UREA by AvKARE UREA cream
UREA by
Drug Labeling and Warnings
UREA by is a Prescription medication manufactured, distributed, or labeled by AvKARE. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
Rx Only
For external use only. Not for ophthalmic use.
Description
Urea 40% is a keratolytic emollient which is a gentle, yet potent, tissue softener for nails and/or skin. Each gram of Urea 40%contains 40% urea as an active ingredient, and the following inactive ingredients: Ceteareth-6, Ceteareth-25, Cetyl Alcohol, Mineral Oil, Propylene Glycol, Purified Water, Sodium Hydroxide, Stearyl Alcohol, Xanthan Gum.
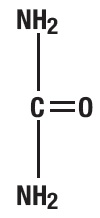
Urea is a diamide of carbonic acid with the following chemical structure:
- Clinical Pharmacology
- Pharmacokinetics
-
Indications and Usage
For debridement and promotion of normal healing of hyperkeratotic surface lesions, particularly where healing is retarded by local infection, necrotic tissue, fibrinous or purulent debris or eschar. Urea is useful for the treatment of hyperkeratotic conditions such as dry, rough skin, dermatitis, psoriasis, xerosis, ichthyosis, eczema, keratosis pilaris, keratosis palmaris, keratoderma, corns and calluses, as well as damaged, ingrown and devitalized nails.
- Contraindications
- Warnings
- Precautions
-
PREGNANCY
PREGNANCY: Pregnancy Category B. Animal reproduction studies have revealed no evidence of harm to the fetus, however, there are no adequate and well-controlled studies in pregnant women. Because animal reproductive studies are not always predictive of human response, Urea 40% should be given to a pregnant woman only if clearly needed.
- NURSING MOTHERS
-
Adverse Reactions
Transient stinging, burning, itching or irritation may occur and normally disappear upon discontinuing the medication.
To report SUSPECTED ADVERSE REACTIONS contact AvKARE, Inc. at 1-855-361-3993; email drugsafety@avkare.com; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- Dosage and Administration
-
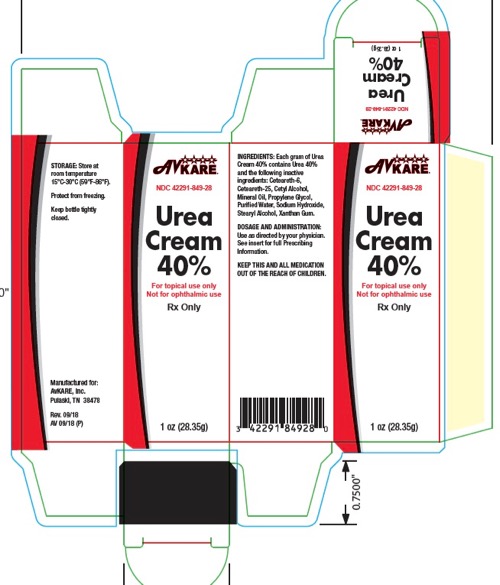
How Supplied
Urea 40% Cream
1 oz. (28.35 g): NDC: 42291-849-28
Urea 40% Cream3 oz. (85 g): NDC: 42291-849-85
Urea 40% Cream7 oz. (198.4 g): NDC: 42291-849-19
Store at room temperature 15°C - 30°C (59°F-86°F).Protect from freezing. Keep bottle tightly closed.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured for:
AvKARE, Inc.
Pulaski, TN 38478
AV Rev. 09/18 (P) Rev. 09/18 - Tube Label
- Carton
-
INGREDIENTS AND APPEARANCE
UREA
urea creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 42291-849 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength UREA (UNII: 8W8T17847W) (UREA - UNII:8W8T17847W) UREA 40 g in 100 g Inactive Ingredients Ingredient Name Strength CETYL ALCOHOL (UNII: 936JST6JCN) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) XANTHAN GUM (UNII: TTV12P4NEE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) PARAFFIN (UNII: I9O0E3H2ZE) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 42291-849-28 1 in 1 CARTON 11/06/2017 1 28 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC: 42291-849-85 1 in 1 CARTON 11/06/2017 2 85 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC: 42291-849-19 1 in 1 CARTON 11/06/2017 3 198 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 11/06/2017 Labeler - AvKARE (796560394)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.