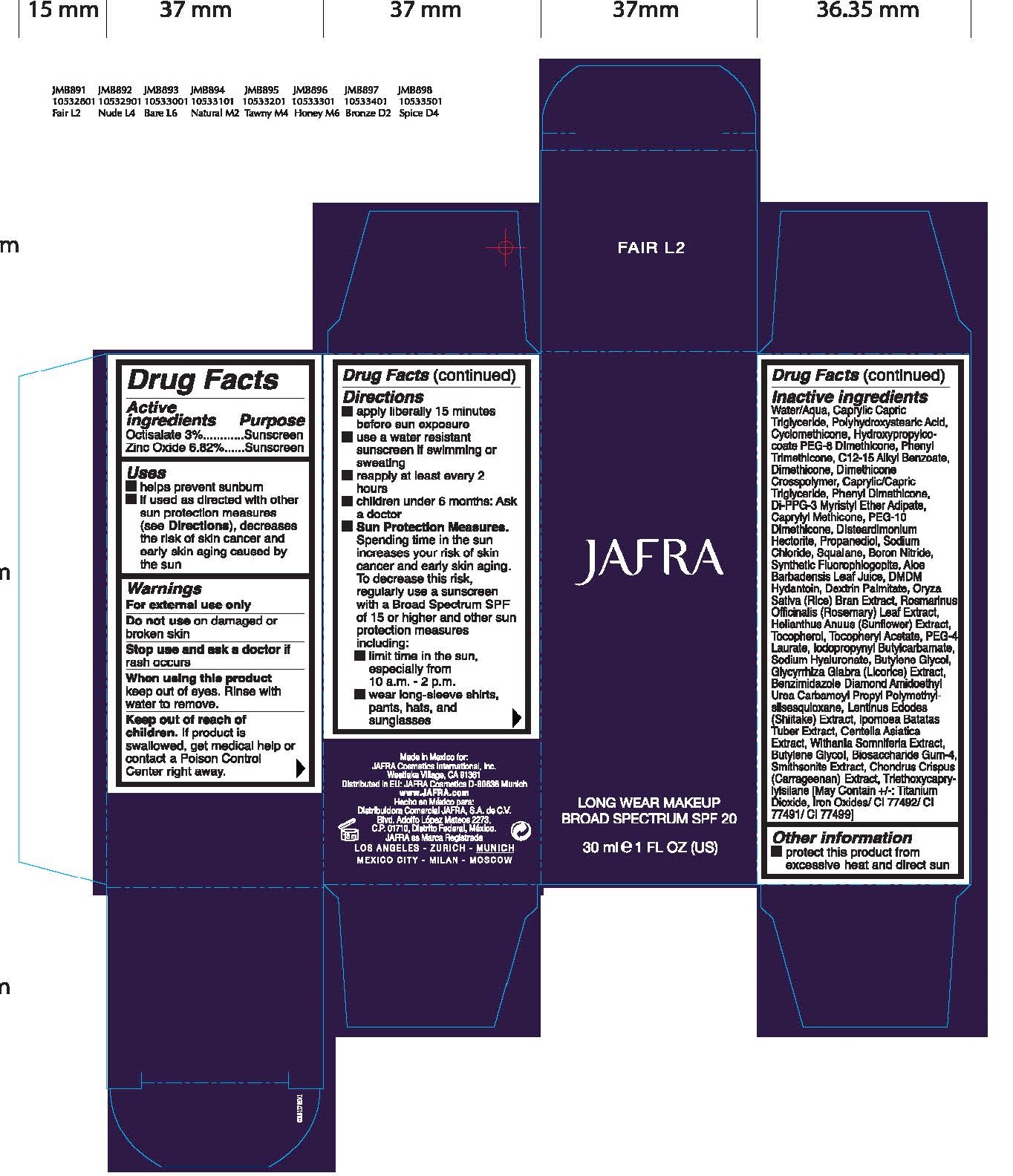
JAFRA LONG WEAR MAKEUP BROAD SPECTRUM SPF 20 FAIR L2
JAFRA LONG WEAR MAKEUP BROAD SPECTRUM SPF 20 FAIR L2 by
Drug Labeling and Warnings
JAFRA LONG WEAR MAKEUP BROAD SPECTRUM SPF 20 FAIR L2 by is a Otc medication manufactured, distributed, or labeled by Absara Cosmetics S.A.P.I DE C.V.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
JAFRA LONG WEAR MAKEUP BROAD SPECTRUM SPF 20 FAIR L2- octisalate, zinc oxide emulsion
Absara Cosmetics S.A.P.I DE C.V.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
JAFRA LONG WEAR MAKEUP BROAD SPECTRUM SPF 20 FAIR L2
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Directions
- apply liberally 15 minutes before use exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. -2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
Inactive ingredients
Water/ Aqua, Caprylic Capric Triglyceride, Polyhydroxystearic Acid, Cyclomethicone, Hydroxypropylcocoate PEG-8 Dimethicone, Phenyl Trimethicone, C12-15 Alkyl Benzoate, Dimethiconem Dimethicone Crosspolymer, Caprylic/Carpic Triglyceride, Phenyl Dimethicone, Di-PPG-3 Myristyl Ether Adipate, Caprylyl Methicone, PEG-10 Dimethicone, Disteardimonium Hectorite, Propanediol, Sodium Chloride, Squalane, Boron Nitride, Synthetic Fluorophlogopite, Aloe Barbadensis Leaf Juice, DMDM Hydantoin, Dextrin Palmitate, Oryza Sativa (Rice) Bran Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Tocopherol, Tocopheryl Acetate, PEG-4 Laurate, Iodopropynyl Butylcarbamate, Sodium Hyaluronate, Butylene Glycol, Glycyrrhiza Glabra (Licoride) Extract, Benzimidazole Diamond Amidoethyl Urea Carbamoyl Propyl Polymethyl Silsesquioxane, Lentinus Edodes (Shiitake) Extract, Ipomoea Batatas Tuber Extract, Centella Asiatica Extract, Withania Somnifera Extract, Butylene Glycol, Biosaccharide Gum-4 Smithsonite Extract, Chondrus Crispus (Carrageenan) Extract, Triethoxycaprylysilane [May Contain +/-: Titanium Dioxide, Iron Oxides/ CI 77492/ CI 77491/ CI 77499]
| JAFRA LONG WEAR MAKEUP BROAD SPECTRUM SPF 20 FAIR L2
octisalate, zinc oxide emulsion |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Absara Cosmetics S.A.P.I DE C.V. (816161236) |