ADSOL RED CELL PRESERVATION SOLUTION SYSTEM IN PLASTIC CONTAINER (PL 146 PLASTIC) (anticoagulant citrate phosphate dextrose- cpd solution and adsol preservation solution kit
ADSOL Red Cell Preservation Solution System in Plastic Container (PL 146 Plastic) by
Drug Labeling and Warnings
ADSOL Red Cell Preservation Solution System in Plastic Container (PL 146 Plastic) by is a Prescription medication manufactured, distributed, or labeled by Fenwal, Inc., Fenwal International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
BLOOD-PACK™ Unit with an Integral SEPACELL™ RS-2000/RZ-2000 Whole Blood Leukocyte Reduction Filter for Collection and Filtration of Whole Blood Using CPD/ADSOL™ Red Cell Preservation Solution
Rx only
Contains Y-Sampling Site for the collection of unanticoagulated whole blood samples for laboratory testing and the DONORCARE™ Needle Guard.
Integral filter unit intended for leukocyte reduction of whole blood up to 72 hours after blood collection. The leukocyte reduced blood products may then be stored for the maximum allowable dating period.
Instructions for Use
Collection Procedure:
Use aseptic technique.
Note: Nominal tubing dimensions of product are 0.118" inner diameter x 0.025" wall thickness.
Note: If the Y-Sampling Site is not used, donor samples may be collected using an alternate method following standard procedures.
Precautions:
- Do not use unless the solutions are clear.
- Before beginning procedure, obtain one access device for each BLOOD-PACK™ unit with Y-Sampling Site to be processed.
- Identify BLOOD-PACK unit using appropriate donor identification system.
- Adjust donor scale to desired collection weight and position primary container on the donor scale as far as possible below donor arm.
- Clamp donor tubing between needle and Y-Sampling Site with hemostat. This step may be performed prior to step 1 or 2.
- Apply pressure to donor’s arm and disinfect site of venipuncture.
- If blood pressure cuff is used, inflate to approximately 60 mmHg.
- Remove needle cover per instructions below:
a. Holding the hub and cover near the tamper-evident seal, twist cover 1/4 turn to break seal.
b. Remove needle cover, being careful not to drag the cover across the needle point.
- Perform venipuncture, appropriately secure donor needle and/or tubing and release clamp on donor tubing.
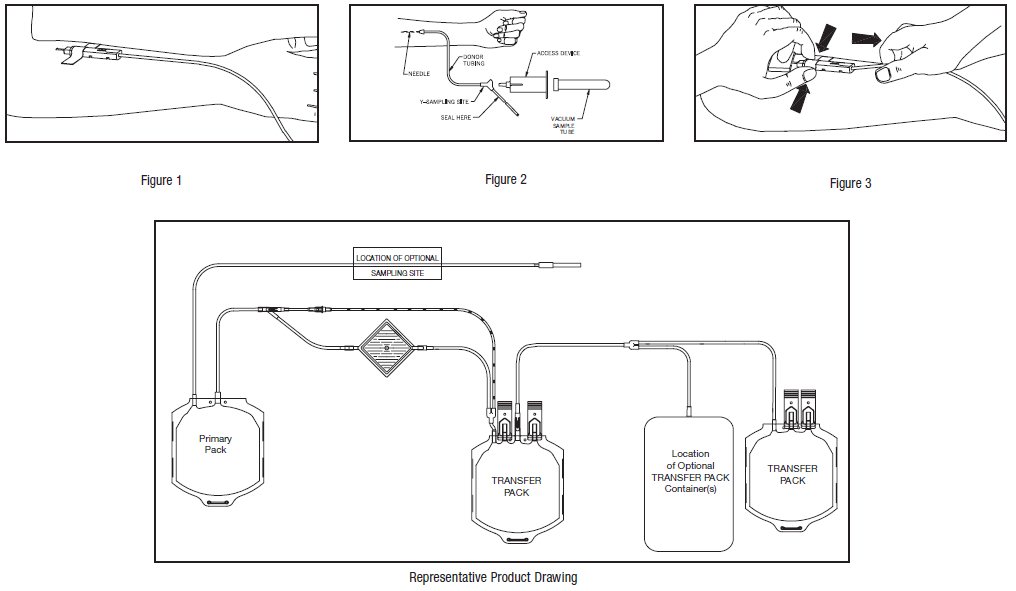
- When good blood flow is established, slide the DONORCARE™ Needle Guard over the needle hub into the engaged position. Leave the front third of the needle hub exposed for access. Stabilize the front of the needle guard to arm with tape. (see Figure 1)
Note: In difficult collection conditions (e.g. slow blood flow), leave the needle guard disengaged, behind the hub during collection. Engage the needle guard at the end of blood collection.
- Mix blood and anticoagulant in primary container at several intervals during collection and immediately after collection.
- Collect the appropriate volume based on BLOOD-PACK unit used.
Note: The volume of anticoagulant is sufficient for the blood collection indicated on BLOOD-PACK unit ± 10%. - Release the pressure on the donor’s arm as appropriate.
Precaution: Do not proceed with the remaining steps until the entire whole blood unit is collected.
- To avoid possible contamination of the whole blood unit, before filling whole blood sample tubes, hermetically seal the donor tubing near the Y-Sampling Site on the side leading to the primary container using a metal clip or appropriate alternate method.
Precaution: Complete steps 13 - 22 within approximately 4 minutes after sealing the donor tubing to avoid possible clot formation in the tubing.
- To collect samples, insert the access device by pushing firmly into the Y-Sampling Site until the membrane seal is penetrated (see Figure 2).
Note: If the access device is assembled such that the outer barrel is screwed onto the Luer, make sure to rotate clockwise upon insertion to avoid barrel detaching from Luer.
- Open the cap on the access device (if applicable).
- Directly align the vacuum sample tube with the internal needle in the access device. Insert vacuum sample tube into device until the stopper is punctured.
- Allow vacuum sample tube to fill with blood then remove from the access device.
- Repeat steps 15 and 16 until the desired number of vacuum sample tubes have been filled.
Notes:
- If the access device needs to be replaced, use a hemostat to clamp the tubing between the needle and the Y-Sampling Site. Then, grasp base of Sampling Site with one hand and pull the access device out with the other hand. Firmly insert the new access device. Remove hemostat and continue sampling.
- If the access device is assembled such that the outer barrel is screwed onto the Luer, make sure to rotate clockwise upon removal to avoid barrel detaching from Luer.
- The access device can only be replaced one time.
Precaution: When replacing the access device, be careful to avoid contact with any blood droplets on the Luer or Sampling Site. Discard used access device appropriately
- Release remaining pressure on donor’s arm.
- If desired, apply hemostat to donor tubing between needle and Y-Sampling Site.
- Withdrawal of Needle (see Figure 3)
Precaution: The needle guard must be held stationary while the needle is withdrawn into it.
·
a. Place folded sterile gauze over puncture site and hold in place with finger tip without exerting pressure.
b. Hold sides of needle guard near the front, between the index finger and thumb. Pull the tubing smoothly until the needle is locked into the needle guard.
c. Confirm the needle lock by:
1. Listen for the 2nd “click” as the needle is drawn into the needle guard.
2. Ensure the tubing cannot be pulled through the needle guard.
- Remove and discard the Y-Sampling Site and needle guard into an appropriate biohazardous waste container following established procedures. If donor tubing is also to be discarded, hermetically seal donor tubing directly above the primary container and remove.
Note: Step 22 may be performed prior to step 21 if desired.
- If the donor tubing is not hermetically sealed directly above the primary container, then strip the blood from the remaining donor tubing into the primary container. Mix and allow tubing to refill; repeat once.
Filtration Procedure:
Precaution: Whole blood collected from certain donors may have extended filtration times and the potential for ineffective filtration and leukoreduction.
Note: The time of whole blood filtration may vary depending on processing option selected.
a. Within 8 hours of collection if whole blood is held at ambient temperature.
b. Within 72 hours of collection if whole blood is refrigerated.
c. Mix unfiltered whole blood thoroughly. Invert the primary container and hang the filter set such that the filter remains vertical. To achieve maximum flow rate, allow set to hang to full length.
Note: The storage container must remain below the level of the filter during filtration.
- Inspect all tubing to insure it hangs freely without kinks. Install clamp on vent line and close. (4R4423 clamp recommended).
- Break the in-line cannula above the filter to start filtration.
Note: Manual or mechanical pressure should not be used to increase the flow rate through the filter.
Note: Tubing below the filter should not be stripped at any time during the filtration process.
Note: If the filtration of whole blood is initiated at ambient temperature and not completed within 8 hours after blood collection, then filtration should be completed between 1 and 6°C.
- When flow stops, open the clamp and gently squeeze the filtered whole blood container until the air is expelled through the vent line and filtered blood fills the donor segment tubing as desired.
- Allow filtration to continue until the inlet side of the filter is filled with air.
- Seal the transfer tubing below the filter. Also seal the vent line tubing directly above the top donor segment number (near the slide clamp). At each location, seal in three places and cut the middle seal being careful to avoid fluid splatter. Discard filter and primary container appropriately.
Note: If a QC sample is desired, thoroughly mix the filtered whole blood and strip the donor segment tubing. Use the last segment (or segments) as the QC sample.
- Make donor segments. Leave segments attached to the filtered whole blood container.
Component Preparation Procedure:
Note: Platelet concentrates are not intended to be made with this product.
- ADSOL™ red cell preservation solution should be added to the red blood cells immediately after the removal of plasma. Preparation of AS-1 red blood cells may vary depending on processing option selected:
a. Within 8 hours of blood collection if whole blood is held at ambient temperature.
b. Within 3 days of blood collection if whole blood is refrigerated.
- Centrifuge filtered whole blood and secondary containers to prepare CPD red blood cells using the appropriate spin condition.
- Place filtered whole blood container in plasma extractor, and express plasma into empty TRANSFER PACK™ container by releasing pressure plate and opening closure in tubing of filtered whole blood container.
- When the desired amount of plasma has been removed, clamp the tubing between Y and plasma container. Seal transfer tubing in three places. Cut the middle seal, being careful to avoid fluid splatter.
- Suspend ADSOL red cell preservation solution container; open closure in tubing and drain contents onto CPD red blood cells. Clamp tubing.
- Seal transfer tubing in three places near the container of filtered red blood cells. Cut the middle seal, being careful to avoid fluid splatter. For double BLOOD-PACK unit codes, discard ADSOL solution container. For all other ADSOL codes, the empty solution container may now be used as a TRANSFER PACK container for further component preparation.
- For further processing of plasma product with multiple BLOOD-PACK units, use standard component processing and storage techniques.
Note: Fresh frozen plasma should be separated from the red blood cells and placed in the freezer at -18°C or colder within 8 hours after blood collection.
- Mix the ADSOL red cell preservation solution and red blood cells thoroughly, producing AS-1 red blood cells, Leukocytes Reduced.
- Store suspended AS-1 red blood cells, Leukocytes Reduced, between 1 and 6°C.
- Infuse AS-1 red blood cells, Leukocytes Reduced, within 42 days of collection.
Warning: Failure to achieve closed system processing conditions negates the extended storage claim and the red cell product must be transfused within 24 hours.
Store at Controlled Room Temperature.
USP Definition of “Controlled Room Temperature”
United States Pharmacopeia, General Notices.
United States Pharmacopeial Convention, Inc.
12601 Twinbrook Parkway, Rockville, MD.
- SPL UNCLASSIFIED SECTION
-
SPL UNCLASSIFIED SECTION
 Fenwal, Inc.
Fenwal, Inc.
Lake Zurich, IL 60047 USAMade in USA
1-800-933-6925
07-19-07-537 REV: A
11/2012FENWAL, BLOOD-PACK, TRANSFER PACK and ADSOL are trademarks of Fenwal, Inc.
SEPACELL is a trademark of Asahi Kasei Medical Co., Ltd.
DONORCARE is a trademark of ITL Corporation.
© 2012 Fenwal, Inc. All rights reserved.
-
PACKAGE/LABEL DISPLAY PANEL
Code 4R3339
10 Units
Fenwal™
Anticoagulant Citrate Phosphate Dextrose Solution, USP (CPD) BLOOD-PACK™ Unit; TRANSFER-PACK™ Container with ADSOL™ Red Cell Preservation Solution; Integral SEPACELL™ Whole Blood Leukocyte Reduction Filter
Triple For the Collection and Processing of 450 mL Blood
Y-Sampling Site, DONORCARETM Needle Guard, 16 ga. Ultra Thin Wall Needle
Rx only
Each unit consists of a PL 146 Plastic primary container with 63 mL of CPD solution containing 1.66 g Sodium Citrate (dihydrate) USP, 1.61 g Dextrose (monohydrate) USP, 188 mg Citric Acid (anhydrous) USP, 140 mg Monobasic Sodium Phosphate (monohydrate) USP, pH may have been adjusted with sodium hydroxide; one 400 mL PL 146 Plastic TRANSFER-PACK container with 100 mL of ADSOL Red Cell Preservation Solution containing 2.2 g Dextrose (monohydrate) USP, 900 mg Sodium Chloride USP, 750 mg Mannitol USP, 27 mg Adenine USP; one empty 400 mL PL 146 Plastic TRANSFER-PACK container; one integral SEPACELL Whole Blood Leukocyte Reduction Filter and one empty 450 mL PL 146 Plastic TRANSFER-PACK container for the storage of AS-1 red blood cells, leukocytes reduced.
Sterile, non-pyrogenic fluid path
See instructions for use.Store at Controlled Room Temperature (refer to direction insert).
- Open pouch by tearing across at notch.
- Direct handling of product surfaces prior to extended storage in the foil pouch, may result in mold growth.
- Unused units in open foil pouch may be kept up to 60 days by folding and securing open end of foil pouch to prevent possible loss of moisture, provided:
- I) Units are not removed from foil pouch, or
- II) Unused units removed from foil pouch are returned to the foil pouch within 12 hours. Units may be removed from the pouch and returned only once.
- Units removed from the foil pouch (that are not returned to the pouch within 12 hours) must be used within 4 days (96 hours). Units out of the foil pouch for longer than 96 hours must be discarded.
FENWAL, BLOOD-PACK, ADSOL, and TRANSFER-PACK are trademarks of Fenwal, Inc.
SEPACELL is a trademark of Asahi Kasei Medical Co., Ltd.
DONORCARE is a trademark of ITL Corporation.
 Fenwal, Inc.
Fenwal, Inc.
Lake Zurich, IL 60047 USAMade in USA
07-28-05-526 REV: A

-
INGREDIENTS AND APPEARANCE
ADSOL RED CELL PRESERVATION SOLUTION SYSTEM IN PLASTIC CONTAINER (PL 146 PLASTIC)
anticoagulant citrate phosphate dextrose (cpd) solution and adsol preservation solution kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0942-6466 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0942-6466-03 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BAG 63 mL Part 2 1 BAG 100 mL Part 1 of 2 CPD
citrate phosphate dextrose solutionProduct Information Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Trisodium Citrate Dihydrate (UNII: B22547B95K) (Anhydrous Citric Acid - UNII:XF417D3PSL) Anhydrous Citric Acid 1.66 g in 63 mL Dextrose Monohydrate (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) Dextrose Monohydrate 1.61 g in 63 mL Anhydrous Citric Acid (UNII: XF417D3PSL) (Anhydrous Citric Acid - UNII:XF417D3PSL) Anhydrous Citric Acid 188 mg in 63 mL Sodium Phosphate, Monobasic, Monohydrate (UNII: 593YOG76RN) (PHOSPHATE ION - UNII:NK08V8K8HR) Sodium Phosphate, Monobasic, Monohydrate 140 mg in 63 mL Inactive Ingredients Ingredient Name Strength Sodium Hydroxide (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 63 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN811104 03/01/2007 Part 2 of 2 ADSOL RED CELL PRESERVATION SOLUTION SYSTEM
adsol red cell preservation solution solutionProduct Information Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Dextrose Monohydrate (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) Dextrose Monohydrate 2.2 g in 100 mL Sodium Chloride (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) Sodium Chloride 900 mg in 100 mL Mannitol (UNII: 3OWL53L36A) (Mannitol - UNII:3OWL53L36A) Mannitol 750 mg in 100 mL Adenine (UNII: JAC85A2161) (Adenine - UNII:JAC85A2161) Adenine 27 mg in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 100 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN811104 03/01/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN811104 03/01/2007 Labeler - Fenwal, Inc. (794519020) Establishment Name Address ID/FEI Business Operations Fenwal International, Inc. 091164590 MANUFACTURE(0942-6466)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
 Manufacturer
Manufacturer