CAMZYOS- mavacamten capsule, gelatin coated
CAMZYOS by
Drug Labeling and Warnings
CAMZYOS by is a Other medication manufactured, distributed, or labeled by Myokardia, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CAMZYOS safely and effectively. See full prescribing information for CAMZYOS.
CAMZYOS® (mavacamten) capsules for oral use
Initial U.S. Approval: 2022WARNING: RISK OF HEART FAILURE
See full prescribing information for complete boxed warning.
- CAMZYOS can cause heart failure due to systolic dysfunction. (5.1)
- Echocardiogram assessments of left ventricular ejection fraction (LVEF) required before and during CAMZYOS use. (2.1)
- Initiation in patients with LVEF <55% not recommended.
- Interrupt if LVEF <50% or if worsening clinical status. (2.1, 5.1)
- Certain CYP450 inhibitors and inducers are contraindicated in patients taking CAMZYOS because of an increased risk of heart failure. (4, 5.2, 7)
- CAMZYOS is available only through a restricted program called the CAMZYOS REMS Program. (5.3)
RECENT MAJOR CHANGES
Boxed Warning
04/2025
Dosage and Administration, Initiation, Maintenance, and Interruption of Treatment (2.1)
04/2025
Dosage and Administration, Concomitant Administration of Weak to Moderate CYP2C19 or Moderate to Strong CYP3A4 Inhibitors (2.2)
04/2025
Warnings and Precautions, Heart Failure (5.1)
04/2025
Warnings and Precautions, Embryo-Fetal Toxicity (5.4)
04/2025
INDICATIONS AND USAGE
CAMZYOS is a cardiac myosin inhibitor indicated for the treatment of adults with symptomatic New York Heart Association (NYHA) class II-III obstructive hypertrophic cardiomyopathy (HCM) to improve functional capacity and symptoms. (1)
DOSAGE AND ADMINISTRATION
Dosage must be individualized based on clinical status and echocardiographic assessment of patient response. Refer to the Full Prescribing Information for instructions. (2.1)
DOSAGE FORMS AND STRENGTHS
Capsules: 2.5 mg, 5 mg, 10 mg, and 15 mg (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Heart Failure: Consider interruption of CAMZYOS in patients with intercurrent illness. (2.1, 5.1)
- Drug Interactions Leading to Heart Failure or Loss of Effectiveness: Advise patients of the potential for drug interactions including with over-the-counter medications. (4, 5.2, 17)
- Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential to use effective contraception until 4 months after the last dose. Avoid concomitant use with a combined hormonal contraceptive that contains a progestin other than norethindrone. (5.4, 7.2, 8.1, 8.3)
ADVERSE REACTIONS
Adverse reactions occurring in >5% of patients and more commonly on CAMZYOS than on placebo were dizziness (27%) and syncope (6%). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bristol-Myers Squibb at 1-800-721-5072 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Weak to moderate CYP2C19 inhibitors and moderate to strong CYP3A4 inhibitors: May increase risk of heart failure. If initiating an inhibitor, CAMZYOS dose reduction and additional monitoring are required. (2.2, 7.1)
- Negative inotropes: Close medical supervision and LVEF monitoring is recommended if a negative inotrope is initiated, or the dose of a negative inotrope is increased. Avoid certain combinations of negative inotropes. (7.3)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 4/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISK OF HEART FAILURE
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Initiation, Maintenance, and Interruption of Treatment
2.2 Concomitant Administration of Weak to Moderate CYP2C19 or Moderate to Strong CYP3A4 Inhibitors
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Heart Failure
5.2 CYP450 Drug Interactions Leading to Heart Failure or Loss of Effectiveness
5.3 CAMZYOS REMS Program
5.4 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect Plasma Concentrations of CAMZYOS
7.2 Potential for CAMZYOS to Affect Plasma Concentrations of Other Drugs
7.3 Drugs That Reduce Cardiac Contractility
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.5 Pharmacogenomics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISK OF HEART FAILURE
CAMZYOS reduces left ventricular ejection fraction (LVEF) and can cause heart failure due to systolic dysfunction [see Warnings and Precautions (5.1)].
Echocardiogram assessments of LVEF are required prior to and during treatment with CAMZYOS. Initiation of CAMZYOS in patients with LVEF <55% is not recommended. Interrupt CAMZYOS if LVEF is <50% at any visit or if the patient experiences heart failure symptoms or worsening clinical status [see Dosage and Administration (2.1) and Warnings and Precautions (5.1)].
Concomitant use of CAMZYOS with certain cytochrome P450 inhibitors or discontinuation of certain cytochrome P450 inducers may increase the risk of heart failure due to systolic dysfunction; therefore, the use of CAMZYOS is contraindicated with the following [see Contraindications (4) and Warnings and Precautions (5.2)]:
- Strong CYP2C19 inhibitors
- Moderate to strong CYP2C19 inducers or moderate to strong CYP3A4 inducers
Because of the risk of heart failure due to systolic dysfunction, CAMZYOS is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called CAMZYOS REMS PROGRAM [see Warnings and Precautions (5.3)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Initiation, Maintenance, and Interruption of Treatment
Confirm absence of pregnancy and usage of effective contraception in females of reproductive potential [see Warnings and Precautions (5.4)].
Initiation or up-titration of CAMZYOS in patients with LVEF <55% is not recommended.
The recommended starting dose is 5 mg orally once daily without regard to food; allowable subsequent doses with titration are 2.5 mg, 5 mg, 10 mg, or 15 mg orally once daily. The maximum recommended dose is 15 mg orally once daily.
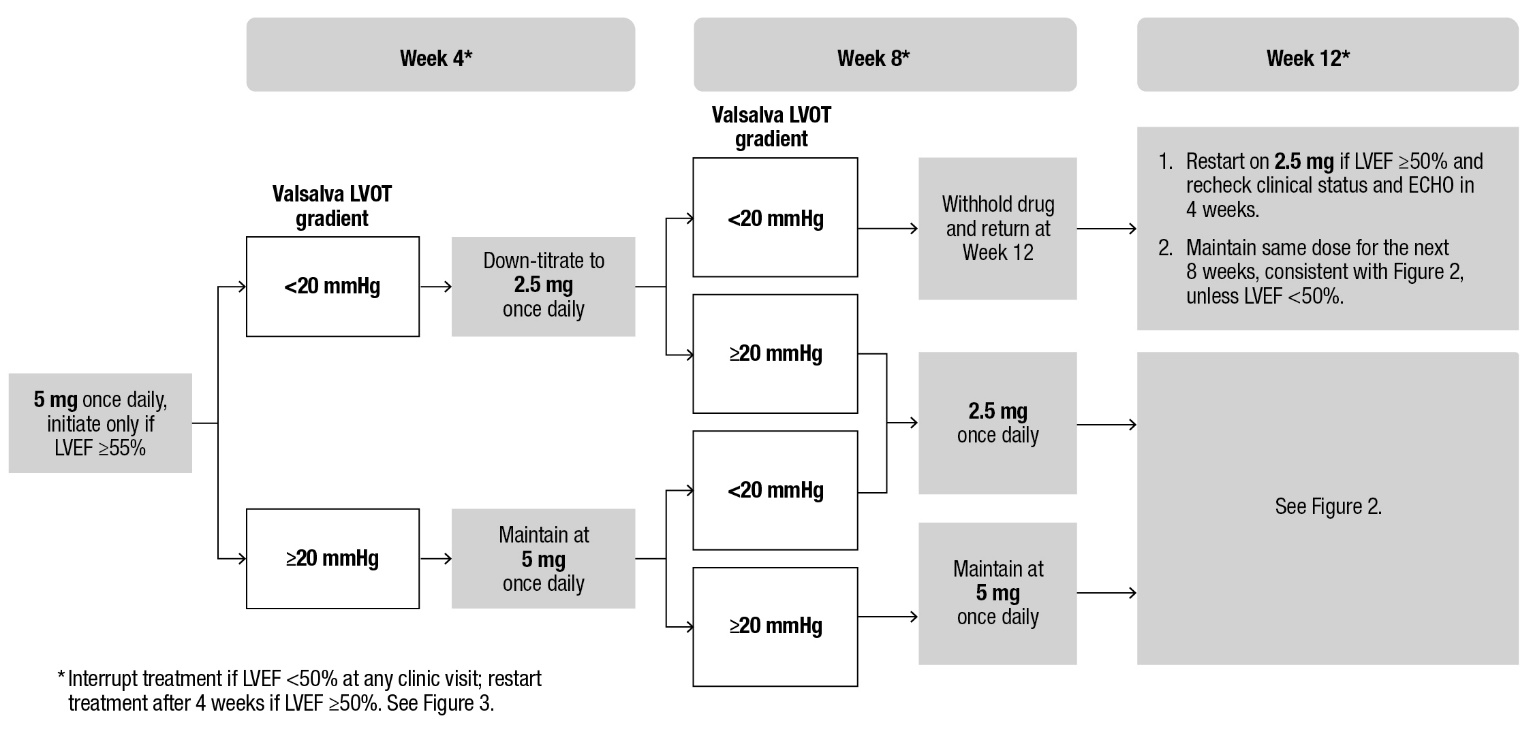
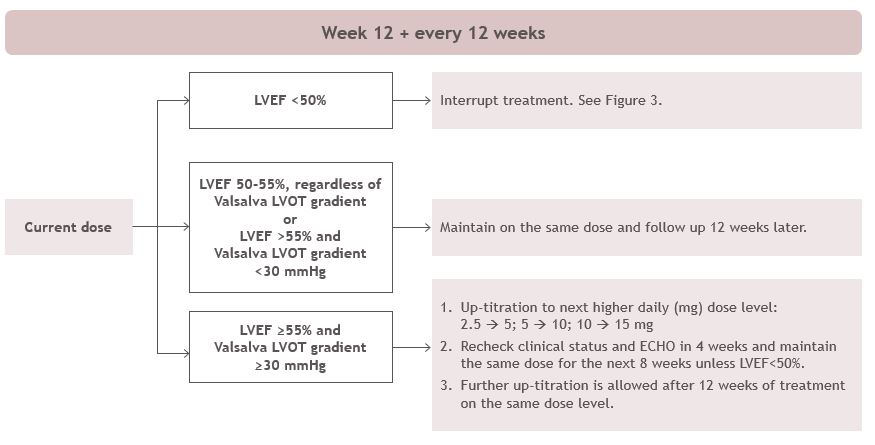
Patients may develop heart failure while taking CAMZYOS. Regular LVEF and Valsalva left ventricular outflow tract (LVOT) gradient assessment is required for careful titration to achieve an appropriate target Valsalva LVOT gradient, while maintaining LVEF ≥50% and avoiding heart failure symptoms (see Figure 1 and Figure 2).
Daily dosing takes weeks to reach steady-state drug levels and therapeutic effects, and genetic variation in metabolism and drug interactions can cause large differences in exposure [see Boxed Warning, Contraindications (4), Warnings and Precautions (5.2), Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
When initiating or titrating CAMZYOS, first consider LVEF then consider the Valsalva LVOT gradient and patient clinical status to guide appropriate CAMZYOS dosing. Assessment of post-exercise LVOT gradient may be considered in symptomatic patients with normal or near normal Valsalva gradients (approximately 30 mmHg) prior to initiating treatment with CAMZYOS. Follow the algorithms for Initiation (Figure 1) and Maintenance (Figure 2) for appropriate CAMZYOS dosing and monitoring schedules.
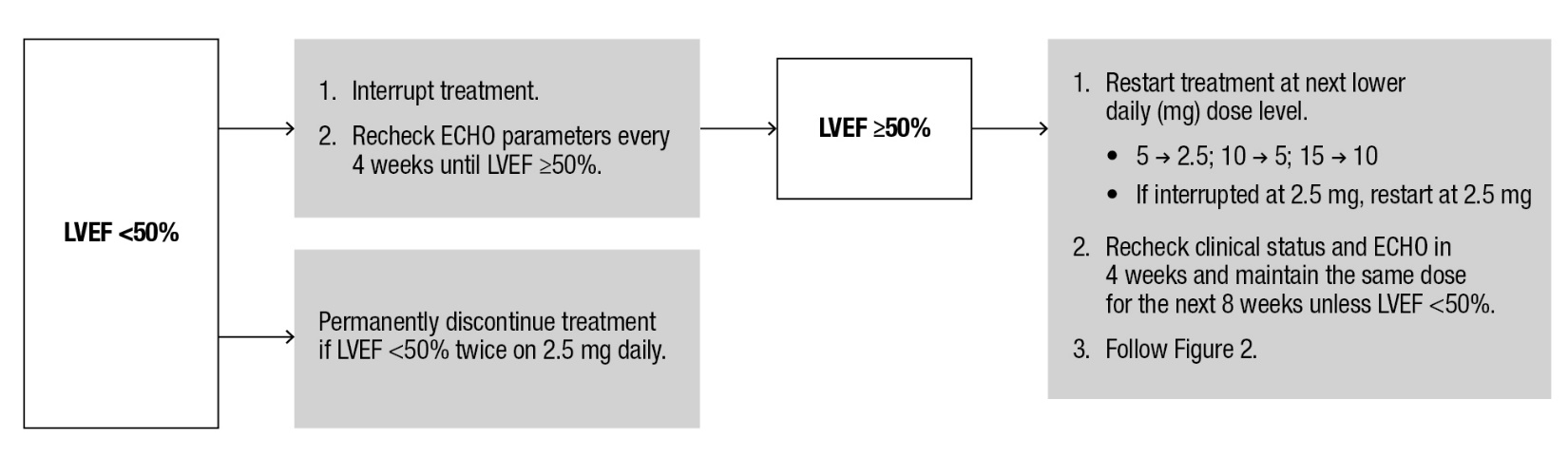
If LVEF <50% while taking CAMZYOS, interrupt treatment. Follow the algorithm for Interruption (Figure 3) for guidance on interrupting, restarting, or discontinuing CAMZYOS. If interrupted at 2.5 mg, either restart at 2.5 mg or discontinue permanently.
Figure 1: Initiation Phase
Figure 2: Maintenance Phase
Figure 3: Treatment Interruption at Any Clinic Visit if LVEF <50%
Delay dose increases when there is intercurrent illness (e.g., serious infection) or arrhythmia (e.g., atrial fibrillation or other uncontrolled tachyarrhythmia) that may impair systolic function. Consider interruption of CAMZYOS in patients with intercurrent illness [see Warnings and Precautions (5.1)].
Missed or delayed doses
If a dose is missed, it should be taken as soon as possible, and the next scheduled dose should be taken at the usual time the following day. Exact timing of dosing during the day is not essential, but two doses should not be taken on the same day.
Swallow capsules whole. Do not break, open, or chew the capsules.
2.2 Concomitant Administration of Weak to Moderate CYP2C19 or Moderate to Strong CYP3A4 Inhibitors
Initiate CAMZYOS at the recommended starting dosage of 5 mg orally once daily in patients who are on stable therapy with a weak CYP2C19 inhibitor or a moderate CYP3A4 inhibitor (see Figure 1).
In patients who are on stable therapy with a moderate CYP2C19 inhibitor or a strong CYP3A4 inhibitor, initiate CAMZYOS at 2.5 mg orally once daily. Interrupt CAMZYOS treatment if Valsalva LVOT gradient is <20 mm Hg at Week 4 or Week 8. Treatment may be resumed after 4 weeks at 2.5 mg once daily if LVEF is ≥50%. If treatment is resumed at Week 12, recheck clinical status, Valsalva LVOT gradient and LVEF in 4 weeks, and maintain the current dose for the next 8 weeks unless LVEF is <50%.
In patients who initiate a weak to moderate CYP2C19 inhibitor or a moderate to strong CYP3A4 inhibitor, reduce dosage of CAMZYOS to the next lower daily (mg) dose level (i.e., 15 mg to 10 mg; 10 mg to 5 mg; or 5 mg to 2.5 mg). Schedule clinical and echocardiographic assessment 4 weeks after inhibitor initiation, and do not up-titrate to the next higher daily (mg) dose level of CAMZYOS until 12 weeks after inhibitor initiation. Avoid initiation of concomitant weak to moderate CYP2C19 and moderate to strong CYP3A4 inhibitors in patients who are on stable treatment with 2.5 mg of CAMZYOS because a lower CAMZYOS once-daily dose is not available [see Dosage and Administration (2.1), Drug Interactions (7.1)].
For short-term use (e.g., 1 week), interrupt CAMZYOS for the duration of treatment with a weak to moderate inhibitor of CYP2C19 or a moderate to strong inhibitor of CYP3A4. Mavacamten may be reinitiated at the previous dose immediately on discontinuation of concomitant therapy.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
CAMZYOS is contraindicated with concomitant use of:
- Strong CYP2C19 inhibitors [see Warnings and Precautions (5.2), Drug Interactions (7.1)]
- Moderate to strong CYP2C19 inducers or moderate to strong CYP3A4 inducers [see Warnings and Precautions (5.2), Drug Interactions (7.1)]
-
5 WARNINGS AND PRECAUTIONS
5.1 Heart Failure
CAMZYOS reduces systolic contraction and can cause heart failure or significantly reduce ventricular function. Patients who experience a serious intercurrent illness (e.g., serious infection) or arrhythmia (e.g., atrial fibrillation or other uncontrolled tachyarrhythmia) are at greater risk of developing systolic dysfunction and heart failure [see Clinical Trial Experience (6.1)].
Assess the patient’s clinical status and LVEF prior to and regularly during treatment and adjust the CAMZYOS dose accordingly [see Dosage and Administration (2.1)]. New or worsening arrhythmia, dyspnea, chest pain, fatigue, palpitations, leg edema, or elevations in N-terminal pro-B-type natriuretic peptide (NT-proBNP) may be signs and symptoms of heart failure and should also prompt an evaluation of cardiac function.
Asymptomatic LVEF reduction, intercurrent illnesses, and arrhythmias require additional dosing considerations [see Dosage and Administration (2.1, 2.2)].
Initiation of CAMZYOS in patients with LVEF <55% is not recommended. Avoid concomitant use of CAMZYOS in patients on disopyramide, ranolazine, verapamil with a beta blocker, or diltiazem with a beta blocker as these medications and combinations increase the risk of left ventricular systolic dysfunction and heart failure symptoms and clinical experience is limited [see Drug Interactions (7)].
5.2 CYP450 Drug Interactions Leading to Heart Failure or Loss of Effectiveness
CAMZYOS is primarily metabolized by CYP2C19 and CYP3A4 enzymes. Concomitant use of CAMZYOS and drugs that interact with these enzymes may lead to life-threatening drug interactions such as heart failure or loss of effectiveness [see Contraindications (4), Warnings and Precautions (5.1), and Drug Interactions (7.1)].
Advise patients of the potential for drug interactions, including with over-the-counter medications (such as omeprazole, esomeprazole, or cimetidine). Advise patients to inform their healthcare provider of all concomitant products prior to and during CAMZYOS treatment [see Drug Interactions (7.1), Patient Counseling Information (17)].
5.3 CAMZYOS REMS Program
CAMZYOS is only available through a restricted program called the CAMZYOS REMS Program because of the risk of heart failure due to systolic dysfunction [see Warnings and Precautions (5.1, 5.2)].
Notable requirements of the CAMZYOS REMS Program include the following:
- Prescribers must be certified by enrolling in the CAMZYOS REMS Program.
- Patients must enroll in the CAMZYOS REMS Program and comply with ongoing monitoring requirements [see Dosage and Administration (2.1)].
- Pharmacies must be certified by enrolling in the CAMZYOS REMS Program and must only dispense to patients who are authorized to receive CAMZYOS.
- Wholesalers and distributors must only distribute to certified pharmacies.
Further information is available at www.CAMZYOSREMS.com or by telephone at 1-833-628-7367.
5.4 Embryo-Fetal Toxicity
CAMZYOS may cause fetal toxicity when administered to a pregnant female, based on findings in animal studies. Confirm absence of pregnancy in females of reproductive potential prior to treatment and advise patients to use effective contraception during treatment with CAMZYOS and for 4 months after the last dose. Combined hormonal contraceptives (CHCs) containing a combination of ethinyl estradiol and norethindrone may be used with mavacamten. However, CAMZYOS may reduce the effectiveness of certain other CHCs. If these CHCs are used, advise patients to add nonhormonal contraception (such as condoms) during concomitant use and for 4 months after the last dose of CAMZYOS [see Drug Interactions (7.2) and Use in Specific Populations (8.1, 8.3)].
-
6 ADVERSE REACTIONS
The following adverse reaction is discussed in other sections of the labeling:
- Heart failure [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of CAMZYOS was evaluated in EXPLORER-HCM, a Phase 3, double-blind, randomized, placebo-controlled trial [see Clinical Studies (14)]. Of the 251 adults with obstructive HCM, 123 patients were treated with CAMZYOS 2.5-15 mg daily and 128 were treated with placebo. CAMZYOS-treated patients had a median duration of exposure of 30 weeks (range: 2-40 weeks).
Syncope (0.8%) was the only adverse drug reaction leading to discontinuation in patients receiving CAMZYOS.
Adverse reactions occurring in >5% of patients and more commonly on CAMZYOS than on placebo were dizziness (27% vs. 18%) and syncope (6% vs. 2%).
The safety of CAMZYOS in patients was further evaluated in VALOR-HCM, a Phase 3, double-blind, randomized, placebo-controlled trial [see Clinical Studies (14)]. Of the 112 adults with symptomatic obstructive HCM, 56 patients were treated with CAMZYOS 2.5-15 mg daily and 55 were treated with placebo. CAMZYOS-treated patients had a median duration of exposure of 17 weeks (range: 3-19 weeks).
There were no new adverse reactions identified in VALOR-HCM.
Effects on Systolic Function
In the EXPLORER-HCM trial, mean (SD) resting LVEF was 74% (6) at baseline in both treatment groups. Consistent with the mechanism of action of CAMZYOS, mean (SD) absolute change from baseline in LVEF was -4% (8) in the CAMZYOS group and 0% (7) in the placebo group over the 30-week treatment period. At Week 38, following an 8-week interruption of trial drug, mean LVEF was similar to baseline for both treatment groups. In the EXPLORER-HCM trial, 7 (6%) patients in the CAMZYOS group and 2 (2%) patients in the placebo group experienced reversible reductions in LVEF to <50% (median 48%: range 35-49%) while on treatment. In 3 of the 7 CAMZYOS patients and 1 of the 2 placebo patients, these reductions were asymptomatic. In all 7 patients treated with CAMZYOS, LVEF recovered following interruption of CAMZYOS [see Warnings and Precautions (5.1)].
-
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect Plasma Concentrations of CAMZYOS
Mavacamten is primarily metabolized by CYP2C19 and to a lesser extent by CYP3A4 and CYP2C9. Inducers and inhibitors of CYP2C19 and moderate to strong inhibitors or inducers of CYP3A4 may affect the exposures of mavacamten [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. (See Table 1)
Table 1: Established and Potentially Significant Pharmacokinetic Drug Interactions with CAMZYOS
Impact of Other Drugs on CAMZYOS
Strong CYP2C19 Inhibitors
Clinical Impact
Concomitant use with a strong CYP2C19 inhibitor increases mavacamten exposure, which may increase the risk of heart failure due to systolic dysfunction [see Contraindications (4), Warnings and Precautions (5.2), Clinical Pharmacology (12.3)].
Prevention or Management
Concomitant use with a strong CYP2C19 inhibitor is contraindicated.
Moderate to Strong CYP2C19 Inducers or Moderate to Strong CYP3A4 Inducers
Clinical Impact
Concomitant use with a moderate to strong CYP2C19 inducer or a moderate to strong CYP3A4 inducer decreases mavacamten exposure, which may reduce CAMZYOS’ efficacy [see Clinical Pharmacology (12.3)]. The risk of heart failure due to systolic dysfunction may increase with discontinuation of these inducers as the levels of induced enzyme normalizes [see Contraindications (4) and Warnings and Precautions (5.2)].
Prevention or Management
Concomitant use of a moderate to strong CYP2C19 inducer or a moderate to strong CYP3A4 inducer is contraindicated.
Weak CYP2C19 Inhibitors or Moderate CYP3A4 Inhibitors
Clinical Impact
Concomitant use with a weak CYP2C19 inhibitor or a moderate CYP3A4 inhibitor increases mavacamten exposure, which may increase the risk of adverse drug reactions [see Warnings and Precautions (5.2)].
Prevention or Management
Initiate CAMZYOS at the recommended starting dosage of 5 mg orally once daily in patients who are on stable therapy with a weak CYP2C19 inhibitor or a moderate CYP3A4 inhibitor.
Reduce dose of CAMZYOS to the next lower daily (mg) dose level (i.e., 15 mg to 10 mg, 10 mg to 5 mg, or 5 mg to 2.5 mg) in patients who are on CAMZYOS treatment and intend to initiate a weak CYP2C19 inhibitor or a moderate CYP3A4 inhibitor. Avoid initiation of concomitant weak CYP2C19 and moderate CYP3A4 inhibitors in patients who are on stable treatment with 2.5 mg of CAMZYOS because a lower dose is not available [see Dosage and Administration (2.2)].
For short-term use (e.g., 1 week), interrupt CAMZYOS for the duration of treatment with a weak inhibitor of CYP2C19 or a moderate inhibitor of CYP3A4. CAMZYOS may be reinitiated at the previous dose immediately on discontinuation of concomitant therapy.
Moderate CYP2C19 Inhibitors or Strong CYP3A4 Inhibitors
Clinical Impact
Concomitant use with a moderate CYP2C19 inhibitor or a strong CYP3A4 inhibitor increases mavacamten exposure, which may increase the risk of adverse drug reactions [see Warnings and Precautions (5.2)]. Discontinuing use of a moderate CYP2C19 inhibitor or strong CYP3A4 inhibitor after long-term concomitant use may decrease mavacamten exposure, which may reduce CAMZYOS’ efficacy [see Clinical Pharmacology (12.3)].
Prevention or Management
Initiate CAMZYOS at a starting dosage of 2.5 mg orally once daily in patients who are on stable therapy with a moderate CYP2C19 inhibitor or a strong CYP3A4 inhibitor.
Reduce dose of CAMZYOS to the next lower daily (mg) dose level (i.e., 15 mg to 10 mg, 10 mg to 5 mg, or 5 mg to 2.5 mg) in patients who are on CAMZYOS and intend to initiate a moderate CYP2C19 inhibitor or a strong CYP3A4 inhibitor. Avoid initiation of concomitant moderate CYP2C19 and strong CYP3A4 inhibitors in patients who are on stable treatment with 2.5 mg of CAMZYOS because a lower dose is not available [see Dosage and Administration (2.2)].
An increase in dose of CAMZYOS may be needed if the moderate inhibitor of CYP2C19 or strong inhibitor of CYP3A4 is discontinued after long-term concomitant use. Monitor for new or worsening symptoms.
For short-term use (e.g., 1 week), interrupt CAMZYOS for the duration of treatment with a moderate inhibitor of CYP2C19 or a strong inhibitor of CYP3A4. CAMZYOS may be reinitiated at the previous dose immediately on discontinuation of concomitant therapy.
7.2 Potential for CAMZYOS to Affect Plasma Concentrations of Other Drugs
Certain CYP3A4, CYP2C9, and CYP2C19 Substrates
Mavacamten is an inducer of CYP3A4, CYP2C9, and CYP2C19. Concomitant use with CYP3A4, CYP2C9, or CYP2C19 substrates may reduce plasma concentration of these drugs [see Clinical Pharmacology (12.3)]. Closely monitor when CAMZYOS is used with concomitant CYP3A4, CYP2C9 or CYP2C19 substrates unless otherwise recommended in the Prescribing Information.
Certain Combined Hormonal Contraceptives
Progestin and ethinyl estradiol are CYP3A4 substrates. Concomitant use of CAMZYOS may decrease exposures of certain progestins [see Clinical Pharmacology (12.3)], which may lead to contraceptive failure. Combined hormonal contraceptives (CHCs) containing a combination of ethinyl estradiol and norethindrone may be used with mavacamten, but if other CHCs are used, advise patients to add nonhormonal contraception (such as condoms) or use an alternative contraceptive method that is not affected by CYP450 enzyme induction (e.g., intrauterine system) during concomitant use and for 4 months after the last dose of CAMZYOS.
7.3 Drugs That Reduce Cardiac Contractility
Expect additive negative inotropic effects of CAMZYOS and other drugs that reduce cardiac contractility. Avoid concomitant use of CAMZYOS in patients on disopyramide, ranolazine, verapamil with a beta blocker, or diltiazem with a beta blocker as these medications and combinations increase the risk of left ventricular systolic dysfunction and heart failure symptoms and clinical experience is limited [see Warnings and Precautions (5.1)].
If concomitant therapy with a negative inotrope is initiated, or if the dose of a negative inotrope is increased, monitor LVEF closely until stable doses and clinical response have been achieved.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on animal data, CAMZYOS may cause fetal harm when administered to a pregnant female. There are no human data on the use of CAMZYOS during pregnancy to evaluate for a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. The underlying maternal condition during pregnancy poses a risk to the mother and fetus (see Clinical Considerations). Advise pregnant females about the potential risk to the fetus with maternal exposure to CAMZYOS during pregnancy.
In animal embryo-fetal development studies, mavacamten-related decreases in mean fetal body weight, reductions in fetal ossification of bones, and increases in post-implantation loss (early and/or late resorptions) were observed in rats and increases in visceral and skeletal malformations were observed in both rabbits and rats at dose exposures similar to that achieved at the maximum recommended human dose (MRHD) (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
There is a pregnancy safety study for CAMZYOS. If CAMZYOS is administered during pregnancy, or if a patient becomes pregnant while receiving CAMZYOS or within 4 months after the last dose of CAMZYOS, healthcare providers should report CAMZYOS exposure by contacting Bristol-Myers Squibb at 1-800-721-5072 or www.bms.com.
Disease-Associated Maternal and Embryo-Fetal Risk
Obstructive HCM in pregnancy has been associated with increased risk for preterm birth.
Animal Data
When mavacamten was administered orally to pregnant rats (0.3 to 1.5 mg/kg/day) during the period of organogenesis, increases in post-implantation loss, decreases in mean fetal body weight, reductions in fetal ossification of bones, and fetal malformations (visceral and skeletal) were observed in the high dose group (1.5 mg/kg/day). Visceral malformations (heart malformation in fetuses, including one total situs inversus) and increased incidences of skeletal malformations (mainly fused sternebrae) were observed at a similar exposure as in humans at the MRHD. Plasma exposure (based on area under the concentration-time curve or AUC) at the no-effect dose for embryo-fetal development in rats is 0.3 times the exposure in humans at the MRHD.
When mavacamten was administered orally to pregnant rabbits (0.6 to 2.0 mg/kg/day) during the period of organogenesis, fetal malformations (visceral and skeletal) were increased at doses of 1.2 mg/kg/day and higher, with similar plasma exposure at 1.2 mg/kg/day as in humans at the MRHD. Visceral findings consisted of malformations of the great vessels (dilatation of pulmonary trunk and/or aortic arch). Skeletal malformations consisted of higher incidences of fused sternebrae at ≥1.2 mg/kg/day. Plasma exposure (AUC) at the no-effect dose for embryo-fetal development in rabbits is 0.4 times the exposure in humans at the MRHD.
In a pre/postnatal development study, mavacamten was administered orally to pregnant rats (0.3, to 1.5 mg/kg/day) from gestation Day 6 to lactation/post-partum Day 20. No adverse effects were observed in the dams or offspring exposed daily from before birth (in utero) through lactation. The no-observed-adverse-effect level (NOAEL) was 1.5 mg/kg/day (the highest dosage level tested), with similar exposure (AUC) as in humans at the MRHD.
8.2 Lactation
Risk Summary
The presence of mavacamten in human or animal milk, the drug’s effects on the breastfed infant, and the effects on milk production are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CAMZYOS and any potential adverse effects on the breastfed child from CAMZYOS or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Based on animal data, CAMZYOS may cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.4) and Use in Specific Populations (8.1)].
Pregnancy Testing
Confirm absence of pregnancy in females of reproductive potential prior to initiation of CAMZYOS.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with CAMZYOS and for 4 months after the last dose. CHCs containing a combination of ethinyl estradiol and norethindrone may be used with CAMZYOS. However, CAMZYOS may reduce the effectiveness of certain other CHCs. If these CHCs are used, advise patients to add nonhormonal contraception (such as condoms) or use an alternative contraceptive method during concomitant use and for 4 months after the last dose of CAMZYOS [see Drug Interactions (7.2)].
8.4 Pediatric Use
The safety and effectiveness of CAMZYOS have not been established in pediatric patients.
8.5 Geriatric Use
Clinical trials included 319 patients dosed with CAMZYOS, 119 of whom were 65 years of age or older (37.3%), and 25 of whom (7.8%) were age 75 years or older. Safety, effectiveness, and pharmacokinetics were similar between patients ≥65 years and younger patients.
8.6 Hepatic Impairment
No dosage adjustment is required in patients with mild (Child-Pugh A) to moderate (Child-Pugh B) hepatic impairment. Mavacamten exposure (AUC) increased up to 220% in patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment compared to patients with normal hepatic function. However, no additional dose adjustment is required in patients with mild to moderate hepatic impairment with the recommended dose titration algorithm and monitoring plan. The effect of severe (Child-Pugh C) hepatic impairment is unknown [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Clinical Experience and Effects
- Cardiovascular effects may include reduced LVEF (left ventricular ejection fraction), heart failure, hypotension, and asystole refractory to medical intervention.
- Neurological effects may include dizziness and syncope.
- An infant death was reported after accidental ingestion of three 15 mg capsules (45 mg).
- An adult administered a single dose of 144 mg developed a vasovagal reaction, hypotension, and asystole, but the subject recovered.
Management
- Discontinue CAMZYOS treatment.
- Provide medically supportive measures to maintain hemodynamic stability and monitor left ventricular function.
- Consider administering activated charcoal (pediatric dose is 1 g/kg; adult dose is 50 g) within 2 hours of ingestion in addition to other supportive measures. The benefit of activated charcoal is negligible after 6 hours [see Clinical Pharmacology (12.3)].
- Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
-
11 DESCRIPTION
CAMZYOS capsules for oral use contain mavacamten, a cardiac myosin inhibitor.
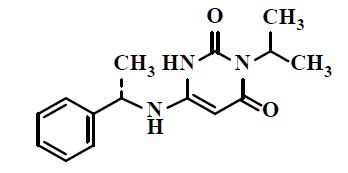
The chemical name of mavacamten is 3-(1-methylethyl)-6-[[(1S)-1-phenylethyl]amino]-2,4(1H,3H)-pyrimidinedione. The molecular formula is C15H19N3O2, and the molecular weight is 273.33 g/mol.
The structural formula of mavacamten is:
Mavacamten is a white to off-white powder that is practically insoluble in water and aqueous buffers at pH 2-10, sparingly soluble in methanol and ethanol, and freely soluble in DMSO and NMP.
CAMZYOS is supplied as immediate release Size 2 hard gelatin capsules, containing 2.5 mg, 5 mg, 10 mg, or 15 mg of mavacamten per capsule as active ingredient and the following inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate (non-bovine), mannitol, and silicon dioxide. The capsule shell contains black edible ink, black iron oxide, gelatin, red iron oxide, titanium dioxide, and yellow iron oxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Mavacamten is an allosteric and reversible inhibitor selective for cardiac myosin. Mavacamten modulates the number of myosin heads that can enter “on actin” (power-generating) states, thus reducing the probability of force-producing (systolic) and residual (diastolic) cross-bridge formation. Excess myosin actin cross-bridge formation and dysregulation of the super-relaxed state are mechanistic hallmarks of HCM. Mavacamten shifts the overall myosin population towards an energy-sparing, recruitable, super-relaxed state. In HCM patients, myosin inhibition with mavacamten reduces dynamic LVOT obstruction and improves cardiac filling pressures.
12.2 Pharmacodynamics
Left Ventricular Ejection Fraction and Left Ventricular Outflow Tract Obstruction
In the EXPLORER-HCM trial, patients achieved reductions in mean resting and provoked (Valsalva) LVOT gradient by Week 4 which were sustained throughout the 30-week trial. At Week 30, the mean (SD) changes from baseline in resting and Valsalva LVOT gradients were -39 (29) mmHg and -49 (34) mmHg, respectively, for the CAMZYOS group and -6 (28) mmHg and -12 (31) mmHg, respectively, for the placebo group. The reductions in Valsalva LVOT gradient were accompanied by decreases in LVEF, generally within the normal range. Eight weeks after discontinuation of CAMZYOS, mean LVEF and Valsalva LVOT gradients were similar to baseline.
Cardiac Structure
In EXPLORER-HCM, echocardiographic measurements of cardiac structure showed a mean (SD) reduction from baseline at Week 30 in left ventricular mass index (LVMI) in the mavacamten group (-7.4 [17.8] g/m2) versus an increase in LVMI in the placebo group (8.9 [15.3] g/m2). There was also a mean (SD) reduction from baseline in left atrial volume index (LAVI) in the mavacamten group (-7.5 [7.8] mL/m2) versus no change in the placebo group (-0.1 [8.7] mL/m2). The clinical significance of these findings is unknown.
Cardiac Biomarkers
In the EXPLORER-HCM trial [see Clinical Studies (14)], reductions in a biomarker of cardiac wall stress, NT-proBNP, were observed by Week 4 and sustained through the end of treatment. At Week 30 compared with baseline, the reduction in NT-proBNP after mavacamten treatment was 80% greater than for placebo (proportion of geometric mean ratio between the two groups, 0.20 [95% CI: 0.17, 0.24]).
In the VALOR-HCM trial [see Clinical Studies (14)], a reduction in NT-proBNP was observed by Week 8 and sustained throughout treatment. At Week 16 compared with baseline, the reduction in NT-proBNP after mavacamten treatment was 67% greater than for placebo (proportion of geometric mean ratio between the two groups, 0.33 [95% CI: 0.27, 0.42]). At Week 16 compared with baseline, a reduction in cardiac troponin I after mavacamten treatment was 47% greater than for placebo (proportion of geometric mean ratio between the two groups, 0.53 [95% CI: 0.41, 0.70]).
The clinical significance of the NT-proBNP and troponin findings is unknown.
Cardiac Electrophysiology
In healthy volunteers receiving multiple doses of CAMZYOS, a concentration-dependent increase in the QTc interval was observed at doses up to 25 mg once daily. No acute QTc changes have been observed at similar exposures during single-dose studies. The mechanism of the QT prolongation effect is not known.
A meta-analysis across clinical studies in HCM patients does not suggest clinically relevant increases in the QTc interval in the therapeutic exposure range. In HCM, the QT interval may be intrinsically prolonged due to the underlying disease, in association with ventricular pacing, or in association with drugs with potential for QT prolongation commonly used in the HCM population. The effect of coadministration of CAMZYOS with other QT-prolonging drugs or in patients with potassium channel variants resulting in a long QT interval have not been characterized.
12.3 Pharmacokinetics
Mavacamten exposure increases dose proportionally following multiple once-daily doses of 1 mg to 15 mg. At the same dose level of CAMZYOS, 170% higher exposures of mavacamten are observed in patients with HCM compared to healthy subjects. Mavacamten accumulation is approximately 100% for Cmax and approximately 600% for AUC in CYP2C19 normal metabolizers (NMs). The accumulation is dependent upon the CYP2C19 metabolism status with the largest accumulation occurring in CYP2C19 poor metabolizers (PMs). At steady-state, the peak-to-trough mavacamten plasma concentration ratio with once daily dosing is approximately 1.5.
Absorption
Mavacamten has an estimated oral bioavailability of at least 85% and median time to maximum concentration (Tmax) of 1 to 2 hours.
Effect of Food
No clinically significant differences in mavacamten AUC were observed following its administration with a high fat meal.
Distribution
Plasma protein binding of mavacamten is between 97 and 98%.
Elimination
Mavacamten has a variable terminal t1/2 that depends on CYP2C19 metabolic status. Mavacamten terminal half‑life is 6 to 9 days in CYP2C19 normal metabolizers (NMs), which is prolonged in CYP2C19 poor metabolizers (PMs) to 23 days.
Metabolism
Mavacamten is extensively metabolized, primarily through CYP2C19 (74%), CYP3A4 (18%), and CYP2C9 (8%).
Excretion
Following a single 25-mg dose of radiolabeled mavacamten, 7% of the dose was recovered in feces (1% unchanged) and 85% in urine (3% unchanged).
Specific Populations
No clinically significant differences in the pharmacokinetics of mavacamten were observed based on age (range: 18-82 years), sex, race, ethnicity, or mild (eGFR: 60 to 89 mL/min/1.73 m2) to moderate (eGFR: 30 to 59 mL/min/1.73 m2) renal impairment. The effects of severe (eGFR: 15 to 30 mL/min/1.73 m2) renal impairment and kidney failure (eGFR: <15 mL/min/1.73 m2; including patients on dialysis) are unknown.
Hepatic Impairment
Mavacamten exposures (AUC) increased up to 220% in patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment. The effect of severe (Child-Pugh C) hepatic impairment is unknown.
Drug Interactions
Clinical Studies and Model-Informed Approaches
Weak CYP2C19 Inhibitors: Concomitant use of mavacamten (15 mg) with omeprazole (20 mg) once daily increased mavacamten AUCinf by 48% with no effect on Cmax in healthy CYP2C19 NMs and rapid metabolizers (RMs; e.g., *1/*17).
Moderate CYP3A4 Inhibitors: Concomitant use of mavacamten (25 mg) with verapamil sustained release (240 mg) increased mavacamten AUCinf by 16% and Cmax by 52% in intermediate metabolizers (IMs; e.g., *1/*2, *1/*3, *2/*17, *3/*17) and NMs of CYP2C19. Concomitant use of mavacamten with diltiazem in CYP2C19 PMs is predicted to increase mavacamten AUC0-24h and Cmax up to 55% and 42%, respectively.
Strong CYP3A4 Inhibitors: Concomitant use of mavacamten (15 mg) with ketoconazole 400 mg once daily in CYP2C19 poor metabolizers is predicted to increase mavacamten AUC0-24 and Cmax up to 130% and 90%, respectively.
Strong CYP2C19 and CYP3A4 Inducers: Concomitant use of mavacamten (a single 15-mg dose) with a strong CYP2C19 and CYP3A4 inducer (rifampin 600-mg daily dose) is predicted to decrease mavacamten AUC0-inf and Cmax by 87% and 22%, respectively, in CYP2C19 NMs, and by 69% and 4%, respectively, in CYP2C19 PMs.
CYP3A4 Substrates: Concomitant use of a 16‑day course of mavacamten (25 mg on Days 1 and 2, followed by 15 mg for 14 days) decreased AUCinf and Cmax of midazolam by 13% and 7%, respectively, in healthy subjects. Following coadministration of mavacamten once daily in HCM patients at the upper end of the therapeutic range, midazolam AUCinf and Cmax are predicted to decrease up to 45% and 24%, respectively.
Certain combined oral contraceptives: No clinically significant differences in ethinyl estradiol and norethindrone exposure were observed in healthy female subjects with CYP2C19 NM phenotype following concomitant use of a combined oral contraceptive containing ethinyl estradiol and norethindrone with a 17-day course of mavacamten (25 mg on days 1 and 2, followed by 15 mg for 15 days). The impact of mavacamten on oral contraceptives containing other progestins is unknown.
CYP2C8 Substrates: Concomitant use of mavacamten once daily in HCM patients is predicted to decrease AUC and Cmax of repaglinide, a CYP2C8 and CYP3A substrate, by up to 27% and 19%, respectively, depending on the dose of mavacamten and CYP2C19 phenotype.
CYP2C9 Substrates: Concomitant use of mavacamten once daily in HCM patients is predicted to decrease AUC and Cmax of tolbutamide, a CYP2C9 substrate, by up to 54% and 23%, respectively, depending on the dose of mavacamten and CYP2C19 phenotype.
CYP2C19 Substrates: Concomitant use of mavacamten once daily in HCM patients is predicted to decrease AUC and Cmax of omeprazole, a CYP2C19 substrate, by up to 48% and 17%, respectively, depending on the dose of mavacamten and CYP2C19 phenotype.
Activated Charcoal: Mavacamten AUC0-72h and AUC0-infinity was reduced by 14% and 34%, respectively, following administration of 50 g activated charcoal with sorbitol 2 hours after ingestion of a single mavacamten 15 mg dose. Administration of activated charcoal 6 hours after the mavacamten dose had minimal effect on mavacamten exposure.
In Vitro Studies
CYP Enzymes: Mavacamten does not inhibit CYP1A2, CYP2B6, CYP2C8, CYP2D6, CYP2C9, CYP2C19, or CYP3A4. Mavacamten is a CYP2B6 inducer.
Transporter Systems: Mavacamten does not inhibit P-gp, BCRP, BSEP, MATE1, MATE2-K, organic anion transporting polypeptides (OATPs), organic cation transporters (OCTs), or organic anion transporters (OATs).
12.5 Pharmacogenomics
Mavacamten AUCinf increased by 241% and Cmax increased by 47% in CYP2C19 poor metabolizers (PMs) compared to normal metabolizers (NMs) following a single dose of 15 mg mavacamten. Mean half-life is prolonged in CYP2C19 PMs compared to NMs (23 days vs. 6 to 9 days, respectively).
Polymorphic CYP2C19 is the main enzyme involved in the metabolism of CAMZYOS. An individual carrying two normal function alleles is a NM (e.g., *1/*1). An individual carrying two no function alleles is a PM (e.g., *2/*2, *2/*3, *3/*3).
The prevalence of CYP2C19 poor metabolizers differs depending on ancestry. Approximately 2% of individuals of European ancestry and 4% of individuals of African ancestry are PMs; the prevalence of PMs is higher in Asian populations (e.g., approximately 13% of East Asians).
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
Mavacamten was not genotoxic in a bacterial reverse mutation test (Ames test), a human in vitro lymphocyte clastogenicity assay, or a rat in vivo micronucleus assay.
There was no evidence of carcinogenicity seen in a 6-month rasH2 transgenic mouse study at mavacamten doses of up to 2.0 mg/kg/day in males and 3.0 mg/kg/day in females, or in a 2-year rat study at mavacamten doses up to 0.6 mg/kg/day. The exposure-based (AUC) safety margins in mice and rats were up to 3X or 0.2X, respectively, compared to AUC exposures in humans at the MRHD.
In reproductive toxicity studies, there was no evidence of effects of mavacamten on mating and fertility in male or female rats at doses up to 1.2 mg/kg/day, or on the viability and fertility of offspring of dams dosed up to 1.5 mg/kg/day. Plasma exposure (AUC) of mavacamten at the highest dose tested was the same as in humans at the MRHD.
-
14 CLINICAL STUDIES
EXPLORER-HCM
The efficacy of CAMZYOS was evaluated in EXPLORER-HCM (NCT-03470545) a Phase 3, double-blind, randomized, placebo-controlled, multicenter, international, parallel-group trial in 251 adults with symptomatic NYHA class II and III obstructive HCM, LVEF ≥55%, and LVOT peak gradient ≥50 mmHg at rest or with provocation.
Patients on dual therapy with beta blocker and calcium channel blocker treatment or monotherapy with disopyramide or ranolazine were excluded. Patients with a known infiltrative or storage disorder causing cardiac hypertrophy that mimicked obstructive HCM, such as Fabry disease, amyloidosis, or Noonan syndrome with left ventricular hypertrophy, were also excluded.
Patients were randomized in a 1:1 ratio to receive either a starting dose of 5 mg of CAMZYOS or placebo once daily for 30 weeks. Treatment assignment was stratified by baseline NYHA functional class, baseline use of beta blockers, and type of ergometer (treadmill or exercise bicycle).
Groups were well matched with respect to age (mean 59 years), BMI (mean 30 kg/m2), heart rate (mean 62 bpm), blood pressure (mean 128/76 mmHg), and race (90% Caucasian). Males comprised 54% of the CAMZYOS group and 65% of the placebo group.
At baseline, approximately 73% of the randomized patients were NYHA class II and 27% were NYHA class III. The mean LVEF was 74%, and the mean Valsalva LVOT gradient was 73 mmHg. About 10% had prior septal reduction therapy, 75% were on beta blockers, 17% were on calcium channel blockers, and 14% had a history of atrial fibrillation.
All patients were initiated on CAMZYOS 5 mg (or matching placebo) once daily, and the dose was periodically adjusted to optimize patient response (decrease in LVOT gradient with Valsalva maneuver) and maintain LVEF ≥50%. The dose was also informed by plasma concentrations of CAMZYOS.
In the CAMZYOS group, at the end of treatment, 49% of patients were receiving the 5-mg dose, 33% were receiving the 10-mg dose, and 11% were receiving the 15-mg dose. Three patients temporarily interrupted their dose due to LVEF <50%, of whom two resumed treatment at the same dose and one had the dose reduced from 10 mg to 5 mg.
Primary endpoint
The primary composite functional endpoint, assessed at 30 weeks, was defined as the proportion of patients who achieved either improvement of peak oxygen consumption (pVO2) by ≥1.5 mL/kg/min plus improvement in NYHA class by at least 1 or improvement of pVO2 by ≥3.0 mL/kg/min plus no worsening in NYHA class.
A greater proportion of patients met the primary endpoint at Week 30 in the CAMZYOS group compared to the placebo group (37% vs. 17%, respectively, p=0.0005; see Table 2).
Table 2: Primary Endpoint at 30 Weeks CAMZYOS
n (%)
N = 123
Placebo
n (%)
N = 128
Difference
(95% CI)
p-value
Total responders
45 (37%)
22 (17%)
19% (9, 30)
0.0005
Change from baseline
pVO2 ≥1.5 mL/kg/min and
decreased NYHA
41 (33%)
18 (14%)
19% (9, 30)
Change from baseline
pVO2 ≥3 mL/kg/min and
NYHA not increased
29 (23%)
14 (11%)
13% (3, 22)
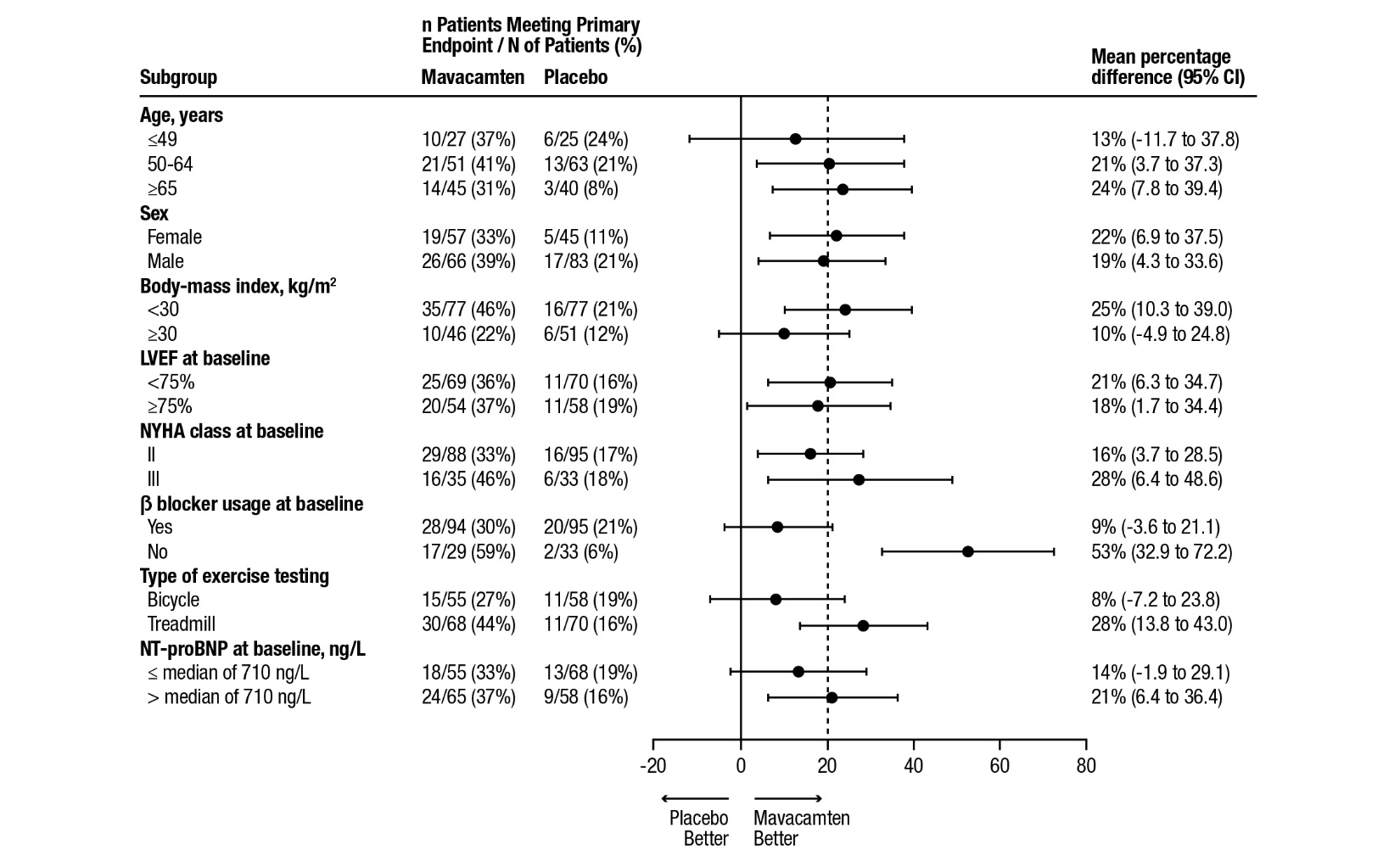
A range of demographic characteristics, baseline disease characteristics, and baseline concomitant medications were examined for their influence on outcomes. Results of the primary analysis consistently favored CAMZYOS across all subgroups analyzed (Figure 4).
Figure 4: Subgroup Analysis of the Primary Composite Functional Endpoint
The dashed vertical line represents the overall treatment effect and the solid vertical line (no effect) indicates no difference between treatment groups.
Note: The figure above presents effects in various subgroups, all of which are baseline characteristics. The 95% confidence limits that are shown do not take into account the number of comparisons made and may not reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.Although the benefit of mavacamten was smaller in patients on background beta blocker therapy compared to those who were not (attenuated improvement in pVO2), analyses of other secondary endpoints (symptoms, LVOT gradient) suggest that patients might benefit from mavacamten treatment regardless of beta blocker use.
Secondary endpoints
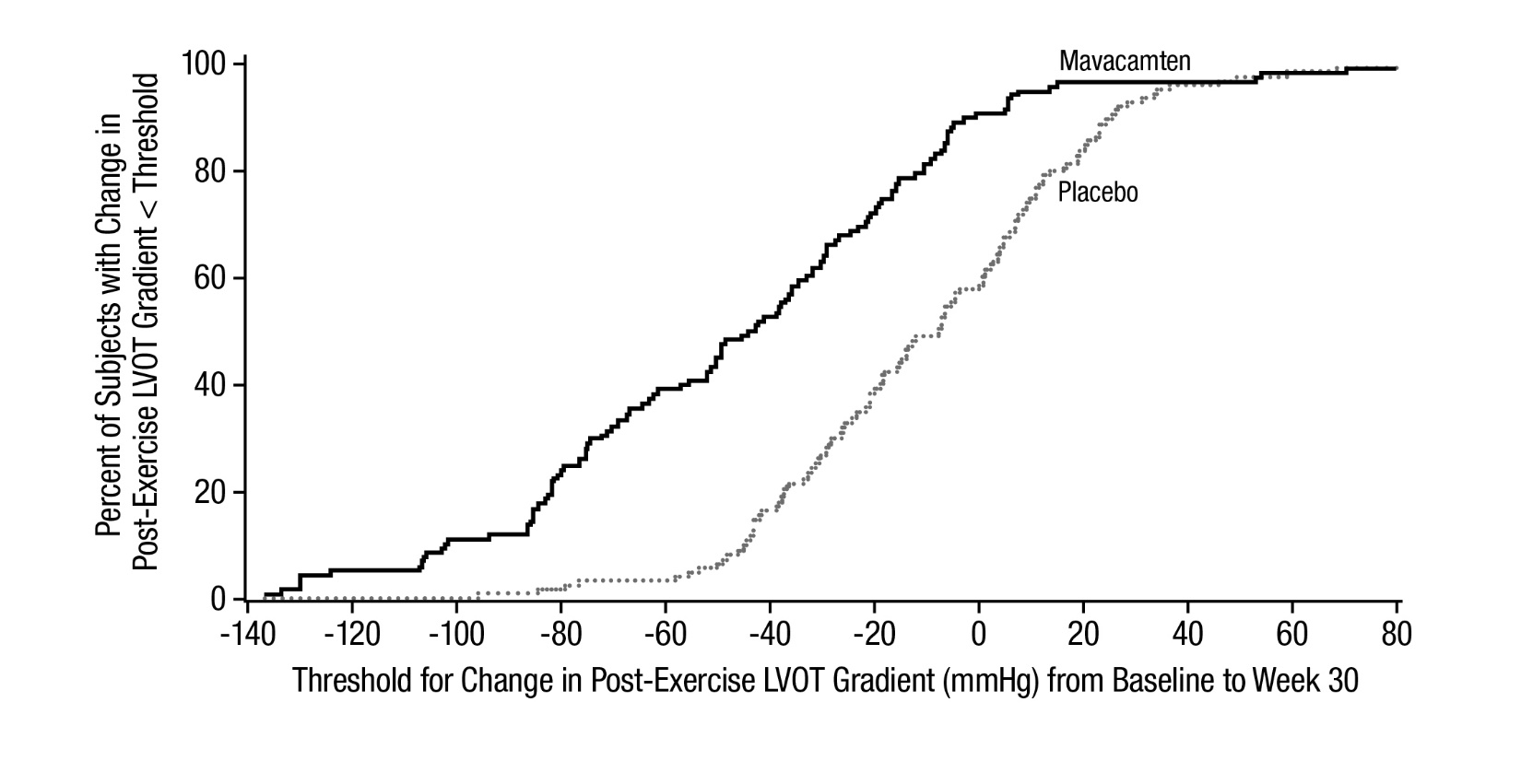
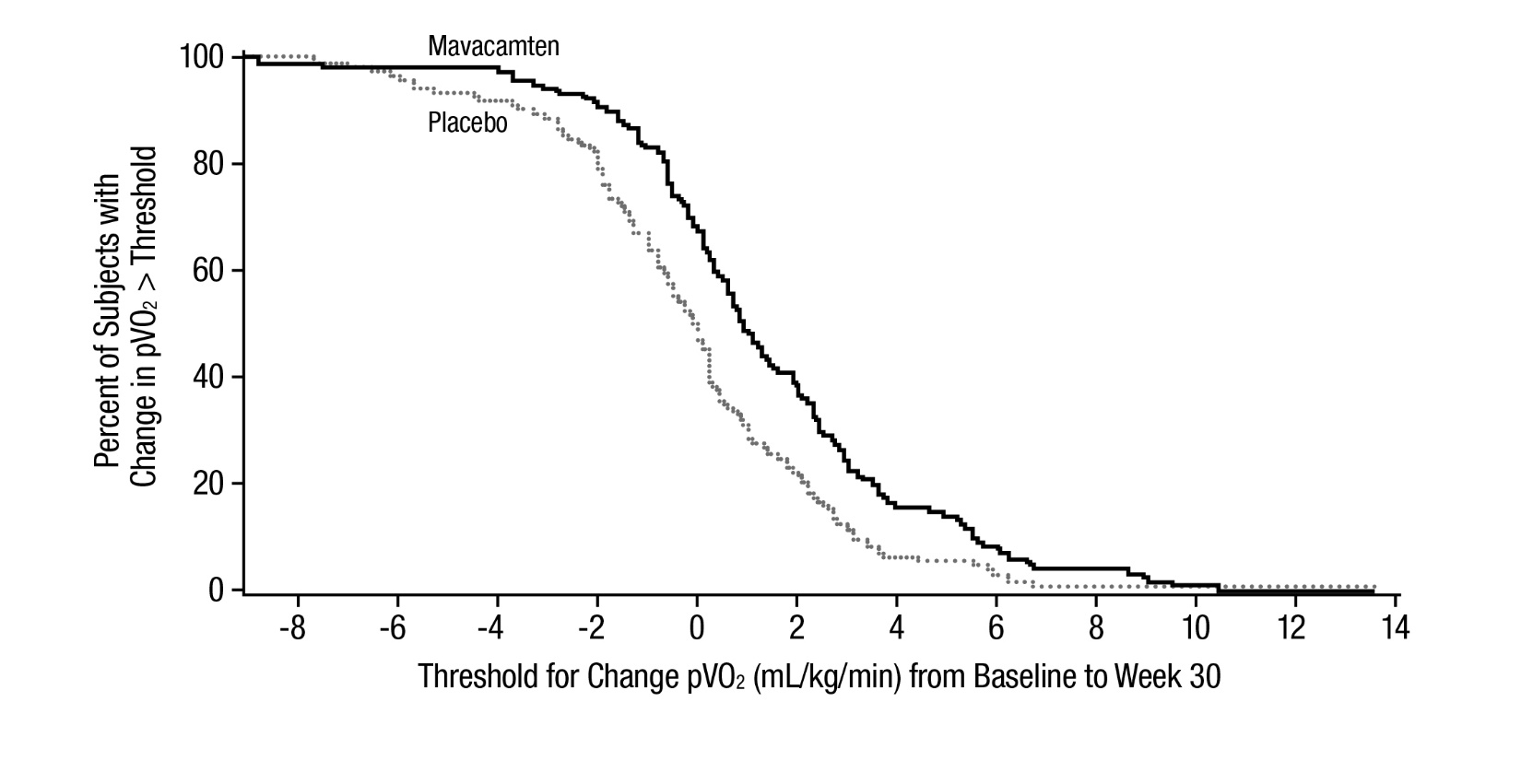
The treatment effects of CAMZYOS on LVOT obstruction, functional capacity, and health status were assessed by change from baseline through Week 30 in post-exercise LVOT peak gradient, change in pVO2, proportion of patients with improvement in NYHA class, Kansas City Cardiomyopathy Questionnaire-23 (KCCQ-23) Clinical Summary Score (CSS), and Hypertrophic Cardiomyopathy Symptom Questionnaire (HCMSQ) Shortness of Breath (SoB) domain score. At Week 30, patients receiving CAMZYOS had greater improvement compared to the placebo group across all secondary endpoints (Table 3, Figure 5, Figure 6, Table 4, and Figures 7-10).
Table 3: Change from Baseline to Week 30 in Post-Exercise LVOT Gradient, pVO2, and NYHA Class CAMZYOS
N=123
Placebo
N=128
Difference (95% CI)
p-value
Post-Exercise LVOT
gradient (mmHg), mean
(SD)
-47 (40)
-10 (30)
-35 (-43, -28)
<0.0001
pVO2 (mL/kg/min), mean
(SD)
1.4 (3.1)
-0.1 (3.0)
1.4 (0.6, 2.1)
<0.0006
Number (%) with NYHA
Class improved ≥1
80 (65%)
40 (31%)
34% (22%, 45%)
<0.0001
Figure 5: Cumulative Distribution of Change from Baseline to Week 30 in LVOT Peak Gradient
Figure 6: Cumulative Distribution of Change from Baseline to Week 30 in pVO2
Table 4: Change from Baseline to Week 30 in KCCQ-23 CSS and HCMSQ SoB Domain †The KCCQ‑23 CSS is derived from the Total Symptom Score (TSS) and the Physical Limitations (PL) score of the KCCQ‑23. The CSS ranges from 0 to 100 with higher scores representing less severe symptoms and/or physical limitations.
‡The HCMSQ SoB domain score measures the frequency and severity of shortness of breath. The HCMSQ SoB domain score ranges from 0 to 18 with lower scores representing less shortness of breath.
Missing data were not imputed to summarize the baseline and change from baseline to Week 30 values. Difference in mean change from baseline between treatment groups was estimated using a mixed model for repeated measures.Baseline, Mean (SD)
Change from Baseline to Week 30, Mean
(SD)
Difference,
LS Mean
(95%CI) and
p-value
CAMZYOS
Placebo
CAMZYOS
Placebo
KCCQ‑23 CSS†
n=99
71 (16)
n=97
71 (19)
14 (14)
4 (14)
9 (5, 13)
p<0.0001
KCCQ-23 TSS
71 (17)
69 (22)
12 (15)
5 (16)
KCCQ-23 PL
70 (18)
72 (19)
15 (17)
4 (15)
HCMSQ SoB‡
n=108
5 (3)
n=109
5 (3)
-3 (3)
-1 (2)
-2 (-2, -1)
p<0.0001
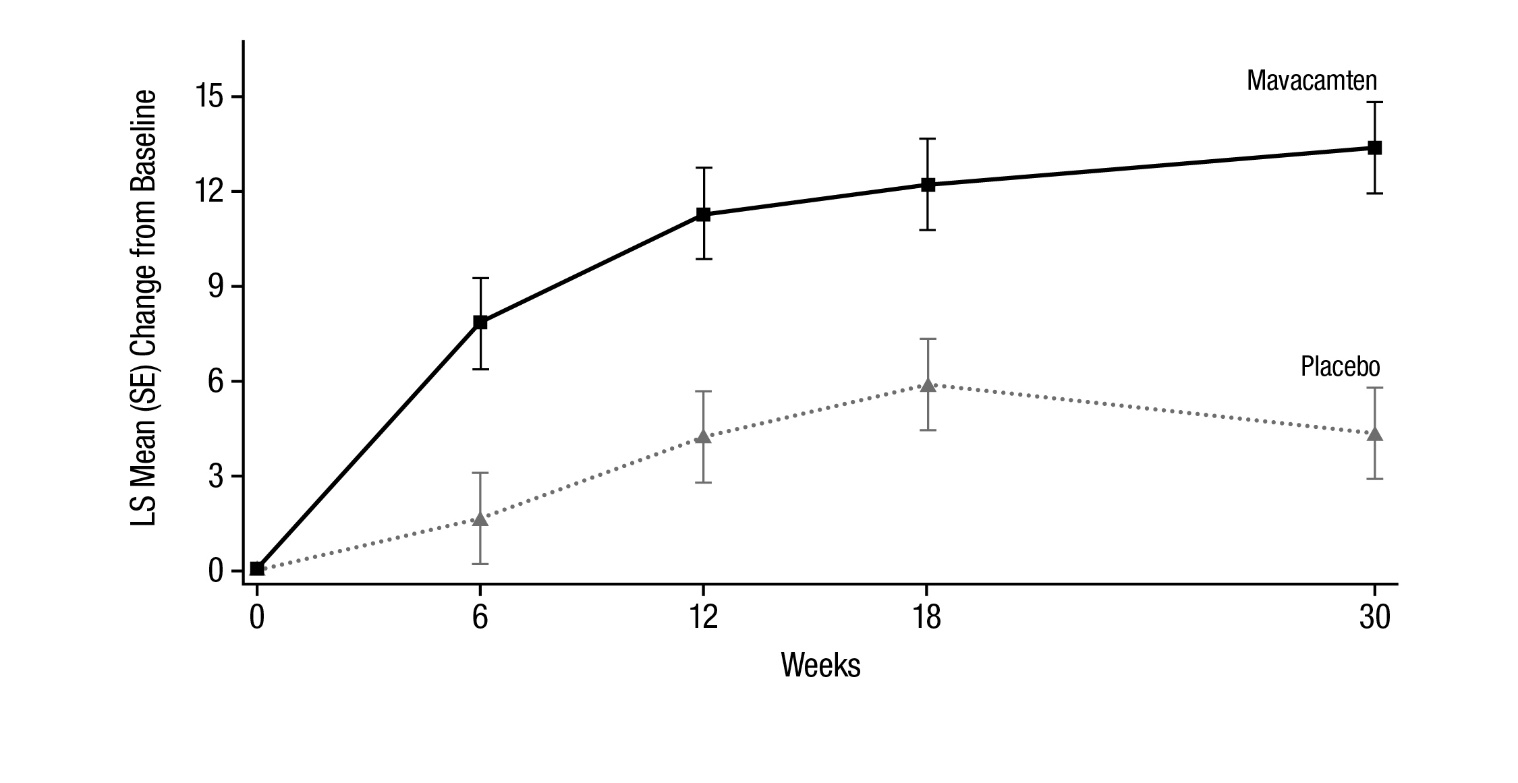
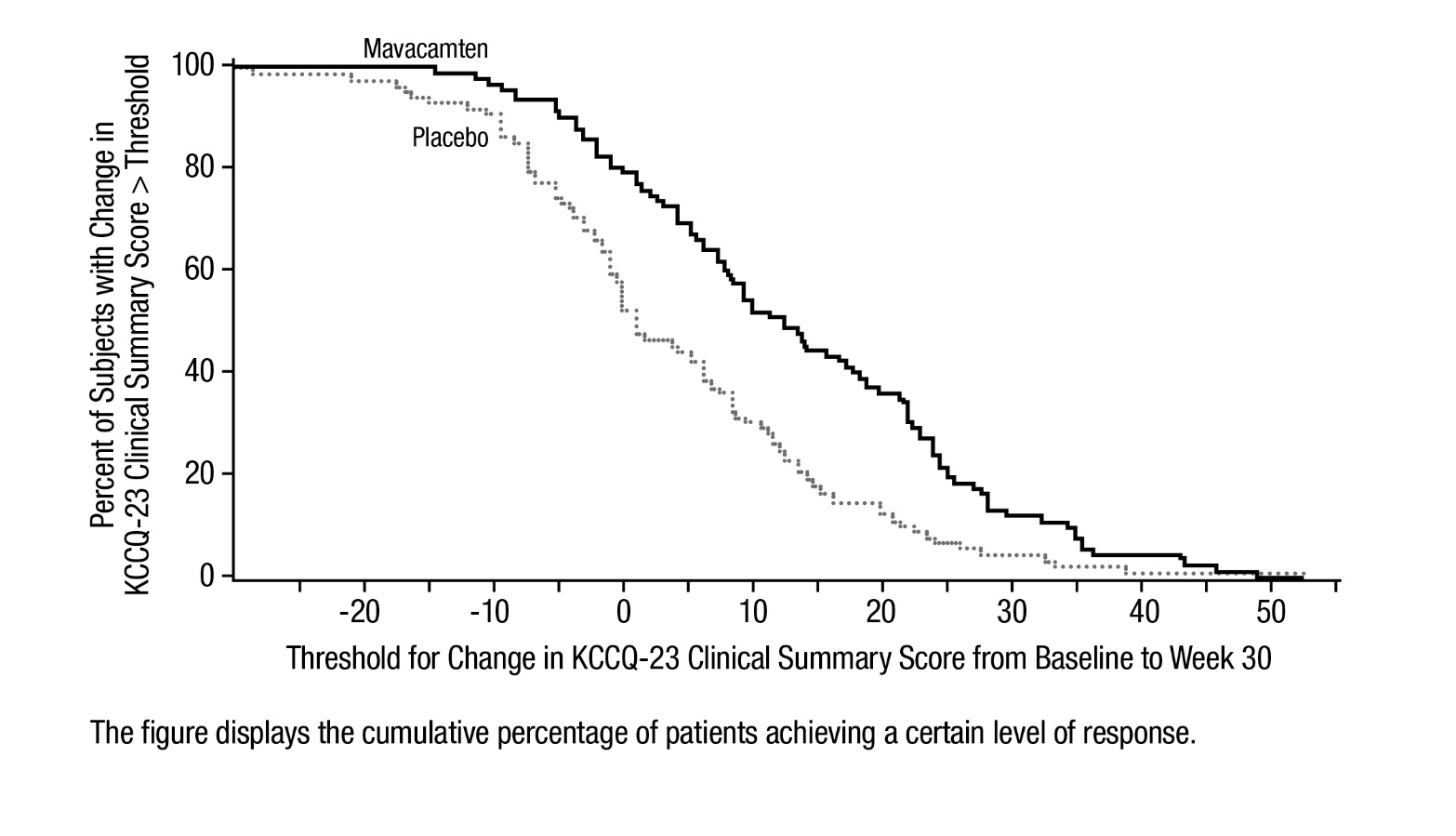
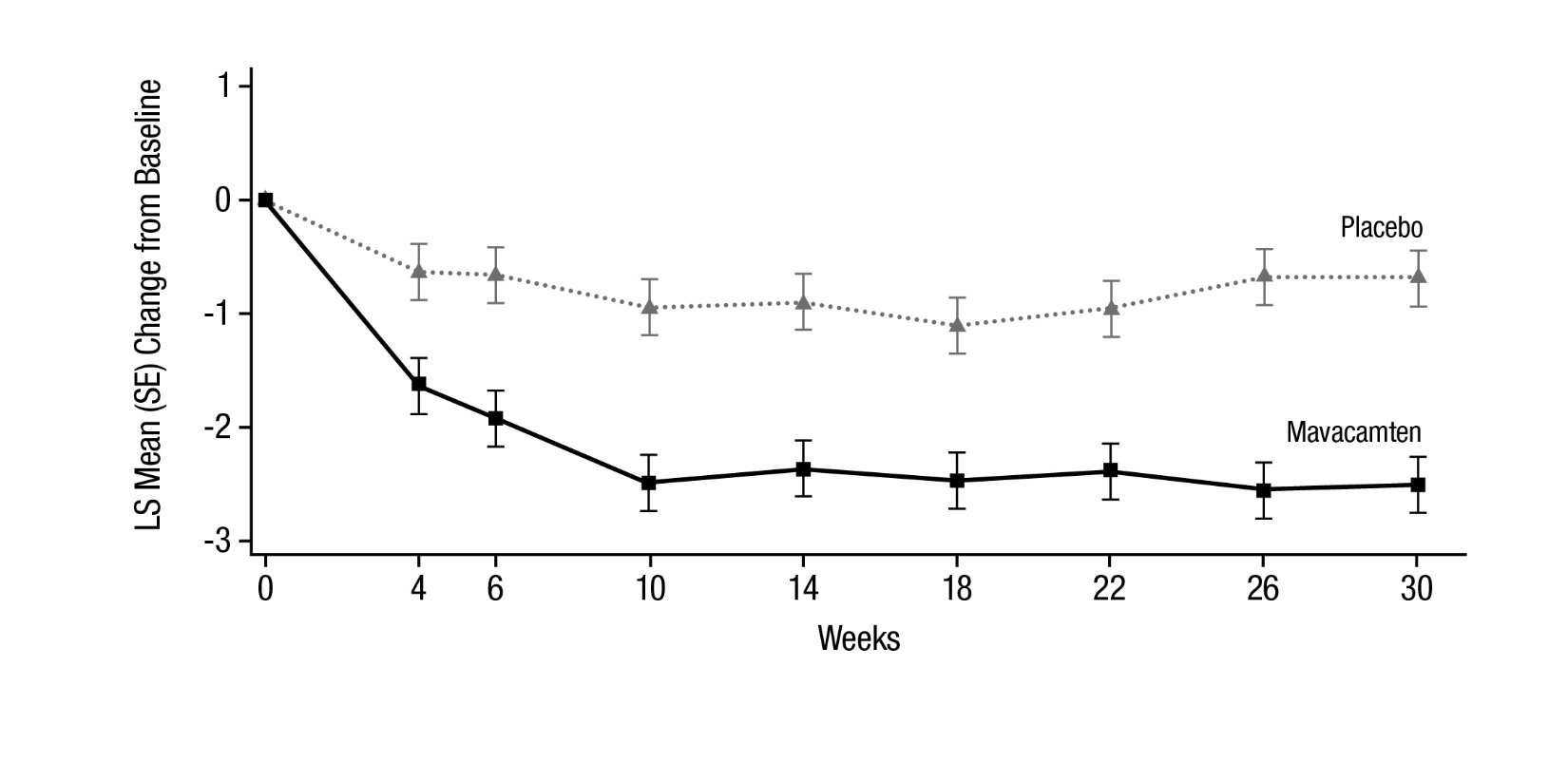
Figure 7 shows the time course for changes in KCCQ-23 CSS. Figure 8 shows the distribution of changes from baseline to Week 30 for KCCQ-23 CSS.
Figure 7: KCCQ-23 Clinical Summary Score: Mean Change from Baseline Over Time
Figure 8: KCCQ-23 Clinical Summary Score: Cumulative Distribution of Change from Baseline to Week 30
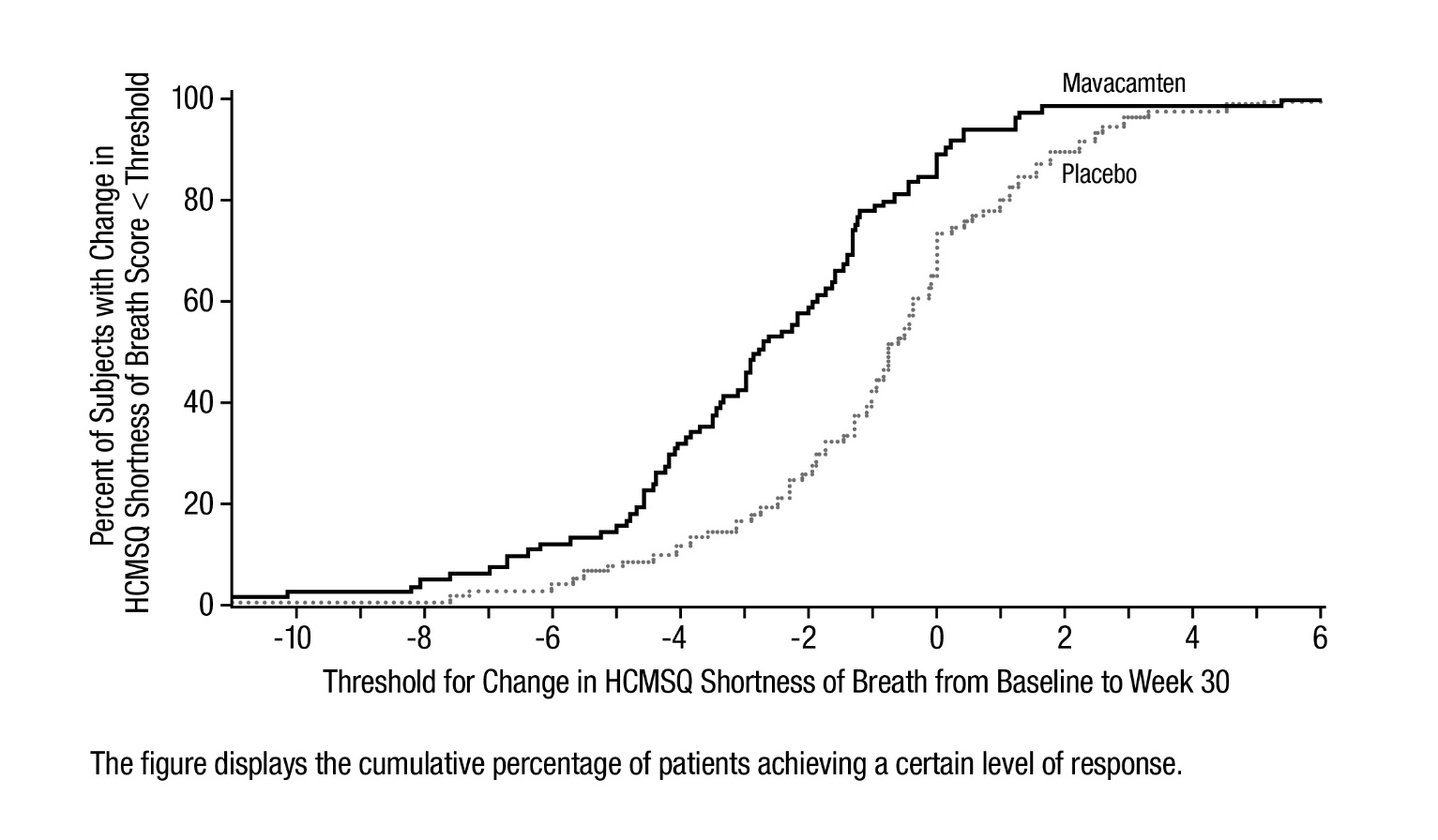
Figure 9 shows the time course for changes in HCMSQ SoB. Figure 10 shows the distribution of changes from baseline to Week 30 for HCMSQ SoB.
Figure 9: HCMSQ Shortness of Breath Domain: Mean Change from Baseline Over Time
Figure 10: HCMSQ Shortness of Breath Domain: Cumulative Distribution of Change from Baseline to Week 30
VALOR-HCM
The efficacy of CAMZYOS was evaluated in VALOR-HCM, a Phase 3, double-blind, randomized, 16-week, placebo-controlled trial in 112 patients (mean age of 60 years; 51% men; 93% ≥NYHA class III) randomized 1:1 to receive treatment with CAMZYOS or placebo. At baseline, all patients had symptomatic obstructive HCM and were SRT eligible.
Patients with severely symptomatic drug-refractory obstructive HCM (including 33% on any combination of beta-blocker, calcium channel blocker and/or disopyramide; 20% were on disopyramide alone or in combination with other treatment), and NYHA class III/IV or class II with exertional syncope or near syncope, were included in the study. Patients were required to have LVOT peak gradient ≥50 mmHg at rest or with provocation, and LVEF ≥60%. Patients must have been referred or under active consideration within the past 12 months for SRT and actively considering scheduling the procedure.
Patients received CAMZYOS (2.5 mg, 5 mg, 10 mg, or 15 mg) or a placebo capsule once daily for 16 weeks. Dose adjustment was based on clinical echocardiogram parameters.
Primary endpoint
CAMZYOS was shown to be superior to placebo in reducing the proportion of patients who met the primary endpoint (the composite of patient decision to proceed with SRT prior to or at Week 16 or met SRT eligibility (LVOT gradient of ≥50 mmHg and NYHA class III-IV, or class II with exertional syncope or near syncope) at Week 16 (18% vs. 77%, respectively, p<0.0001; see Table 5).
Table 5: Primary Endpoint at 16 Weeks *NYHA Class III or IV, or Class II with exertion induced syncope or near syncope and dynamic LVOT gradient at rest or with provocation (i.e., Valsalva or exercise) ≥50 mmHg. CAMZYOS
n (%)
n=56
Placebo
n (%)
n=56
Treatment difference
(95% CI)
p-value
Primary efficacy composite endpoint
10 (18)
43 (77)
59%
(44%, 74%)<0.0001
Patient decision to proceed with SRT
2 (3.6)
2 (3.6)
SRT-eligible based on guideline
criteria*8 (14)
39 (70)
SRT status not evaluable (imputed as
meeting guideline criteria)0
2 (3.6)
Secondary endpoints
The treatment effects of CAMZYOS on LVOT obstruction, functional capacity, and health status were assessed by change from baseline through Week 16 in post-exercise LVOT gradient, proportion of patients with improvement in NYHA class, and KCCQ-23 CSS.
Table 6: Change from Baseline to Week 16 in Secondary Endpoints †The KCCQ-23 CSS is derived from the Total Symptom Score (TSS) and the Physical Limitations (PL) score of the KCCQ-23. The CSS ranges from 0 to 100 with higher scores representing less severe symptoms and/or physical limitations. CAMZYOS
n=56
Placebo
n=56
Difference
(95% CI)
p-value
Post-Exercise LVOT gradient (mmHg), mean (SD)
-39
(37)
-2
(29)
-38
(-49, -28)
<0.0001
Number (%) with NYHA Class improved ≥1
35
(63%)
12
(21%)
41%
(25%, 58%)
<0.0001
KCCQ-23 CSS†, mean (SD)
10
(16)
2
(12)
9
(5, 14)
<0.0001
- KCCQ-23 TSS, mean (SD)
10
(16)
2
(14)
10
(5, 15)
- KCCQ-23 PL, mean (SD)
10
(19)
2
(17)
10
(5, 16)
Figure 11 shows the time course for changes in KCCQ-23 CSS. Figure 12 shows the distribution of changes from baseline to Week 16 for KCCQ-23 CSS.
Figure 11: KCCQ-23 Clinical Summary Score: Mean Change from Baseline Over Time
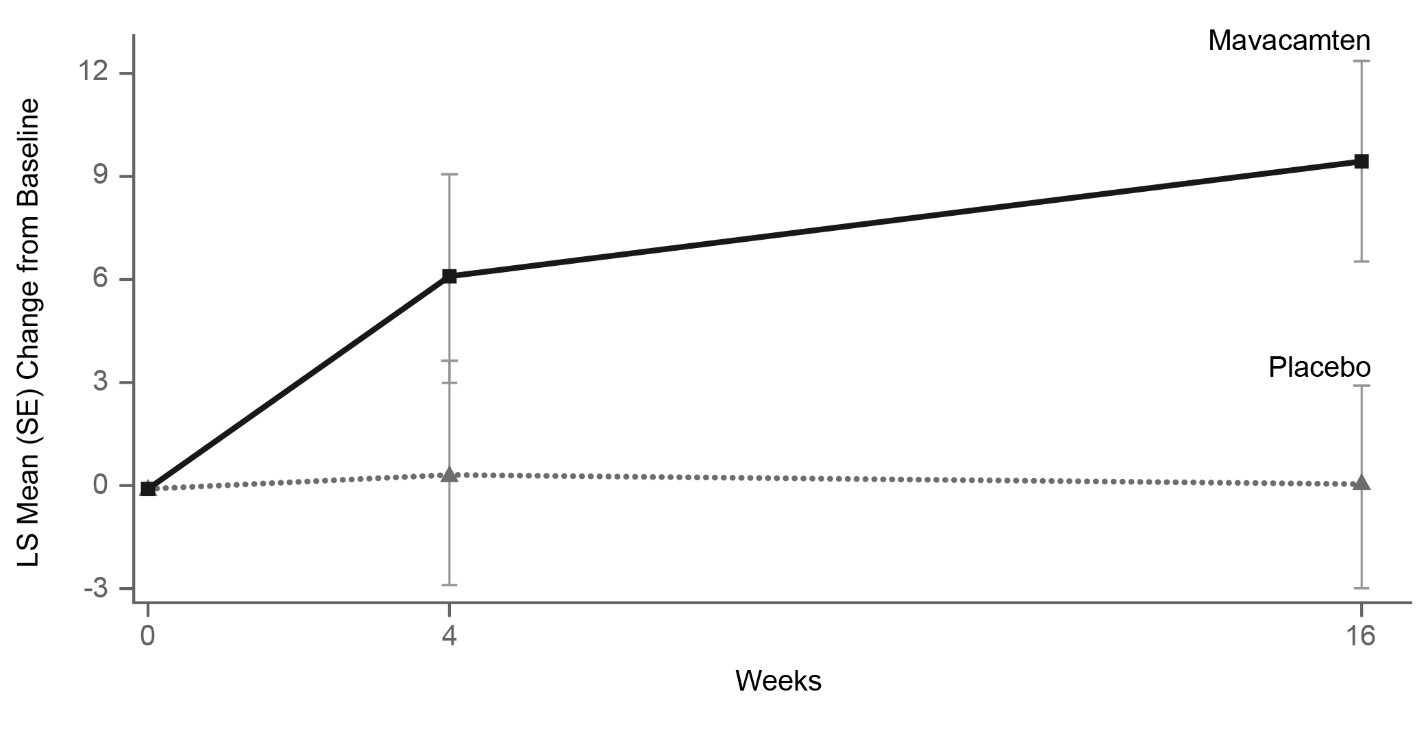
Figure 12: KCCQ-23 Clinical Summary Score: Cumulative Distribution of Change from Baseline to Week 16
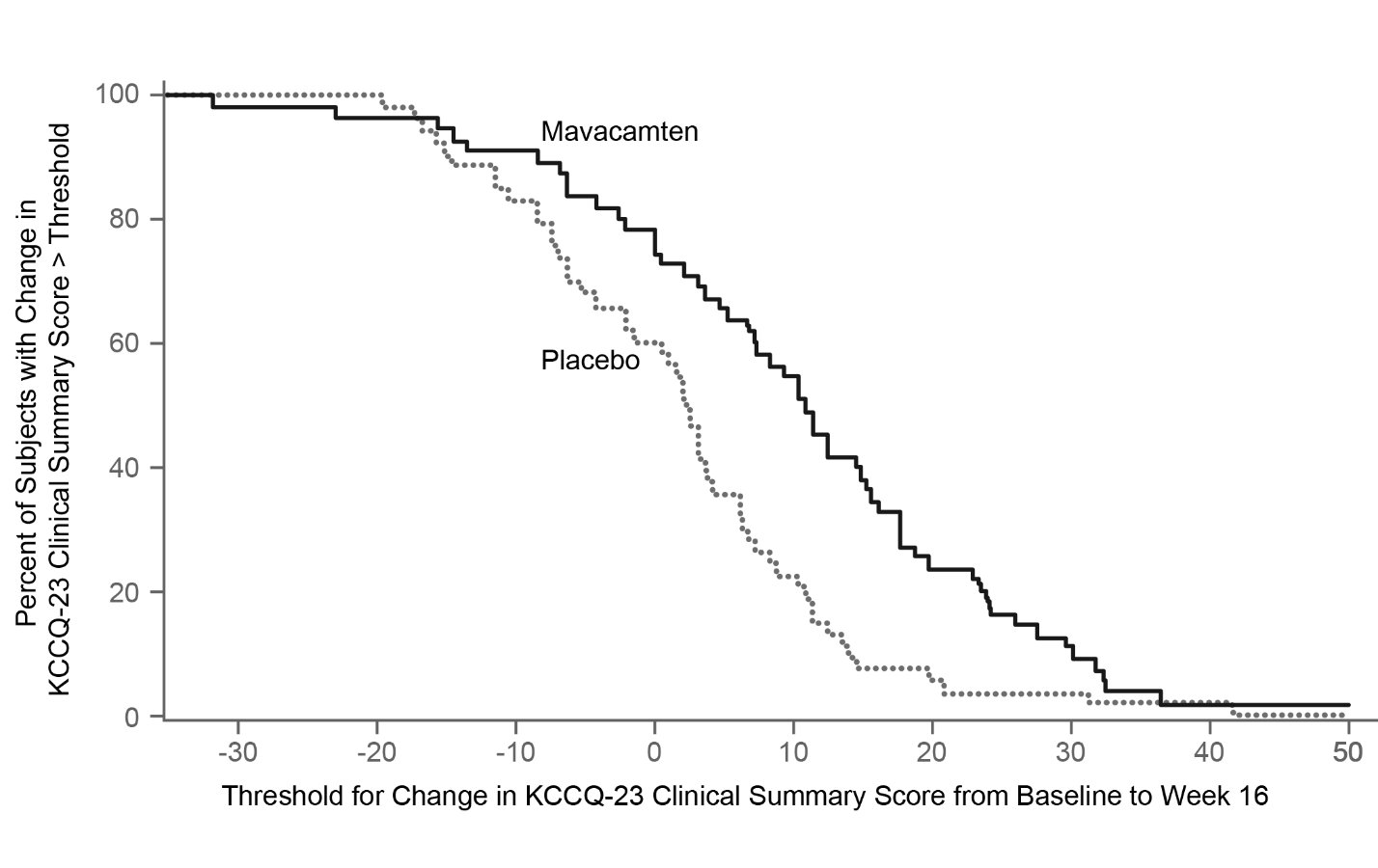
The figure displays the cumulative percentage of patients achieving a certain level of response. -
16 HOW SUPPLIED/STORAGE AND HANDLING
CAMZYOS® is supplied as immediate release Size 2 hard gelatin capsules containing 2.5 mg, 5 mg, 10 mg, or 15 mg of mavacamten. White opaque capsule bodies are imprinted with “Mava”, and the opaque cap is imprinted with the strength. The capsule contains white to off-white powder. CAMZYOS capsules are available in bottles of 30 capsules, as listed in the table below:
Strength
Capsule Cap
NDC Number
2.5 mg
Light purple
73625-111-11
5 mg
Yellow
73625-112-11
10 mg
Pink
73625-113-11
15 mg
Gray
73625-114-11
Storage
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F) [see USP Controlled Room Temperature].
-
17 PATIENT COUNSELING INFORMATION
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide).
Heart Failure
Inform patients that cardiac function monitoring must be performed using echocardiography to monitor for heart failure [see Warnings and Precautions (5.1)]. Advise patients to report any signs or symptoms of heart failure immediately to their healthcare provider.
Drug Interactions
Advise patients to inform their healthcare providers of all concomitant products, including over-the-counter medications (such as omeprazole, esomeprazole, or cimetidine) and supplements, prior to and during CAMZYOS treatment.
CAMZYOS REMS Program
CAMZYOS is available only through a restricted program called the CAMZYOS REMS Program [see Warnings and Precautions (5.3)]. Inform the patient of the following notable requirements:
- Patients must enroll in the program and comply with ongoing monitoring requirements [see Warnings and Precautions 5.1 and 5.3].
CAMZYOS is only prescribed by certified healthcare providers and only dispensed from certified pharmacies participating in the program. Provide patients with the telephone number and website for information on how to obtain the product [see Warnings and Precautions (5.3)].
Embryo-Fetal Toxicity
Advise pregnant females and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.4) and Use in Specific Populations (8.1, 8.3)].
Advise females of reproductive potential to use effective contraception during treatment with CAMZYOS and for 4 months after the last dose.
CHCs containing a combination of ethinyl estradiol and norethindrone may be used with mavacamten. However, CAMZYOS may reduce the effectiveness of certain other combined hormonal contraceptives (CHC). If these CHCs are used, advise patients to add nonhormonal contraception (such as condoms) during concomitant use and for 4 months after the last dose of CAMZYOS [see Drug Interactions (7.2) and Use in Specific Populations (8.3)].
Advise females who are exposed to CAMZYOS during pregnancy that there is a pregnancy safety study that monitors pregnancy outcomes. Encourage these patients to report their pregnancies to Bristol-Myers Squibb at 1-800-721-5072 or www.bms.com.
Instructions for Taking CAMZYOS
CAMZYOS capsules should be swallowed whole. Advise patients that if they miss a dose of CAMZYOS, to take the dose as soon as possible that day and the next scheduled dose should be taken at the usual time the following day. The patient should not take two doses in the same day.
Marketed by:
Bristol-Myers Squibb Company
Princeton, NJ 08543 USACAMZYOS® is a trademark of MyoKardia, Inc., a Bristol Myers Squibb company.
-
Medication Guide
MEDICATION GUIDE
CAMZYOS® (kam-zai-ōs)
(mavacamten)
capsules, for oral use
What is the most important information I should know about CAMZYOS?
CAMZYOS may cause serious side effects, including:
- Heart failure, a condition where the heart cannot pump with enough force. Heart failure is a serious condition that can lead to death. You must have echocardiograms before you take your first dose and during your treatment with CAMZYOS to help your healthcare provider understand how your heart is responding to CAMZYOS. People who develop a serious infection or irregular heartbeat have a greater risk of heart failure during treatment with CAMZYOS.
- Tell your healthcare provider or get medical help right away if you develop new or worsening:
o shortness of breath
o swelling in your legs
o chest pain
o a racing sensation in your heart (palpitations)
o fatigue
o rapid weight gain
- The risk of heart failure is also increased when CAMZYOS is taken with certain other medicines. Tell your healthcare provider about the medicines you take, both prescribed and obtained over-the-counter, before and during treatment with CAMZYOS.
-
Because of the serious risk of heart failure, CAMZYOS is only available through a restricted program called the CAMZYOS Risk Evaluation and Mitigation Strategy (REMS) Program.
- o Your healthcare provider must be enrolled in the CAMZYOS REMS Program in order for you to be prescribed CAMZYOS.
- o Before you take CAMZYOS, you must enroll in the CAMZYOS REMS Program. Talk to your healthcare provider about how to enroll in the CAMZYOS REMS Program. You will be given information about the program when you enroll.
- o Before you take CAMZYOS, your healthcare provider and pharmacist will make sure you understand how to take CAMZYOS safely, which will include returning for echocardiograms when advised by your healthcare provider.
- CAMZYOS can only be dispensed by a certified pharmacy that participates in the CAMZYOS REMS Program. Your healthcare provider can give you information on how to find a certified pharmacy. You will not be able to get CAMZYOS at a local pharmacy.
-
- o If you have questions about the CAMZYOS REMS Program, ask your healthcare provider, go to www.CAMZYOSREMS.com, or call 1-833-628-7367.
See “What are the possible side effects of CAMZYOS?” for information about side effects.
What is CAMZYOS?
CAMZYOS is a prescription medicine used to treat adults with symptomatic obstructive hypertrophic cardiomyopathy (HCM). CAMZYOS may improve your symptoms and your ability to be active. It is not known if CAMZYOS is safe and effective in children.
Before taking CAMZYOS, tell your healthcare provider about all of your medical conditions, including if you:
- are pregnant or plan to become pregnant. CAMZYOS may harm your unborn baby. Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with CAMZYOS. You may also report your pregnancy by calling Bristol-Myers Squibb at 1-800-721-5072 or www.bms.com.
- If you are a female and able to become pregnant:
- o Your healthcare provider will do a pregnancy test before you start treatment with CAMZYOS.
- o You should use effective birth control (contraception) during treatment with CAMZYOS and for 4 months after your last dose of CAMZYOS.
- o CAMZYOS may reduce how well some hormonal birth control works. Talk to your healthcare provider about the use of effective forms of birth control during treatment with CAMZYOS.
- are breastfeeding or plan to breastfeed. It is not known if CAMZYOS passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby during treatment with CAMZYOS.
Before and during CAMZYOS treatment, tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking CAMZYOS with certain medicines or grapefruit juice may cause heart failure. Do not stop or change the dose of a medicine or start a new medicine without telling your healthcare provider.
Especially tell your healthcare provider if you:
- Take over-the-counter medicines such as omeprazole (for example, Prilosec), esomeprazole (for example, Nexium), or cimetidine (for example, Tagamet).
- Take other medicines to treat your obstructive HCM disease.
- Develop an infection.
How should I take CAMZYOS?
- Take CAMZYOS exactly as your healthcare provider tells you to take it.
- Do not change your dose of CAMZYOS without talking to your healthcare provider first.
- Take CAMZYOS 1 time a day.
- Swallow the capsule whole. Do not break, open, or chew the capsule.
- If you miss a dose of CAMZYOS, take it as soon as possible and take your next dose at your regularly scheduled time the next day. Do not take 2 doses on the same day to make up for a missed dose.
- Your healthcare provider may change your dose, temporarily stop, or permanently stop your treatment with CAMZYOS if you have certain side effects.
- If you take too much CAMZYOS, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of CAMZYOS?
CAMZYOS may cause serious side effects, including:
- Heart failure. See “What is the most important information I should know about CAMZYOS?”
The most common side effects of CAMZYOS include: dizziness and fainting (syncope).
These are not all of the possible side effects of CAMZYOS.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Bristol-Myers Squibb at 1-800-721-5072.
How should I store CAMZYOS?
Store CAMZYOS at room temperature between 68°F to 77°F (20°C to 25°C).
Keep CAMZYOS and all medicines out of the reach of children.
General information about the safe and effective use of CAMZYOS.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CAMZYOS for a condition for which it was not prescribed. Do not give CAMZYOS to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about CAMZYOS that is written for health professionals.
What are the ingredients in CAMZYOS?
Active ingredient: mavacamten
Inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate (non-bovine), mannitol, and silicon dioxide. The capsule contains black iron oxide, gelatin, red iron oxide, titanium dioxide, and yellow iron oxide.
Marketed by:
Bristol-Myers Squibb Company
Princeton, NJ 08543 USACAMZYOS® is a trademark of MyoKardia, Inc., a Bristol Myers Squibb company.
For more information, go to www.CAMZYOS.com or call 1-800-721-5072.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 04/2025
-
CAMZYOS 2.5 mg Label
NDC: 73625-111-11
Rx only
CAMZYOS™
(mavacamten)
capsules
2.5 mg
Dispense the enclosed Medication Guide to each patient.
Recommended Dosage: See Prescribing Information
Keep out of reach of children.
Do not use if inner seal bottle is broken or missing.
30 Capsules
Bristol Myers Squibb
-
CAMZYOS 5 mg Label
NDC: 73625-112-11
Rx only
CAMZYOS™
(mavacamten)
capsules
5 mg
Dispense the enclosed Medication Guide to each patient.
Recommended Dosage: See Prescribing Information
Keep out of reach of children.
Do not use if inner seal bottle is broken or missing.
30 Capsules
Bristol Myers Squibb
-
CAMZYOS 10 mg Label
NDC: 73625-113-11
Rx only
CAMZYOS™
(mavacamten)
capsules
10 mg
Dispense the enclosed Medication Guide to each patient.
Recommended Dosage: See Prescribing Information
Keep out of reach of children.
Do not use if inner seal bottle is broken or missing.
30 Capsules
Bristol Myers Squibb
-
CAMZYOS 15 mg Label
NDC: 73625-114-11
Rx only
CAMZYOS™
(mavacamten)
capsules
15 mg
Dispense the enclosed Medication Guide to each patient.
Recommended Dosage: See Prescribing Information
Keep out of reach of children.
Do not use if inner seal bottle is broken or missing.
30 Capsules
Bristol Myers Squibb
-
INGREDIENTS AND APPEARANCE
CAMZYOS
mavacamten capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73625-111 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAVACAMTEN (UNII: QX45B99R3J) (MAVACAMTEN - UNII:QX45B99R3J) MAVACAMTEN 2.5 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color PURPLE (Light purple capsule cap) Score no score Shape CAPSULE (hard gelatin) Size 2mm Flavor Imprint Code Mava;one;side;strength;on;other Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73625-111-11 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/28/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA214998 04/28/2022 CAMZYOS
mavacamten capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73625-112 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAVACAMTEN (UNII: QX45B99R3J) (MAVACAMTEN - UNII:QX45B99R3J) MAVACAMTEN 5 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color YELLOW (capsule cap) Score no score Shape CAPSULE (hard gelatin) Size 2mm Flavor Imprint Code Mava;one;side;strength;on;other Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73625-112-11 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/28/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA214998 04/28/2022 CAMZYOS
mavacamten capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73625-113 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAVACAMTEN (UNII: QX45B99R3J) (MAVACAMTEN - UNII:QX45B99R3J) MAVACAMTEN 10 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color PINK (capsule cap) Score no score Shape CAPSULE (hard gelatin) Size 2mm Flavor Imprint Code Mava;one;side;strength;on;other Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73625-113-11 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/28/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA214998 04/28/2022 CAMZYOS
mavacamten capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73625-114 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAVACAMTEN (UNII: QX45B99R3J) (MAVACAMTEN - UNII:QX45B99R3J) MAVACAMTEN 15 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color GRAY (capsule cap) Score no score Shape CAPSULE (hard gelatin) Size 2mm Flavor Imprint Code Mava;one;side;strength;on;other Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73625-114-11 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/28/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA214998 04/28/2022 Labeler - Myokardia, Inc. (078698088)
Trademark Results [CAMZYOS]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 CAMZYOS 98478203 not registered Live/Pending |
MyoKardia, Inc. 2024-04-01 |
 CAMZYOS 97539194 not registered Live/Pending |
MyoKardia, Inc. 2022-08-08 |
 CAMZYOS 90463748 not registered Live/Pending |
MyoKardia, Inc. 2021-01-13 |
 CAMZYOS 88693134 not registered Live/Pending |
MyoKardia, Inc. 2019-11-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.