundice, lethargy, or flu-like symptoms.
JUXTAPID REMS Program [see Warnings and Precautions (5.2)]
JUXTAPID is only available through a restricted program called JUXTAPID REMS Program and therefore, JUXTAPID is only available from certified pharmacies that are enrolled in the program.
Embryofetal Toxicity [see Warnings and Precautions (5.3), Drug Interactions (7.2), Use in Specific Populations (8.1, 8.3)]
- JUXTAPID is contraindicated in pregnancy since it may cause fetal harm. Advise female patients who become pregnant to discontinue JUXTAPID and inform their healthcare provider of a known or suspected pregnancy.
- Advise female patients of reproductive potential to use effective contraception during treatment with JUXTAPID and for two weeks after the final dose.
- Advise patients who are taking oral contraceptives and experience vomiting or diarrhea while taking JUXTAPID to use an effective alternative contraceptive method until 7 days after resolution of symptoms.
Lactation [see Use in Specific Populations (8.2)]
Advise female patients not to breastfeed during treatment with JUXTAPID.
Dietary Supplements [see Warnings and Precautions (5.4)]
- Inform patients or their caregiver(s) that JUXTAPID may reduce the absorption of fat-soluble nutrients. Instruct patients or their caregiver(s) to take daily nutritional supplements that contain the dosages of vitamin E and essential fatty acids recommended in the Dosage and Administration Section (2.2).
Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.5)]
- Inform patients or their caregiver(s) that gastrointestinal adverse reactions are common with JUXTAPID. These include, but are not limited to, diarrhea, nausea/vomiting, abdominal pain/discomfort, flatulence, and constipation. Advise patients or their caregiver(s) to adhere to low-fat diet supplying <20% of energy from fat or less than 30 grams of fat, whichever is less, to reduce the risk of gastrointestinal adverse reactions.
- Instruct patients or their caregiver(s) to stop JUXTAPID and contact their healthcare provider if severe diarrhea occurs or if they experience symptoms of volume depletion such as lightheadedness, decreased urine output, or tiredness.
- Inform patients or their caregiver(s) that taking JUXTAPID with food may adversely impact gastrointestinal tolerability; therefore, they should take JUXTAPID at least 2 hours after the evening meal, swallowing each capsule whole, with water. Inform patients or their caregiver(s) that if the patient is unable to swallow the intact capsule(s), the capsule(s) can be opened and the contents sprinkled on 1 tablespoon of apple sauce or mashed banana, which are fat free. For younger patients who require a smaller spoon for administration, inform the patient or their caregiver(s) to sprinkle the capsule content onto 1 tablespoon of apple sauce or mashed banana and administer the entire amount using the smaller spoon.
- Absorption of oral medications may be affected in patients who develop diarrhea or vomiting. For example, hormone absorption from oral contraceptives may be incomplete, warranting the use of additional contraceptive methods. Patients who develop these symptoms should seek advice from their healthcare provider.
Drug Interactions [see Warnings and Precautions (5.6, 5.7) and Drug Interactions (7)]
- Instruct patients or their caregiver(s) to avoid food or drinks containing grapefruit during JUXTAPID treatment.
- Because multiple drug-drug interactions have been described with JUXTAPID, advise patients or their caregiver(s) to tell their healthcare provider(s) about all medications, nutritional supplements, and vitamins that they are taking or may be taking while taking JUXTAPID.
- Advise patients taking simvastatin or lovastatin that JUXTAPID may cause myopathy. Instruct patients to promptly report any unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever.
Missed Doses
- If a dose of JUXTAPID is missed, the normal dose should be taken at the usual time the next day. If dosing is interrupted for more than a week, tell patients or their caregiver(s) to contact their healthcare provider before restarting treatment.
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 02/2026
70015629-017465-00Medication Guide
JUXTAPID® (JUKS-tuh-pid)
(lomitapide)
capsules, for oral useWhat is the most important information I should know about JUXTAPID? - JUXTAPID is available only through certified pharmacies that are enrolled in the JUXTAPID REMS Program. Your healthcare provider must be enrolled in the program in order for you to be prescribed JUXTAPID.
- There is a registry that collects information about the effects of taking JUXTAPID over time. Ask your healthcare provider for more information about this registry or visit www.JUXTAPID.com or call 1-877-902-4099.
JUXTAPID may cause serious side effects including: -
Liver problems. JUXTAPID can cause liver problems such as increased liver enzymes or increased fat in the liver.
- Your healthcare provider should do blood tests to check your liver before you start JUXTAPID, if your dose is increased, and while you take JUXTAPID. If your tests show some liver problems, your healthcare provider may adjust your dose of JUXTAPID or stop it altogether.
- Tell your healthcare provider if you have had liver problems, including liver problems while taking other medicines.
- JUXTAPID may cause nausea, vomiting, and stomach pain, especially if you do not eat a low-fat diet. These side effects can also be symptoms of liver problems.
- Tell your healthcare provider right away if you have any of these symptoms of liver problems while taking JUXTAPID:
- Nausea, vomiting, or stomach pain that gets worse, does not go away, or changes
- fever
- flu-like symptoms
- yellowing of your eyes or skin
- you are more tired than usual
- Drinking alcohol may increase your chance of having liver problems or make your liver problems worse. You should not have more than 1 alcoholic drink each day while taking JUXTAPID.
-
Harm to your unborn baby. JUXTAPID may cause harm to your unborn baby.
- If you are pregnant, think you may be pregnant, or are planning to become pregnant, do not take JUXTAPID.
- If you are a female who can get pregnant, you should have a pregnancy test before you start taking JUXTAPID. Your pregnancy test must be negative for you to get JUXTAPID.
- Do not have sex while taking JUXTAPID unless you are using effective birth control.
- Talk with your healthcare provider or nurse to find the best method of birth control for you.
- Birth control pills may not work as well if you have diarrhea or vomiting.
- If you start taking birth control pills while you are taking JUXTAPID, tell your healthcare provider. Your healthcare provider might need to change your dose of JUXTAPID.
- Continue taking birth control during JUXTAPID treatment and for 2 weeks after your last dose of JUXTAPID.
- If you become pregnant while taking JUXTAPID, stop taking JUXTAPID and call your healthcare provider right away.
- There is a registry that monitors outcomes in women exposed to JUXTAPID during pregnancy. If you become pregnant while taking JUXTAPID, call 1-877-902-4099 or visit www.JUXTAPID.com for more information about the Lomitapide Observational Worldwide Exposure Registry (LOWER).
What is JUXTAPID?
JUXTAPID is a prescription medicine used along with a low-fat diet, exercise, and other low-density lipoprotein cholesterol (LDL-C) lowering medicines to reduce LDL-C in adults and children 2 years of age and older with a type of high cholesterol called homozygous familial hypercholesterolemia (HoFH).
It is not known if JUXTAPID is safe and effective in people with kidney problems including people with end-stage kidney disease who are not on dialysis.
It is not known if JUXTAPID is safe and effective when used in children under the age of 2.Who should not take JUXTAPID?
Do not take JUXTAPID if you:- are pregnant, think you may be pregnant, or are planning to become pregnant. See "What is the most important information I should know about JUXTAPID?"
- take medicines that are strong or moderate CYP3A4 inhibitors, which affect how the body breaks down JUXTAPID. Check with your healthcare provider or pharmacist to see if you are taking any of these medicines. These may include certain medicines intended to treat bacterial, fungal, or viral infections, and medicines to treat depression, high blood pressure, or chest pain (angina).
- drink grapefruit juice
- have moderate or severe liver problems or active liver disease, including people who have unexplained abnormal liver tests.
What should I tell my doctor before taking JUXTAPID?
Before you take JUXTAPID, tell your healthcare provider about all of your medical conditions, including if you:- have liver problems.
- have kidney problems.
- have intestine or bowel problems.
- drink alcohol
- are breastfeeding or plan to breastfeed. It is not known if JUXTAPID passes into your breast milk. You and your healthcare provider should decide if you will take JUXTAPID or breastfeed. You should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Before starting a new medicine while taking JUXTAPID, even if you will only be taking it for a short time, ask your healthcare provider or pharmacist if it is safe to take with JUXTAPID.
JUXTAPID may affect the way other medicines work, and other medicines may affect how JUXTAPID works.
Certain medicines can affect how your liver breaks down other medicines.
Especially tell your healthcare provider if you take:- a blood thinner called warfarin
- medicines for high cholesterol, including statins such as lovastatin or simvastatin and bile acid binding medicines such as colesevelam or cholestyramine
- medicines for bacteria, fungus, or viral infection (including HIV and hepatitis C)
- medicines for depression, high blood pressure, or angina
- birth control pills
How should I take JUXTAPID? - Take JUXTAPID exactly as your healthcare provider tells you to take it.
- Your healthcare provider will tell you how much JUXTAPID to take and when to take it.
- Your healthcare provider may change your dose of JUXTAPID if needed.
- Do not change your JUXTAPID dose yourself.
- Take JUXTAPID 1 time each day with water, at least 2 hours after your evening meal.
- You should not take JUXTAPID with food. Taking JUXTAPID with food may cause stomach problems.
- Swallow JUXTAPID capsules whole.
- If you cannot swallow JUXTAPID capsules whole, you can open the capsule and sprinkle the contents on 1 tablespoon of apple sauce or mashed banana.
- If you or your child require a smaller spoon, sprinkle the contents onto 1 tablespoon of apple sauce or mashed banana and use a teaspoon to take the full amount.
- If you take a medicine that lowers cholesterol by binding bile acids, such as colesevelam or cholestyramine, take it at least 4 hours before or 4 hours after you take JUXTAPID. Ask your healthcare provider if you are not sure if you take these medicines.
- To help lower the chance of stomach problems, stay on a low-fat diet. Ask your healthcare provider about talking to a dietician to learn what you should eat while taking JUXTAPID.
- JUXTAPID makes it harder for some nutrients to get into your body. Your healthcare provider will tell you how much Vitamin E and essential fatty acids supplements to take each day while taking JUXTAPID.
- If you take too much JUXTAPID, call your healthcare provider right away.
- Do not stop JUXTAPID unless your healthcare provider tells you to stop it.
- If you miss a dose of JUXTAPID, take your usual dose the next day at the usual time. If you stop taking JUXTAPID for more than a week, talk to your healthcare provider before restarting treatment.
What are the possible side effects of JUXTAPID?
JUXTAPID can cause serious side effects, including:- See "What is the most important information I should know about JUXTAPID?"
- problems absorbing certain nutrients. JUXTAPID may decrease your ability to absorb fat-soluble nutrients such as Vitamin E and fatty acids. You should take supplements each day that contain fat-soluble vitamins. People with bowel or pancreas problems may have an increased chance of not being able to absorb these nutrients. See "How should I take JUXTAPID?"
- gastrointestinal symptoms. Diarrhea, nausea, vomiting, and stomach pain or discomfort are very common when taking JUXTAPID. Strictly following a low-fat diet may help lower the chance of having these symptoms. Stop taking JUXTAPID and tell your healthcare provider if you have severe diarrhea, especially if you also have lightheadedness, decreased urine output, or tiredness.
- muscle pain, tenderness and weakness (myopathy). This can happen when JUXTAPID is taken with certain doses of simvastatin and lovastatin. Tell your doctor right away if you have unexplained muscle pain, tenderness, or weakness, especially if you have a fever or feel more tired than usual while you take JUXTAPID.
- increased levels of certain blood thinners. JUXTAPID can increase the level of the blood thinner, warfarin. If you take warfarin, your healthcare provider should check your blood clotting times frequently, especially after your dose of JUXTAPID changes.
- liver problems caused by certain drugs. Certain medicines can cause liver problems, including isotretinoin, acetaminophen, methotrexate, tetracyclines, and tamoxifen. If you take these medicines with JUXTAPID your healthcare provider may do blood tests more often to check your liver.
The most common side effects of JUXTAPID in adults and children 5 years and older include: - diarrhea
- nausea
- stomach (abdominal) pain
- indigestion
- vomiting
- increases in liver enzymes
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of JUXTAPID. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store JUXTAPID? - Store JUXTAPID at room temperature, between 68°F and 77°F (20°C and 25°C).
- Keep JUXTAPID in a tightly closed container.
- Keep JUXTAPID capsules dry.
- Safely throw away medicine that is out of date or no longer needed.
General information about the safe and effective use of JUXTAPID.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use JUXTAPID for a condition for which it was not prescribed. Do not give JUXTAPID to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about JUXTAPID. You can ask your pharmacist or healthcare provider for information about JUXTAPID that is written for health professionals.
For more information, go to www.JUXTAPID.com or call 1-85-JUXTAPID (1-855-898-2743).What are the ingredients in JUXTAPID?
Active ingredient: lomitapide mesylate
Inactive ingredients: pregelatinized starch, sodium starch glycolate, microcrystalline cellulose, lactose monohydrate, silicon dioxide, magnesium stearate
Capsule shell: The capsule shells of all strengths contain gelatin and titanium dioxide; the 2 mg capsules contain black iron oxide; the 5 mg, 10 mg and 30 mg capsules also contain red iron oxide; and the 30 mg capsules also contain yellow iron oxide. The imprinting ink contains shellac, black iron oxide, and propylene glycol.Chiesi JUXTAPID is a registered trademark owned by the Chiesi Group ©2026 Chiesi USA. All rights reserved.
Manufactured for: Chiesi Farmaceutici S.p.A., Parma, Italy. - PRINCIPAL DISPLAY PANEL - 2 mg Capsule Bottle Label
-
PRINCIPAL DISPLAY PANEL - 2 mg Capsule Bottle Carton
NDC: 10122-402-28
Rx Only28 capsules
Juxtapid®
(lomitapide) capsules2 mg
Attention Pharmacist:
Dispense the accompanying
Medication Guide to each patient.
- PRINCIPAL DISPLAY PANEL - 5 mg Capsule Bottle Label
-
PRINCIPAL DISPLAY PANEL - 5 mg Capsule Bottle Carton
NDC: 10122-405-28
Rx Only28 capsules
Juxtapid®
(lomitapide) capsules5 mg
Attention Pharmacist:
Dispense the accompanying
Medication Guide to each patient.Chiesi
- PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Label
-
PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Carton
NDC: 10122-410-28
Rx Only28 capsules
Juxtapid®
(lomitapide) capsules10 mg
Attention Pharmacist:
Dispense the accompanying
Medication Guide to each patient.Chiesi
- PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label
-
PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Carton
NDC: 10122-420-28
Rx Only28 capsules
Juxtapid®
(lomitapide) capsules20 mg
Attention Pharmacist:
Dispense the accompanying
Medication Guide to each patient.Chiesi
- PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Label
-
PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Carton
NDC: 10122-430-28
Rx Only28 capsules
Juxtapid®
(lomitapide) capsules30 mg
Attention Pharmacist:
Dispense the accompanying
Medication Guide to each patient.Chiesi
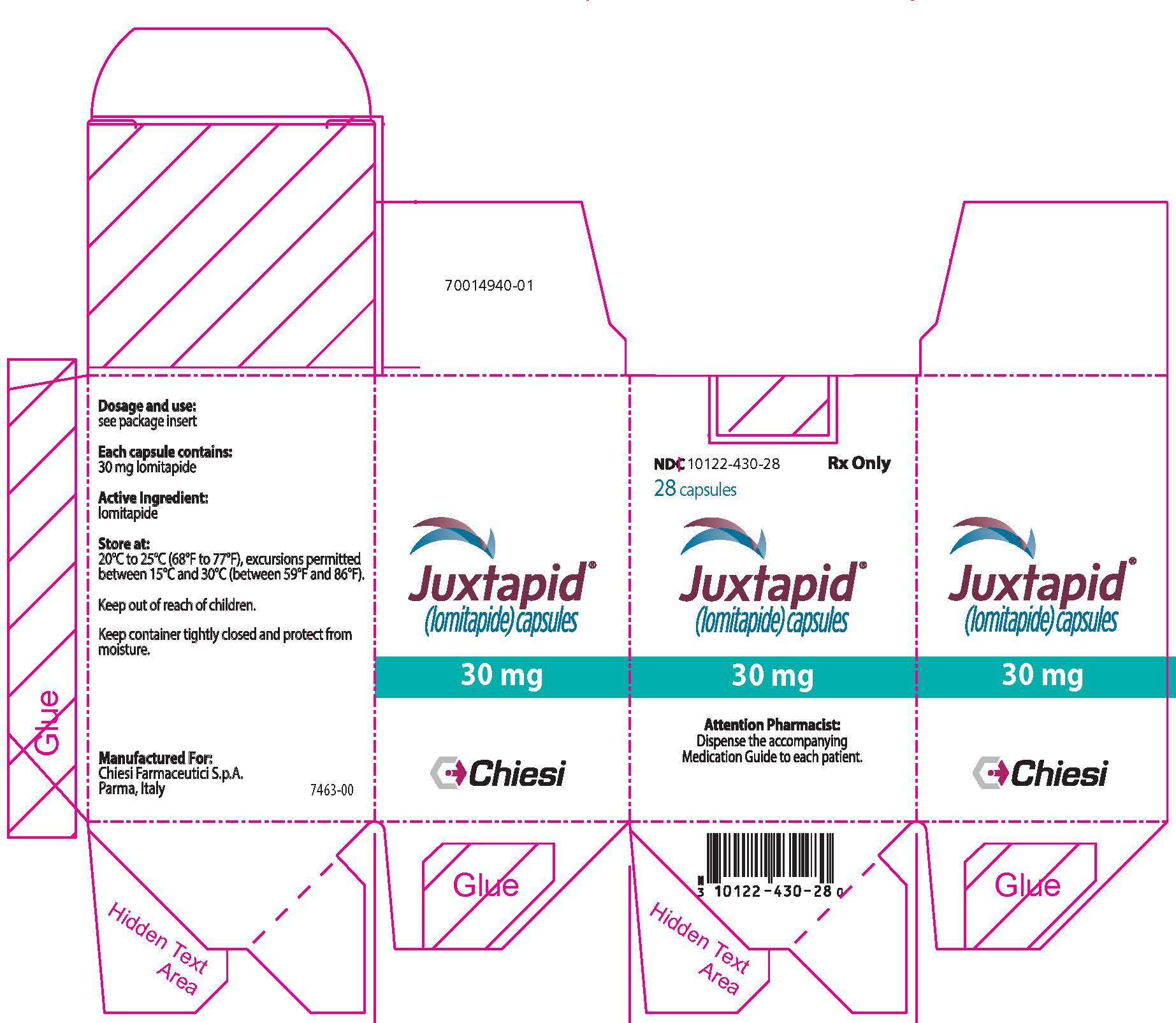
-
INGREDIENTS AND APPEARANCE
JUXTAPID
lomitapide mesylate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 10122-402 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOMITAPIDE MESYLATE (UNII: X4S83CP54E) (LOMITAPIDE - UNII:82KUB0583F) LOMITAPIDE 2 mg Product Characteristics Color GRAY (Swedish Grey) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code A733;2mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10122-402-28 28 in 1 BOTTLE; Type 0: Not a Combination Product 02/25/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203858 02/25/2026 JUXTAPID
lomitapide mesylate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 10122-405 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOMITAPIDE MESYLATE (UNII: X4S83CP54E) (LOMITAPIDE - UNII:82KUB0583F) LOMITAPIDE 5 mg Product Characteristics Color orange (Swedish Orange) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code A733;5mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10122-405-28 28 in 1 BOTTLE; Type 0: Not a Combination Product 01/28/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203858 01/03/2013 JUXTAPID
lomitapide mesylate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 10122-410 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOMITAPIDE MESYLATE (UNII: X4S83CP54E) (LOMITAPIDE - UNII:82KUB0583F) LOMITAPIDE 10 mg Product Characteristics Color orange (Swedish Orange) , white Score no score Shape CAPSULE Size 19mm Flavor Imprint Code A733;10mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10122-410-28 28 in 1 BOTTLE; Type 0: Not a Combination Product 01/28/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203858 01/03/2013 JUXTAPID
lomitapide mesylate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 10122-420 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOMITAPIDE MESYLATE (UNII: X4S83CP54E) (LOMITAPIDE - UNII:82KUB0583F) LOMITAPIDE 20 mg Product Characteristics Color white Score no score Shape CAPSULE Size 19mm Flavor Imprint Code A733;20mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10122-420-28 28 in 1 BOTTLE; Type 0: Not a Combination Product 01/28/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203858 01/03/2013 JUXTAPID
lomitapide mesylate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 10122-430 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOMITAPIDE MESYLATE (UNII: X4S83CP54E) (LOMITAPIDE - UNII:82KUB0583F) LOMITAPIDE 30 mg Product Characteristics Color yellow, orange (Swedish Orange) Score no score Shape CAPSULE Size 22mm Flavor Imprint Code A733;30mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10122-430-28 28 in 1 BOTTLE; Type 0: Not a Combination Product 06/22/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203858 01/03/2013 Labeler - Chiesi USA, Inc. (088084228)
Trademark Results [Juxtapid]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 JUXTAPID 97193314 not registered Live/Pending |
Amryt Pharmaceuticals Inc. 2021-12-28 |
 JUXTAPID 97193306 not registered Live/Pending |
Amryt Pharmaceuticals Inc. 2021-12-28 |
 JUXTAPID 85981114 4512713 Live/Registered |
Aegerion Pharmaceuticals, Inc. 2013-02-22 |
 JUXTAPID 85980934 4508190 Live/Registered |
Aegerion Pharmaceuticals, Inc. 2012-12-14 |
 JUXTAPID 85857475 not registered Dead/Abandoned |
Aegerion Pharmaceuticals, Inc. 2013-02-22 |
 JUXTAPID 85802929 not registered Dead/Abandoned |
Aegerion Pharmaceuticals, Inc. 2012-12-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.