Premier Value 44-525-Discontinued
Cold and Allergy by
Drug Labeling and Warnings
Cold and Allergy by is a Otc medication manufactured, distributed, or labeled by Chain Drug Consortium, LNK International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
COLD AND ALLERGY- chlorpheniramine maleate, phenylephrine hcl tablet
Chain Drug Consortium
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Premier Value 44-525-Discontinued
Uses
- temporarily relieves these symptoms due to hay fever (allergic rhinitis) or other respiratory allergies:
- runny nose
- itchy, watery eyes
- itching of the nose or throat
- sneezing
- nasal congestion
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
- nasal congestion
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- high blood pressure
- heart disease
- thyroid disease
- diabetes
- glaucoma
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
When using this product
- do not exceed recommended dose
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Directions
- adults and children 12 years and over: take 1 tablet every 4 hours. Do not take more than 6 tablets in 24 hours.
- children under 12 years: do not use this product in children under 12 years of age
Other information
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients
croscarmellose sodium, lactose, magnesium stearate, microcrystalline cellulose, silica gel, stearic acid
Principal Display Panel
Premier
Value®
NDC: 68016-684-24
Cold & Allergy
Chlorpheniramine maleate - ANTIHISTAMINE
Phenylephrine HCl - NASAL DECONGESTANT
Relieves: Nasal Congestion Runny Nose,
Sneezing & Itchy, Watery Eyes
24 Tablets
One Tablet Dose
INDEPENDENTLY TESTED
PV
SATISFACTION GUARANTEED
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
50844 ORG061252508
DISTRIBUTED BY
CHAIN DRUG CONSORTIUM, LLC
UPARC, BLDG. A3, SUITE 338
1020 WILLIAM PITT WAY
PITTSBURGH, PA 15238
www.chaindrugconsortium.com
If for any reason you are not satisfied with
this product, please return it to the store
where purchased for a full refund.
Premier Value 44-525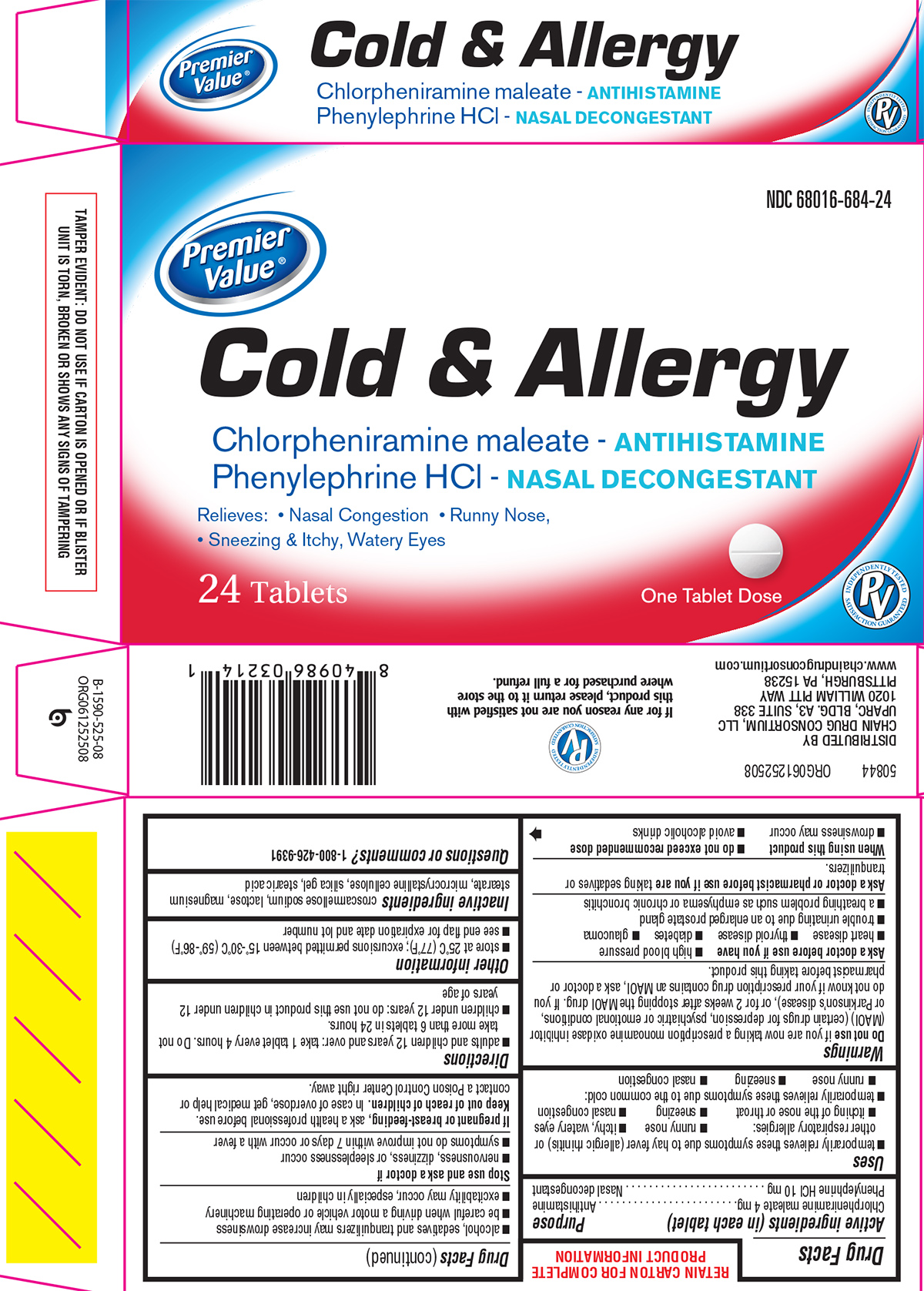
| COLD AND ALLERGY
chlorpheniramine maleate, phenylephrine hcl tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Chain Drug Consortium (101668460) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(68016-684) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | MANUFACTURE(68016-684) | |