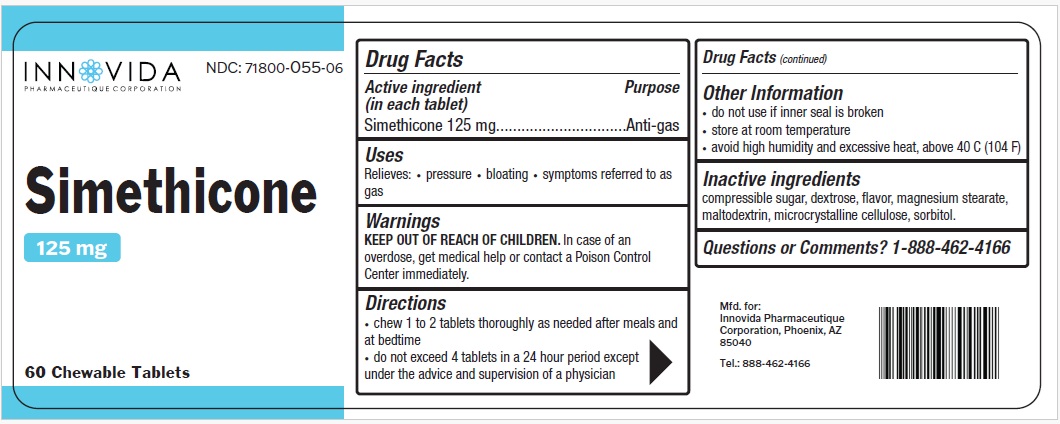
Simethicone by Innovida Pharmaeutique Corporation
Simethicone by
Drug Labeling and Warnings
Simethicone by is a Otc medication manufactured, distributed, or labeled by Innovida Pharmaeutique Corporation. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
SIMETHICONE- dimethicone tablet, chewable
Innovida Pharmaeutique Corporation
----------
Warnings
KEEP OUT OF REACH OF CHILDREN. In case of an overdose, get medical help or contact a Poison Control Center immediately
Directions
- chew 1 to 2 tablets thoroughly as needed after meals and at bedtime
- do not exceed 4 tablets in a 24 hour period except under the advice and supervision of a physician
Other Information
- do not use if inner seal is broken
- store at room temperature
- avoid high humidity and excessive heat, above 40°C (104° F)
| SIMETHICONE
dimethicone tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Innovida Pharmaeutique Corporation (080892908) |
Revised: 12/2025
Document Id: 46e11d8b-7a1f-e5cb-e063-6394a90a35a2
Set id: 6638d4a9-605b-4409-ae4a-522b1da8aee6
Version: 2
Effective Time: 20251226