COARTEM- artemether and lumefantrine tablet
COARTEM by
Drug Labeling and Warnings
COARTEM by is a Prescription medication manufactured, distributed, or labeled by Central Texas Community Health Centers. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use COARTEM Tablets safely and effectively. See full prescribing information for COARTEM Tablets.
COARTEM® (artemether/lumefantrine) Tablets
Initial U.S. Approval: 2009
INDICATIONS AND USAGE
- Coartem (artemether and lumefantrine) Tablets are indicated for treatment of acute, uncomplicated malaria infections due to Plasmodium falciparum in patients of 5 kg bodyweight and above. (1)
- Coartem Tablets have been shown to be effective in geographical regions where resistance to chloroquine has been reported. (1)
- Coartem Tablets should not be used to treat severe malaria or to prevent malaria. (1)
DOSAGE AND ADMINISTRATION
- Coartem Tablets should be taken with food. (2.1, 5.2)
- Tablets may be crushed and mixed with 1 to 2 teaspoons of water immediately prior to administration to patients, including children. (2.1)
- Coartem Tablets should be administered over 3 days for a total of 6 doses: an initial dose, second dose after 8 hours and then twice-daily (morning and evening) for the following 2 days. (2.2, 2.3)
- The adult dosage for patients with bodyweight of 35 kg and above is 4 tablets per dose for a total of 6 doses. (2.2)
- The number of tablets per dose for children is determined by bodyweight, as shown in the chart below. (2.3)
Tablets per dose by bodyweight; total of 6 doses over 3 days 5 to <15 kg 1 tablet 15 to <25 kg 2 tablets 25 to <35 kg 3 tablets 35 kg and over 4 tablets DOSAGE FORMS AND STRENGTHS
Tablets are scored and contain 20 mg artemether and 120 mg lumefantrine. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Avoid use in patients with known QT prolongation, those with hypokalemia or hypomagnesemia, and those taking other drugs that prolong the QT interval. (5.1, 12.6)
- Halofantrine and Coartem Tablets should not be administered within one month of each other due to potential additive effects on the QT interval. (5.1, 5.2, 12.3)
- Antimalarials should not be given concomitantly, unless there is no other treatment option, due to limited safety data. (5.2)
- QT prolonging drugs, including quinine and quinidine, should be used cautiously following Coartem Tablets. (5.1, 5.2, 7.7, 12.3)
- Substrates, inhibitors, or inducers of CYP3A4, including antiretroviral medications, should be used cautiously with Coartem Tablets, due to a potential loss of efficacy of the concomitant drug or additive QT prolongation. (5.3, 7.2, 7.3)
ADVERSE REACTIONS
The most common adverse reactions in adults (>30%) are headache, anorexia, dizziness, asthenia, arthralgia and myalgia. The most common adverse reactions in children (>12%) are pyrexia, cough, vomiting, anorexia, and headache. (6.2)
To report SUSPECTED ADVERSE REACTIONS, contact Novartis Pharmaceuticals Corporation at 1-888-669-6682 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatchDRUG INTERACTIONS
- CYP3A4 Inducers: Potential for loss of antimalarial efficacy. (4, 5.3, 7.1, 12.3)
- CYP3A4 Inhibitors: Use cautiously due to potential for QT prolongation. (5.3, 7.2, 12.3)
- Antiretrovirals: Use cautiously due to potential for QT prolongation, loss of antiviral efficacy, or loss of antimalarial efficacy of Coartem Tablets. (5.3, 7.3, 12.3)
- Mefloquine: If used immediately before treatment, monitor for decreased efficacy of Coartem Tablets and encourage food consumption. (2.1, 7.4, 12.3)
- Hormonal Contraceptives: Effectiveness may be reduced; use an additional method of birth control. (5.3, 7.5, 12.3)
- CYP2D6 Substrates: Monitor for adverse reactions and potential QT prolongation. (5.1, 5.4, 7.6)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2015
- Coartem (artemether and lumefantrine) Tablets are indicated for treatment of acute, uncomplicated malaria infections due to Plasmodium falciparum in patients of 5 kg bodyweight and above. (1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Administration Instructions
2.2 Dosage in Adult Patients (>16 years of age)
2.3 Dosage in Pediatric Patients
2.4 Dosage in Patients with Hepatic or Renal Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Prolongation of the QT Interval
5.2 Use of QT Prolonging Drugs and Other Antimalarials
5.3 Drug Interactions with CYP3A4
5.4 Drug Interactions with CYP2D6
5.5 Recrudescence
5.6 Hepatic and Renal Impairment
5.7 Plasmodium vivax Infection
6 ADVERSE REACTIONS
6.1 Serious Adverse Reactions
6.2 Clinical Studies Experience
6.3 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Rifampin
7.2 Ketoconazole
7.3 Antiretroviral Drugs
7.4 Prior Use of Mefloquine
7.5 Hormonal Contraceptives
7.6 CYP2D6 Substrates
7.7 Sequential Use of Quinine
7.8 Interaction with Drugs that are Known to Prolong the QT Interval
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic and Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
12.4 Microbiology
12.6 Effects on the Electrocardiogram
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Treatment of Acute, Uncomplicated P. falciparum Malaria
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
Coartem (artemether/lumefantrine) Tablets are indicated for treatment of acute, uncomplicated malaria infections due to Plasmodium falciparum in patients of 5 kg bodyweight and above. Coartem Tablets have been shown to be effective in geographical regions where resistance to chloroquine has been reported [see Clinical Studies (14.1)].
Limitations of Use:
- Coartem Tablets are not approved for patients with severe or complicated P. falciparum malaria.
- Coartem Tablets are not approved for the prevention of malaria.
- Coartem Tablets are not approved for patients with severe or complicated P. falciparum malaria.
-
2 DOSAGE AND ADMINISTRATION
2.1 Administration Instructions
Coartem Tablets should be taken with food. Patients with acute malaria are frequently averse to food. Patients should be encouraged to resume normal eating as soon as food can be tolerated since this improves absorption of artemether and lumefantrine.
For patients who are unable to swallow the tablets such as infants and children, Coartem Tablets may be crushed and mixed with a small amount of water (1 to 2 teaspoons) in a clean container for administration immediately prior to use. The container can be rinsed with more water and the contents swallowed by the patient. The crushed tablet preparation should be followed whenever possible by food/drink (e.g., milk, formula, pudding, broth, and porridge).
In the event of vomiting within 1 to 2 hours of administration, a repeat dose should be taken. If the repeat dose is vomited, the patient should be given an alternative antimalarial for treatment.
2.2 Dosage in Adult Patients (>16 years of age)
A 3-day treatment schedule with a total of 6 doses is recommended for adult patients with a bodyweight of 35 kg and above:
Four tablets as a single initial dose, 4 tablets again after 8 hours and then 4 tablets twice-daily (morning and evening) for the following 2 days (total course of 24 tablets).
For patients weighing less than 35 kg, see Dosage in Pediatric Patients (2.3).
2.3 Dosage in Pediatric Patients
A 3-day treatment schedule with a total of 6 doses is recommended as below:
5 kg to less than 15 kg bodyweight: One tablet as an initial dose, 1 tablet again after 8 hours and then 1 tablet twice-daily (morning and evening) for the following 2 days (total course of 6 tablets).
15 kg to less than 25 kg bodyweight: Two tablets as an initial dose, 2 tablets again after 8 hours and then 2 tablets twice-daily (morning and evening) for the following 2 days (total course of 12 tablets).
25 kg to less than 35 kg bodyweight: Three tablets as an initial dose, 3 tablets again after 8 hours and then 3 tablets twice-daily (morning and evening) for the following 2 days (total course of 18 tablets).
35 kg bodyweight and above: Four tablets as a single initial dose, 4 tablets again after 8 hours and then 4 tablets twice-daily (morning and evening) for the following 2 days (total course of 24 tablets).
2.4 Dosage in Patients with Hepatic or Renal Impairment
No specific pharmacokinetic studies have been carried out in patients with hepatic or renal impairment. Most patients with acute malaria present with some degree of related hepatic and/or renal impairment. In clinical studies, the adverse event profile did not differ in patients with mild or moderate hepatic impairment compared to patients with normal hepatic function. No specific dose adjustments are needed for patients with mild or moderate hepatic impairment.
In clinical studies, the adverse event profile did not differ in patients with mild or moderate renal impairment compared to patients with normal renal function. There were few patients with severe renal impairment in clinical studies. There is no significant renal excretion of lumefantrine, artemether and dihydroartemisinin (DHA) in healthy volunteers and while clinical experience in this population is limited, no dose adjustment is recommended.
Caution should be exercised when administering Coartem Tablets in patients with severe hepatic or renal impairment [see Warnings and Precautions (5.6)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Hypersensitivity
- Known hypersensitivity to artemether, lumefantrine, or to any of the excipients of Coartem Tablets [see Adverse Reactions (6.3)].
Strong CYP3A4 Inducers
- Coadministration of strong inducers of CYP3A4 such as rifampin, carbamazepine, phenytoin, and St. John's wort with Coartem Tablets can result in decreased concentrations of artemether and/or lumefantrine and loss of antimalarial efficacy [see Warnings and Precautions (5.3), Drug Interactions (7.1), and Clinical Pharmacology (12.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Prolongation of the QT Interval
Some antimalarials (e.g., halofantrine, quinine, quinidine) including Coartem Tablets have been associated with prolongation of the QT interval on the electrocardiogram.
Coartem Tablets should be avoided in patients:
- with congenital prolongation of the QT interval (e.g., long QT syndrome) or any other clinical condition known to prolong the QTc interval such as patients with a history of symptomatic cardiac arrhythmias, with clinically relevant bradycardia or with severe cardiac disease.
- with a family history of congenital prolongation of the QT interval or sudden death.
- with known disturbances of electrolyte balance, e.g., hypokalemia or hypomagnesemia.
- receiving other medications that prolong the QT interval, such as class IA (quinidine, procainamide, disopyramide), or class III (amiodarone, sotalol) antiarrhythmic agents; antipsychotics (pimozide, ziprasidone); antidepressants; certain antibiotics (macrolide antibiotics, fluoroquinolone antibiotics, imidazole, and triazole antifungal agents) [see Clinical Pharmacology (12.6)].
- receiving medications that are metabolized by the cytochrome enzyme CYP2D6 which also have cardiac effects (e.g., flecainide, imipramine, amitriptyline, clomipramine) [see Warnings and Precautions (5.4), Drug Interactions (7.6), and Clinical Pharmacology (12.3)].
5.2 Use of QT Prolonging Drugs and Other Antimalarials
Halofantrine and Coartem Tablets should not be administered within 1 month of each other due to the long elimination half-life of lumefantrine (3 to 6 days) and potential additive effects on the QT interval [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
Antimalarials should not be given concomitantly with Coartem Tablets, unless there is no other treatment option, due to limited safety data.
Drugs that prolong the QT interval, including antimalarials such as quinine and quinidine, should be used cautiously following Coartem Tablets, due to the long elimination half-life of lumefantrine (3 to 6 days) and the potential for additive effects on the QT interval; ECG monitoring is advised if use of drugs that prolong the QT interval is medically required [see Warnings and Precautions (5.1), Drug Interactions (7.7), and Clinical Pharmacology (12.3)].
If mefloquine is administered immediately prior to Coartem Tablets there may be a decreased exposure to lumefantrine, possibly due to a mefloquine-induced decrease in bile production. Therefore, patients should be monitored for decreased efficacy and food consumption should be encouraged while taking Coartem Tablets [see Dosage and Administration (2.1), Drug Interactions (7.4), and Clinical Pharmacology (12.3)].
5.3 Drug Interactions with CYP3A4
When Coartem Tablets are coadministered with substrates of CYP3A4 it may result in decreased concentrations of the substrate and potential loss of substrate efficacy. When Coartem Tablets are coadministered with an inhibitor of CYP3A4, including grapefruit juice it may result in increased concentrations of artemether and/or lumefantrine and potentiate QT prolongation. When Coartem Tablets are coadministered with inducers of CYP3A4 it may result in decreased concentrations of artemether and/or lumefantrine and loss of antimalarial efficacy [see Contraindications (4) and Drug Interactions (7)].
Drugs that have a mixed effect on CYP3A4, especially antiretroviral drugs such as HIV protease inhibitors and non-nucleoside reverse transcriptase inhibitors, and those that have an effect on the QT interval should be used with caution in patients taking Coartem Tablets [see Drug Interactions (7.3, 7.7)].
Coartem Tablets may reduce the effectiveness of hormonal contraceptives. Therefore, patients using oral, transdermal patch, or other systemic hormonal contraceptives should be advised to use an additional non-hormonal method of birth control [see Drug Interactions (7.5)].
5.4 Drug Interactions with CYP2D6
Administration of Coartem Tablets with drugs that are metabolized by CYP2D6 may significantly increase plasma concentrations of the coadministered drug and increase the risk of adverse effects. Many of the drugs metabolized by CYP2D6 can prolong the QT interval and should not be administered with Coartem Tablets due to the potential additive effect on the QT interval (e.g., flecainide, imipramine, amitriptyline, clomipramine) [see Warnings and Precautions (5.1), Drug Interactions (7.6), and Clinical Pharmacology (12.3)].
5.5 Recrudescence
Food enhances absorption of artemether and lumefantrine following administration of Coartem Tablets. Patients who remain averse to food during treatment should be closely monitored as the risk of recrudescence may be greater [see Dosage and Administration (2.1)].
In the event of recrudescent P. falciparum infection after treatment with Coartem Tablets, patients should be treated with a different antimalarial drug.
5.6 Hepatic and Renal Impairment
Coartem Tablets have not been studied for efficacy and safety in patients with severe hepatic and/or renal impairment [see Dosage and Administration (2.4)].
5.7 Plasmodium vivax Infection
Coartem Tablets have been shown in limited data (43 patients) to be effective in treating the erythrocytic stage of P. vivax infection. However, relapsing malaria caused by P. vivax requires additional treatment with other antimalarial agents to achieve radical cure i.e., eradicate any hypnozoites forms that may remain dormant in the liver.
- with congenital prolongation of the QT interval (e.g., long QT syndrome) or any other clinical condition known to prolong the QTc interval such as patients with a history of symptomatic cardiac arrhythmias, with clinically relevant bradycardia or with severe cardiac disease.
-
6 ADVERSE REACTIONS
6.1 Serious Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in greater detail in other sections of labeling:
- Hypersensitivity Reactions [see Contraindications (4) and Adverse Reactions (6.3)].
6.2 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rate observed in practice.
The data described below reflect exposure to a 6-dose regimen of Coartem Tablets in 1,979 patients including 647 adults (older than 16 years) and 1,332 children (16 years and younger). For the 6-dose regimen, Coartem Tablets was studied in active-controlled (366 patients) and non-controlled, open-label trials (1,613 patients). The 6-dose Coartem Tablets population was patients with malaria between ages 2 months and 71 years: 67% (1,332) were 16 years and younger and 33% (647) were older than 16 years. Males represented 73% and 53% of the adult and pediatric populations, respectively. The majority of adult patients were enrolled in studies in Thailand, while the majority of pediatric patients were enrolled in Africa.
Tables 1 and 2 show the most frequently reported adverse reactions (≥3%) in adults and children respectively who received the 6-dose regimen of Coartem Tablets. Adverse reactions collected in clinical trials included signs and symptoms at baseline but only treatment emergent adverse events, defined as events that appeared or worsened after the start of treatment, are presented below. In adults, the most frequently reported adverse reactions were headache, anorexia, dizziness, and asthenia. In children, the adverse reactions were pyrexia, cough, vomiting, anorexia, and headache. Most adverse reactions were mild, did not lead to discontinuation of study medication, and resolved.
In limited comparative studies, the adverse reaction profile of Coartem Tablets appeared similar to that of another antimalarial regimen.
Discontinuation of Coartem Tablets due to adverse drug reactions occurred in 1.1% of patients treated with the 6-dose regimen overall: 0.2% (1/647) in adults and 1.6% (21/1,332) in children.
Table 1: Adverse Reactions Occurring in 3% or More of Adult Patients Treated in Clinical Trials with the 6-dose Regimen of Coartem Tablets System Organ Class Preferred Term Adults*
N=647 (%)Nervous system disorders Headache 360 (56) Dizziness 253 (39) Metabolism and nutrition disorders Anorexia 260 (40) General disorders and administration site conditions Asthenia 243 (38) Pyrexia 159 (25) Chills 147 (23) Fatigue 111 (17) Malaise 20 (3) Musculoskeletal and connective tissue disorders Arthralgia 219 (34) Myalgia 206 (32) Gastrointestinal disorders Nausea 169 (26) Vomiting 113 (17) Abdominal pain 112 (17) Diarrhea 46 (7) Psychiatric disorders Sleep disorder 144 (22) Insomnia 32 (5) Cardiac disorders Palpitations 115 (18) Hepatobiliary disorders Hepatomegaly 59 (9) Blood and lymphatic system disorders Splenomegaly 57 (9) Anemia 23 (4) Respiratory, thoracic and mediastinal disorders Cough 37 (6) Skin and subcutaneous tissue disorders Pruritus 24 (4) Rash 21 (3) Ear and labyrinth disorders Vertigo 21 (3) Infections and infestations Malaria 18 (3) Nasopharyngitis 17 (3) * Adult patients defined as >16 years of age
Table 2: Adverse Reactions Occurring in 3% or More of Pediatric Patients Treated in Clinical Trials with the 6-dose Regimen of Coartem Tablets System Organ Class Preferred Term Children*
N=1,332 (%)General disorders and administration site conditions Pyrexia 381 (29) Chills 72 (5) Asthenia 63 (5) Fatigue 46 (3) Respiratory, thoracic and mediastinal disorders Cough 302 (23) Gastrointestinal disorders Vomiting 242 (18) Abdominal pain 112 (8) Diarrhea 100 (8) Nausea 61 (5) Infections and infestations Plasmodium falciparum infection 224 (17) Rhinitis 51 (4) Metabolism and nutrition disorders Anorexia 175 (13) Nervous system disorders Headache 168 (13) Dizziness 56 (4) Blood and lymphatic system disorders Splenomegaly 124 (9) Anemia 115 (9) Hepatobiliary disorders Hepatomegaly 75 (6) Investigations Aspartate aminotransferase increased 51 (4) Musculoskeletal and connective tissue disorders Arthralgia 39 (3) Myalgia 39 (3) Skin and subcutaneous tissue disorders Rash 38 (3) * Children defined as patients ≤16 years of age
Clinically significant adverse reactions reported in adults and/or children treated with the 6-dose regimen of Coartem Tablets which occurred in clinical studies at <3% regardless of causality are listed below:
Blood and lymphatic system disorders: eosinophilia
Ear and labyrinth disorders: tinnitus
Eye disorders: conjunctivitis
Gastrointestinal disorders: constipation, dyspepsia, dysphagia, peptic ulcer
General disorders: gait disturbance
Infections and infestations: abscess, acrodermatitis, bronchitis, ear infection, gastroenteritis, helminthic
infection, hookworm infection, impetigo, influenza, lower respiratory tract infection, malaria,
nasopharyngitis, oral herpes, pneumonia, respiratory tract infection, subcutaneous abscess, upper
respiratory tract infection, urinary tract infectionInvestigations: alanine aminotransferase increased, aspartate aminotransferase increased, hematocrit
decreased, lymphocyte morphology abnormal, platelet count decreased, platelet count increased, white
blood cell count decreased, white blood cell count increasedMetabolism and nutrition disorders: hypokalemia
Musculoskeletal and connective tissue disorders: back pain
Nervous system disorders: ataxia, clonus, fine motor delay, hyperreflexia, hypoesthesia, nystagmus,
tremorPsychiatric disorders: agitation, mood swings
Renal and urinary disorders: hematuria, proteinuria
Respiratory, thoracic and mediastinal disorders: asthma, pharyngo-laryngeal pain
Skin and subcutaneous tissue disorders: urticaria
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Coartem Tablets. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypersensitivity reactions: anaphylaxis, urticaria, angioedema, and serious skin reactions (bullous eruption) have been reported.
-
7 DRUG INTERACTIONS
7.1 Rifampin
Oral administration of rifampin, a strong CYP3A4 inducer, with Coartem Tablets resulted in significant decreases in exposure to artemether, dihydroartemisinin (DHA, metabolite of artemether) and lumefantrine by 89%, 85% and 68%, respectively, when compared to exposure values after Coartem Tablets alone. Concomitant use of strong inducers of CYP3A4 such as rifampin, carbamazepine, phenytoin, and St. John's wort is contraindicated with Coartem Tablets [see Contraindications (4) and Clinical Pharmacology (12.3)].
7.2 Ketoconazole
Concurrent oral administration of ketoconazole, a potent CYP3A4 inhibitor, with a single dose of Coartem Tablets resulted in a moderate increase in exposure to artemether, DHA, and lumefantrine in a study of 15 healthy subjects. No dose adjustment of Coartem Tablets is necessary when administered with ketoconazole or other potent CYP3A4 inhibitors. However, due to the potential for increased concentrations of lumefantrine which could lead to QT prolongation, Coartem Tablets should be used cautiously with drugs that inhibit CYP3A4 [see Warnings and Precautions (5.1, 5.3) and Clinical Pharmacology (12.3)].
7.3 Antiretroviral Drugs
Both artemether and lumefantrine are metabolized by CYP3A4. Antiretroviral drugs, such as protease inhibitors and non-nucleoside reverse transcriptase inhibitors, are known to have variable patterns of inhibition, induction or competition for CYP3A4. Therefore, the effects of antiretroviral drugs on the exposure to artemether, DHA, and lumefantrine are also variable [see Clinical Pharmacology (12.3)]. Coartem Tablets should be used cautiously in patients on antiretroviral drugs because decreased artemether, DHA, and/or lumefantrine concentrations may result in a decrease of antimalarial efficacy of Coartem Tablets, and increased lumefantrine concentrations may cause QT prolongation [see Warnings and Precautions (5.3)].
7.4 Prior Use of Mefloquine
Administration of 3 doses of mefloquine followed 12 hours later by a 6-dose regimen of Coartem Tablets in 14 healthy volunteers demonstrated no effect of mefloquine on plasma concentrations of artemether or the artemether/DHA ratio. However, exposure to lumefantrine was reduced, possibly due to lower absorption secondary to a mefloquine-induced decrease in bile production. Patients should be monitored for decreased efficacy and food consumption should be encouraged with administration of Coartem Tablets [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
7.5 Hormonal Contraceptives
In vitro, the metabolism of ethinyl estradiol and levonorgestrel was not induced by artemether, DHA, or lumefantrine. However, artemether has been reported to weakly induce, in humans, the activity of CYP2C19, CYP2B6, and CYP3A. Therefore, Coartem Tablets may potentially reduce the effectiveness of hormonal contraceptives. Patients using oral, transdermal patch, or other systemic hormonal contraceptives should be advised to use an additional non-hormonal method of birth control [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
7.6 CYP2D6 Substrates
Lumefantrine inhibits CYP2D6 in vitro. Administration of Coartem Tablets with drugs that are metabolized by CYP2D6 may significantly increase plasma concentrations of the coadministered drug and increase the risk of adverse effects. Many of the drugs metabolized by CYP2D6 can prolong the QT interval and should not be administered with Coartem Tablets due to the potential additive effect on the QT interval (e.g., flecainide, imipramine, amitriptyline, clomipramine) [see Warnings and Precautions (5.1, 5.4) and Clinical Pharmacology (12.3)].
7.7 Sequential Use of Quinine
A single dose of intravenous quinine (10 mg/kg bodyweight) concurrent with the final dose of a 6-dose regimen of Coartem Tablets demonstrated no effect of intravenous quinine on the systemic exposure of DHA or lumefantrine. Quinine exposure was also not altered. Exposure to artemether was decreased. This decrease in artemether exposure is not thought to be clinically significant. However, quinine and other drugs that prolong the QT interval should be used cautiously following treatment with Coartem Tablets due to the long elimination half-life of lumefantrine and the potential for additive QT effects; ECG monitoring is advised if use of drugs that prolong the QT interval is medically required [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
7.8 Interaction with Drugs that are Known to Prolong the QT Interval
Coartem Tablets are to be used with caution when coadministered with drugs that may cause prolonged QT interval such as antiarrhythmics of classes IA and III, neuroleptics and antidepressant agents, certain antibiotics including some agents of the following classes: macrolides, fluoroquinolones, imidazole, and triazole antifungal agents [see Warnings and Precautions (5.1, 5.2)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
Safety data from an observational pregnancy study of approximately 500 pregnant women who were exposed to Coartem Tablets (including a third of patients who were exposed in the first trimester), and published data of over 1,000 pregnant patients who were exposed to artemisinin derivatives, did not show an increase in adverse pregnancy outcomes or teratogenic effects over background rate.
The efficacy of Coartem Tablets in the treatment of acute, uncomplicated malaria in pregnant women has not been established.
Coartem Tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pregnant rats dosed during the period of organogenesis at or higher than a dose of about half the highest clinical dose of 1120 mg artemether-lumefantrine per day (based on body surface area comparisons), showed increases in fetal loss, early resorptions and post implantation loss. No adverse effects were observed in animals dosed at about one-third the highest clinical dose. Similarly, dosing in pregnant rabbits at about 3 times the clinical dose (based on body surface area comparisons) resulted in abortions, preimplantation loss, post implantation loss and decreases in the number of live fetuses. No adverse reproductive effects were detected in rabbits at 2 times the clinical dose. Embryo-fetal loss is a significant reproductive toxicity. Other artemisinins are known to be embryotoxic in animals. However, because metabolic profiles in animals and humans are dissimilar, artemether exposures in animals may not be predictive of human exposures [see Nonclinical Toxicology (13.2)]. These data cannot rule out an increased risk for early pregnancy loss or fetal defects in humans.
8.3 Nursing Mothers
It is not known whether artemether or lumefantrine is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Coartem Tablets are administered to a nursing woman. Animal data suggest both artemether and lumefantrine are excreted into breast milk. The benefits of breastfeeding to mother and infant should be weighed against potential risk from infant exposure to artemether and lumefantrine through breast milk.
8.4 Pediatric Use
The safety and effectiveness of Coartem Tablets have been established for the treatment of acute, uncomplicated malaria in studies involving pediatric patients weighing 5 kg or more [see Clinical Studies (14.1)]. The safety and efficacy have not been established in pediatric patients who weigh less than 5 kg. Children from non-endemic countries were not included in clinical trials.
8.5 Geriatric Use
Clinical studies of Coartem Tablets did not include sufficient numbers of subjects aged 65 years and over to determine they respond differently from younger subjects. In general, the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy in elderly patients should be considered when prescribing Coartem Tablets.
8.6 Hepatic and Renal Impairment
No specific pharmacokinetic studies have been performed in patients with either hepatic or renal impairment. Coartem Tablets have not been studied for efficacy and safety in patients with severe hepatic and/or renal impairment. Based on the pharmacokinetic data in 16 healthy subjects showing no or insignificant renal excretion of lumefantrine, artemether and DHA, no dose adjustment for the use of Coartem Tablets in patients with renal impairment is advised. No dosage adjustment is necessary in patients with mild to moderate hepatic impairment [see Dosage and Administration (2.4) and Warnings and Precautions (5.6)].
- 10 OVERDOSAGE
-
11 DESCRIPTION
Coartem Tablets contain a fixed combination of 2 antimalarial active ingredients, artemether, an artemisinin derivative, and lumefantrine. Both components are blood schizontocides. The chemical name of artemether is (3R,5aS,6R,8aS,9R,10S,12R,12aR)-10-methoxy-3,6,9-trimethyldecahydro-3,12-epoxypyrano[4,3-j]-1,2-benzodioxepine. Artemether is a white, crystalline powder that is freely soluble in acetone, soluble in methanol and ethanol, and practically insoluble in water. It has the empirical formula C16H26O5 with a molecular weight of 298.4, and the following structural formula:
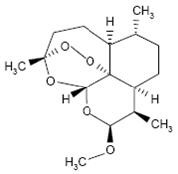
The chemical name of lumefantrine is (1RS)-2-(dibutylamino)-1-{(9Z)-2,7-dichloro-9-[(4-chlorophenyl)methylene]-9H-fluorene-4-yl}ethanol. Lumefantrine is a yellow, crystalline powder that is freely soluble in N,N-dimethylformamide, chloroform, and ethyl acetate; soluble in dichloromethane; slightly soluble in ethanol and methanol; and insoluble in water. It has the empirical formula C30H32Cl3NO with a molecular weight of 528.9, and the following structural formula:
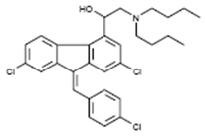
Coartem Tablets are for oral administration. Each Coartem Tablet contains 20 mg of artemether and 120 mg lumefantrine. The inactive ingredients are colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, and polysorbate 80.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Coartem Tablets, a fixed dose combination of artemether and lumefantrine in the ratio of 1:6, is an antimalarial agent [see Clinical Pharmacology (12.4)].
12.3 Pharmacokinetics
Absorption
Following administration of Coartem Tablets to healthy volunteers and patients with malaria, artemether is absorbed with peak plasma concentrations reached about 2 hours after dosing. Absorption of lumefantrine, a highly lipophilic compound, starts after a lag-time of up to 2 hours, with peak plasma concentrations about 6 to 8 hours after administration. The single dose (4 tablets) pharmacokinetic parameters for artemether, dihydroartemisinin (DHA), an active antimalarial metabolite of artemether, and lumefantrine in adult Caucasian healthy volunteers are given in Table 3. Multiple dose data after the 6-dose regimen of Coartem Tablets in adult malaria patients are given in Table 4.
Table 3: Single Dose Pharmacokinetic Parametersa for Artemether, Dihydroartemisinin (DHA), and Lumefantrine under Fed Conditions Study 2102
(n=50)Study 2104
(n=48)Artemether Cmax (ng/mL) 60.0 ± 32.5 83.8 ± 59.7 tmax (h) 1.50 2.00 AUClast (ng∙h/mL) 146 ± 72.2 259 ± 150 t½ (h) 1.6 ± 0.7 2.2 ± 1.9 DHA Cmax (ng/mL) 104 ± 35.3 90.4 ± 48.9 tmax (h) 1.76 2.00 AUClast (ng∙h/mL) 284 ± 83.8 285 ± 98.0 t½ (h) 1.6 ± 0.6 2.2 ± 1.5 Lumefantrine Cmax (µg/mL) 7.38 ± 3.19 9.80 ± 4.20 tmax (h) 6.01 8.00 AUClast (µg∙h/mL) 158 ± 70.1 243 ± 117 t½ (h) 101 ± 35.6 119 ± 51.0 aMean ± SD Cmax, AUClast, t½ and Median tmax
Food enhances the absorption of both artemether and lumefantrine. In healthy volunteers, the relative bioavailability of artemether was increased between 2- to 3-fold, and that of lumefantrine 16-fold when Coartem Tablets were taken after a high-fat meal compared under fasted conditions. Patients should be encouraged to take Coartem Tablets with a meal as soon as food can be tolerated [see Dosage and Administration (2.1)].
Distribution
Artemether and lumefantrine are both highly bound to human serum proteins in vitro (95.4% and 99.7%, respectively). Dihydroartemisinin is also bound to human serum proteins (47% to 76%). Protein binding to human plasma proteins is linear.
Biotransformation
In human liver microsomes and recombinant CYP450 enzymes, the metabolism of artemether was catalyzed predominantly by CYP3A4/5. Dihydroartemisinin (DHA) is an active metabolite of artemether. The metabolism of artemether was also catalyzed to a lesser extent by CYP2B6, CYP2C9 and CYP2C19. In vitro studies with artemether at therapeutic concentrations revealed no significant inhibition of the metabolic activities of CYP1A2, CYP2A6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, CYP3A4/5, and CYP4A9/11. In vitro studies with artemether, DHA, and lumefantrine at therapeutic concentrations revealed no significant induction of the metabolic activities of CYP1A1, CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP3A4, or CYP3A5.
During repeated administration of Coartem Tablets, systemic exposure of artemether decreased significantly, while concentrations of DHA increased, although not to a statistically significant degree. The artemether/DHA AUC ratio is 1.2 after a single dose and 0.3 after 6 doses given over 3 days. This suggests that there was induction of enzymes responsible for the metabolism of artemether.
In human liver microsomes and in recombinant CYP450 enzymes, lumefantrine was metabolized mainly by CYP3A4 to desbutyl-lumefantrine. The systemic exposure to the metabolite desbutyl-lumefantrine was less than 1% of the exposure to the parent compound. In vitro, lumefantrine significantly inhibits the activity of CYP2D6 at therapeutic plasma concentrations.
Caution is recommended when combining Coartem Tablets with substrates, inhibitors, or inducers of CYP3A4, especially antiretroviral drugs and those that prolong the QT interval (e.g., macrolide antibiotics, pimozide) [see Contraindications (4), Warnings and Precautions (5.1, 5.2, 5.3), and Drug Interactions (7)].
Coadministration of Coartem Tablets with CYP2D6 substrates may result in increased plasma concentrations of the CYP2D6 substrate and increase the risk of adverse reactions. In addition, many of the drugs metabolized by CYP2D6 can prolong the QT interval and should not be administered with Coartem Tablets due to the potential additive effect on the QT interval (e.g., flecainide, imipramine, amitriptyline, clomipramine) [see Warnings and Precautions (5.1, 5.4) and Drug Interactions (7.6)].
Elimination
Artemether and DHA are cleared from plasma with an elimination half-life of about 2 hours. Lumefantrine is eliminated more slowly, with an elimination half-life of 3 to 6 days in healthy volunteers and in patients with falciparum malaria. Demographic characteristics such as sex and weight appear to have no clinically relevant effects on the pharmacokinetics of artemether and lumefantrine.
In 16 healthy volunteers, neither lumefantrine nor artemether was found in the urine after administration of Coartem Tablets, and urinary excretion of DHA amounted to less than 0.01% of the artemether dose.
Hepatic and Renal Impairment
No specific pharmacokinetic studies have been performed in patients with either hepatic or renal impairment. There is no significant renal excretion of lumefantrine, artemether and DHA in healthy volunteers and while clinical experience in this population is limited, no dose adjustment in renal impairment is recommended [see Dosage and Administration (2.4)].
Pediatric Patients
The PK of artemether, DHA, and lumefantrine were obtained in 2 pediatric studies by sparse sampling using a population-based approach. PK estimates derived from a composite plasma concentration profile for artemether, DHA, and lumefantrine are provided in Table 4.
Systemic exposure to artemether, DHA, and lumefantrine, when dosed on a mg/kg body weight basis in pediatric patients (≥5 to <35 kg body weight), is comparable to that of the recommended dosing regimen in adult patients.
Table 4: Summary of Pharmacokinetic Parameters for Lumefantrine, Artemether and DHA in Pediatric and Adult Patients with Malaria Following Administration of a 6-dose Regimen of Coartem Tablets Adults1 Pediatric patients (body weight, kg)2 Drug 5 to <15 15 to <25 25 to <35 Lumefantrine Mean Cmax, range (mcg/mL) 5.60 - 9.0 4.71 – 12.6 Not Available Mean AUClast, range (mcg∙h/mL) 410 - 561 372 – 699 Not Available Artemether Mean Cmax ± SD (ng/mL) 186 ± 125 223 ± 309 198 ± 179 174 ± 145 Dihydroartemisinin Mean Cmax ± SD (ng/mL) 101 ± 58 54.7 ± 58.9 79.8 ± 80.5 65.3 ± 23.6 1 There are a total of 181 adults for lumefantrine pharmacokinetic parameters and a total of 25 adults for artemether and dihydroartemisinin pharmacokinetic parameters.
2 There are 477 children for the lumefantrine pharmacokinetic parameters; for artemether and dihydroartemisinin pharmacokinetic parameters there are 55, 29, and 8 children
for the 5 to <15, 15 to <25 and the 25 to <35 kg groups, respectively.Geriatric Patients
No specific pharmacokinetic studies have been performed in patients older than 65 years of age.
Drug Interactions
Rifampin (strong CYP3A4 inducer)
Oral administration of rifampin (600 mg daily), a strong CYP3A4 inducer, with Coartem Tablets (6-dose regimen over 3 days) in 6 HIV-1 and tuberculosis co-infected adults without malaria resulted in significant decreases in exposure, in terms of AUC, to artemether, DHA and lumefantrine by 89%, 85% and 68%, respectively, when compared to exposure values after Coartem Tablets alone. Concomitant use of strong inducers of CYP3A4 such as rifampin, carbamazepine, phenytoin, and St. John's wort is contraindicated with Coartem Tablets [see Contraindications (4)].
Ketoconazole (potent CYP3A4 inhibitor)
Concurrent oral administration of ketoconazole (400 mg on day 1 followed by 200 mg on days 2, 3, 4 and 5) with Coartem Tablets (single dose of 4 tablets of 20 mg artemether/120 mg lumefantrine per tablet) with a meal led to an increase in exposure, in terms of area under the curve (AUC), of artemether (2.3-fold), DHA (1.5-fold), and lumefantrine (1.6-fold) in 13 healthy subjects. The pharmacokinetics of ketoconazole was not evaluated. Based on this study, dose adjustment of Coartem Tablets is considered unnecessary when administered with ketoconazole or other CYP3A4 inhibitors. However, due to the potential for increased concentrations of lumefantrine which could lead to QT prolongation, Coartem Tablets should be used cautiously with other drugs that inhibit CYP3A4 (e.g., antiretroviral drugs, macrolide antibiotics, antidepressants, imidazole antifungal agents) [see Warnings and Precautions (5.1, 5.3)].
Antimalarials
The oral administration of mefloquine in 14 healthy volunteers administered as 3 doses of 500 mg, 250 mg and 250 mg, followed 12 hours later by Coartem Tablets (6 doses of 4 tablets of 20 mg artemether/120 mg lumefantrine per tablet), had no effect on plasma concentrations of artemether or the artemether/DHA ratio. In the same study, there was a 30% reduction in Cmax and 40% reduction in AUC of lumefantrine, possibly due to lower absorption secondary to a mefloquine-induced decrease in bile production.
Intravenous administration of a single dose of quinine (10 mg/kg bodyweight) concurrent with the last dose of a 6-dose regimen of Coartem Tablets had no effect on systemic exposure of DHA, lumefantrine or quinine in 14 healthy volunteers. Mean AUC of artemether were 46% lower when administered with quinine compared to Coartem Tablets alone. This decrease in artemether exposure is not thought to be clinically significant. However, quinine should be used cautiously in patients following treatment with Coartem Tablets due to the long elimination half-life of lumefantrine and the potential for additive effects on the QT interval; ECG monitoring is advised if use of quinine is medically required [see Warnings and Precautions (5.2)].
Antiretroviral Drugs
The oral administration of lopinavir/ritonavir (400 mg/100 mg twice daily for 26 days) in 10 healthy volunteers coadministered with Coartem Tablets (6-dose regimen over 3 days), resulted in a decrease in systemic exposures, in terms of AUC, to artemether and DHA by approximately 40%, but an increase in exposure to lumefantrine by approximately 2.3-fold. The oral administration of efavirenz (600 mg once daily for 26 days) in 12 healthy volunteers coadministered with Coartem Tablets (6-dose regimen over 3 days), resulted in a decrease in exposures to artemether, DHA, and lumefantrine by approximately 50%, 45%, and 20%, respectively. Exposures to lopinavir/ritonavir and efavirenz were not significantly affected by concomitant use of Coartem Tablets. Coartem Tablets should be used cautiously in patients on antiretroviral drugs such as HIV protease inhibitors and non-nucleoside reverse transcriptase inhibitors because decreased artemether, DHA, and/or lumefantrine concentrations may result in a decrease of antimalarial efficacy of Coartem Tablets, and increased lumefantrine concentrations may cause QT prolongation [see Warnings and Precautions (5.3) and Drug Interactions (7.3)].
Hormonal Contraceptives
No clinical drug-drug interaction studies between Coartem Tablets and hormonal contraceptives have been performed. In vitro studies revealed that the metabolism of ethinyl estradiol and levonorgestrel was not induced by artemether, DHA or lumefantrine. However, artemether has been reported to weakly induce, in humans, the activity of CYP2C19, CYP2B6, and CYP3A. Therefore, coadministration of Coartem Tablets may potentially reduce the effectiveness of hormonal contraceptives [see Warnings and Precautions (5.3) and Drug Interactions (7.5)].
12.4 Microbiology
Mechanism of Action
Coartem Tablets, a fixed ratio of 1:6 parts of artemether and lumefantrine, respectively, is an antimalarial agent. Artemether is rapidly metabolized into an active metabolite dihydroartemisinin (DHA). The antimalarial activity of artemether and DHA has been attributed to endoperoxide moiety. The exact mechanism by which lumefantrine exerts its antimalarial effect is not well defined. Available data suggest lumefantrine inhibits the formation of β-hematin by forming a complex with hemin. Both artemether and lumefantrine were shown to inhibit nucleic acid and protein synthesis.
Activity In Vitro and In Vivo
Artemether and lumefantrine are active against the erythrocytic stages of Plasmodium falciparum.
Drug Resistance
Strains of P. falciparum with a moderate decrease in susceptibility to artemether or lumefantrine alone can be selected in vitro or in vivo, but not maintained in the case of artemether. The clinical relevance of such an effect is not known.
12.6 Effects on the Electrocardiogram
In a healthy adult volunteer parallel-group study including a placebo and moxifloxacin control-group (n=42 per group), the administration of the 6-dose regimen of Coartem Tablets was associated with prolongation of QTcF (Fridericia). Following administration of a 6-dose regimen of Coartem Tablets consisting of 4 tablets per dose (total of 4 tablets of 80 mg artemether/480 mg lumefantrine) taken with food, the maximum mean change from baseline and placebo adjusted QTcF was 7.5 msec (1-sided 95% Upper CI: 11 msec). There was a concentration-dependent increase in QTcF for lumefantrine.
In clinical trials conducted in children, no patient had QTcF >500 msec. Over 5% of patients had an increase in QTcF of over 60 msec.
In clinical trials conducted in adults, QTcF prolongation of >500 msec was reported in 3 (0.3%) patients. Over 6% of adults had a QTcF increase of over 60 msec from baseline.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies were not conducted.
Mutagenesis
No evidence of mutagenicity was detected. The artemether: lumefantrine combination was evaluated using the Salmonella and Escherichia/mammalian-microsome mutagenicity test, the gene mutation test with Chinese hamster cells V79, the cytogenetic test on Chinese hamster cells in vitro, and the rat micronucleus test, in vivo.
Impairment of Fertility
Pregnancy rates were reduced by about one-half in female rats dosed for 2 to 4 weeks with the artemether-lumefantrine combination at 1000 mg/kg (about 9 times the clinical dose based on body surface area comparisons). Male rats dosed for 70 days showed increases in abnormal sperm (87% abnormal) and increased testes weights at 30 mg/kg doses (about one-third the clinical dose). Higher doses (about 9 times the clinical dose) resulted in decreased sperm motility and 100% abnormal sperm cells.
13.2 Animal Toxicology and/or Pharmacology
Neonatal rats (7 to 21 days old) were more sensitive to the toxic effects of artemether (a component of Coartem Tablets) than older juvenile rats or adults. Mortality and severe clinical signs were observed in neonatal rats at doses which were well tolerated in pups above 22 days old.
-
14 CLINICAL STUDIES
14.1 Treatment of Acute, Uncomplicated P. falciparum Malaria
The efficacy of Coartem Tablets was evaluated for the treatment of acute, uncomplicated malaria caused by P. falciparum in HIV negative patients in 8 clinical studies. Uncomplicated malaria was defined as symptomatic P. falciparum malaria without signs and symptoms of severe malaria or evidence of vital organ dysfunction. Baseline parasite density ranged from 500/mcL to 200,000/mcL (0.01% to 4% parasitemia) in the majority of patients. Studies were conducted in partially immune and non-immune adults and children (≥5kg body weight) with uncomplicated malaria in China, Thailand, sub-Saharan Africa, Europe, and South America. Patients who had clinical features of severe malaria, severe cardiac, renal, or hepatic impairment were excluded.
The studies include two 4-dose studies assessing the efficacy of the components of the regimen, a study comparing a 4-dose versus a 6-dose regimen, and 5 additional 6-dose regimen studies.
Coartem Tablets were administered at 0, 8, 24, and 48 hours in the 4-dose regimen, and at 0, 8, 24, 36, 48, and 60 hours in the 6-dose regimen. Efficacy endpoints consisted of:
- 28-day cure rate, defined as clearance of asexual parasites (the erythrocytic stage) within 7 days without recrudescence by day 28
- parasite clearance time (PCT), defined as time from first dose until first total and continued disappearance of asexual parasite which continues for a further 48 hours
- fever clearance time (FCT), defined as time from first dose until the first time body temperature fell below 37.5°C and remained below 37.5°C for at least a further 48 hours (only for patients with temperature >37.5°C at baseline)
The modified intent to treat (mITT) population includes all patients with malaria diagnosis confirmation who received at least 1 dose of study drug. Evaluable patients generally are all patients who had a day 7 and a day 28 parasitological assessment or experienced treatment failure by day 28.
Studies 1 and 2: The 2 studies which assessed the efficacy of Coartem Tablets (4 doses of 4 tablets of 20 mg artemether/120 mg lumefantrine) compared to each component alone were randomized, double-blind, comparative, single center, conducted in China. The efficacy results (Table 5) support that the combination of artemether and lumefantrine in Coartem Tablets had a significantly higher 28-day cure rate compared to artemether and had a significantly faster parasite clearance time (PCT) and fever clearance time (FCT) compared to lumefantrine.
Table 5: Clinical Efficacy of Coartem Tablets versus Components (mITT Population)1 Study No.
Region/patient ages28-day cure rate2
n/N (%) patientsMedian FCT3
[25th,75th percentile]Median PCT
[25th,75th percentile]Study 1
China, ages 13 to 57 yearsCoartem Tablets 50/51 (98.0) 24 hours [9, 48] 30 hours [24, 36] Artemether4 24/52 (46.2) 21 hours [12, 30] 30 hours [24, 33] Lumefantrine5 47/52 (90.4) 60 hours [36, 78] 54 hours [45, 66] Study 2
China, ages 12 to 65 yearsCoartem Tablets 50/52 (96.2) 21 hours [6, 33] 30 hours [24, 36] Lumefantrine 6 45/51 (88.2) 36 hours [12, 60] 48 hours [42, 60] 1In mITT analysis, patients whose status was uncertain were classified as treatment failures.
2Efficacy cure rate based on blood smear microscopy.
3For patients who had a body temperature >37.5°C at baseline only
495% CI (Coartem Tablets – artemether) on 28-day cure rate: 37.8%, 66.0%
5P-value comparing Coartem Tablets to lumefantrine on parasite clearance time (PCT) and fever clearance time (FCT): <0.001
6P-value comparing Coartem Tablets to lumefantrine on parasite clearance time (PCT): <0.001 and on fever clearance time (FCT): <0.05Results of 4-dose studies conducted in areas with high resistance such as Thailand during 1995-96 showed lower efficacy results than the above studies. Therefore, Study 3 was conducted.
Study 3: Study 3 was a randomized, double-blind, 2-center study conducted in Thailand in adults and children (aged ≥2 years), which compared the 4-dose regimen (administered over 48 hours) of Coartem Tablets to a 6-dose regimen (administered over 60 hours). Twenty-eight day cure rate in mITT subjects was 81% (96/118) for the Coartem Tablets 6-dose arm as compared to 71% (85/120) in the 4-dose arm.
Studies 4, 5, 6, 7, and 8: In these studies, Coartem Tablets were administered as the 6-dose regimen.
In study 4, a total of 150 adults and children aged ≥2 years received Coartem Tablets. In study 5, a total 164 adults and children ≥12 years received Coartem Tablets. Both studies were conducted in Thailand.
Study 6 was a study of 165 non-immune adults residing in regions non-endemic for malaria (Europe and Colombia) who contracted acute uncomplicated falciparum malaria when traveling in endemic regions.
Study 7 was conducted in Africa in 310 infants and children aged 2 months to 9 years, weighing 5 kg to 25 kg, with an axillary temperature ≥37.5ºC.
Study 8 was conducted in Africa in 452 infants and children, aged 3 months to 12 years, weighing 5 kg to <35 kg, with fever (≥37.5°C axillary or ≥38°C rectally) or history of fever in the preceding 24 hours.
Results of 28-day cure rate, median parasite clearance time (PCT), and fever clearance time (FCT) for Studies 3 to 8 are reported in Table 6.
Table 6: Clinical Efficacy of 6-dose Regimen of Coartem Tablets Study No. Region/ages 28-day cure rate1 n/N (%) patients Median FCT2
[25th, 75th percentile]Median PCT
[25th, 75th percentile]mITT3 Evaluable Study 3 Thailand, ages 3 – 62 years 96/118 (81.4) 93/96 (96.9) 35 hours
[20, 46]44 hours
[22, 47]Early failure4 0 0 Late failure5 4 (3.4) 3 (3.1) Lost to follow-up 18 (15.3) Other6 0 Study 4 Thailand, ages 2 – 63 years 130/149 (87.2) 130/134 (97.0) 22 hours
[19, 44]NA Early failure4 0 0 Late failure5 4 (2.7) 4 (3.0) Lost to follow-up 13 (8.7) Other6 2 (1.3) Study 5 Thailand, ages 12 – 71 years 148/164 (90.2) 148/155 (95.5) 29 hours
[8, 51]29 hours
[18, 40]Early failure4 0 0 Late failure5 7 (4.3) 7 (4.5) Lost to follow-up 9 (5.5) Other6 0 Study 6
Europe/Columbia, ages 16 – 66 years120/162 (74.1) 119/124 (96.0) 37 hours
[18, 44]42 hours
[34, 63]Early failure4 6 (3.7) 1 (0.8) Late failure5 3 (1.9) 3 (2.4) Lost to follow-up 17 (10.5) Other6 16 (9.9) 1 (0.8) Study 7 Africa, ages 2 months – 9 years 268/310 (86.5) 267/300 (89.0) 8 hours
[8, 24]24 hours
[24, 36]Early failure4 2 (0.6) 0 Late failure5 34 (11.0) 33 (11.0) Lost to follow-up 2 (0.6) Other6 4 (1.3) Study 8 Africa, ages 3 months – 12 years 374/452 (82.7) 370/419 (88.3) 8 hours
[8, 23]35 hours
[24, 36]Early failure4 13 (2.9) 0 Late failure5 49 (10.8) 49 (11.7) Lost to follow-up 6 (1.3) Other6 10 (2.2) 1 Efficacy cure rate based on blood smear microscopy
2 For patients who had a body temperature >37.5°C at baseline only
3 In mITT analysis, patients whose status was uncertain were classified as treatment failures.
4 Early failures were usually defined as patients withdrawn for unsatisfactory therapeutic effect within the first 7 days or because they received another antimalarial medication within the first 7 days
5 Late failures were defined as patients achieving parasite clearance within 7 days but having parasite reappearance including recrudescence or new infection during the 28-day follow-up period
6 Other includes withdrawn due to protocol violation or non-compliance, received additional medication after day 7, withdrew consent, missing day 7 or 28 assessmentIn all studies, patients' signs and symptoms of malaria resolved when parasites were cleared.
In studies conducted in areas with high transmission rates, such as Africa, reappearance of P. falciparum parasites may be due to recrudescence or a new infection.
The efficacy by body weight category for studies 7 and 8 is summarized in Table 7.
Table 7: Clinical Efficacy by Weight for Pediatric Studies Study No.
Age categoryCoartem Tablets 6-dose Regimen mITT population1 Evaluable population Median PCT
[25th,75th percentile]28-day cure rate2
n/N (%) patients28-day cure rate2
n/N (%) patientsStudy 7 5 to <10 kg 24 [24, 36] 133/154 (86.4) 133/149 (89.3) 10 to <15 kg 35 [24, 36] 94/110 (85.5) 94/107 (87.9) 15 to 25 kg 24 [24, 36] 41/46 (89.1) 40/44 (90.9) Study 83 5 to <10 kg 36 [24, 36] 61/83 (73.5) 61/69 (88.4) 10 to <15 kg 35 [24, 36] 160/190 (84.2) 157/179 (87.7) 15 to <25 kg 35 [24, 36] 123/145 (84.8) 123/140 (87.9) 25 to <35 kg 26 [24, 36] 30/34 (88.2) 29/31 (93.5) 1 In mITT analysis, patients whose status was uncertain were classified as treatment failures.
2 Efficacy cure rate based on blood smear microscopy
3 Coartem Tablets administered as crushed tabletsThe efficacy of Coartem Tablets for the treatment P. falciparum infections mixed with P. vivax was assessed in a small number of patients. Coartem Tablets are only active against the erythrocytic phase of P. vivax malaria. Of the 43 patients with mixed infections at baseline, all cleared their parasitemia within 48 hours. However, parasite relapse occurred commonly (14/43; 33%). Relapsing malaria caused by P. vivax requires additional treatment with other antimalarial agents to achieve radical cure i.e., eradicate any hypnozoite forms that may remain dormant in the liver.
- 28-day cure rate, defined as clearance of asexual parasites (the erythrocytic stage) within 7 days without recrudescence by day 28
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Coartem (artemether/lumefantrine) Tablets
20 mg/120 mg Tablets - yellow, round flat tablets with beveled edges and scored on one side. Tablets are imprinted with N/C on one side and CG on the other.
Bottle of 24 NDC: 0078-0568-45
Store at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature].
Dispense in tight container (USP).
-
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-Approved Patient Labeling.
Information for Safe Use
- Instruct patients to take Coartem Tablets with food. Patients who do not have an adequate intake of food are at risk for recrudescence of malaria.
- Patients with known hypersensitivity to artemether, lumefantrine, or to any of the excipients should not receive Coartem Tablets.
- Instruct patients to inform their physician of any personal or family history of QT prolongation or proarrhythmic conditions such as hypokalemia, bradycardia, or recent myocardial ischemia.
- Instruct patients to inform their physician if they are taking any other medications that prolong the QT interval, such as class IA (quinidine, procainamide, disopyramide), or class III (amiodarone, sotalol) antiarrhythmic agents; antipsychotics (pimozide, ziprasidone); antidepressants; certain antibiotics (macrolide antibiotics, fluoroquinolone antibiotics, imidazole, and triazole antifungal agents).
- Instruct patients to notify their physicians if they have any symptoms of prolongation of the QT interval, including prolonged heart palpitations or a loss of consciousness.
- Instruct patients to avoid medications that are metabolized by the cytochrome enzyme CYP2D6 while receiving Coartem Tablets since these drugs also have cardiac effects (e.g., flecainide, imipramine, amitriptyline, clomipramine).
- Inform patients that based on animal data, Coartem Tablets administered during pregnancy may result in fetal loss. Fetal defects have been reported when artemisinins are administered to animals.
- Halofantrine and Coartem Tablets should not be administered within 1 month of each other due to potential additive effects on the QT interval.
- Antimalarials should not be given concomitantly with Coartem Tablets, unless there is no other treatment option, due to limited safety data.
- QT prolonging drugs, including quinine and quinidine, should be used cautiously following Coartem Tablets due to the long elimination half-life of lumefantrine and the potential for additive effects on the QT interval. ECG monitoring is advised if use of drugs that prolong the QT interval is medically required.
- Closely monitor food intake in patients who received mefloquine immediately prior to treatment with Coartem Tablets.
- Use Coartem Tablets cautiously in patients receiving other drugs that are substrates, inhibitors or inducers of CYP3A4, including grapefruit juice, especially those that prolong the QT interval or are antiretroviral drugs.
- Coadministration of strong inducers of CYP3A4 such as rifampin, carbamazepine, phenytoin, and St. John's wort is contraindicated with Coartem Tablets.
- Coartem Tablets may reduce the effectiveness of hormonal contraceptives. Therefore, patients using oral, transdermal patch, or other systemic hormonal contraceptives should be advised to use an additional non-hormonal method of birth control.
- Inform patients that Coartem Tablets can cause hypersensitivity reactions. Instruct patients to discontinue the drug at the first sign of a skin rash, hives or other skin reactions, a rapid heartbeat, difficulty in swallowing or breathing, any swelling suggesting angioedema (e.g., swelling of the lips, tongue, face, tightness of the throat, hoarseness), or other symptoms of an allergic reaction.
T2015-44
March 2015 - Instruct patients to take Coartem Tablets with food. Patients who do not have an adequate intake of food are at risk for recrudescence of malaria.
-
PATIENT PACKAGE INSERT
FDA-Approved Patient Labeling
Patient Information
Coartem®
(co-AR-tem)
(artemether and lumefantrine)
Tablets
Read this patient information before you start taking Coartem. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What is Coartem?
Coartem is a prescription medicine used to treat uncomplicated malaria in adults and children who weigh at least 11 pounds (5 kg).
Who should not take Coartem?
Do not take Coartem if you are allergic to any of the ingredients. See the end of this leaflet for a complete list of ingredients in Coartem.
Do not take Coartem if you are taking rifampin (medicine to treat leprosy or tuberculosis), certain medicines used to treat epilepsy (such as carbamazepine, phenytoin), or St. John's wort (Hypericum perforatum, a medicinal plant or extract of this medicinal plant).
What should I tell my healthcare provider before taking Coartem?
Before you take Coartem, tell your healthcare provider about all your medical conditions including if you:
- have heart disease or a family history of heart problems or heart disease
- have liver or kidney problems
- have recently taken other medicines used to treat malaria
- are pregnant or are planning to become pregnant. Coartem may increase your risk for loss of pregnancy. Fetal defects have been reported when artemisinins are administered to animals. Talk to your healthcare provider before taking Coartem.
- are breastfeeding. It is not known if Coartem passes into your breast milk. You and your doctor will decide the best way to feed your baby if you take Coartem.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Coartem and other medicines may affect each other causing side effects. Coartem may affect the way other medicines work and other medicines may affect how Coartem works.
Especially tell your doctor if you take:
- any other medicines to treat or prevent malaria
- medicines for your heart
- antipsychotic medicines
- antidepressants
- medicines for seizures or trigeminal neuralgia (facial nerve pain)
- antibiotics (including medicines to treat tuberculosis)
- medicines to treat HIV-infection
- hormonal methods of birth control (for example, birth control pills or patch). If you are taking a hormonal birth control medicine, you should also use an additional method of birth control.
Ask your healthcare provider if you are not sure if your medicine is 1 that is listed above. Know the medicines you take. Keep a list of your medicines with you to show your healthcare providers when you get a new medicine.
How should I take Coartem?
- Take Coartem exactly as prescribed.
- If you weigh 77 pounds (35 kg) or more, 1 dose of Coartem is 4 tablets.
- If you weigh less than 77 pounds (35 kg), your healthcare provider will tell you how many tablets to take for each dose.
- A full course of treatment is 6 doses of Coartem taken over 3 days:
Day 1: take 1 dose; 8 hours later take 1 dose
Day 2: take 1 dose in the morning, 1 dose in the evening
Day 3: take 1 dose in the morning, 1 dose in the evening
Take Coartem for 3 days even if you are feeling better.
- Every dose of Coartem should be taken with food, such as milk, infant formula, pudding, porridge, or broth. It is important for you to eat as soon as you can so that your malaria will go away and not get worse.
- Do not drink grapefruit juice while you take Coartem. Drinking grapefruit juice during treatment with Coartem can cause you to have too much medicine in your blood.
- Coartem may be crushed and mixed with 1 to 2 teaspoons of water in a clean container.
- If you vomit within 1 hour of taking Coartem you should take another dose of Coartem. If you vomit the second dose, tell your healthcare provider. A different medicine may need to be prescribed for you.
Tell your healthcare provider right away if:
- your malaria does not get better
- you vomited any of your doses of Coartem
- you are not able to eat
- you get flu-like symptoms (chills, fever, muscle pains, or headaches) again after you have finished your treatment with Coartem.
- you have any change in the way your heart beats or a loss of consciousness (fainting).
What are the possible side effects of Coartem?
Coartem can cause serious side effects including:
-
A heart problem called QT prolongation that can cause an abnormal heartbeat can happen in people who take Coartem. The chance of this happening is higher in people with a family history of prolonged QT interval, low potassium (hypokalemia), and in people who take medicines to control heartbeats.
- Allergic reactions. Symptoms of an allergic reaction include: rash, hives, fast heartbeat, trouble swallowing or breathing, swelling of lips, tongue, face, tightness of the throat, or trouble speaking. If you have a serious allergic reaction, stop taking Coartem and get emergency medical help right away.
The most common side effects in adults are:
- headache
- feeling dizzy
- feeling weak
- loss of appetite
- muscle and joint pain or stiffness
- feeling tired
- chills
- fever
The most common side effects in children are:
- fever
- cough
- vomiting
- headache
- loss of appetite
These are not all the possible side effects of Coartem. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Coartem?
Store Coartem between 59ºF to 86ºF (15ºC to 30ºC).
Keep Coartem and all medicines out of the reach of children.
General information about the safe and effective use of Coartem.
Medicines are sometimes prescribed for purposes other than those listed in patient information leaflets. Do not use Coartem for a condition for which it was not prescribed. Do not give Coartem to other people, even if they have the same symptoms that you have. It may harm them.
This patient information leaflet summarizes the most important information about Coartem. If you would like more information about Coartem talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Coartem that is written for health professionals. For more information call 1-855-262-7836.
What are the ingredients in Coartem?
Active ingredients include: artemether, lumefantrine
Inactive ingredients include: colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, polysorbate 80
Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
© Novartis
T2015-45
March 2015 - have heart disease or a family history of heart problems or heart disease
-
PRINCIPAL DISPLAY PANEL - 20/120 mg Tablet Bottle Label
Austin/Travis Co. Health & Human Services Dept.
1000 Toyath St Austin, TX 78703 512-972-6206Coartem
20/120 mg
#24Date:
Name:
Dr.First day, 4 tablet by mouth every 8 hours for 2 doses, then 4 tablet
every 12 hours on days 2 and 302/2018
F0171W1
Coartem 20/120 mg tabs #24 NDC: 76413-154-24
Batch: 08231704
Lot: F0171W1
Exp: 02/2018
NOVARTISFederal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.

-
INGREDIENTS AND APPEARANCE
COARTEM
artemether and lumefantrine tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76413-154(NDC:0078-0568) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARTEMETHER (UNII: C7D6T3H22J) (ARTEMETHER - UNII:C7D6T3H22J) ARTEMETHER 20 mg LUMEFANTRINE (UNII: F38R0JR742) (LUMEFANTRINE - UNII:F38R0JR742) LUMEFANTRINE 120 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYSORBATE 80 (UNII: 6OZP39ZG8H) Product Characteristics Color YELLOW Score 2 pieces Shape ROUND (round and flat tablet with bevelled edges) Size 9mm Flavor Imprint Code N;C;CG Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76413-154-24 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/07/2009 2 NDC: 76413-154-18 18 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/07/2009 3 NDC: 76413-154-12 12 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/07/2009 4 NDC: 76413-154-06 6 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/07/2009 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022268 04/07/2009 Labeler - Central Texas Community Health Centers (079674019) Establishment Name Address ID/FEI Business Operations Central Texas Community Health Centers 079674019 REPACK(76413-154) , RELABEL(76413-154)
Trademark Results [COARTEM]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 COARTEM 78678097 not registered Dead/Abandoned |
Novartis AG 2005-07-26 |
 COARTEM 77646579 3713280 Live/Registered |
Novartis AG 2009-01-09 |
 COARTEM 75345324 not registered Dead/Abandoned |
NOVARTIS AG 1997-08-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.