YUVEZZI- carbachol and brimonidine tartrate solution/ drops
YUVEZZI by
Drug Labeling and Warnings
YUVEZZI by is a Prescription medication manufactured, distributed, or labeled by Visus Therapeutics, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use YUVEZZI™ safely and effectively. See full prescribing information for YUVEZZI.
YUVEZZI (carbachol and brimonidine tartrate ophthalmic solution) 2.75% / 0.1%, for topical ophthalmic use
Initial U.S. Approval: 2026INDICATIONS AND USAGE
YUVEZZI is a combination of carbachol, a cholinergic agonist, and brimonidine, an alpha-adrenergic receptor agonist, indicated for the treatment of presbyopia in adults. ( 1)
DOSAGE AND ADMINISTRATION
Instill one drop in each eye once daily. ( 2.1)
DOSAGE FORMS AND STRENGTHS
Ophthalmic solution: carbachol 2.75% and brimonidine tartrate 0.1% in a single-dose vial. ( 3)
CONTRAINDICATIONS
Hypersensitivity. ( 4)
WARNINGS AND PRECAUTIONS
- Blurred Vision: Advise patients not to drive or operate machinery if vision is not clear (e.g., blurred vision). Exercise caution in night driving and other hazardous activities in poor illumination. ( 5.1)
- Risk of Retinal Detachment: Rare cases of retinal tear and detachment have been reported with miotics. Individuals with preexisting retinal disease are at increased risk. Therefore, examination of the retina is advised in all patients prior to initiation of therapy. Patients should be advised to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss. ( 5.2)
- Iritis: Caution is advised in patients with iritis. ( 5.3)
- Potentiation of Vascular Insufficiency: Use with caution in patients with depression, cerebral or coronary insufficiency, Raynaud's phenomenon, orthostatic hypotension, or thromboangiitis obliterans. ( 5.4)
ADVERSE REACTIONS
Most common adverse reactions (incidence >5%) are eye pain upon instillation, visual impairment, eye irritation upon instillation, and headache. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Visus Therapeutics, Inc., at 1-888-735-0821 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Antihypertensives/cardiac glycosides may lower blood pressure. ( 7.1)
Use with CNS depressants may result in an additive or potentiating effect. ( 7.2)
Tricyclic antidepressants may potentially blunt the hypotensive effect of systemic clonidine. ( 7.3)
Monoamine oxidase inhibitors may result in increased hypotension. ( 7.4)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Blurred Vision
5.2 Risk of Retinal Detachment
5.3 Iritis
5.4 Potentiation of Vascular Insufficiency
5.5 Potential for Eye Injury or Contamination
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Antihypertensives/Cardiac Glycosides
7.2 CNS Depressants
7.3 Tricyclic Antidepressants
7.4 Monoamine Oxidase Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.2 Administration Instructions
Remove contact lenses prior to instillation of YUVEZZI. Wait 10 minutes after dosing before reinserting contact lenses.
If more than one topical ophthalmic medication is being used, the medicines must be administered at least 5 minutes apart.
One single-dose vial can be used to dose both eyes. Discard the open single-dose vial and any remaining contents immediately after use.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Blurred Vision
Miotics may cause accommodative spasm. Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision).
In addition, patients may experience temporary dim or dark vision with miotics. Advise patients to exercise caution in night driving and other hazardous activities in poor illumination.
5.2 Risk of Retinal Detachment
Rare cases of retinal tear and detachment have been reported with miotics. Individuals with pre-existing retinal disease are at increased risk. Examination of the retina is advised in all patients prior to the initiation of therapy.
Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss.
5.3 Iritis
Sequelae of ocular inflammation, i.e., adhesions (synechiae) between the iris and the lens, may be exacerbated with miotic use, therefore, YUVEZZI is not recommended to be used when iritis is present.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in labeling:
- Hypersensitivity [see Contraindications (4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
YUVEZZI was evaluated in 536 patients with presbyopia in two randomized, double-masked, controlled studies. BRIO™ I (NCT05270863) was a single-dose crossover study in which 178 patients received YUVEZZI. BRIO II (NCT05135286) included 358 patients who received YUVEZZI in the long-term safety portion of the study (up to 12 months duration, including a 6-month open-label extension). Adverse reactions reported in > 5% to 7% of participants were eye pain upon instillation and visual impairment. Adverse reactions reported in > 10% to 16% of participants were eye irritation upon instillation and headache. The majority of adverse events were mild, transient, and self-resolving.
-
7 DRUG INTERACTIONS
7.1 Antihypertensives/Cardiac Glycosides
Because brimonidine tartrate, a component of YUVEZZI, may reduce blood pressure, caution in using drugs such as antihypertensives and/or cardiac glycosides with YUVEZZI is advised.
7.2 CNS Depressants
Although specific drug interaction studies have not been conducted with YUVEZZI, the possibility of an additive or potentiating effect with central nervous system (CNS) depressants (alcohol, opiates, barbiturates, sedatives, or anesthetics) should be considered.
7.3 Tricyclic Antidepressants
Tricyclic antidepressants have been reported to blunt the hypotensive effect of systemic clonidine. It is not known whether the concurrent use of these agents with YUVEZZI in humans can lead to resulting interference with pupil constriction effects. Caution is advised in patients taking tricyclic antidepressants, which can affect the metabolism and uptake of circulating amines.
7.4 Monoamine Oxidase Inhibitors
Monoamine oxidase (MAO) inhibitors may theoretically interfere with the metabolism of brimonidine tartrate and potentially result in an increased systemic side effect such as hypotension. Caution is advised in patients taking MAO inhibitors which can affect the metabolism and uptake of circulating amines.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of YUVEZZI administration in pregnant women to inform a drug-associated risk. Topical ocular administration of YUVEZZI to pregnant rats throughout organogenesis did not produce adverse effects up to 3 times the maximum recommended human ocular dose (MRHOD). Topical ocular administration of YUVEZZI to pregnant rabbits throughout organogenesis at approximately 3 times the MRHOD resulted in maternal toxicity with no effect on the fetus, including reduced maternal food consumption, body weight change, and related moribundity in 2/25 animals during the dosing period. Body weight and food consumption effects recovered after the dosing stopped ( see Data).
Because animal reproductive studies are not always predictive of human response, YUVEZZI should be administered during pregnancy only if the potential benefit justifies the potential risk to the pregnant mother and fetus. The background risk of major birth defects and miscarriage for the indicated population is unknown. However, the background risk in the U.S. general population of major birth defects is 2 to 4%, and of miscarriage is 15 to 20%, of clinically recognized pregnancies.
Human Data
No adequate and well-controlled trials of YUVEZZI have been conducted in pregnant women.
Animal Data
YUVEZZI was not teratogenic in embryofetal development studies when given by topical ocular administration to pregnant rats or rabbits throughout organogenesis at dose levels up to approximately 3 times the MRHOD of 1.815 mg/day carbachol and 0.066 mg/day brimonidine tartrate based on body surface area scaling. Topical ocular administration of YUVEZZI to pregnant rats on gestation days (GD) 6 to 17 at dose levels of up to 0.224 mg/day carbachol and 0.008 mg/day brimonidine tartrate (approximately 3 times the MRHOD, based on body surface area) produced no maternal toxicity or embryofetal effects. Topical ocular administration of YUVEZZI to pregnant rabbits on GD 7 to 19 at the dose level of 1.12 mg/day carbachol and 0.04 mg/day brimonidine tartrate (approximately 3 times the MRHOD, based on body surface area scaling) led to maternal toxicity with no embryofetal effects. Maternal findings included significantly reduced food consumption (41%) and body weight change (-87%), with related moribundity in 2/25 animals during the dosing period, compared with the vehicle control group. Body weight and food consumption effects resolved after a 10-day recovery period.
8.2 Lactation
Risk Summary
There is no information regarding the presence of carbachol and brimonidine tartrate in human milk, the effects on the breastfed infants, or the effects on milk production to inform risk of YUVEZZI to an infant during lactation.
Systemic levels of carbachol and brimonidine tartrate following topical ocular administration of YUVEZZI are low [see Clinical Pharmacology (12.3)] , and it is not known whether measurable levels of carbachol and brimonidine tartrate would be present in maternal milk following topical ocular administration.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for YUVEZZI and any potential adverse effects on the breastfed child from YUVEZZI.
-
10 OVERDOSAGE
Systemic toxicity following topical ocular administration of carbachol is rare, but patients who are sensitive may occasionally develop sweating and gastrointestinal overactivity. Accidental ingestion can produce sweating, salivation, nausea, and a decrease in blood pressure. In moderate overdosage, spontaneous recovery is to be expected and is aided by intravenous fluids to compensate for dehydration. For patients demonstrating severe poisoning, use atropine, the pharmacologic antagonist to carbachol.
Very limited information exists on accidental ingestion of brimonidine in adults; the only adverse reaction reported to date has been hypotension. Treatment of an oral overdose includes supportive and symptomatic therapy; a patent airway should be maintained.
-
11 DESCRIPTION
YUVEZZI (carbachol and brimonidine tartrate ophthalmic solution) 2.75% / 0.1% is a fixed-dose combination of carbachol, a cholinergic agonist, and brimonidine, an alpha-adrenergic receptor agonist, for topical ophthalmic use. YUVEZZI is formulated as an isotonic, sterile, clear, greenish-yellow ophthalmic solution. YUVEZZI does not contain an anti-microbial preservative.
The chemical name for carbachol is 2-carbamoyloxyethyl(trimethyl)azanium chloride. The molecular weight is 182.648 g/mol and the molecular formula is C 6H 15ClN 2O 2. Its structural formula is:
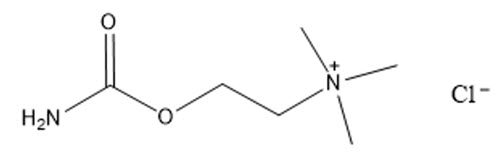
The chemical name for brimonidine tartrate is 5-bromo-N-(4,5-dihydro-1H-imidazol-2-yl)-6-quinoxalinamine L-tartrate. The molecular weight is 442.226 g/mol and the molecular formula is C 15H 16BrN 5O 6. Its structural formula is:
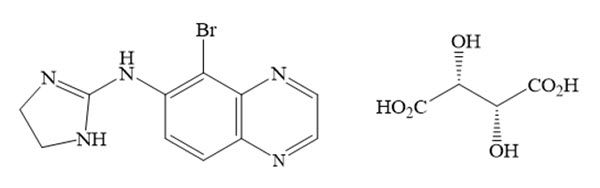
Each mL of YUVEZZI contains carbachol 2.75% (27.5 mg/mL) and brimonidine tartrate 0.1% (1 mg/mL) as the active ingredients. Inactive ingredients are: hydroxypropyl methylcellulose (HPMC), sodium phosphate monobasic monohydrate, sodium phosphate dibasic heptahydrate, water for injection, and may also include hydrochloric acid and/or sodium hydroxide to adjust pH, if necessary.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The proposed mechanism of action of this fixed-dose combination of carbachol and brimonidine in presbyopia is pupillary constriction and the creation of a pinhole effect that improves near visual acuity and depth of focus. Carbachol is a cholinergic agent which produces constriction of the iris sphincter and ciliary body. Brimonidine tartrate, an alpha-adrenergic agonist, blocks contraction of the iris dilator muscle and relaxes tonic contraction of the ciliary muscle, enhancing selectivity for the pupil and increasing bioavailability of carbachol in the aqueous humor.
12.2 Pharmacodynamics
Pupil size was assessed using automated digital pupillometry under mesopic conditions. Average pupil diameter reduction following YUVEZZI was significantly superior to both active monotherapies from pre-dose baseline to Hour 0.5 through Hour 10 in the BRIO I study, and to vehicle from pre-dose baseline to Hour 0.5 through Hour 8 in the BRIO II study.
12.3 Pharmacokinetics
Absorption
Systemic exposures to brimonidine tartrate and carbachol were evaluated in participants with presbyopia who were administered 1 drop of YUVEZZI to each eye. Brimonidine tartrate plasma concentrations were quantifiable in 5 of 14 subjects, with C maxand T maxup to 0.066 ng/mL and 0.52 hours respectively. Carbachol plasma concentrations were quantifiable in 6 of 14 subjects, with C maxand T maxup to 1.02 ng/mL and 4.42 hours respectively.
Distribution
Brimonidine is approximately 29% bound to plasma proteins in healthy subjects. The protein binding of carbachol has not been studied.
Elimination
Metabolism
In humans, brimonidine is extensively metabolized by the liver. Metabolism of carbachol has not been studied.
Excretion
Urinary excretion is the major route of elimination of brimonidine and its metabolites. Approximately 87% of an orally administered radioactive dose of brimonidine was eliminated within 120 hours, with 74% found in the urine. Excretion of carbachol has not been studied.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long term studies in animals have not been performed to evaluate carcinogenic potential of YUVEZZI or carbachol.
No brimonidine tartrate–related carcinogenic effects were observed with dietary administration at dose levels up to 2.5 mg/kg/day in mice for 21 months or 1 mg/kg/day in rats for 24 months.
Mutagenesis
Carbachol was not mutagenic or clastogenic in a battery of in vitro and in vivo studies including the Ames bacterial reverse mutation test, an in vitro micronucleus assay in TK6 cells, and an in vivo rat bone marrow micronucleus assay.
Brimonidine tartrate was not mutagenic or clastogenic in a series of in vitro and in vivo studies, including the Ames bacterial reverse mutation test, a chromosomal aberration assay in Chinese Hamster Ovary (CHO) cells, and 3 in vivo studies in CD-1 mice: a host-mediated assay, cytogenic study, and dominant lethal assay.
Impairment of Fertility
Fertility studies of YUVEZZI or carbachol have not been conducted.
A reproduction and fertility study in rats with brimonidine tartrate demonstrated no adverse effect on male or female fertility at oral doses up to 1 mg/kg.
-
14 CLINICAL STUDIES
The efficacy of YUVEZZI for the treatment of presbyopia was demonstrated in two Phase 3, randomized, double‐masked, controlled studies in participants 45 to 80 years old with presbyopia. BRIO I was a crossover study in which 182 participants received a single administration of 1 drop of YUVEZZI, carbachol, and brimonidine tartrate in each eye with at least 3 days between treatments and was designed to evaluate the superiority of YUVEZZI over either active monotherapy. In BRIO II, 436 participants were randomized for a single administration of 1 drop of YUVEZZI or vehicle (218 to each group) in each eye. In addition, participants administered 1 drop of YUVEZZI or vehicle once daily for 6 months followed by a 6-month open-label extension. Both studies included participants who were post-refractive surgery and/or pseudophakic.
The proportions of participants achieving a gain from baseline of 3 lines (15 letters) or more in binocular uncorrected near visual acuity (BUCNVA) without a loss of 1 line (5 letters) or more in binocular uncorrected distance visual acuity (BUCDVA) under mesopic conditions were evaluated.
Table 1 demonstrates the effect on presbyopia, from 30 minutes post dose to 8 hours.
Table 1. Proportion of participants from BRIO I and BRIO II studies achieving a gain from baseline of 3 lines (15 letters) or more in BUCNVA without a loss of 1 line (5 letters) or more in BUCDVA Timepoint BRIO I BRIO II YUVEZZI
(N=182)Brimonidine tartrate
(N=182)Carbachol
(N=182)YUVEZZI
(N=218)Vehicle
(N=218)P-values are for comparisons to YUVEZZI.
Abbreviations: N: number of participants in the study arm.Hour 0.5 27.0% 10.8%
(p<0.01)24.9%
(p=0.54)19.4% 1.8%
(p<0.01)Hour 1 49.4% 22.7%
(p<0.01)35.0%
(p<0.01)38.2% 3.7%
(p<0.01)Hour 2 51.1% 26.7%
(p<0.01)39.8%
(p<0.01)34.1% 3.7%
(p<0.01)Hour 4 32.0% 22.2%
(p=0.02)21.6%
(p=0.01)20.3% 2.8%
(p<0.01)Hour 6 22.5% 14.8%
(p=0.04)13.7%
(p<0.01)13.8% 2.8%
(p<0.01)Hour 8 13.5% 12.5%
(p=0.73)8.6%
(p=0.10)10.2% 3.2%
(p=0.01) -
16 HOW SUPPLIED/STORAGE AND HANDLING
YUVEZZI (carbachol and brimonidine tartrate ophthalmic solution) 2.75% / 0.1% is supplied as a clear, greenish-yellow ophthalmic solution in a low-density polyethylene (LDPE) single-dose vial, as follows:
Carton box of 30 single-dose vials (6 pouches each containing 5 vials) NDC: 84892-275-30 -
17 PATIENT COUNSELING INFORMATION
Night Driving
Advise patients that they may experience temporary dim or dark vision. Advise patients to exercise caution with night driving and when hazardous activities are undertaken in poor illumination [see Warnings and Precautions (5.1)] .
Blurred Vision
Temporary problems when changing focus between near objects and distant objects may occur. Advise patients not to drive or use machinery if vision is not clear [see Warnings and Precautions (5.1)] .
When to Seek Physician Advice
Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss [see Warnings and Precautions (5.2)] .
Contact Lens Wear
Advise patients to remove contact lenses prior to the instillation of YUVEZZI. Wait 10 minutes after dosing before reinserting contact lenses [see Dosage and Administration (2.2)] .
Potential for Eye Injury or Contamination of the Product
Advise patients to avoid touching the tip of the single-dose vial to the eye, eyelids, or to any other surface to prevent eye injury or contamination [see Warnings and Precautions (5.5)] . Advise patients to discard the opened single-dose vial and any remaining contents immediately after use.
Concomitant Topical Ocular Therapy
Advise patients that if more than one topical ophthalmic medication is being used, the medicines must be administered at least 5 minutes apart [see Dosage and Administration (2.2)] .
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 0.16 mL Vial Pouch Carton
NDC: 84892-275-30
YUVEZZI™
(carbachol and brimonidine tartrate
ophthalmic solution) 2.75%/0.1%Rx Only
For topical ophthalmic use
6 pouches each containing 5 single-dose vials (0.16 mL each)
tenpoint
THERAPEUTICS
-
INGREDIENTS AND APPEARANCE
YUVEZZI
carbachol and brimonidine tartrate solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84892-275 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBACHOL (UNII: 8Y164V895Y) (CARBAMOYLCHOLINE - UNII:54Z8M50D6Q) CARBACHOL 27.5 mg in 1 mL BRIMONIDINE TARTRATE (UNII: 4S9CL2DY2H) (BRIMONIDINE - UNII:E6GNX3HHTE) BRIMONIDINE TARTRATE 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84892-275-30 6 in 1 CARTON 02/18/2026 1 NDC: 84892-275-02 5 in 1 POUCH 1 NDC: 84892-275-01 0.16 mL in 1 VIAL, SINGLE-DOSE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC: 84892-275-99 1 in 1 CARTON 02/18/2026 2 NDC: 84892-275-02 5 in 1 POUCH 2 NDC: 84892-275-01 0.16 mL in 1 VIAL, SINGLE-DOSE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220142 02/18/2026 Labeler - Visus Therapeutics, Inc. (117431652)
Trademark Results [YUVEZZI]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 YUVEZZI 97760492 not registered Live/Pending |
Visus Therapeutics Inc. 2023-01-19 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.