INVOKAMET- canagliflozin and metformin hydrochloride tablet, film coated INVOKAMET XR- canagliflozin and metformin hydrochloride tablet, film coated, extended release
INVOKAMET XR by
Drug Labeling and Warnings
INVOKAMET XR by is a Prescription medication manufactured, distributed, or labeled by Janssen Pharmaceuticals, Inc., Janssen Ortho LLC, Janssen Pharmaceutica NV, Farmhispania, Granules India Limited, Janssen Pharmaceuticals Inc, Ajinomoto OmniChem, Janssen Cilag SpA, Rolabo Outsourcing S.L. (Spain), Johnson & Johnson Private Limited (Mumbai), Patheon Puerto Rico, Inc. (Manati), PHAST Gesellschaft für Pharmazeutische Qualitätsstandards mbH. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use INVOKAMET/INVOKAMET XR safely and effectively. See full prescribing information for INVOKAMET/INVOKAMET XR.
INVOKAMET® (canagliflozin and metformin hydrochloride tablet), for oral use
INVOKAMET® XR (canagliflozin and metformin hydrochloride extended-release tablets), for oral use
Initial U.S. Approval: 2014WARNING: LACTIC ACIDOSIS and LOWER LIMB AMPUTATION
See full prescribing information for complete boxed warning.
Lactic Acidosis
- Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. Symptoms included malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Laboratory abnormalities included elevated blood lactate levels, anion gap acidosis, increased lactate/pyruvate ratio; and metformin plasma levels generally >5 mcg/mL. (5.1)
- Risk factors include renal impairment, concomitant use of certain drugs, age >65 years old, radiological studies with contrast, surgery and other procedures, hypoxic states, excessive alcohol intake, and hepatic impairment. Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the Full Prescribing Information. (5.1)
- If lactic acidosis is suspected, discontinue INVOKAMET/INVOKAMET XR and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended. (5.1)
Risk of Lower Limb Amputation
- In patients with type 2 diabetes who have established cardiovascular disease (CVD) or at risk for CVD, canagliflozin, a component of INVOKAMET/INVOKAMET XR, has been associated with lower limb amputations, most frequently of the toe and midfoot; some also involved the leg. (5.2)
- Before initiating, consider factors that may increase the risk of amputation. Monitor patients receiving INVOKAMET/INVOKAMET XR for infections or ulcers of the lower limbs, and discontinue if these occur. (5.2)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
INVOKAMET and INVOKAMET XR are a combination of canagliflozin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor, and metformin hydrochloride (HCl), a biguanide, indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus (1)
- Canagliflozin is indicated to reduce the risk of major adverse cardiovascular events in adults with type 2 diabetes mellitus and established cardiovascular disease (1)
- Canagliflozin is indicated to reduce the risk of end-stage kidney disease, doubling of serum creatinine, cardiovascular death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria (1).
Limitations of Use:
- Not for treatment of type 1 diabetes mellitus or diabetic ketoacidosis (1)
DOSAGE AND ADMINISTRATION
- Individualize starting dose based on the patient's current regimen and renal function (2.2, 2.3, 2.4)
- Initiation of INVOKAMET or INVOKAMET XR is not recommended in patients with an eGFR less than 45 mL/min/1.73 m2 (2.4)
- INVOKAMET: one tablet, twice daily with meals, recommended starting dose of canagliflozin is 50 mg twice daily and metformin HCl 500 mg twice daily (2.2)
- INVOKAMET XR: two tablets, once daily with the morning meal. Swallow whole. Never crush, cut, or chew (2.2)
- Canagliflozin dose can be increased to a total daily dose of 300 mg in patients tolerating 100 mg who have an eGFR of 60 mL/min/1.73 m2 or greater and require additional glycemic control. Do not exceed a total daily canagliflozin dose of 300 mg (2.2)
- Gradually escalate metformin HCl dose to reduce the gastrointestinal side effects while not exceeding a total daily dose of 2,000 mg (2.1)
- Assess renal function before initiating and periodically thereafter (2.1, 2.3)
- Dose adjustment for patients with renal impairment may be required (2.4)
- Adjust canagliflozin dose when taken with an UDP-Glucuronosyltransferase (UGT) inducer (2.5, 7.1)
- May need to be discontinued at time of, or prior to, iodinated contrast imaging procedures (2.6)
DOSAGE FORMS AND STRENGTHS
INVOKAMET tablets:
- Canagliflozin 50 mg and metformin HCl 500 mg
- Canagliflozin 50 mg and metformin HCl 1,000 mg
- Canagliflozin 150 mg and metformin HCl 500 mg
- Canagliflozin 150 mg and metformin HCl 1,000 mg (3)
INVOKAMET XR extended-release tablets:
- Canagliflozin 50 mg and metformin HCl 500 mg
- Canagliflozin 50 mg and metformin HCl 1,000 mg
- Canagliflozin 150 mg and metformin HCl 500 mg
- Canagliflozin 150 mg and metformin HCl 1,000 mg (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Hypotension: Before initiating, assess volume status and correct hypovolemia in patients with renal impairment, the elderly, in patients with low systolic blood pressure, or on diuretics, ACEi, or ARB. Monitor for signs and symptoms during therapy (5.3)
- Ketoacidosis: Assess patients who present with signs and symptoms of metabolic acidosis for ketoacidosis, regardless of blood glucose level. If suspected, discontinue, evaluate and treat promptly. Before initiating, consider risk factors for ketoacidosis. Patients may require monitoring and temporary discontinuation in clinical situations known to predispose to ketoacidosis (5.4)
- Acute kidney injury: Consider temporarily discontinuing in settings of reduced oral intake or fluid losses. If acute kidney injury occurs, discontinue and promptly treat. Monitor renal function during therapy (5.5)
- Urosepsis and pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated (5.6)
- Hypoglycemia: Consider a lower dose of insulin or insulin secretagogue to reduce the risk of hypoglycemia when used in combination (5.7)
- Necrotizing fasciitis of the perineum (Fournier's gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment (5.8)
- Genital mycotic infections: Monitor and treat if indicated (5.9)
- Hypersensitivity reactions: Discontinue and monitor until signs and symptoms resolve (5.10)
- Bone fracture: Consider factors that contribute to fracture risk before initiating INVOKAMET/INVOKAMET XR (5.11)
- Vitamin B12 deficiency: Metformin HCl may lower vitamin B12 levels. Measure hematological parameters annually and vitamin B12 at 2- to 3-year intervals and manage any abnormalities (5.12)
ADVERSE REACTIONS
- Most common adverse reactions associated with canagliflozin (5% or greater incidence): female genital mycotic infections, urinary tract infection, and increased urination (6.1)
- Most common adverse reactions associated with metformin HCl (5% or greater incidence) are diarrhea, nausea, vomiting, flatulence, asthenia, indigestion, abdominal discomfort, and headache (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-526-7736 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Carbonic anhydrase inhibitors may increase risk of lactic acidosis. Consider more frequent monitoring (7.1)
- Drugs that reduce metformin clearance (such as ranolazine, vandetanib, dolutegravir, and cimetidine) may increase the accumulation of metformin. Consider the benefits and risks of concomitant use (7.1)
- Alcohol: Warn patients against excessive intake (7.1)
- UGT inducers (e.g., rifampin): Canagliflozin exposure is reduced. Adjust canagliflozin dose (2.5, 7.2)
- Digoxin: Monitor digoxin levels (7.2)
USE IN SPECIFIC POPULATIONS
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters (8.1)
- Lactation: Not recommended when breastfeeding (8.2)
- Females and Males of Reproductive Potential: Advise premenopausal females of the potential for an unintended pregnancy (8.3)
- Geriatrics: Higher incidence of adverse reactions related to reduced intravascular volume. Assess renal function more frequently (5.3, 6.1, 8.5)
- Renal impairment: Higher incidence of adverse reactions related to reduced intravascular volume and renal function (2.4, 5.5, 8.6)
- Hepatic Impairment: Avoid use in patients with hepatic impairment (8.7)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 1/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: LACTIC ACIDOSIS and LOWER LIMB AMPUTATION
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Prior to Initiation of INVOKAMET/INVOKAMET XR
2.2 Dosage Overview
2.3 Starting Dose Based on Patient's Current Regimen
2.4 Recommended Dosage for Patients with Renal Impairment
2.5 Concomitant Use with UDP-Glucuronosyltransferase Enzyme Inducers
2.6 Discontinuation for Iodinated Contrast Imaging Procedures
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Lactic Acidosis
5.2 Lower Limb Amputation
5.3 Hypotension
5.4 Ketoacidosis
5.5 Acute Kidney Injury
5.6 Urosepsis and Pyelonephritis
5.7 Hypoglycemia with Concomitant Use of Sulfonylurea or Insulin
5.8 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene)
5.9 Genital Mycotic Infections
5.10 Hypersensitivity Reactions
5.11 Bone Fracture
5.12 Vitamin B12 Levels
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Drug Interactions with Metformin
7.2 Drug Interactions with Canagliflozin
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Glycemic Control Trials in Adults with Type 2 Diabetes Mellitus
14.2 Canagliflozin Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
14.3 Canagliflozin Renal and Cardiovascular Outcomes in Patients with Diabetic Nephropathy and Albuminuria
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: LACTIC ACIDOSIS and LOWER LIMB AMPUTATION
Lactic Acidosis
- Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1)].
- Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment [see Warnings and Precautions (5.1)].
- Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2, 2.3), Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7), and Use in Specific Populations (8.6, 8.7)].
- If metformin-associated lactic acidosis is suspected, immediately discontinue INVOKAMET/INVOKAMET XR and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1)].
Risk of Lower Limb Amputation
- An increased risk of lower limb amputations associated with canagliflozin, a component of INVOKAMET/INVOKAMET XR, versus placebo was observed in CANVAS (5.9 vs 2.8 events per 1000 patient-years) and CANVAS-R (7.5 vs 4.2 events per 1000 patient-years), two large, randomized, placebo-controlled trials in patients with type 2 diabetes who had established cardiovascular disease (CVD) or were at risk for CVD [see Warnings and Precautions (5.2)].
- Amputations of the toe and midfoot were most frequent; however, amputations involving the leg were also observed. Some patients had multiple amputations, some involving both limbs [see Warnings and Precautions (5.2)].
- Before initiating, consider factors that may increase the risk of amputation, such as a history of prior amputation, peripheral vascular disease, neuropathy, and diabetic foot ulcers [see Warnings and Precautions (5.2)].
- Monitor patients receiving INVOKAMET/INVOKAMET XR for infection, new pain or tenderness, sores or ulcers involving the lower limbs, and discontinue if these complications occur [see Warnings and Precautions (5.2)].
-
1 INDICATIONS AND USAGE
INVOKAMET and INVOKAMET XR are a combination of canagliflozin and metformin hydrochloride (HCl) indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Canagliflozin is indicated to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD).
Canagliflozin is indicated to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria > 300 mg/day.
-
2 DOSAGE AND ADMINISTRATION
2.1 Prior to Initiation of INVOKAMET/INVOKAMET XR
- Assess renal function before initiating INVOKAMET/INVOKAMET XR and periodically thereafter [see Warnings and Precautions (5.1, 5.5)], Contraindications (4)].
- In patients with volume depletion not previously treated with canagliflozin, normalize volume status before initiating INVOKAMET/INVOKAMET XR [see Warnings and Precautions (5.3), Use in Specific Populations (8.5, 8.6)].
2.2 Dosage Overview
- INVOKAMET dosing is one tablet, twice daily with meals.
- INVOKAMET XR dosing is two tablets, once daily with the morning meal. Swallow each tablet whole and never crush, cut, or chew.
- The starting and maintenance dose of INVOKAMET/INVOKAMET XR is based on the patient's current regimen and renal function [see Dosage and Administration (2.3, 2.4)].
- The maximum recommended daily dose is canagliflozin 300 mg and metformin HCl 2,000 mg in patients with an estimated glomerular filtration rate (eGFR) of 60 mL/min/1.73 m2 or greater. The recommended dosage for patients with renal impairment is shown in Table 1 [see Dosage and Administration (2.4)].
2.3 Starting Dose Based on Patient's Current Regimen
Individualize the starting dose of INVOKAMET/INVOKAMET XR based on the patient's current regimen and renal function [see Dosage and Administration (2.4)]:
Patients Currently not Treated with Either Canagliflozin or Metformin HCl
- INVOKAMET: One tablet, twice daily, each tablet containing canagliflozin 50 mg and metformin HCl 500 mg;
- INVOKAMET XR: Two tablets, once daily, each tablet containing canagliflozin 50 mg and metformin HCl 500 mg.
Patients on Metformin HCl
- INVOKAMET: One tablet, twice daily, where the total daily dose contains canagliflozin 100 mg and the same, or nearest appropriate, daily dose of metformin HCl;
- INVOKAMET XR: Two tablets, once daily, where the total daily dose contains canagliflozin 100 mg and the patient's same, or nearest appropriate, daily dose of metformin HCl.
Patients on Canagliflozin
- INVOKAMET: One tablet, twice daily, where the total daily dose contains the same daily dose of canagliflozin and metformin HCl 1,000 mg;
- INVOKAMET XR: Two tablets, once daily, where the total daily dose contains the patient's same daily dose of canagliflozin and metformin HCl 1,000 mg.
Patients Already Treated with Canagliflozin and Metformin HCl
- INVOKAMET: One tablet, twice daily, where the total daily dose contains the same daily dose of canagliflozin and the same, or nearest appropriate, daily dose of metformin HCl;
- INVOKAMET XR: Two tablets, once daily, where the total daily dose contains the same daily dose of canagliflozin and the same, or nearest appropriate, daily dose of metformin HCl.
Patients with eGFR of 60 mL/min/1.73 m2 or Greater and Require Additional Glycemic Control
- INVOKAMET: For patients tolerating canagliflozin 50 mg twice daily, increase the dose of canagliflozin to 150 mg twice daily, with gradual metformin HCl dose escalation based on tolerability [see Adverse Reactions (6.1)];
- INVOKAMET XR: For patients tolerating a daily dose of canagliflozin 100 mg, increase the dose to 300 mg once daily, with gradual metformin HCl dose escalation based on tolerability [see Adverse Reactions (6.1)].
2.4 Recommended Dosage for Patients with Renal Impairment
- Initiation of INVOKAMET or INVOKAMET XR is not recommended in patients with an eGFR <45 mL/min/1.73 m2.
- Table 1 has dosage recommendations based on eGFR [see Use in Specific Populations (8.6), Clinical Studies (14.3)].
Table 1: Recommended Dosage estimated Glomerular Filtration Rate
eGFR (mL/min/1.73 m2)Recommended Dosage - * with albuminuria >300 mg/day.
eGFR 60 or greater [See Dosage and Administration (2.3)]. eGFR 45 to less than 60 Limit the dose of canagliflozin component to 100 mg daily (two 50 mg tablets). eGFR 30 to less than 45* Assess the benefit risk of continuing INVOKAMET or INVOKAMET XR; limit the dose of canagliflozin to 100 mg daily (two 50 mg tablets). eGFR less than 30 Contraindicated; discontinue INVOKAMET or INVOKAMET XR [see Contraindications (4)]. On dialysis Contraindicated [see Contraindications]. 2.5 Concomitant Use with UDP-Glucuronosyltransferase Enzyme Inducers
Patients with eGFR 60 mL/min/1.73 m2 or greater
If an inducer of UDP-Glucuronosyltransferase (UGT) (e.g., rifampin, phenytoin, phenobarbital, ritonavir) is co-administered with INVOKAMET/INVOKAMET XR, increase the total daily dose of canagliflozin to 200 mg in patients currently tolerating a total daily dose of canagliflozin 100 mg. The total daily dose of canagliflozin may be increased to a maximum of 300 mg in patients currently tolerating canagliflozin 200 mg and who require additional glycemic control [see Drug Interactions (7.2)].
2.6 Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue INVOKAMET/INVOKAMET XR at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR of less than 60 mL/min/1.73 m2; in patients with a history of liver disease, alcoholism or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart INVOKAMET/INVOKAMET XR if renal function is stable [see Warnings and Precautions (5.1)].
-
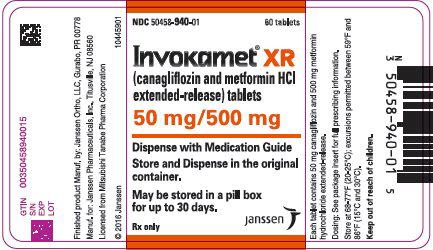
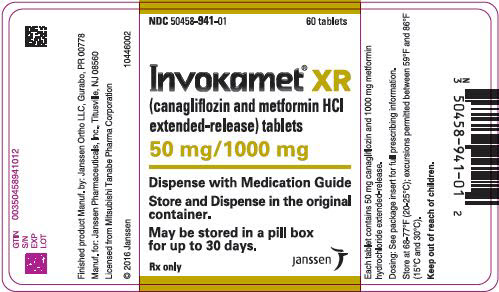
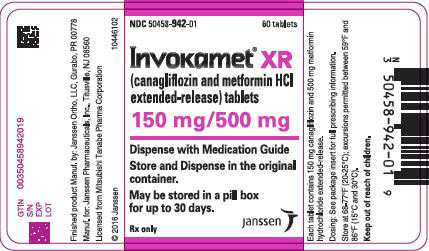
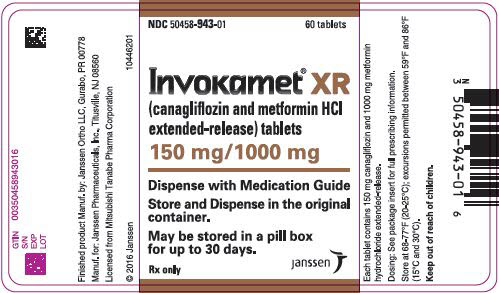
3 DOSAGE FORMS AND STRENGTHS
INVOKAMET (canagliflozin and metformin HCl) tablets are available as follows:
Canagliflozin Strength Metformin HCl Strength Color/Shape Tablet Identifiers* - * Embossing appears on both sides of tablet.
50 mg 500 mg white/capsule-shaped CM 155 50 mg 1,000 mg beige/capsule-shaped CM 551 150 mg 500 mg yellow/capsule-shaped CM 215 150 mg 1,000 mg purple/capsule-shaped CM 611 INVOKAMET XR (canagliflozin and metformin HCl) extended-release tablets are available as follows:
Canagliflozin Strength Metformin HCl Strength Color/Shape Tablet Identifiers* - * Embossing appears on one side only of tablet.
50 mg 500 mg almost white to light orange/oblong, biconvex CM1 50 mg 1,000 mg pink/oblong, biconvex CM3 150 mg 500 mg orange/oblong, biconvex CM2 150 mg 1,000 mg reddish brown/oblong, biconvex CM4 -
4 CONTRAINDICATIONS
INVOKAMET/INVOKAMET XR is contraindicated in patients:
- With severe renal impairment (eGFR less than 30 mL/min/1.73 m2) or on dialysis [see Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
- With acute or chronic metabolic acidosis, including diabetic ketoacidosis [see Warnings and Precautions (5.4)].
- With serious hypersensitivity reaction to canagliflozin or metformin HCl, such as anaphylaxis or angioedema [see Warnings and Precautions (5.10) and Adverse Reactions (6)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Lactic Acidosis
There have been post-marketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension and resistant bradyarrhythmias have occurred with severe acidosis. Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate:pyruvate ratio; metformin plasma levels generally >5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of INVOKAMET/INVOKAMET XR. In INVOKAMET/INVOKAMET XR-treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin is dialyzable, with a clearance of up to 170 mL/minute under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue INVOKAMET/INVOKAMET XR and report these symptoms to their healthcare provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
Renal Impairment: The postmarketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient's renal function include [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
- Before initiating INVOKAMET/INVOKAMET XR, obtain an estimated glomerular filtration rate (eGFR).
- INVOKAMET/INVOKAMET XR is contraindicated in patients with an eGFR less than 30 mL/minute/1.73 m2 [see Contraindications (4)].
- Obtain an eGFR at least annually in all patients taking INVOKAMET/INVOKAMET XR. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
Drug Interactions: The concomitant use of INVOKAMET/INVOKAMET XR with specific drugs may increase the risk of metformin-associated lactic acidosis: those that impair renal function, result in significant hemodynamic change, interfere with acid-base balance or increase metformin accumulation (e.g. cationic drugs) [see Drug Interactions (7)]. Therefore, consider more frequent monitoring of patients.
Age 65 or Greater: The risk of metformin-associated lactic acidosis increases with the patient's age because elderly patients have a greater likelihood of having hepatic, renal, or cardiac impairment than younger patients. Assess renal function more frequently in elderly patients [see Use in Specific Populations (8.5)].
Radiological Studies with Contrast: Administration of intravascular iodinated contrast agents in metformin-treated patients has led to an acute decrease in renal function and the occurrence of lactic acidosis. Stop INVOKAMET/INVOKAMET XR at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR less than 60 mL/min/1.73 m2; in patients with a history of hepatic impairment, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and restart INVOKAMET/INVOKAMET XR if renal function is stable.
Surgery and Other Procedures: Withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension and renal impairment.
INVOKAMET/INVOKAMET XR should be temporarily discontinued while patients have restricted food and fluid intake.
Hypoxic States: Several of the postmarketing cases of metformin-associated lactic acidosis occurred in the setting of acute congestive heart failure (particularly when accompanied by hypoperfusion and hypoxemia). Cardiovascular collapse (shock), acute myocardial infarction, sepsis, and other conditions associated with hypoxemia have been associated with lactic acidosis and may also cause pre-renal azotemia. When such events occur, discontinue INVOKAMET/INVOKAMET XR.
Excessive Alcohol Intake: Alcohol potentiates the effect of metformin on lactate metabolism and this may increase the risk of metformin-associated lactic acidosis. Warn patients against excessive alcohol intake while receiving INVOKAMET/INVOKAMET XR.
Hepatic Impairment: Patients with hepatic impairment have developed metformin-associated lactic acidosis. This may be due to impaired lactate clearance resulting in higher lactate blood levels. Therefore, avoid use of INVOKAMET/INVOKAMET XR in patients with clinical or laboratory evidence of hepatic disease.
5.2 Lower Limb Amputation
An increased risk of lower limb amputations associated with canagliflozin, a component of INVOKAMET/INVOKAMET XR, versus placebo was observed in CANVAS (5.9 vs 2.8 events per 1000 patient-years) and CANVAS-R (7.5 vs 4.2 events per 1000 patient-years), two randomized, placebo-controlled trials evaluating patients with type 2 diabetes who had either established cardiovascular disease or were at risk for cardiovascular disease. The risk of lower limb amputations was observed at both the 100 mg and 300 mg once daily dosage regimens. The amputation data for CANVAS and CANVAS-R are shown in Tables 3 and 4, respectively [see Adverse Reactions (6.1)].
Amputations of the toe and midfoot (99 out of 140 patients with amputations receiving canagliflozin in the two trials) were the most frequent; however, amputations involving the leg, below and above the knee, were also observed (41 out of 140 patients with amputations receiving canagliflozin in the two trials). Some patients had multiple amputations, some involving both lower limbs.
Lower limb infections, gangrene, and diabetic foot ulcers were the most common precipitating medical events leading to the need for an amputation. The risk of amputation was highest in patients with a baseline history of prior amputation, peripheral vascular disease, and neuropathy.
Before initiating INVOKAMET/INVOKAMET XR, consider factors in the patient history that may predispose to the need for amputations, such as a history of prior amputation, peripheral vascular disease, neuropathy and diabetic foot ulcers. Counsel patients about the importance of routine preventative foot care. Monitor patients receiving INVOKAMET/INVOKAMET XR for signs and symptoms of infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and discontinue INVOKAMET/INVOKAMET XR if these complications occur.
5.3 Hypotension
Canagliflozin causes intravascular volume contraction. Symptomatic hypotension can occur after initiating INVOKAMET/INVOKAMET XR [see Adverse Reactions (6.1)] particularly in patients with impaired renal function (eGFR less than 60 mL/min/1.73 m2), elderly patients, patients on either diuretics or medications that interfere with the renin-angiotensin-aldosterone system (e.g., angiotensin-converting-enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs]), or patients with low systolic blood pressure. Before initiating INVOKAMET/INVOKAMET XR in patients with one or more of these characteristics who were not already on canagliflozin, volume status should be assessed and corrected. Monitor for signs and symptoms after initiating therapy.
5.4 Ketoacidosis
Reports of ketoacidosis, a serious life-threatening condition requiring urgent hospitalization have been identified in patients with type 1 and type 2 diabetes mellitus receiving sodium glucose co-transporter-2 (SGLT2) inhibitors, including canagliflozin. Fatal cases of ketoacidosis have been reported in patients taking canagliflozin. INVOKAMET/INVOKAMET XR is not indicated for the treatment of patients with type 1 diabetes mellitus [see Indications and Usage (1)].
Patients treated with INVOKAMET/INVOKAMET XR who present with signs and symptoms consistent with severe metabolic acidosis should be assessed for ketoacidosis regardless of presenting blood glucose levels, as ketoacidosis associated with INVOKAMET/INVOKAMET XR may be present even if blood glucose levels are less than 250 mg/dL. If ketoacidosis is suspected, INVOKAMET/INVOKAMET XR should be discontinued, patient should be evaluated, and prompt treatment should be instituted. Treatment of ketoacidosis may require insulin, fluid and carbohydrate replacement.
In many of the postmarketing reports, and particularly in patients with type 1 diabetes, the presence of ketoacidosis was not immediately recognized and institution of treatment was delayed because presenting blood glucose levels were below those typically expected for diabetic ketoacidosis (often less than 250 mg/dL). Signs and symptoms at presentation were consistent with dehydration and severe metabolic acidosis and included nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. In some but not all cases, factors predisposing to ketoacidosis such as insulin dose reduction, acute febrile illness, reduced caloric intake, surgery, pancreatic disorders suggesting insulin deficiency (e.g., type 1 diabetes, history of pancreatitis or pancreatic surgery), and alcohol abuse were identified.
Before initiating INVOKAMET/INVOKAMET XR consider factors in the patient history that may predispose to ketoacidosis including pancreatic insulin deficiency from any cause, caloric restriction, and alcohol abuse.
For patients who undergo scheduled surgery, consider temporarily discontinuing INVOKAMET/INVOKAMET XR for at least 3 days prior to surgery [see Clinical Pharmacology (12.2, 12.3)].
Consider monitoring for ketoacidosis and temporarily discontinuing INVOKAMET/INVOKAMET XR in other clinical situations known to predispose to ketoacidosis (e.g., prolonged fasting due to acute illness or post-surgery). Ensure risk factors for ketoacidosis are resolved prior to restarting INVOKAMET/INVOKAMET XR.
Educate patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue INVOKAMET/INVOKAMET XR and seek medical attention immediately if signs and symptoms occur.
5.5 Acute Kidney Injury
Canagliflozin causes intravascular volume contraction [see Warnings and Precautions (5.3)] and can cause acute kidney injury. There have been postmarketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients receiving SGLT2 inhibitors, including canagliflozin.
Increases in serum creatinine and decreases in estimated GFR may also be observed with initiation of INVOKAMET/INVOKAMET XR [see Adverse Reactions (6.1) and Clinical Pharmacology (12.1)].
Before initiating INVOKAMET/INVOKAMET XR, consider factors that may predispose patients to acute kidney injury including hypovolemia, chronic renal insufficiency, congestive heart failure, and concomitant medications (diuretics, ACE inhibitors, ARBs, NSAIDs). Consider temporarily discontinuing INVOKAMET/INVOKAMET XR in the setting of reduced oral intake (such as acute illness or fasting) or fluid losses (such as gastrointestinal illness or excessive heat exposure); monitor patients for signs and symptoms of acute kidney injury. If acute kidney injury occurs, discontinue INVOKAMET/INVOKAMET XR promptly and institute treatment.
Renal function should be evaluated prior to initiation of INVOKAMET/INVOKAMET XR and monitored periodically thereafter.
5.6 Urosepsis and Pyelonephritis
There have been postmarketing reports of serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization in patients receiving SGLT2 inhibitors, including canagliflozin. Treatment with SGLT2 inhibitors increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6)].
5.7 Hypoglycemia with Concomitant Use of Sulfonylurea or Insulin
Insulin and insulin secretagogues are known to cause hypoglycemia. INVOKAMET/INVOKAMET XR may increase the risk of hypoglycemia when combined with insulin or an insulin secretagogue [see Adverse Reactions (6.1)]. Therefore, a lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with INVOKAMET/INVOKAMET XR.
5.8 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene)
Reports of necrotizing fasciitis of the perineum (Fournier's gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including canagliflozin. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with INVOKAMET/INVOKAMET XR presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue INVOKAMET/INVOKAMET XR, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
5.9 Genital Mycotic Infections
Canagliflozin increases the risk of genital mycotic infections. Patients with a history of genital mycotic infections and uncircumcised males were more likely to develop genital mycotic infections [see Adverse Reactions (6.1)]. Monitor and treat appropriately.
5.10 Hypersensitivity Reactions
Hypersensitivity reactions, including angioedema and anaphylaxis, have been reported with canagliflozin. These reactions generally occurred within hours to days after initiating canagliflozin. If hypersensitivity reactions occur, discontinue use of INVOKAMET/INVOKAMET XR; treat and monitor until signs and symptoms resolve [see Contraindications (4) and Adverse Reactions (6.1, 6.2)].
5.11 Bone Fracture
An increased risk of bone fracture, occurring as early as 12 weeks after treatment initiation, was observed in patients using canagliflozin in the CANVAS trial [see Clinical Studies (14.2)]. Consider factors that contribute to fracture risk prior to initiating INVOKAMET/INVOKAMET XR [see Adverse Reactions (6.1)].
5.12 Vitamin B12 Levels
In metformin HCl clinical trials of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin HCl or vitamin B12 supplementation. Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. Measure hematologic parameters on an annual basis and vitamin B12 at 2- to 3-year intervals in patients on INVOKAMET/INVOKAMET XR and manage any abnormalities [see Adverse Reactions (6.1)].
-
6 ADVERSE REACTIONS
The following important adverse reactions are also discussed elsewhere in the labeling:
- Lactic Acidosis [see Boxed Warning and Warnings and Precautions (5.1, 5.5)]
- Lower Limb Amputation [see Boxed Warning and Warnings and Precautions (5.2)]
- Hypotension [see Warnings and Precautions (5.3)]
- Ketoacidosis [see Warnings and Precautions (5.4)]
- Acute Kidney Injury [see Warnings and Precautions (5.5)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.6)]
- Hypoglycemia with Concomitant Use of Sulfonylurea or Insulin [see Warnings and Precautions (5.7)]
- Necrotizing Fasciitis of the Perineum (Fournier's gangrene) [see Warnings and Precautions (5.8)]
- Genital Mycotic Infections [see Warnings and Precautions (5.9)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.10)]
- Bone Fracture [see Warnings and Precautions (5.11)]
- Vitamin B12 Deficiency [see Warnings and Precautions (5.12)]
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Pool of Placebo-Controlled Trials for Glycemic Control
Canagliflozin
The data in Table 2 is derived from four 26-week placebo-controlled trials where canagliflozin was used as monotherapy in one trial and as add-on therapy in three trials. These data reflect exposure of 1,667 patients to canagliflozin and a mean duration of exposure to canagliflozin of 24 weeks with 1,275 patients exposed to a combination of canagliflozin and metformin HCl. Patients received canagliflozin 100 mg (N=833), canagliflozin 300 mg (N=834) or placebo (N=646) once daily. The mean daily dose of metformin HCl was 2,138 mg (SD 337.3) for the 1,275 patients in the three placebo-controlled metformin HCl add-on trials. The mean age of the population was 56 years and 2% were older than 75 years of age. Fifty percent (50%) of the population was male and 72% were Caucasian, 12% were Asian, and 5% were Black or African American. At baseline the population had diabetes for an average of 7.3 years, had a mean HbA1C of 8.0% and 20% had established microvascular complications of diabetes. Baseline renal function was normal or mildly impaired (mean eGFR 88 mL/min/1.73 m2).
Table 2 shows common adverse reactions associated with the use of canagliflozin. These adverse reactions were not present at baseline, occurred more commonly on canagliflozin than on placebo, and occurred in at least 2% of patients treated with either canagliflozin 100 mg or canagliflozin 300 mg.
Table 2: Adverse Reactions from Pool of Four 26–Week Placebo-Controlled Studies Reported in ≥ 2% of Canagliflozin-Treated Patients* Adverse Reaction Placebo
N=646Canagliflozin
100 mg
N=833Canagliflozin
300 mg
N=834Note: Percentages were weighted by studies. Study weights were proportional to the harmonic mean of the three treatment sample sizes. - * The four placebo-controlled trials included one monotherapy trial and three add-on combination trials with metformin HCl, metformin HCl and sulfonylurea, or metformin HCl and pioglitazone.
- † Urinary tract infections include the following adverse reactions: Urinary tract infection, Cystitis, Kidney infection, and Urosepsis.
- ‡ Increased urination includes the following adverse reactions: Polyuria, Pollakiuria, Urine output increased, Micturition urgency, and Nocturia.
- § Thirst includes the following adverse reactions: Thirst, Dry mouth, and Polydipsia.
- ¶ Female genital mycotic infections include the following adverse reactions: Vulvovaginal candidiasis, Vulvovaginal mycotic infection, Vulvovaginitis, Vaginal infection, Vulvitis, and Genital infection fungal.
- # Male genital mycotic infections include the following adverse reactions: Balanitis or Balanoposthitis, Balanitis candida, and Genital infection fungal.
Urinary tract infections† 3.8% 5.9% 4.4% Increased urination‡ 0.7% 5.1% 4.6% Thirst§ 0.1% 2.8% 2.4% Constipation 0.9% 1.8% 2.4% Nausea 1.6% 2.1% 2.3% N=312 N=425 N=430 Female genital mycotic infections¶ 2.8% 10.6% 11.6% Vulvovaginal pruritus 0.0% 1.6% 3.2% N=334 N=408 N=404 Male genital mycotic infections# 0.7% 4.2% 3.8% Abdominal pain was also more commonly reported in patients taking canagliflozin 100 mg (1.8%), 300 mg (1.7%) than in patients taking placebo (0.8%).
Canagliflozin and Metformin HCl
The incidence and type of adverse reactions in the three 26-week placebo-controlled metformin HCl tablets add-on trials, representing a majority of data from the four 26-week placebo-controlled trials, was similar to the adverse reactions described in Table 2. There were no additional adverse reactions identified in the pooling of these three placebo-controlled trials that included metformin HCl tablets relative to the four placebo-controlled trials.
In a trial with canagliflozin as initial combination therapy with metformin HCl [see Clinical Studies (14.1)], an increased incidence of diarrhea was observed in the canagliflozin and metformin HCl combination groups (4.2%) compared to canagliflozin or metformin HCl monotherapy groups (1.7%).
Placebo-Controlled Trial in Diabetic Nephropathy
The occurrence of adverse reactions for canagliflozin was evaluated in patients participating in CREDENCE, a study in patients with type 2 diabetes mellitus and diabetic nephropathy with albuminuria ˃ 300 mg/day [see Clinical Studies (14.3)]. These data reflect exposure of 2,201 patients to canagliflozin and a mean duration of exposure to canagliflozin of 137 weeks.
The rate of lower limb amputations associated with the use of canagliflozin 100 mg relative to placebo was 12.3 vs 11.2 events per 1000 patient-years, respectively] in CREDENCE, an outcomes study of patients with type 2 diabetes and diabetic nephropathy, with 2.6 years mean duration of follow-up [see Clinical Studies (14.3)].
In CREDENCE, incidence rates of adjudicated events of diabetic ketoacidosis (DKA) were 0.21 (0.5%, 12/2,200) and 0.03 (0.1%, 2/2,197) per 100 patient-years of follow-up with canagliflozin 100 mg and placebo, respectively [see Warnings and Precautions (5.4)]. The incidence of acute kidney injury was similar between canagliflozin 100 mg and placebo in CREDENCE [see Warnings and Precautions (5.5)].
In CREDENCE, the incidence of hypotension was 2.8% and 1.5% on canagliflozin 100 mg and placebo, respectively [see Warnings and Precautions (5.3)].
Pool of Placebo- and Active-Controlled Trials for Glycemic Control and Cardiovascular Outcomes
The occurrence of adverse reactions for canagliflozin was evaluated in patients participating in placebo- and active-controlled trials and in an integrated analysis of two cardiovascular trials, CANVAS and CANVAS-R.
The types and frequency of common adverse reactions observed in the pool of eight clinical trials (which reflect an exposure of 6,177 patients to canagliflozin) were consistent with those listed in Table 2. Percentages were weighted by studies. Study weights were proportional to the harmonic mean of the three treatment sample sizes. In this pool, canagliflozin was also associated with the adverse reactions of fatigue (1.8%, 2.2%, and 2.0% with comparator, canagliflozin 100 mg, and canagliflozin 300 mg, respectively) and loss of strength or energy (i.e., asthenia) (0.6%, 0.7%, and 1.1% with comparator, canagliflozin 100 mg, and canagliflozin 300 mg, respectively).
In the pool of eight clinical trials, the incidence rate of pancreatitis (acute or chronic) was 0.1%, 0.2%, and 0.1% receiving comparator, canagliflozin 100 mg, and canagliflozin 300 mg, respectively.
In the pool of eight clinical trials, hypersensitivity-related adverse reactions (including erythema, rash, pruritus, urticaria, and angioedema) was 3.0%, 3.8%, and 4.2% of patients receiving comparator, canagliflozin 100 mg, and canagliflozin 300 mg, respectively. Five patients experienced serious adverse reactions of hypersensitivity with canagliflozin, which included 4 patients with urticaria and 1 patient with a diffuse rash and urticaria occurring within hours of exposure to canagliflozin. Among these patients, 2 patients discontinued canagliflozin. One patient with urticaria had recurrence when canagliflozin was re-initiated.
Photosensitivity-related adverse reactions (including photosensitivity reaction, polymorphic light eruption, and sunburn) occurred in 0.1%, 0.2%, and 0.2% of patients receiving comparator, canagliflozin 100 mg, and canagliflozin 300 mg, respectively.
Other adverse reactions occurring more frequently on canagliflozin than on comparator were:
Lower Limb Amputation
An increased risk of lower limb amputations associated with canagliflozin was observed in CANVAS (5.9 vs 2.8 events per 1000 patient-years) and CANVAS-R (7.5 vs 4.2 events per 1000 patient-years), two randomized, placebo-controlled trials evaluating patients with type 2 diabetes who had either established cardiovascular disease or were at risk for cardiovascular disease. Patients in CANVAS and CANVAS-R were followed for an average of 5.7 and 2.1 years, respectively [see Clinical Studies (14.2)]. The amputation data for CANVAS and CANVAS-R are shown in Tables 3 and 4, respectively [see Warnings and Precautions (5.2)].
Table 3: CANVAS Amputations Placebo
N=1441Canagliflozin
100 mg
N=1445Canagliflozin
300 mg
N=1441Canagliflozin
(Pooled)
N=2886Note: Incidence is based on the number of patients with at least one amputation, and not the total number of amputation events. A patient's follow-up is calculated from Day 1 to the first amputation event date. Some patients had more than one amputation. Patients with an amputation, n (%) 22 (1.5) 50 (3.5) 45 (3.1) 95 (3.3) Total amputations 33 83 79 162 Amputation incidence rate
(per 1000 patient-years)2.8 6.2 5.5 5.9 Hazard Ratio (95% CI) -- 2.24 (1.36, 3.69) 2.01 (1.20, 3.34) 2.12 (1.34, 3.38) Table 4: CANVAS-R Amputations Placebo
N=2903Canagliflozin
100 mg
(with up-titration to 300 mg)
N=2904Note: Incidence is based on the number of patients with at least one amputation, and not the total number of amputation events. A patient's follow-up is calculated from Day 1 to the first amputation event date. Some patients had more than one amputation. Patients with an amputation, n (%) 25 (0.9) 45 (1.5) Total amputations 36 59 Amputation incidence rate
(per 1000 patient-years)4.2 7.5 Hazard Ratio (95% CI) -- 1.80 (1.10, 2.93) Renal Cell Carcinoma
In the CANVAS trial (mean duration of follow-up of 5.7 years) [see Clinical Studies (14.2)], the incidence of renal cell carcinoma was 0.15% (2/1331) and 0.29% (8/2716) for placebo and canagliflozin, respectively, excluding patients with less than 6 months of follow-up, less than 90 days of treatment, or a history of renal cell carcinoma. A causal relationship to canagliflozin could not be established due to the limited number of cases.
Volume Depletion-Related Adverse Reactions
Canagliflozin results in an osmotic diuresis, which may lead to reductions in intravascular volume. In clinical trials for glycemic control, treatment with canagliflozin was associated with a dose-dependent increase in the incidence of volume depletion-related adverse reactions (e.g., hypotension, postural dizziness, orthostatic hypotension, syncope, and dehydration). An increased incidence was observed in patients on the 300 mg dose. The three factors associated with the largest increase in volume depletion-related adverse reactions in these trials were the use of loop diuretics, moderate renal impairment (eGFR 30 to less than 60 mL/min/1.73 m2), and age 75 years and older (Table 5) [see Dosage and Administration (2.4), Warnings and Precautions (5.3), and Use in Specific Populations (8.5 and 8.6)].
Table 5: Patients With at Least One Volume Depletion-Related Adverse Reaction (Pooled Results from 8 Clinical Trials for Glycemic Control) Baseline Characteristic Comparator Group*
%Canagliflozin 100 mg
%Canagliflozin 300 mg
%- * Includes placebo and active-comparator groups
- † Patients could have more than 1 of the listed risk factors
Overall population 1.5% 2.3% 3.4% 75 years of age and older† 2.6% 4.9% 8.7% eGFR less than 60 mL/min/1.73 m2† 2.5% 4.7% 8.1% Use of loop diuretic† 4.7% 3.2% 8.8% Falls
In a pool of nine clinical trials with mean duration of exposure to canagliflozin of 85 weeks, the proportion of patients who experienced falls was 1.3%, 1.5%, and 2.1% with comparator, canagliflozin 100 mg, and canagliflozin 300 mg, respectively. The higher risk of falls for patients treated with canagliflozin was observed within the first few weeks of treatment.
Genital Mycotic Infections
In the pool of four placebo-controlled clinical trials for glycemic control, female genital mycotic infections (e.g., vulvovaginal mycotic infection, vulvovaginal candidiasis, and vulvovaginitis) occurred in 2.8%, 10.6%, and 11.6% of females treated with placebo, canagliflozin 100 mg, and canagliflozin 300 mg, respectively. Patients with a history of genital mycotic infections were more likely to develop genital mycotic infections on canagliflozin. Female patients who developed genital mycotic infections on canagliflozin were more likely to experience recurrence and require treatment with oral or topical antifungal agents and anti-microbial agents. In females, discontinuation due to genital mycotic infections occurred in 0% and 0.7% of patients treated with placebo and canagliflozin, respectively [see Warnings and Precautions (5.9)].
In the pool of four placebo-controlled clinical trials, male genital mycotic infections (e.g., candidal balanitis, balanoposthitis) occurred in 0.7%, 4.2%, and 3.8% of males treated with placebo, canagliflozin 100 mg, and canagliflozin 300 mg, respectively. Male genital mycotic infections occurred more commonly in uncircumcised males and in males with a prior history of balanitis or balanoposthitis. Male patients who developed genital mycotic infections on canagliflozin were more likely to experience recurrent infections (22% on canagliflozin versus none on placebo), and require treatment with oral or topical antifungal agents and anti-microbial agents than patients on comparators. In males, discontinuations due to genital mycotic infections occurred in 0% and 0.5% of patients treated with placebo and canagliflozin, respectively.
In the pooled analysis of 8 randomized trials evaluating glycemic control, phimosis was reported in 0.3% of uncircumcised male patients treated with canagliflozin and 0.2% required circumcision to treat the phimosis [see Warnings and Precautions (5.9)].
Hypoglycemia
In canagliflozin glycemic control trials, hypoglycemia was defined as any event regardless of symptoms, where biochemical hypoglycemia was documented (any glucose value below or equal to 70 mg/dL). Severe hypoglycemia was defined as an event consistent with hypoglycemia where the patient required the assistance of another person to recover, lost consciousness, or experienced a seizure (regardless of whether biochemical documentation of a low glucose value was obtained). In individual clinical trials of glycemic control [see Clinical Studies (14.1)], episodes of hypoglycemia occurred at a higher rate when canagliflozin was co-administered with insulin or sulfonylureas (Table 6) [see Warnings and Precautions (5.7)].
Table 6: Incidence of Hypoglycemia* in Randomized Clinical Studies of Glycemic Control - * Number of patients experiencing at least one event of hypoglycemia based on either biochemically documented episodes or severe hypoglycemic events in the intent-to-treat population
- † Severe episodes of hypoglycemia were defined as those where the patient required the assistance of another person to recover, lost consciousness, or experienced a seizure (regardless of whether biochemical documentation of a low glucose value was obtained)
- ‡ Phase 2 clinical study with twice daily dosing (50 mg or 150 mg twice daily in combination with metformin HCl)
- § Subgroup of patients (N=287) from insulin substudy on canagliflozin in combination with metformin HCl and insulin (with or without other antiglycemic agents)
Monotherapy
(26 weeks)Placebo
(N=192)Canagliflozin 100 mg
(N=195)Canagliflozin 300 mg
(N=197)Overall [N (%)] 5 (2.6) 7 (3.6) 6 (3.0) In Combination with Metformin HCl
(26 weeks)Placebo + Metformin HCl
(N=183)Canagliflozin 100 mg + Metformin HCl
(N=368)Canagliflozin 300 mg + Metformin HCl
(N=367)Overall [N (%)] 3 (1.6) 16 (4.3) 17 (4.6) Severe [N (%)]† 0 (0) 1 (0.3) 1 (0.3) In Combination with Metformin HCl
(18 weeks)‡Placebo
(N=93)Canagliflozin 100 mg
(N=93)Canagliflozin 300 mg
(N=93)Overall [N (%)] 3 (3.2) 4 (4.3) 3 (3.2) In Combination with Metformin HCl + Sulfonylurea
(26 weeks)Placebo + Metformin HCl + Sulfonylurea
(N=156)Canagliflozin 100 mg + Metformin HCl + Sulfonylurea
(N=157)Canagliflozin 300 mg + Metformin HCl + Sulfonylurea
(N=156)Overall [N (%)] 24 (15.4) 43 (27.4) 47 (30.1) Severe [N (%)]† 1 (0.6) 1 (0.6) 0 In Combination with Metformin HCl + Pioglitazone
(26 weeks)Placebo + Metformin HCl + Pioglitazone
(N=115)Canagliflozin 100 mg + Metformin HCl + Pioglitazone
(N=113)Canagliflozin 300 mg + Metformin HCl + Pioglitazone
(N=114)Overall [N (%)] 3 (2.6) 3 (2.7) 6 (5.3) In Combination with Insulin
(18 weeks)Placebo
(N=565)Canagliflozin 100 mg
(N=566)Canagliflozin 300 mg
(N=587)Overall [N (%)] 208 (36.8) 279 (49.3) 285 (48.6) Severe [N (%)]† 14 (2.5) 10 (1.8) 16 (2.7) In Combination with Insulin and Metformin HCl (18 weeks)§ Placebo
(N=145)Canagliflozin 100 mg
(N=139)Canagliflozin 300 mg
(N=148)Overall [N (%)] 66 (45.5) 58 (41.7) 70 (47.3) Severe [N (%)]† 4 (2.8) 1 (0.7) 3 (2.0) Bone Fracture
In the CANVAS trial [see Clinical Studies (14.2)], the incidence rates of all adjudicated bone fracture were 1.09, 1.59, and 1.79 events per 100 patient-years of follow-up to placebo, canagliflozin 100 mg, and canagliflozin 300 mg, respectively. The fracture imbalance was observed within the first 26 weeks of therapy and remained through the end of the trial. Fractures were more likely to be low trauma (e.g., fall from no more than standing height), and affect the distal portion of upper and lower extremities [see Warnings and Precautions (5.11)].
Metformin HCl
The most common adverse reactions (5% or greater incidence) due to initiation of metformin HCl are diarrhea, nausea, vomiting, flatulence, asthenia, indigestion, abdominal discomfort, and headache.
Long-term treatment with metformin HCl has been associated with a decrease in vitamin B12, which may result in clinically significant vitamin B12 deficiency (e.g., megaloblastic anemia) [see Warnings and Precautions (5.12)].
Laboratory and Imaging Tests
Increases in Serum Creatinine and Decreases in eGFR
Initiation of canagliflozin causes an increase in serum creatinine and decrease in estimated GFR. In patients with moderate renal impairment, the increase in serum creatinine generally does not exceed 0.2 mg/dL, occurs within the first 6 weeks of starting therapy, and then stabilizes. Increases that do not fit this pattern should prompt further evaluation to exclude the possibility of acute kidney injury [see Warnings and Precautions (5.5) and Mechanism of Action (12.1)]. The acute effect on eGFR reverses after treatment discontinuation suggesting acute hemodynamic changes may play a role in the renal function changes observed with canagliflozin.
Increases in Serum Potassium
In a pooled population of patients (N=723) in glycemic control trials with moderate renal impairment (eGFR 45 to less than 60 mL/min/1.73 m2), increases in serum potassium to greater than 5.4 mEq/L and 15% above occurred in 5.3%, 5.0%, and 8.8% of patients treated with placebo, canagliflozin 100 mg, and canagliflozin 300 mg, respectively. Severe elevations (greater than or equal to 6.5 mEq/L) occurred in 0.4% of patients treated with placebo, no patients treated with canagliflozin 100 mg, and 1.3% of patients treated with canagliflozin 300 mg.
In these patients, increases in potassium were more commonly seen in those with elevated potassium at baseline. Among patients with moderate renal impairment, approximately 84% were taking medications that interfere with potassium excretion, such as potassium-sparing diuretics, angiotensin-converting-enzyme inhibitors, and angiotensin-receptor blockers [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)].
In CREDENCE, no difference in serum potassium, no increase in adverse events of hyperkalemia, and no increase in absolute (> 6.5 mEq/L) or relative (> upper limit of normal and > 15% increase from baseline) increases in serum potassium were observed with canagliflozin 100 mg relative to placebo.
Increases in Low-Density Lipoprotein Cholesterol (LDL-C) and non-High-Density Lipoprotein Cholesterol (non-HDL-C)
In the pool of four glycemic control placebo-controlled trials, dose-related increases in LDL-C with canagliflozin were observed. Mean changes (percent changes) from baseline in LDL-C relative to placebo were 4.4 mg/dL (4.5%) and 8.2 mg/dL (8.0%) with canagliflozin 100 mg and canagliflozin 300 mg, respectively. The mean baseline LDL-C levels were 104 to 110 mg/dL across treatment groups.
Dose-related increases in non-HDL-C with canagliflozin were observed. Mean changes (percent changes) from baseline in non-HDL-C relative to placebo were 2.1 mg/dL (1.5%) and 5.1 mg/dL (3.6%) with canagliflozin 100 mg and 300 mg, respectively. The mean baseline non-HDL-C levels were 140 to 147 mg/dL across treatment groups.
Increases in Hemoglobin
In the pool of four placebo-controlled trials of glycemic control, mean changes (percent changes) from baseline in hemoglobin were -0.18 g/dL (-1.1%) with placebo, 0.47 g/dL (3.5%) with canagliflozin 100 mg, and 0.51 g/dL (3.8%) with canagliflozin 300 mg. The mean baseline hemoglobin value was approximately 14.1 g/dL across treatment groups. At the end of treatment, 0.8%, 4.0%, and 2.7% of patients treated with placebo, canagliflozin 100 mg, and canagliflozin 300 mg, respectively, had hemoglobin above the upper limit of normal.
Decreases in Bone Mineral Density
Bone mineral density (BMD) was measured by dual-energy X-ray absorptiometry in a clinical trial of 714 older adults (mean age 64 years). At 2 years, patients randomized to canagliflozin 100 mg and canagliflozin 300 mg had placebo-corrected declines in BMD at the total hip of 0.9% and 1.2%, respectively, and at the lumbar spine of 0.3% and 0.7%, respectively. Additionally, placebo-adjusted BMD declines were 0.1% at the femoral neck for both canagliflozin doses and 0.4% at the distal forearm for patients randomized to canagliflozin 300 mg. The placebo-adjusted change at the distal forearm for patients randomized to canagliflozin 100 mg was 0%.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of canagliflozin. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Canagliflozin
Ketoacidosis
Acute Kidney Injury
Anaphylaxis, Angioedema
Urosepsis and Pyelonephritis
Necrotizing Fasciitis of the Perineum (Fournier's gangrene)
Metformin HCl
Cholestatic, hepatocellular, and mixed hepatocellular liver injury
-
7 DRUG INTERACTIONS
7.1 Drug Interactions with Metformin
Table 7: Clinically Significant Drug Interactions with Metformin Carbonic Anhydrase Inhibitors Clinical Impact: Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with INVOKAMET/INVOKAMET XR may increase the risk for lactic acidosis. Intervention: Consider more frequent monitoring of these patients. Examples: Topiramate or other carbonic anhydrase inhibitors (e.g., zonisamide, acetazolamide or dichlorphenamide) Drugs That Reduce Metformin Clearance Clinical Impact: Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3)]. Intervention: Consider the benefits and risks of concomitant use. Examples: Ranolazine, vandetanib, dolutegravir, and cimetidine Alcohol Clinical Impact: Alcohol is known to potentiate the effect of metformin HCl on lactate metabolism. Intervention: Warn patients against excessive alcohol intake while receiving INVOKAMET/INVOKAMET XR. Insulin Secretagogues or Insulin Clinical Impact: Coadministration of INVOKAMET/INVOKAMET XR with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia. Intervention: Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin. Drugs Affecting Glycemic Control Clinical Impact: Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. Intervention: When such drugs are administered to a patient receiving INVOKAMET/INVOKAMET XR, monitor for loss of blood glucose control. When such drugs are withdrawn from a patient receiving INVOKAMET/INVOKAMET XR, monitor for hypoglycemia. Examples: Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid. 7.2 Drug Interactions with Canagliflozin
Table 8: Clinically Significant Drug Interactions with Canagliflozin UGT Enzyme Inducers Clinical Impact: May reduce the efficacy of INVOKAMET/INVOKAMET XR. Intervention: For patients with eGFR 60 mL/min/1.73 m2 or greater, if an inducer of UGTs is co-administered with INVOKAMET/INVOKAMET XR, increase the total daily dose of canagliflozin to 200 mg in patients currently tolerating INVOKAMET/INVOKAMET XR with a total daily dose of canagliflozin 100 mg. The total daily dose of canagliflozin may be increased to 300 mg in patients currently tolerating canagliflozin 200 mg and who require additional glycemic control.
For patients with eGFR less than 60 mL/min/1.73 m2, if an inducer of UGTs is co-administered with INVOKAMET/INVOKAMET XR, increase the total daily dose of canagliflozin to 200 mg in patients currently tolerating canagliflozin 100 mg [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].Examples: Rifampin, phenytoin, phenobarbital, ritonavir Digoxin Clinical Impact: Canagliflozin increased digoxin exposure [see Clinical Pharmacology (12.3)]. Intervention: Monitor patients taking INVOKAMET/INVOKAMET XR with concomitant digoxin for a need to adjust dose of digoxin. Drug/Laboratory Test Interference Positive Urine Glucose Test Intervention: Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors as SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Intervention: Monitoring glycemic control with 1,5-AG assay is not recommended as measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on animal data showing adverse renal effects from canagliflozin, INVOKAMET/INVOKAMET XR is not recommended during the second and third trimesters of pregnancy.
Limited data with INVOKAMET, INVOKAMET XR or canagliflozin in pregnant women are not sufficient to determine a drug-associated risk for major birth defects or miscarriage. Published studies with metformin HCl use during pregnancy have not reported a clear association with metformin HCl and major birth defect or miscarriage risk (see Data). There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy (see Clinical Considerations).
In animal studies, adverse renal pelvic and tubule dilatations that were not reversible were observed in rats when canagliflozin was administered at an exposure 0.5-times the 300 mg clinical dose, based on AUC during a period of renal development corresponding to the late second and third trimesters of human pregnancy. No adverse developmental effects were observed when metformin HCl was administered to pregnant Sprague Dawley rats and rabbits during the period of organogenesis at doses up to 2- and 6-times, respectively, a 2000 mg clinical dose, based on body surface area (see Data).
The estimated background risk of major birth defects is 6–10% in women with pre-gestational diabetes with an HbA1C >7 and has been reported to be as high as 20–25% in women with a HbA1C >10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
Data
Human Data
Published data from post-marketing studies have not reported a clear association with metformin HCl and major birth defects, miscarriage, or adverse maternal or fetal outcomes when metformin HCl was used during pregnancy. However, these studies cannot definitely establish the absence of any metformin-associated risk because of methodological limitations, including small sample size and inconsistent comparator groups.
Animal Data
Canagliflozin
Canagliflozin dosed directly to juvenile rats from postnatal day (PND) 21 until PND 90 at doses of 4, 20, 65, or 100 mg/kg increased kidney weights and dose dependently increased the incidence and severity of renal pelvic and tubular dilatation at all doses tested. Exposure at the lowest dose was greater than or equal to 0.5-times the 300 mg clinical dose, based on AUC. These outcomes occurred with drug exposure during periods of renal development in rats that correspond to the late second and third trimester of human renal development. The renal pelvic dilatations observed in juvenile animals did not fully reverse within a 1 month recovery period.
In embryo-fetal development studies in rats and rabbits, canagliflozin was administered for intervals coinciding with the first trimester period of organogenesis in humans. No developmental toxicities independent of maternal toxicity were observed when canagliflozin was administered at doses up to 100 mg/kg in pregnant rats and 160 mg/kg in pregnant rabbits during embryonic organogenesis or during a study in which maternal rats were dosed from gestation day (GD) 6 through PND 21, yielding exposures up to approximately 19-times the 300 mg clinical dose, based on AUC.
Metformin HCl
Metformin HCl did not cause adverse developmental effects when administered to pregnant Sprague Dawley rats and rabbits up to 600 mg/kg/day during the period of organogenesis. This represents an exposure of about 2- and 6-times a 2000 mg clinical dose based on body surface area (mg/m2) for rats and rabbits, respectively.
Canagliflozin and Metformin HCl
No adverse developmental effects were observed when canagliflozin and metformin HCl were co-administered to pregnant rats during the period of organogenesis at exposures up to 11 and 13 times, respectively, the 300 mg and 2000 mg clinical doses of canagliflozin and metformin HCl based on AUC.
8.2 Lactation
Risk Summary
There is no information regarding the presence of INVOKAMET, INVOKAMET XR or canagliflozin in human milk, the effects on the breastfed infant, or the effects on milk production. Limited published studies report that metformin is present in human milk (see Data). However, there is insufficient information on the effects of metformin HCl on the breastfed infant and no available information on the effects of metformin HCl on milk production. Canagliflozin is present in the milk of lactating rats (see Data). Since human kidney maturation occurs in utero and during the first 2 years of life when lactational exposure may occur, there may be risk to the developing human kidney.
Because of the potential for serious adverse reactions in a breastfed infant, advise women that use of INVOKAMET/INVOKAMET XR is not recommended while breastfeeding.
Data
Published clinical lactation studies report that metformin is present in human milk which resulted in infant doses approximately 0.11% to 1% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 0.13 and 1. However, the studies were not designed to definitely establish the risk of use of metformin HCl during lactation because of small sample size and limited adverse event data collected in infants.
Radiolabeled canagliflozin administered to lactating rats on day 13 post-partum was present at a milk/plasma ratio of 1.40, indicating that canagliflozin and its metabolites are transferred into milk at a concentration comparable to that in plasma. Juvenile rats directly exposed to canagliflozin showed a risk to the developing kidney (renal pelvic and tubular dilatations) during maturation.
8.3 Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as therapy with metformin HCl may result in ovulation in some anovulatory women.
8.4 Pediatric Use
Safety and effectiveness of INVOKAMET/INVOKAMET XR in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
INVOKAMET and INVOKAMET XR
Because renal function abnormalities can occur after initiating canagliflozin, metformin is substantially excreted by the kidney, and aging can be associated with reduced renal function, monitor renal function more frequently after initiating INVOKAMET/INVOKAMET XR in the elderly and then adjust dose based on renal function [see Dosage and Administration (2.4) and Warnings and Precautions (5.1, 5.5)].
Canagliflozin
In 13 clinical trials of canagliflozin, 2,294 patients 65 years and older, and 351 patients 75 years and older were exposed to canagliflozin. Of these patients, 1,534 patients 65 years and older and 196 patients 75 years and older were exposed to the combination of canagliflozin and metformin HCl [see Clinical Studies (14)]. Patients 65 years and older had a higher incidence of adverse reactions related to reduced intravascular volume with canagliflozin (such as hypotension, postural dizziness, orthostatic hypotension, syncope, and dehydration), particularly with the 300 mg daily dose, compared to younger patients; a more prominent increase in the incidence was seen in patients who were 75 years and older [see Dosage and Administration (2.1) and Adverse Reactions (6.1)]. Smaller reductions in HbA1C with canagliflozin relative to placebo were seen in older (65 years and older; -0.61% with canagliflozin 100 mg and -0.74% with canagliflozin 300 mg relative to placebo) compared to younger patients (-0.72% with canagliflozin 100 mg and -0.87% with canagliflozin 300 mg relative to placebo).
Metformin HCl
Controlled clinical trials of metformin HCl did not include sufficient numbers of elderly patients to determine whether they respond differently from younger patients, although other reported clinical experience has not identified differences in responses between the elderly and younger patients. The initial and maintenance dosing of metformin HCl should be conservative in patients with advanced age due to the potential for decreased renal function in this population. Any dose adjustment should be based on a careful assessment of renal function [see Contraindications (4), Warnings and Precautions (5.5), and Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Canagliflozin
The efficacy and safety of canagliflozin for glycemic control were evaluated in a trial that included patients with moderate renal impairment (eGFR 30 to less than 50 mL/min/1.73 m2) [see Clinical Studies (14.1)]. These patients had less overall glycemic efficacy, and patients treated with canagliflozin 300 mg per day had increases in serum potassium, which were transient and similar by the end of study. Patients with renal impairment using canagliflozin for glycemic control may also be more likely to experience hypotension and may be at higher risk for acute kidney injury [see Warnings and Precautions (5.5)].
Efficacy and safety studies with canagliflozin did not enroll patients with ESKD on dialysis or patients with an eGFR less than 30 mL/min/1.73 m2. Canagliflozin is contraindicated in patients with ESKD on dialysis and in patients with severe renal impairment (eGFR less than 30 mL/min/1.73 m2) who are being treated for glycemic control [see Contraindications (4) and Clinical Pharmacology (12.1)].
Metformin HCl
Metformin is substantially excreted by the kidney, and the risk of metformin accumulation and lactic acidosis increases with the degree of renal impairment. INVOKAMET/INVOKAMET XR is contraindicated in severe renal impairment (eGFR less than 30 mL/min/1.73 m2) or in patients on dialysis [see Dosage and Administration (2.4), Contraindications (4), Warnings and Precautions (5.1), and Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Use of metformin HCl in patients with hepatic impairment has been associated with some cases of lactic acidosis. INVOKAMET/INVOKAMET XR is not recommended in patients with hepatic impairment [see Warnings and Precautions (5.1)].
-
10 OVERDOSAGE
Overdose of metformin HCl has occurred, including ingestion of amounts greater than 50 grams. Hypoglycemia was reported in approximately 10% of cases, but no causal association with metformin HCl use has been established. Lactic acidosis has been reported in approximately 32% of metformin HCl overdose cases [see Warnings and Precautions (5.1)].
In the event of an overdose with INVOKAMET/INVOKAMET XR, contact the Poison Control Center. Employ the usual supportive measures (e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring, and institute supportive treatment) as dictated by the patient's clinical status. Canagliflozin was negligibly removed during a 4-hour hemodialysis session. Canagliflozin is not expected to be dialyzable by peritoneal dialysis. Metformin is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions. Therefore, hemodialysis may be useful partly for removal of accumulated metformin from patients in whom INVOKAMET/INVOKAMET XR overdosage is suspected.
-
11 DESCRIPTION
INVOKAMET® (canagliflozin and metformin HCl) and INVOKAMET® XR (canagliflozin and metformin hydrochloride extended-release tablets) contain canagliflozin and metformin HCl.
Canagliflozin
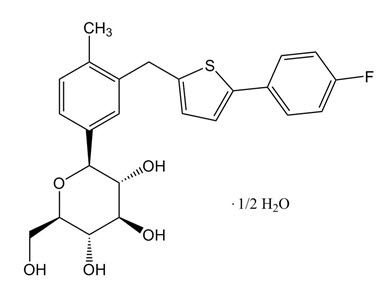
Canagliflozin is an inhibitor of sodium-glucose co-transporter 2 (SGLT2), the transporter responsible for reabsorbing the majority of glucose filtered by the kidney. Canagliflozin is chemically known as (1S)-1,5-anhydro-1-[3-[[5-(4-fluorophenyl)-2-thienyl]methyl]-4-methylphenyl]-D-glucitol hemihydrate and its molecular formula and weight are C24H25FO5S∙1/2 H2O and 453.53, respectively. The structural formula for canagliflozin is:
Canagliflozin is practically insoluble in aqueous media from pH 1.1 to 12.9.
Metformin HCl
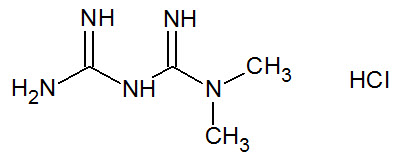
Metformin HCl is a biguanide chemically known as 1,1-Dimethylbiguanide hydrochloride and its molecular formula and weight are C4H11N5 ∙ HCl and 165.62, respectively. The structural formula for metformin HCl is:
INVOKAMET and INVOKAMET XR
- INVOKAMET/INVOKAMET XR are supplied as film-coated tablets for oral administration. Each 50 mg/500 mg tablet and 50 mg/1,000 mg tablet contains 51 mg of canagliflozin equivalent to 50 mg canagliflozin (anhydrous) and 500 mg or 1,000 mg metformin HCl (equivalent to metformin 389.93 mg and 779.86 mg, respectively).
- Each 150 mg/500 mg tablet and 150 mg/1,000 mg tablet contains 153 mg of canagliflozin equivalent to 150 mg canagliflozin (anhydrous) and 500 mg or 1,000 mg metformin HCl (equivalent to metformin 389.93 mg and 779.86 mg, respectively).
INVOKAMET contains the following inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate, and microcrystalline cellulose. The magnesium stearate is vegetable-sourced. The tablets are finished with a commercially available film-coating consisting of the following inactive ingredients: macrogol/PEG3350, polyvinyl alcohol (partially hydrolyzed), talc, titanium dioxide, iron oxide yellow (50 mg/1,000 mg and 150 mg/500 mg tablets only), iron oxide red (50 mg/1,000 mg, 150 mg/500 mg and 150 mg/1,000 mg tablets only), and iron oxide black (150 mg/1,000 mg tablets only).
INVOKAMET XR contains the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, hypromellose, lactose anhydrous, magnesium stearate (vegetable-sourced), microcrystalline cellulose, polyethylene oxide, and silicified microcrystalline cellulose (50 mg/500 mg and 50 mg/1,000 mg tablets only). The tablets are finished with a commercially available film-coating consisting of the following inactive ingredients: macrogol/PEG3350, polyvinyl alcohol (partially hydrolyzed), talc, titanium dioxide, iron oxide red, iron oxide yellow, and iron oxide black (50 mg/1,000 mg and 150 mg/1,000 mg tablets only).
INVOKAMET XR tablets provide canagliflozin for immediate-release and metformin HCl for extended-release. Each bilayer tablet is compressed from two separate granulates, one for each active ingredient of the tablet, and finished with a film-coating. The metformin HCl extended-release layer is based on a polymer matrix which controls the drug release by passive diffusion through the swollen matrix in combination with tablet erosion.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Canagliflozin
Sodium-glucose co-transporter 2 (SGLT2), expressed in the proximal renal tubules, is responsible for the majority of the reabsorption of filtered glucose from the tubular lumen. Canagliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, canagliflozin reduces reabsorption of filtered glucose and lowers the renal threshold for glucose (RTG), and thereby increases urinary glucose excretion (UGE).
Canagliflozin increases the delivery of sodium to the distal tubule by blocking SGLT2-dependent glucose and sodium reabsorption. This is believed to increase tubuloglomerular feedback and reduce intraglomerular pressure.
Metformin HCl
Metformin HCl is an antihyperglycemic agent which improves glucose tolerance in patients with type 2 diabetes, lowering both basal and postprandial plasma glucose. Metformin HCl decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may decrease.
12.2 Pharmacodynamics
Canagliflozin
Following single and multiple oral doses of canagliflozin in patients with type 2 diabetes, dose-dependent decreases in RTG and increases in urinary glucose excretion were observed. From a starting RTG value of approximately 240 mg/dL, canagliflozin at 100 mg and 300 mg once daily suppressed RTG throughout the 24-hour period. Data from single oral doses of canagliflozin in healthy volunteers indicate that, on average, the elevation in urinary glucose excretion approaches baseline by about 3 days for doses up to 300 mg once daily. Maximal suppression of mean RTG over the 24-hour period was seen with the 300 mg daily dose to approximately 70 to 90 mg/dL in patients with type 2 diabetes in Phase 1 trials. The reductions in RTG led to increases in mean UGE of approximately 100 g/day in patients with type 2 diabetes treated with either 100 mg or 300 mg of canagliflozin. The 24-h mean RTG at steady state was similar following once daily and twice daily dosing regimens at the same total daily dose of 100 mg or 300 mg. In patients with type 2 diabetes given 100 to 300 mg once daily over a 16-day dosing period, reductions in RTG and increases in urinary glucose excretion were observed over the dosing period. In this trial, plasma glucose declined in a dose-dependent fashion within the first day of dosing.
Cardiac Electrophysiology
In a randomized, double-blind, placebo-controlled, active-comparator, 4-way crossover trial, 60 healthy subjects were administered a single oral dose of canagliflozin 300 mg, canagliflozin 1,200 mg (4 times the maximum recommended dose), moxifloxacin, and placebo. No meaningful changes in QTc interval were observed with either the recommended dose of 300 mg or the 1,200 mg dose.
12.3 Pharmacokinetics
INVOKAMET
Administration of INVOKAMET 150 mg/1,000 mg fixed-dose combination with food resulted in no change in overall exposure of canagliflozin. There was no change in metformin AUC; however, the mean peak plasma concentration of metformin was decreased by 16% when administered with food. A delayed time to peak plasma concentration was observed for both components (a delay of 2 hours for canagliflozin and 1 hour for metformin) under fed conditions. These changes are not likely to be clinically meaningful.
INVOKAMET XR
After administration of INVOKAMET XR tablets with a high-fat breakfast, the peak (Cmax) and total (AUC) exposure of canagliflozin were not altered relative to dosing in the fasted state. However, the AUC of metformin increased by approximately 61% and Cmax increased by approximately 13%.
Canagliflozin
The pharmacokinetics of canagliflozin is essentially similar in healthy subjects and patients with type 2 diabetes. Following single-dose oral administration of 100 mg and 300 mg of canagliflozin, peak plasma concentrations (median Tmax) of canagliflozin occurs within 1 to 2 hours post-dose. Plasma Cmax and AUC of canagliflozin increased in a dose-proportional manner from 50 mg to 300 mg. The apparent terminal half-life (t1/2) was 10.6 hours and 13.1 hours for the 100 mg and 300 mg doses, respectively. Steady-state was reached after 4 to 5 days of once-daily dosing with canagliflozin 100 mg to 300 mg. Canagliflozin does not exhibit time-dependent pharmacokinetics and accumulated in plasma up to 36% following multiple doses of 100 mg and 300 mg. The mean systemic exposure (AUC) at steady state was similar following once daily and twice daily dosing regimens at the same total daily dose of 100 mg or 300 mg.
Absorption
Metformin
The absolute bioavailability of a metformin HCl 500 mg tablet given under fasting conditions is approximately 50% to 60%. Trials using single oral doses of metformin HCl 500 to 1,500 mg, and 850 to 2,550 mg, indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an alteration in elimination.
Following a single oral dose of 1,000 mg metformin HCl extended-release tablets (two 500 mg tablets) after a meal, the time to reach maximum plasma metformin concentration (Tmax) is achieved at approximately 7–8 hours. In both single and multiple-dose trials in healthy subjects, once daily 1,000 mg (two 500 mg tablets) dosing results in up to 35% higher Cmax, of metformin relative to the immediate-release given as 500 mg twice daily without any change in overall systemic exposure, as measured by AUC.
Distribution
Canagliflozin
The mean steady-state volume of distribution of canagliflozin following a single intravenous infusion in healthy subjects was 83.5 L, suggesting extensive tissue distribution. Canagliflozin is extensively bound to proteins in plasma (99%), mainly to albumin. Protein binding is independent of canagliflozin plasma concentrations. Plasma protein binding is not meaningfully altered in patients with renal or hepatic impairment.
Metformin
The apparent volume of distribution (V/F) of metformin following single oral doses of metformin HCl 850 mg immediate-release tablets averaged 654 ± 358 L. Metformin is negligibly bound to plasma proteins, in contrast to sulfonylureas, which are more than 90% protein bound. Metformin partitions into erythrocytes, most likely as a function of time.
Metabolism
Excretion
Canagliflozin
Following administration of a single oral [14C] canagliflozin dose to healthy subjects, 41.5%, 7.0%, and 3.2% of the administered radioactive dose was recovered in feces as canagliflozin, a hydroxylated metabolite, and an O-glucuronide metabolite, respectively. Enterohepatic circulation of canagliflozin was negligible.
Approximately 33% of the administered radioactive dose was excreted in urine, mainly as O-glucuronide metabolites (30.5%). Less than 1% of the dose was excreted as unchanged canagliflozin in urine. Renal clearance of canagliflozin 100 mg and 300 mg doses ranged from 1.30 to 1.55 mL/min.
Mean systemic clearance of canagliflozin was approximately 192 mL/min in healthy subjects following intravenous administration.
Metformin
Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Specific Populations
Trials characterizing the pharmacokinetics of canagliflozin and metformin after administration of INVOKAMET/INVOKAMET XR were not conducted in patients with renal and hepatic impairment. Descriptions of the individual components in this patient population are described below.
Renal Impairment
Canagliflozin
A single-dose, open-label trial evaluated the pharmacokinetics of canagliflozin 200 mg in subjects with varying degrees of renal impairment (classified using the MDRD-eGFR formula) compared to healthy subjects.
Renal impairment did not affect the Cmax of canagliflozin. Compared to healthy subjects (N=3; eGFR greater than or equal to 90 mL/min/1.73 m2), plasma AUC of canagliflozin was increased by approximately 15%, 29%, and 53% in subjects with mild (N=10), moderate (N=9), and severe (N=10) renal impairment, respectively, (eGFR 60 to less than 90, 30 to less than 60, and 15 to less than 30 mL/min/1.73 m2, respectively), but was similar for ESRD (N=8) subjects and healthy subjects. Increases in canagliflozin AUC of this magnitude are not considered clinically relevant. The glucose lowering pharmacodynamic response to canagliflozin declines with increasing severity of renal impairment [see Contraindications (4) and Warnings and Precautions (5.5)].
Canagliflozin was negligibly removed by hemodialysis.
Metformin
In patients with decreased renal function, the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased [see Contraindications (4) and Warnings and Precautions (5.1)].
Following a single dose administration of metformin HCl extended-release tablets 500 mg in patients with mild and moderate renal failure (based on measured creatinine clearance), the oral and renal clearance of metformin were decreased by 33% and 50% and 16% and 53%, respectively [see Warnings and Precautions (5.5)]. Metformin peak and systemic exposure was 27% and 61% greater, respectively in mild renal impaired and 74% and 2.36-fold greater in moderate renal impaired patients as compared to healthy subjects [see Contraindications (4) and Warnings and Precautions (5.1)].
Hepatic Impairment
Canagliflozin
Relative to subjects with normal hepatic function, the geometric mean ratios for Cmax and AUC∞ of canagliflozin were 107% and 110%, respectively, in subjects with Child-Pugh class A (mild hepatic impairment) and 96% and 111%, respectively, in subjects with Child-Pugh class B (moderate hepatic impairment) following administration of a single 300 mg dose of canagliflozin.
These differences are not considered to be clinically meaningful. There is no clinical experience in patients with Child-Pugh class C (severe) hepatic impairment [see Warnings and Precautions (5.1)].
Metformin
No pharmacokinetic trials of metformin HCl tablets have been conducted in patients with hepatic insufficiency [see Warnings and Precautions (5.1)].
Pharmacokinetic Effects of Age, Body Mass Index (BMI)/Weight, Gender and Race
Canagliflozin
Based on the population PK analysis with data collected from 1526 subjects, age, body mass index (BMI)/weight, gender, and race do not have a clinically meaningful effect on the pharmacokinetics of canagliflozin [see Use in Specific Populations (8.5)].
Metformin
Metformin pharmacokinetic parameters did not differ significantly between normal subjects and patients with type 2 diabetes when analyzed according to gender.
No trials of metformin pharmacokinetic parameters according to race have been performed.
Canagliflozin
Age had no clinically meaningful effect on the pharmacokinetics of canagliflozin based on a population pharmacokinetic analysis [see Adverse Reactions (6.1) and Use in Specific Populations (8.5)].
Metformin
Limited data from controlled pharmacokinetic trials of metformin HCl tablets in healthy elderly subjects suggest that total plasma clearance of metformin is decreased, the half-life is prolonged, and Cmax is increased, compared with healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function [see Warnings and Precautions (5.1, 5.5) and Use in Specific Populations (8.5)].
Drug-Drug Interactions
INVOKAMET and INVOKAMET XR
Pharmacokinetic drug interaction trials with INVOKAMET/INVOKAMET XR have not been performed; however, such trials have been conducted with the individual components canagliflozin and metformin HCl.
Co-administration of multiple doses of canagliflozin (300 mg) and metformin HCl (2,000 mg) given once daily did not meaningfully alter the pharmacokinetics of either canagliflozin or metformin in healthy subjects.
Canagliflozin
In Vitro Assessment of Drug Interactions
Canagliflozin did not induce CYP450 enzyme expression (3A4, 2C9, 2C19, 2B6, and 1A2) in cultured human hepatocytes. Canagliflozin did not inhibit the CYP450 isoenzymes (1A2, 2A6, 2C19, 2D6, or 2E1) and weakly inhibited CYP2B6, CYP2C8, CYP2C9, and CYP3A4 based on in vitro studies with human hepatic microsomes. Canagliflozin is a weak inhibitor of P-gp.
Canagliflozin is also a substrate of drug transporters P-glycoprotein (P-gp) and MRP2.
In Vivo Assessment of Drug Interactions
Table 9: Effect of Co–Administered Drugs on Systemic Exposures of Canagliflozin Co-Administered Drug Dose of Co-Administered Drug* Dose of Canagliflozin* Geometric Mean Ratio
(Ratio With/Without Co-Administered Drug)
No Effect = 1.0AUC†
(90% CI)Cmax
(90% CI)QD = once daily; BID = twice daily - * Single dose unless otherwise noted
- † AUCinf for drugs given as a single dose and AUC24h for drugs given as multiple doses
See Drug Interactions (7.2) for the clinical relevance of the following: Rifampin 600 mg QD for 8 days 300 mg 0.49
(0.44; 0.54)0.72
(0.61; 0.84)No dose adjustments of canagliflozin required for the following: Cyclosporine 400 mg 300 mg QD for 8 days 1.23
(1.19; 1.27)1.01
(0.91; 1.11)Ethinyl estradiol and levonorgestrel 0.03 mg ethinyl estradiol and 0.15 mg levonorgestrel 200 mg QD for 6 days 0.91
(0.88; 0.94)0.92
(0.84; 0.99)Hydrochlorothiazide 25 mg QD for 35 days 300 mg QD for 7 days 1.12
(1.08; 1.17)1.15
(1.06; 1.25)Metformin HCl 2,000 mg 300 mg QD for 8 days 1.10
(1.05; 1.15)1.05
(0.96; 1.16)Probenecid 500 mg BID for 3 days 300 mg QD for 17 days 1.21
(1.16; 1.25)1.13
(1.00; 1.28)Table 10: Effect of Canagliflozin on Systemic Exposure of Co-Administered Drugs Co-Administered Drug Dose of Co-Administered Drug* Dose of Canagliflozin* Geometric Mean Ratio
(Ratio With/Without Co-Administered Drug)
No Effect = 1.0AUC†
(90% CI)Cmax
(90% CI)QD = once daily; BID = twice daily; INR = International Normalized Ratio - * Single dose unless otherwise noted
- † AUCinf for drugs given as a single dose and AUC24h for drugs given as multiple doses
- ‡ AUC0–12h
See Drug Interactions (7.2) for the clinical relevance of the following: Digoxin 0.5 mg QD first day followed by 0.25 mg QD for 6 days 300 mg QD for 7 days Digoxin 1.20
(1.12; 1.28)1.36
(1.21; 1.53)No dose adjustments of co-administered drug required for the following: Acetaminophen 1,000 mg 300 mg BID for 25 days Acetaminophen 1.06‡
(0.98; 1.14)1.00
(0.92; 1.09)Ethinyl estradiol and levonorgestrel 0.03 mg ethinyl estradiol and 0.15 mg levonorgestrel 200 mg QD for 6 days ethinyl estradiol 1.07
(0.99; 1.15)1.22
(1.10; 1.35)Levonorgestrel 1.06
(1.00; 1.13)1.22
(1.11; 1.35)Glyburide 1.25 mg 200 mg QD for 6 days Glyburide 1.02
(0.98; 1.07)0.93
(0.85; 1.01)3-cis-hydroxy-glyburide 1.01
(0.96; 1.07)0.99
(0.91; 1.08)4-trans-hydroxy-glyburide 1.03
(0.97; 1.09)0.96
(0.88; 1.04)Hydrochlorothiazide 25 mg QD for 35 days 300 mg QD for 7 days Hydrochlorothiazide 0.99
(0.95; 1.04)0.94
(0.87; 1.01)Metformin HCl 2,000 mg 300 mg QD for 8 days Metformin 1.20
(1.08; 1.34)1.06
(0.93; 1.20)Simvastatin 40 mg 300 mg QD for 7 days Simvastatin 1.12
(0.94; 1.33)1.09
(0.91; 1.31)simvastatin acid 1.18
(1.03; 1.35)1.26
(1.10; 1.45)Warfarin 30 mg 300 mg QD for 12 days (R)-warfarin 1.01
(0.96; 1.06)1.03
(0.94; 1.13)(S)-warfarin 1.06
(1.00; 1.12)1.01
(0.90; 1.13)INR 1.00
(0.98; 1.03)1.05
(0.99; 1.12)Metformin
Table 11: Effect of Co–Administered Drugs on Plasma Metformin Systemic Exposures Co-Administered Drug Dose of Co-Administered Drug* Dose of Metformin HCl* Geometric Mean Ratio
(Ratio With/Without Co-Administered Drug)
No Effect = 1.00AUC† Cmax - * Single dose unless otherwise noted
- † AUC = AUC0–∞
- ‡ Metformin HCl extended-release tablets 500 mg
- § Ratio of arithmetic means
- ¶ Healthy volunteer study at steady state with topiramate 100 mg every 12 hours and metformin 500 mg every 12 hours for 7 days. Study conducted to assess pharmacokinetics only
- # Steady state AUC0–12h.
No dose adjustments required for the following: Glyburide 5 mg 500 mg‡ 0.98§ 0.99§ Furosemide 40 mg 850 mg 1.09§ 1.22§ Nifedipine 10 mg 850 mg 1.16 1.21 Propranolol 40 mg 850 mg 0.90 0.94 Ibuprofen 400 mg 850 mg 1.05§ 1.07§ Drugs that are eliminated by renal tubular secretion increase the accumulation of metformin [see Warnings and Precautions (5) and Drug Interactions (7)] Cimetidine 400 mg 850 mg 1.40 1.61 Carbonic anhydrase inhibitors may cause metabolic acidosis [see Warnings and Precautions (5) and Drug Interactions (7)] Topiramate¶ 100 mg 500 mg 1.25# 1.18 Table 12: Effect of Metformin HCl on Co–Administered Drug Systemic Exposures Co-Administered Drug Dose of Co-Administered Drug* Dose of Metformin HCl* Geometric Mean Ratio
(Ratio With/Without Co-Administered Drug)
No Effect = 1.00AUC† Cmax - * Single dose unless otherwise noted
- † AUC = AUC0–∞
- ‡ AUC0–24 hr reported
- § Ratio of arithmetic means, p-value of difference <0.05
- ¶ Ratio of arithmetic means.
No dose adjustments required for the following: Glyburide 5 mg 500 mg‡ 0.78§ 0.63§ Furosemide 40 mg 850 mg 0.87§ 0.69§ Nifedipine 10 mg 850 mg 1.10‡ 1.08 Propranolol 40 mg 850 mg 1.01‡ 0.94 Ibuprofen 400 mg 850 mg 0.97¶ 1.01¶ Cimetidine 400 mg 850 mg 0.95‡ 1.01 Metformin is negligibly bound to plasma proteins and is, therefore, less likely to interact with highly protein-bound drugs such as salicylates, sulfonamides, chloramphenicol, and probenecid.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
INVOKAMET and INVOKAMET XR
No animal studies have been conducted with the combined products in INVOKAMET/INVOKAMET XR to evaluate carcinogenesis, mutagenesis, or impairment of fertility. The following data are based on findings in studies with canagliflozin and metformin HCl individually.
Canagliflozin
Carcinogenesis
Carcinogenicity was evaluated in 2-year studies conducted in CD1 mice and Sprague-Dawley rats. Canagliflozin did not increase the incidence of tumors in mice dosed at 10, 30, or 100 mg/kg (less than or equal to 14 times exposure from a 300 mg clinical dose).
Testicular Leydig cell tumors, considered secondary to increased luteinizing hormone (LH), increased significantly in male rats at all doses tested (10, 30, and 100 mg/kg). In a 12-week clinical trial, LH did not increase in males treated with canagliflozin.
Renal tubular adenoma and carcinoma increased significantly in male and female rats dosed at 100 mg/kg, or approximately 12-times exposure from a 300 mg clinical dose. Also, adrenal pheochromocytoma increased significantly in males and numerically in females dosed at 100 mg/kg. Carbohydrate malabsorption associated with high doses of canagliflozin was considered a necessary proximal event in the emergence of renal and adrenal tumors in rats. Clinical trials have not demonstrated carbohydrate malabsorption in humans at canagliflozin doses of up to 2-times the recommended clinical dose of 300 mg.
Mutagenesis
Canagliflozin was not mutagenic with or without metabolic activation in the Ames assay. Canagliflozin was mutagenic in the in vitro mouse lymphoma assay with but not without metabolic activation. Canagliflozin was not mutagenic or clastogenic in an in vivo oral micronucleus assay in rats and an in vivo oral Comet assay in rats.
Metformin HCl
Carcinogenesis
Long-term carcinogenicity studies have been performed in rats (dosing duration of 104 weeks) and mice (dosing duration of 91 weeks) at doses up to and including 900 mg/kg/day and 1500 mg/kg/day, respectively. These doses are both approximately 4 times the maximum recommended human daily dose of 2000 mg based on body surface area comparisons. No evidence of carcinogenicity with metformin HCl was found in either male or female mice. Similarly, there was no tumorigenic potential observed with metformin HCl in male rats. There was, however, an increased incidence of benign stromal uterine polyps in female rats treated with 900 mg/kg/day.
Impairment of Fertility
Canagliflozin had no effects on the ability of rats to mate and sire or maintain a litter up to the high dose of 100 mg/kg (approximately 14 times and 18 times the 300 mg clinical dose in males and females, respectively), although there were minor alterations in a number of reproductive parameters (decreased sperm velocity, increased number of abnormal sperm, slightly fewer corpora lutea, fewer implantation sites, and smaller litter sizes) at the highest dosage administered.
Fertility of male or female rats was unaffected by metformin HCl when administered at doses as high as 600 mg/kg/day, which is approximately 3 times the maximum recommended human daily dose based on body surface area comparisons.
-
14 CLINICAL STUDIES
14.1 Glycemic Control Trials in Adults with Type 2 Diabetes Mellitus
Canagliflozin has been studied in combination with metformin HCl alone, metformin HCl and sulfonylurea, metformin HCl and sitagliptin, metformin HCl and a thiazolidinedione (i.e., pioglitazone), and metformin HCl and insulin (with or without other anti-hyperglycemic agents). The efficacy of canagliflozin was compared to a dipeptidyl peptidase-4 (DPP-4) inhibitor (sitagliptin), both as add-on combination therapy with metformin HCl and sulfonylurea, and a sulfonylurea (glimepiride), both as add-on combination therapy with metformin HCl.
There have been no clinical efficacy trials conducted with INVOKAMET/INVOKAMET XR; however, bioequivalence of INVOKAMET/INVOKAMET XR to canagliflozin and metformin HCl co-administered as individual tablets was demonstrated in healthy subjects.
Canagliflozin as Initial Combination Therapy with Metformin HCl
A total of 1,186 patients with type 2 diabetes inadequately controlled with diet and exercise participated in a 26-week double-blind, active-controlled, parallel-group, 5-arm, multicenter trial to evaluate the efficacy and safety of initial therapy with canagliflozin in combination with metformin HCl XR. The median age was 56 years, 48% of patients were men, and the mean baseline eGFR was 87.6 mL/min/1.73 m2. The median duration of diabetes was 1.6 years, and 72% of patients were treatment naïve. After completing a 2-week single-blind placebo run-in period, patients were randomly assigned for a double-blind treatment period of 26 weeks to 1 of 5 treatment groups (Table 13). The metformin HCl XR dose was initiated at 500 mg/day for the first week of treatment and then increased to 1,000 mg/day. Metformin HCl XR or matching placebo was up-titrated every 2–3 weeks during the next 8 weeks of treatment to a maximum daily dose of 1,500 to 2,000 mg/day, as tolerated; about 90% of patients reached 2,000 mg/day.
At the end of treatment, canagliflozin 100 mg and canagliflozin 300 mg in combination with metformin HCl XR resulted in a statistically significant greater improvement in HbA1C compared to their respective canagliflozin doses (100 mg and 300 mg) alone or metformin HCl XR alone.
Table 13: Results from 26-Week Active-Controlled Clinical Study of Canagliflozin Alone or Canagliflozin as Initial Combination Therapy with Metformin HCl XR* Efficacy Parameter Metformin HCl XR
(N=237)Canagliflozin 100 mg
(N=237)Canagliflozin 300 mg
(N=238)Canagliflozin 100 mg + Metformin HCl XR
(N=237)Canagliflozin 300 mg + Metformin HCl XR
(N=237)HbA1C (%) - * Intent-to-treat population
- † There were 121 patients without week 26 efficacy data. Analyses addressing missing data gave consistent results with the results provided in this table.
- ‡ Least squares mean adjusted for covariates including baseline value and stratification factor
- § Adjusted p=0.001
- ¶ Adjusted p<0.05
Baseline (mean) 8.81 8.78 8.77 8.83 8.90 Change from baseline (adjusted mean)† -1.30 -1.37 -1.42 -1.77 -1.78 Difference from canagliflozin 100 mg (adjusted mean) (95% CI)‡ -0.40§
(-0.59, -0.21)Difference from canagliflozin 300 mg (adjusted mean) (95% CI)‡ -0.36§
(-0.56, -0.17)Difference from metformin HCl XR (adjusted mean) (95% CI)‡ -0.46§
(-0.66, -0.27)-0.48§
(-0.67, -0.28)Percent of patients achieving HbA1C < 7% 38 34 39 47¶ 51¶ Canagliflozin as Add-on Combination Therapy with Metformin HCl
A total of 1,284 patients with type 2 diabetes inadequately controlled on metformin HCl monotherapy (greater than or equal to 2,000 mg/day or at least 1,500 mg/day if higher dose not tolerated) participated in a 26-week, double-blind, placebo- and active-controlled trial to evaluate the efficacy and safety of canagliflozin in combination with metformin HCl. The mean age was 55 years, 47% of patients were men, and the mean baseline eGFR was 89 mL/min/1.73 m2. Patients already on the required metformin HCl dose (N=1009) were randomized after completing a 2-week, single-blind, placebo run-in period. Patients taking less than the required metformin HCl dose or patients on metformin HCl in combination with another antihyperglycemic agent (N=275) were switched to metformin HCl monotherapy (at doses described above) for at least 8 weeks before entering the 2-week, single-blind, placebo run-in. After the placebo run-in period, patients were randomized to canagliflozin 100 mg, canagliflozin 300 mg, sitagliptin 100 mg, or placebo, administered once daily as add-on therapy to metformin HCl.
At the end of treatment, canagliflozin 100 mg and 300 mg once daily resulted in a statistically significant improvement in HbA1C (p<0.001 for both doses) compared to placebo when added to metformin HCl. Canagliflozin 100 mg and 300 mg once daily also resulted in a greater proportion of patients achieving an HbA1C less than 7%, in significant reduction in fasting plasma glucose (FPG), in improved postprandial glucose (PPG), and in percent body weight reduction compared to placebo when added to metformin HCl (see Table 14). Statistically significant (p<0.001 for both doses) mean changes from baseline in systolic blood pressure relative to placebo were -5.4 mmHg and -6.6 mmHg with canagliflozin 100 mg and 300 mg, respectively.
Table 14: Results from 26-Week Placebo-Controlled Clinical Study of Canagliflozin in Combination with Metformin HCl* Efficacy Parameter Placebo + Metformin HCl
(N=183)Canagliflozin 100 mg + Metformin HCl
(N=368)Canagliflozin 300 mg + Metformin HCl
(N=367)- * Intent-to-treat population using last observation in study prior to glycemic rescue therapy
- † Least squares mean adjusted for baseline value and stratification factors
- ‡ p<0.001
HbA1C (%) Baseline (mean) 7.96 7.94 7.95 Change from baseline (adjusted mean) -0.17 -0.79 -0.94 Difference from placebo (adjusted mean) (95% CI)† -0.62‡
(-0.76, -0.48)-0.77‡
(-0.91, -0.64)Percent of patients achieving HbA1C < 7% 30 46‡ 58‡ Fasting Plasma Glucose (mg/dL) Baseline (mean) 164 169 173 Change from baseline (adjusted mean) 2 -27 -38 Difference from placebo (adjusted mean) (95% CI)† -30‡
(-36, -24)-40‡
(-46, -34)2-hour Postprandial Glucose (mg/dL) Baseline (mean) 249 258 262 Change from baseline (adjusted mean) -10 -48 -57 Difference from placebo (adjusted mean) (95% CI)† -38‡
(-49, -27)-47‡
(-58, -36)Body Weight Baseline (mean) in kg 86.7 88.7 85.4 % change from baseline (adjusted mean) -1.2 -3.7 -4.2 Difference from placebo (adjusted mean) (95% CI)† -2.5‡
(-3.1, -1.9)-2.9‡
(-3.5, -2.3)Canagliflozin Compared to Glimepiride, Both as Add-on Combination Therapy with Metformin HCl
A total of 1,450 patients with type 2 diabetes inadequately controlled on metformin HCl monotherapy (greater than or equal to 2,000 mg/day or at least 1,500 mg/day if higher dose not tolerated) participated in a 52-week, double-blind, active-controlled trial to evaluate the efficacy and safety of canagliflozin in combination with metformin HCl.
The mean age was 56 years, 52% of patients were men, and the mean baseline eGFR was 90 mL/min/1.73 m2. Patients tolerating maximally required metformin HCl dose (N=928) were randomized after completing a 2-week, single-blind, placebo run-in period. Other patients (N=522) were switched to metformin HCl monotherapy (at doses described above) for at least 10 weeks, then completed a 2-week single-blind run-in period. After the 2-week run-in period, patients were randomized to canagliflozin 100 mg, canagliflozin 300 mg, or glimepiride (titration allowed throughout the 52-week trial to 6 or 8 mg), administered once daily as add-on therapy to metformin HCl.
As shown in Table 15 and Figure 1, at the end of treatment, canagliflozin 100 mg provided similar reductions in HbA1C from baseline compared to glimepiride when added to metformin HCl therapy. Canagliflozin 300 mg provided a greater reduction from baseline in HbA1C compared to glimepiride, and the relative treatment difference was -0.12% (95% CI: −0.22; −0.02). As shown in Table 15, treatment with canagliflozin 100 mg and 300 mg daily provided greater improvements in percent body weight change, relative to glimepiride.
Table 15: Results from 52–Week Clinical Study Comparing Canagliflozin to Glimepiride in Combination with Metformin HCl* Efficacy Parameter Canagliflozin 100 mg + Metformin HCl
(N=483)Canagliflozin 300 mg + Metformin HCl
(N=485)Glimepiride (titrated) + Metformin HCl
(N=482)- * Intent-to-treat population using last observation in study prior to glycemic rescue therapy
- † Least squares mean adjusted for baseline value and stratification factors
- ‡ Canagliflozin + metformin HCl is considered non-inferior to glimepiride + metformin HCl because the upper limit of this confidence interval is less than the pre-specified non-inferiority margin of < 0.3%.
- § p<0.001
HbA1C (%) Baseline (mean) 7.78 7.79 7.83 Change from baseline (adjusted mean) -0.82 -0.93 -0.81 Difference from glimepiride (adjusted mean) (95% CI)† -0.01‡
(-0.11, 0.09)-0.12‡
(-0.22, -0.02)Percent of patients achieving HbA1C < 7% 54 60 56 Fasting Plasma Glucose (mg/dL) Baseline (mean) 165 164 166 Change from baseline (adjusted mean) -24 -28 -18 Difference from glimepiride (adjusted mean) (95% CI)† -6
(-10, -2)-9
(-13, -5)Body Weight Baseline (mean) in kg 86.8 86.6 86.6 % change from baseline (adjusted mean) -4.2 -4.7 1.0 Difference from glimepiride (adjusted mean) (95% CI)† -5.2§
(-5.7, -4.7)-5.7§
(-6.2, -5.1)Figure 1: Mean HbA1C Change at Each Time Point (Completers) and at Week 52 Using Last Observation Carried Forward (mITT Population)
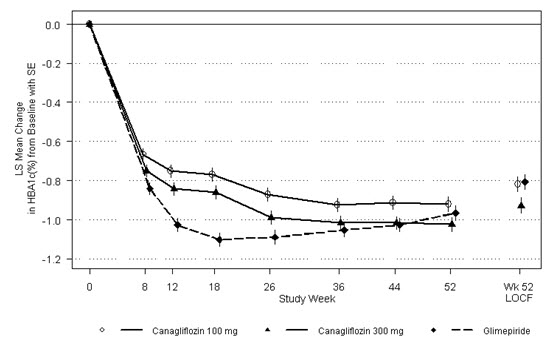
Canagliflozin as Add-on Combination Therapy with Metformin HCl and Sitagliptin
A total of 217 patients with type 2 diabetes inadequately controlled on the combination of metformin HCl (greater than or equal to 1,500 mg/day) and sitagliptin 100 mg/day (or equivalent fixed-dose combination) participated in a 26-week, double-blind, placebo-controlled trial to evaluate the efficacy and safety of canagliflozin in combination with metformin HCl and sitagliptin. The mean age was 57 years, 58% of patients were men, 73% of patients were Caucasian, 15% were Asian, and 12% were Black or African-American. The mean baseline eGFR was 90 mL/min/1.73 m2 and the mean baseline BMI was 32 kg/m2. The mean duration of diabetes was 10 years. Eligible patients entered a 2-week, single-blind, placebo run-in period and were subsequently randomized to canagliflozin 100 mg or placebo, administered once daily as add-on to metformin HCl and sitagliptin. Patients with a baseline eGFR of 70 mL/min/1.73 m2 or greater who were tolerating canagliflozin 100 mg and who required additional glycemic control (fasting finger stick 100 mg/dL or greater at least twice within 2 weeks) were up-titrated to canagliflozin 300 mg. While up-titration occurred as early as Week 4, most (90%) patients randomized to canagliflozin were up-titrated to canagliflozin 300 mg by 6 to 8 weeks.
At the end of 26 weeks, canagliflozin once daily resulted in a statistically significant improvement in HbA1C (p<0.001) compared to placebo when added to metformin HCl and sitagliptin (see Table 16).
Table 16: Results from 26–Week Placebo-Controlled Clinical Study of Canagliflozin in Combination with Metformin HCl and Sitagliptin Efficacy Parameter Placebo + Metformin HCl and Sitagliptin
(N=108*)Canagliflozin + Metformin HCl and Sitagliptin
(N=109*)- * To preserve the integrity of randomization, all randomized patients were included in the analysis. The patient who was randomized once to each arm was analyzed on canagliflozin.
- † Early treatment discontinuation before week 26, occurred in 11.0% and 24.1% of canagliflozin and placebo patients, respectively.
- ‡ Estimated using a multiple imputation method modeling a "wash-out" of the treatment effect for patients having missing data who discontinued treatment. Missing data was imputed only at week 26 and analyzed using ANCOVA.
- § p<0.001
- ¶ Patients without week 26 efficacy data were considered as non-responders when estimating the proportion achieving HbA1C < 7%.
- # Estimated using a multiple imputation method modeling a "wash-out" of the treatment effect for patients having missing data who discontinued treatment. A mixed model for repeated measures was used to analyze the imputed data.
HbA1C (%) Baseline (mean) 8.40 8.50 Change from baseline (adjusted mean) -0.03 -0.83 Difference from placebo (adjusted mean) (95% CI)†‡ -0.81§
(-1.11; -0.51)Percent of patients achieving HbA1C < 7%¶ 9 28 Fasting Plasma Glucose (mg/dL)# Baseline (mean) 180 185 Change from baseline (adjusted mean) -3 -28 Difference from placebo (adjusted mean) (95% CI) -25§
(-39; -11)Canagliflozin as Add-on Combination Therapy with Metformin HCl and Sulfonylurea
A total of 469 patients with type 2 diabetes inadequately controlled on the combination of metformin HCl (greater than or equal to 2,000 mg/day or at least 1,500 mg/day if higher dose not tolerated) and sulfonylurea (maximal or near-maximal effective dose) participated in a 26-week, double-blind, placebo-controlled trial to evaluate the efficacy and safety of canagliflozin in combination with metformin HCl and sulfonylurea. The mean age was 57 years, 51% of patients were men, and the mean baseline eGFR was 89 mL/min/1.73 m2. Patients already on the protocol-specified doses of metformin HCl and sulfonylurea (N=372) entered a 2-week, single-blind, placebo run-in period. Other patients (N=97) were required to be on a stable protocol-specified dose of metformin HCl and sulfonylurea for at least 8 weeks before entering the 2-week run-in period. Following the run-in period, patients were randomized to canagliflozin 100 mg, canagliflozin 300 mg, or placebo administered once daily as add-on to metformin HCl and sulfonylurea.
At the end of treatment, canagliflozin 100 mg and 300 mg once daily resulted in a statistically significant improvement in HbA1C (p<0.001 for both doses) compared to placebo when added to metformin HCl and sulfonylurea. Canagliflozin 100 mg and 300 mg once daily also resulted in a greater proportion of patients achieving an HbA1C less than 7.0%, in a significant reduction in fasting plasma glucose (FPG), and in percent body weight reduction compared to placebo when added to metformin HCl and sulfonylurea (see Table 17).
Table 17: Results from 26–Week Placebo-Controlled Clinical Study of Canagliflozin in Combination with Metformin HCl and Sulfonylurea* Efficacy Parameter Placebo + Metformin HCl and Sulfonylurea
(N=156)Canagliflozin 100 mg + Metformin HCl
and Sulfonylurea
(N=157)Canagliflozin 300 mg + Metformin HCl
and Sulfonylurea
(N=156)- * Intent-to-treat population using last observation in study prior to glycemic rescue therapy
- † Least squares mean adjusted for baseline value and stratification factors
- ‡ p<0.001
HbA1C (%) Baseline (mean) 8.12 8.13 8.13 Change from baseline (adjusted mean) -0.13 -0.85 -1.06 Difference from placebo (adjusted mean) (95% CI)† -0.71‡
(-0.90, -0.52)-0.92‡
(-1.11, -0.73)Percent of patients achieving HbA1C < 7% 18 43‡ 57‡ Fasting Plasma Glucose (mg/dL) Baseline (mean) 170 173 168 Change from baseline (adjusted mean) 4 -18 -31 Difference from placebo (adjusted mean) (95% CI)† -22‡
(-31, -13)-35‡
(-44, -25)Body Weight Baseline (mean) in kg 90.8 93.5 93.5 % change from baseline (adjusted mean) -0.7 -2.1 -2.6 Difference from placebo (adjusted mean) (95% CI)† -1.4‡
(-2.1, -0.7)-2.0‡
(-2.7, -1.3)Canagliflozin Compared to Sitagliptin, Both as Add-on Combination Therapy with Metformin HCl and Sulfonylurea
A total of 755 patients with type 2 diabetes inadequately controlled on the combination of metformin HCl (greater than or equal to 2,000 mg/day or at least 1,500 mg/day if higher dose not tolerated) and sulfonylurea (near-maximal or maximal effective dose) participated in a 52 week, double-blind, active-controlled trial to compare the efficacy and safety of canagliflozin 300 mg versus sitagliptin 100 mg in combination with metformin HCl and sulfonylurea. The mean age was 57 years, 56% of patients were men, and the mean baseline eGFR was 88 mL/min/1.73 m2. Patients already on protocol-specified doses of metformin HCl and sulfonylurea (N=716) entered a 2-week single-blind, placebo run-in period. Other patients (N=39) were required to be on a stable protocol-specified dose of metformin HCl and sulfonylurea for at least 8 weeks before entering the 2-week run-in period. Following the run-in period, patients were randomized to canagliflozin 300 mg or sitagliptin 100 mg as add-on to metformin HCl and sulfonylurea.
As shown in Table 18 and Figure 2, at the end of treatment, canagliflozin 300 mg provided greater HbA1C reduction compared to sitagliptin 100 mg when added to metformin HCl and sulfonylurea (p<0.05). Canagliflozin 300 mg resulted in a mean percent change in body weight from baseline of -2.5% compared to +0.3% with sitagliptin 100 mg. A mean change in systolic blood pressure from baseline of -5.06 mmHg was observed with canagliflozin 300 mg compared to +0.85 mmHg with sitagliptin 100 mg.
Table 18: Results from 52−Week Clinical Study Comparing Canagliflozin to Sitagliptin in Combination with Metformin HCl and Sulfonylurea* Efficacy Parameter Canagliflozin 300 mg + Metformin HCl and Sulfonylurea
(N=377)Sitagliptin 100 mg + Metformin HCl and Sulfonylurea
(N=378)- * Intent-to-treat population using last observation in study prior to glycemic rescue therapy
- † Least squares mean adjusted for baseline value and stratification factors
- ‡ Canagliflozin + metformin HCl + sulfonylurea is considered non-inferior to sitagliptin + metformin HCl + sulfonylurea because the upper limit of this confidence interval is less than the pre-specified non-inferiority margin of < 0.3%.
- § p<0.001
HbA1C (%) Baseline (mean) 8.12 8.13 Change from baseline (adjusted mean) -1.03 -0.66 Difference from sitagliptin (adjusted mean) (95% CI)† -0.37‡
(-0.50, -0.25)Percent of patients achieving HbA1C < 7% 48 35 Fasting Plasma Glucose (mg/dL) Baseline (mean) 170 164 Change from baseline (adjusted mean) -30 -6 Difference from sitagliptin (adjusted mean) (95% CI)† -24
(-30, -18)Body Weight Baseline (mean) in kg 87.6 89.6 % change from baseline (adjusted mean) -2.5 0.3 Difference from sitagliptin (adjusted mean) (95% CI)† -2.8§
(-3.3, -2.2)Figure 2: Mean HbA1C Change at Each Time Point (Completers) and at Week 52 Using Last Observation Carried Forward (mITT Population)
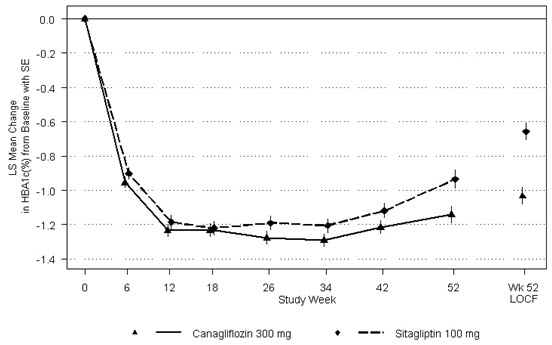
Canagliflozin as Add-on Combination Therapy with Metformin HCl and Pioglitazone
A total of 342 patients with type 2 diabetes inadequately controlled on the combination of metformin HCl (greater than or equal to 2,000 mg/day or at least 1,500 mg/day if higher dose not tolerated) and pioglitazone (30 or 45 mg/day) participated in a 26-week, double--blind, placebo-controlled trial to evaluate the efficacy and safety of canagliflozin in combination with metformin HCl and pioglitazone. The mean age was 57 years, 63% of patients were men, and the mean baseline eGFR was 86 mL/min/1.73 m2. Patients already on protocol-specified doses of metformin HCl and pioglitazone (N=163) entered a 2-week, single-blind, placebo run-in period. Other patients (N=181) were required to be on stable protocol-specified doses of metformin HCl and pioglitazone for at least 8 weeks before entering the 2-week run-in period. Following the run-in period, patients were randomized to canagliflozin 100 mg, canagliflozin 300 mg, or placebo, administered once daily as add-on to metformin HCl and pioglitazone.
At the end of treatment, canagliflozin 100 mg and 300 mg once daily resulted in a statistically significant improvement in HbA1C (p<0.001 for both doses) compared to placebo when added to metformin HCl and pioglitazone. Canagliflozin 100 mg and 300 mg once daily also resulted in a greater proportion of patients achieving an HbA1C less than 7%, in significant reduction in fasting plasma glucose (FPG), and in percent body weight reduction compared to placebo when added to metformin HCl and pioglitazone (see Table 19). Statistically significant (p<0.05 for both doses) mean changes from baseline in systolic blood pressure relative to placebo were -4.1 mmHg and -3.5 mmHg with canagliflozin 100 mg and 300 mg, respectively.
Table 19: Results from 26–Week Placebo-Controlled Clinical Study of Canagliflozin in Combination with Metformin HCl and Pioglitazone* Efficacy Parameter Placebo + Metformin HCl and Pioglitazone
(N=115)Canagliflozin 100 mg + Metformin HCl and Pioglitazone
(N=113)Canagliflozin 300 mg + Metformin HCl and Pioglitazone
(N=114)- * Intent-to-treat population using last observation in study prior to glycemic rescue therapy
- † Least squares mean adjusted for baseline value and stratification factors
- ‡ p<0.001
HbA1C (%) Baseline (mean) 8.00 7.99 7.84 Change from baseline (adjusted mean) -0.26 -0.89 -1.03 Difference from placebo (adjusted mean) (95% CI)† -0.62‡
(-0.81, -0.44)-0.76‡
(-0.95, -0.58)Percent of patients achieving HbA1C < 7% 33 47‡ 64‡ Fasting Plasma Glucose (mg/dL) Baseline (mean) 164 169 164 Change from baseline (adjusted mean) 3 -27 -33 Difference from placebo (adjusted mean) (95% CI)† -29‡
(-37, -22)-36‡
(-43, -28)Body Weight Baseline (mean) in kg 94.0 94.2 94.4 % change from baseline (adjusted mean) -0.1 -2.8 -3.8 Difference from placebo (adjusted mean) (95% CI)† -2.7‡
(-3.6, -1.8)-3.7‡
(-4.6, -2.8)Canagliflozin as Add-on Combination Therapy with Insulin (With or Without Other Anti-Hyperglycemic Agents, Including Metformin HCl)
A total of 1,718 patients with type 2 diabetes inadequately controlled on insulin greater than or equal to 30 units/day or insulin in combination with other antihyperglycemic agents participated in an 18-week, double-blind, placebo-controlled substudy of a cardiovascular trial to evaluate the efficacy and safety of canagliflozin in combination with insulin. Of these patients, a subgroup of 432 patients with inadequate glycemic control received canagliflozin or placebo plus metformin HCl and ≥ 30 units/day of insulin over 18 weeks.
In this subgroup, the mean age was 61 years, 67% of patients were men, and the mean baseline eGFR was 81 mL/min/1.73 m2. Patients on metformin HCl in combination with basal, bolus, or basal/bolus insulin for at least 10 weeks entered a 2-week, single-blind, placebo run-in period. Approximately 74% of these patients were on a background of metformin HCl and basal/bolus insulin regimen. After the run-in period, patients were randomized to canagliflozin 100 mg, canagliflozin 300 mg, or placebo, administered once daily as add-on to metformin HCl and insulin. The mean daily insulin dose at baseline was 93 units, which was similar across treatment groups.
At the end of treatment, canagliflozin 100 mg and 300 mg once daily resulted in a statistically significant improvement in HbA1C (p<0.001 for both doses) compared to placebo when added to metformin HCl and insulin. Canagliflozin 100 mg and 300 mg once daily also resulted in a greater proportion of patients achieving an HbA1C less than 7%, in significant reductions in fasting plasma glucose (FPG), and in percent body weight reductions compared to placebo (see Table 20). Statistically significant (p=0.023 for the 100 mg and p<0.001 for the 300 mg dose) mean change from baseline in systolic blood pressure relative to placebo was –3.5 mmHg and –6 mmHg with canagliflozin 100 mg and 300 mg, respectively. Fewer patients on canagliflozin in combination with metformin HCl and insulin required glycemic rescue therapy: 3.6% of patients receiving canagliflozin 100 mg, 2.7% of patients receiving canagliflozin 300 mg, and 6.2% of patients receiving placebo. An increased incidence of hypoglycemia was observed in this trial, which is consistent with the expected increase of hypoglycemia when an agent not associated with hypoglycemia is added to insulin [see Warnings and Precautions (5.7) and Adverse Reactions (6.1)].
Table 20: Results from 18–Week Placebo-Controlled Clinical Study of Canagliflozin in Combination with Metformin HCl and Insulin ≥ 30 Units/Day* Efficacy Parameter Placebo + Metformin HCl + Insulin
(N=145)Canagliflozin 100 mg + Metformin HCl + Insulin
(N=139)Canagliflozin 300 mg + Metformin HCl + Insulin
(N=148)- * Intent-to-treat population using last observation in study prior to glycemic rescue therapy
- † Least squares mean adjusted for baseline value and stratification factors
- ‡ p≤0.001
- § p≤0.01
HbA1C (%) Baseline (mean) 8.15 8.20 8.22 Change from baseline (adjusted mean) 0.03 -0.64 -0.79 Difference from placebo (adjusted mean) (95% CI)† -0.66‡
(-0.81, -0.51)-0.82‡
(-0.96, -0.67)Percent of patients achieving HbA1C < 7% 9 19§ 29‡ Fasting Plasma Glucose (mg/dL) Baseline 163 168 167 Change from baseline (adjusted mean) 1 -16 -24 Difference from placebo (adjusted mean) (97.5% CI)† -16‡
(-28, -5)-25‡
(-36, -14)Body Weight Baseline (mean) in kg 102.3 99.7 101.1 % change from baseline (adjusted mean) 0.0 -1.7 -2.7 Difference from placebo (adjusted mean) (97.5% CI)† -1.7‡
(-2.4, -1.0)-2.7‡
(-3.4, -2.0)14.2 Canagliflozin Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
Canagliflozin is indicated to reduce the risk of major adverse cardiovascular events in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD).
The CANVAS and CANVAS-R trials were multicenter, multi-national, randomized, double-blind parallel group, with similar inclusion and exclusion criteria. Patients eligible for enrollment in both CANVAS and CANVAS-R trials were: 30 years of age or older and had established, stable, cardiovascular, cerebrovascular, peripheral artery disease (66% of the enrolled population) or were 50 years of age or older and had two or more other specified risk factors for cardiovascular disease (34% of the enrolled population).
The integrated analysis of the CANVAS and CANVAS-R trials compared the risk of Major Adverse Cardiovascular Event (MACE) between canagliflozin and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease. The primary endpoint, MACE, was the time to first occurrence of a three-part composite outcome which included cardiovascular death, non-fatal myocardial infarction and non-fatal stroke.
In CANVAS, patients were randomly assigned 1:1:1 to canagliflozin 100 mg, canagliflozin 300 mg, or matching placebo. In CANVAS-R, patients were randomly assigned 1:1 to canagliflozin 100 mg or matching placebo, and titration to 300 mg was permitted at the investigator's discretion (based on tolerability and glycemic needs) after Week 13. Concomitant antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 10,134 patients were treated (4,327 in CANVAS and 5,807 in CANVAS-R; total of 4,344 randomly assigned to placebo and 5,790 to canagliflozin) for a mean exposure duration of 149 weeks (223 weeks [4.3 years] in CANVAS and 94 weeks [1.8 years] in CANVAS-R). Approximately 78% of the trial population was Caucasian, 13% was Asian, and 3% was Black. The mean age was 63 years and approximately 64% were male.
The mean HbA1C at baseline was 8.2% and mean duration of diabetes was 13.5 years with 70% of patients having had diabetes for 10 years or more. Approximately 31%, 21% and 17% reported a past history of neuropathy, retinopathy and nephropathy, respectively, and the mean eGFR 76 mL/min/1.73 m2). At baseline, patients were treated with one (19%) or more (80%) antidiabetic medications including metformin (77%), insulin (50%), and sulfonylurea (43%).
At baseline, the mean systolic blood pressure was 137 mmHg, the mean diastolic blood pressure was 78 mmHg, the mean LDL was 89 mg/dL, the mean HDL was 46 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 115 mg/g. At baseline, approximately 80% of patients were treated with renin angiotensin system inhibitors, 53% with beta-blockers, 13% with loop diuretics, 36% with non-loop diuretics, 75% with statins, and 74% with antiplatelet agents (mostly aspirin). During the trial, investigators could modify anti-diabetic and cardiovascular therapies to achieve local standard of care treatment targets with respect to blood glucose, lipid, and blood pressure. More patients receiving canagliflozin compared to placebo initiated anti-thrombotics (5.2% vs 4.2%) and statins (5.8% vs 4.8%) during the trial.
For the primary analysis, a stratified Cox proportional hazards model was used to test for non-inferiority against a pre-specified risk margin of 1.3 for the hazard ratio of MACE.
In the integrated analysis of CANVAS and CANVAS-R trials, canagliflozin reduced the risk of first occurrence of MACE. The estimated hazard ratio (95% CI) for time to first MACE was 0.86 (0.75, 0.97). Refer to Table 21. Vital status was obtained for 99.6% of patients across the trials. The Kaplan-Meier curve depicting time to first occurrence of MACE is shown in Figure 3.
Table 21: Treatment Effect for the Primary Composite Endpoint, MACE, and its Components in the Integrated Analysis of CANVAS and CANVAS-R studies* Placebo
N=4347 (%)Canagliflozin
N=5795 (%)Hazard ratio
(95% CI)†- * Intent-To-Treat Analysis Set
- † Stratified Cox-proportional hazards model with treatment as a factor and stratified by study and by prior CV disease
- ‡ P-value for superiority (2-sided) = 0.0158
- § Number and percentage of first events
- ¶ Due to pooling of unequal randomization ratios, Cochran-Mantel-Haenszel weights were applied to calculate percentages
Composite of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke
(time to first occurrence)‡, §, ¶,426 (10.4) 585 (9.2) 0.86 (0.75, 0.97) Non-fatal myocardial infarction§, ¶ 159 (3.9) 215 (3.4) 0.85 (0.69, 1.05) Non-fatal Stroke§, ¶ 116 (2.8) 158 (2.5) 0.90 (0.71, 1.15) Cardiovascular Death§, ¶ 185 (4.6) 268 (4.1) 0.87 (0.72, 1.06) Figure 3: Time to First Occurrence of MACE
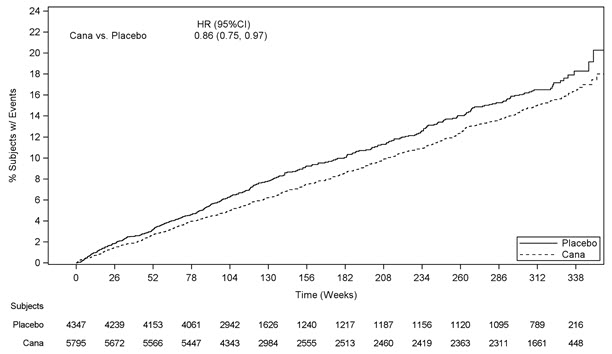
14.3 Canagliflozin Renal and Cardiovascular Outcomes in Patients with Diabetic Nephropathy and Albuminuria
Canagliflozin is indicated to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria > 300 mg/day.
The Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation Trial (CREDENCE) was a multinational, randomized, double-blind, placebo-controlled trial comparing canagliflozin with placebo in patients with type 2 diabetes mellitus, an eGFR ≥ 30 to < 90 mL/min/1.73 m2 and albuminuria (urine albumin/creatinine ˃ 300 to ≤ 5000 mg/g) who were receiving standard of care including a maximum-tolerated, labeled daily dose of an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB).
The primary objective of CREDENCE was to assess the efficacy of canagliflozin relative to placebo in reducing the composite endpoint of end stage kidney disease (ESKD), doubling of serum creatinine, and renal or CV death.
Patients were randomized to receive canagliflozin 100 mg (N=2,202) or placebo (N=2,199) and treatment was continued until the initiation of dialysis or renal transplantation.
The median follow-up duration for the 4,401 randomized subjects was 137 weeks. Vital status was obtained for 99.9% of subjects.
The population was 67% White, 20% Asian, and 5% Black; 32% were of Hispanic or Latino ethnicity. The mean age was 63 years and 66% were male.
At randomization, the mean HbA1c was 8.3%, the median urine albumin/creatinine was 927 mg/g, the mean eGFR was 56.2 mL/min/1.73 m2, 50% had prior CV disease, and 15% reported a history of heart failure. The most frequent antihyperglycemic agents (AHA) medications used at baseline were insulin (66%), biguanides (58%), and sulfonylureas (29%). Nearly all subjects (99.9%) were on ACEi or ARB at randomization, approximately 60% were taking an anti-thrombotic agent (including aspirin), and 69% were on a statin.
The primary composite endpoint in the CREDENCE study was the time to first occurrence of ESKD (defined as an eGFR < 15 mL/min/1.73 m2, initiation of chronic dialysis or renal transplant), doubling of serum creatinine, and renal or CV death. Canagliflozin 100 mg significantly reduced the risk of the primary composite endpoint based on a time-to-event analysis [HR: 0.70; 95% CI: 0.59, 0.82; p<0.0001] (see Figure 4). The treatment effect reflected a reduction in progression to ESKD, doubling of serum creatinine and cardiovascular death as shown in Table 22 and Figure 4. There were few renal deaths during the trial. Canagliflozin 100 mg also significantly reduced the risk of hospitalization for heart failure [HR: 0.61; 95% CI: 0.47 to 0.80; p<0.001].
Table 22: Analysis of Primary Endpoint (including the Individual Components) and Secondary Endpoints from the CREDENCE Study Placebo canagliflozin Endpoint N=2,199
(%)Event Rate* N=2,202
(%)Event Rate* HR†
(95% CI)Intent-To-Treat Analysis Set (time to first occurrence)
The individual components do not represent a breakdown of the composite outcomes, but rather the total number of subjects experiencing an event during the course of the study.- * Event rate per 100 patient-years.
- † Hazard ratio (canagliflozin compared to placebo), 95% CI and p-value are estimated using a stratified Cox proportional hazards model including treatment as the explanatory variable and stratified by screening eGFR (≥ 30 to < 45, ≥ 45 to < 60, ≥ 60 to < 90 mL/min/1.73 m2). HR is not presented for renal death due to the small number of events in each group.
- ‡ P-value <0.0001
- § P-value <0.001
- ¶ P-value <0.02
Primary Composite Endpoint (ESKD, doubling of serum creatinine, renal death, or CV death) 340 (15.5) 6.1 245 (11.1) 4.3 0.70
(0.59, 0.82) ‡ESKD 165 (7.5) 2.9 116 (5.3) 2.0 0.68
(0.54, 0.86)Doubling of serum creatinine 188 (8.5) 3.4 118 (5.4) 2.1 0.60
(0.48, 0.76)Renal death 5 (0.2) 0.1 2 (0.1) 0.0 CV death 140 (6.4) 2.4 110 (5.0) 1.9 0.78
(0.61, 1.00)CV death or hospitalization for heart failure 253 (11.5) 4.5 179 (8.1) 3.1 0.69
(0.57, 0.83) §CV death, non-fatal myocardial infarction or non-fatal stroke 269 (12.2) 4.9 217 (9.9) 3.9 0.80
(0.67, 0.95) ¶Non-fatal myocardial infarction 87 (4.0) 1.6 71 (3.2) 1.3 0.81
(0.59, 1.10)Non-fatal stroke 66 (3.0) 1.2 53 (2.4) 0.9 0.80
(0.56, 1.15)Hospitalization for heart failure 141 (6.4) 2.5 89 (4.0) 1.6 0.61
(0.47, 0.80) §ESKD, doubling of serum creatinine or renal death 224 (10.2) 4.0 153 (6.9) 2.7 0.66
(0.53, 0.81) ‡The Kaplan-Meier curve (Figure 4) shows time to first occurrence of the primary composite endpoint of ESKD, doubling of serum creatinine, renal death, or CV death. The curves begin to separate by Week 52 and continue to diverge thereafter.
Figure 4: CREDENCE: Time to First Occurrence of the Primary Composite Endpoint

-
16 HOW SUPPLIED/STORAGE AND HANDLING
INVOKAMET® tablets are available in bottles of 60 in the strengths listed below:
INVOKAMET TABLET STRENGTH canagliflozin/metformin HCl tablets 50 mg/500 mg 50 mg/1000 mg 150 mg/500 mg 150 mg/1000 mg Color White Beige Yellow Purple Tablet Identification CM CM CM CM 155 551 215 611 Capsule-shaped, film-coated tablets NDC 50458-540-60 50458-541-60 50458-542-60 50458-543-60 INVOKAMET® XR tablets are available in bottles of 60 in the strengths listed below:
INVOKAMET XR TABLET STRENGTH canagliflozin/metformin HCl extended-release tablets 50 mg/500 mg 50 mg/1000 mg 150 mg/500 mg 150 mg/1000 mg Color Almost White to Light Orange Pink Orange Reddish Brown Tablet Identification CM1 CM3 CM2 CM4 Oblong, biconvex, film-coated tablets, a thin line on the tablet side may be visible. NDC 50458-940-01 50458-941-01 50458-942-01 50458-943-01 Storage and Handling
Keep out of reach of children.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store and dispense in the original container. Storage in a pill box or pill organizer is allowed for up to 30 days.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-Approved Patient Labeling (Medication Guide).
Lactic Acidosis:
Explain the risks of lactic acidosis, its symptoms, and conditions that predispose to its development, as noted in Warnings and Precautions (5.1). Advise patients to discontinue INVOKAMET/INVOKAMET XR immediately and to promptly notify their healthcare provider if unexplained hyperventilation, myalgias, malaise, unusual somnolence or other nonspecific symptoms occur. Once a patient is stabilized on INVOKAMET/INVOKAMET XR, gastrointestinal symptoms, which are common during initiation of metformin HCl, are unlikely to recur. Later occurrence of gastrointestinal symptoms could be due to lactic acidosis or other serious disease.
Counsel patients against excessive alcohol intake while receiving INVOKAMET/INVOKAMET XR.
Inform patients about importance of regular testing of renal function and hematological parameters while receiving INVOKAMET/INVOKAMET XR.
Instruct patients to inform their doctor that they are taking INVOKAMET/INVOKAMET XR prior to any surgical or radiological procedure, as temporary discontinuation of INVOKAMET/INVOKAMET XR may be required until renal function has been confirmed to be normal [see Warnings and Precautions (5.1)].
Lower Limb Amputation:
Inform patients that INVOKAMET/INVOKAMET XR is associated with an increased risk of amputations. Counsel patients about the importance of routine preventative foot care. Instruct patients to monitor for new pain or tenderness, sores or ulcers, or infections involving the leg or foot and to seek medical advice immediately if such signs or symptoms develop [see Boxed Warning and Warnings and Precautions (5.2)].
Hypotension:
Inform patients that symptomatic hypotension may occur with INVOKAMET/INVOKAMET XR and advise them to contact their doctor if they experience such symptoms [see Warnings and Precautions (5.3)]. Inform patients that dehydration may increase the risk for hypotension and to have adequate fluid intake.
Ketoacidosis:
Inform patients that ketoacidosis is a serious life-threatening condition and that cases of ketoacidosis have been reported during use of canagliflozin, sometimes associated with illness or surgery among other risk factors. Instruct patients to check ketones (when possible) if symptoms consistent with ketoacidosis occur even if blood glucose is not elevated. If symptoms of ketoacidosis (including nausea, vomiting, abdominal pain, tiredness, and labored breathing) occur, instruct patients to discontinue INVOKAMET/INVOKAMET XR and seek medical attention immediately [see Warnings and Precautions (5.4)].
Acute Kidney Injury:
Inform patients that acute kidney injury has been reported during use of canagliflozin. Advise patients to seek medical advice immediately if they have reduced oral intake (such as due to acute illness or fasting), or increased fluid losses (such as due to vomiting, diarrhea, or excessive heat exposure), as it may be appropriate to temporarily discontinue INVOKAMET/INVOKAMET XR use in those settings [see Warnings and Precautions (5.5)].
Serious Urinary Tract Infections:
Inform patients of the potential for urinary tract infections, which may be serious. Provide them with information on the symptoms of urinary tract infections. Advise them to seek medical advice if such symptoms occur [see Warnings and Precautions (5.6)].
Necrotizing Fasciitis of the Perineum (Fournier's Gangrene):
Inform patients that necrotizing infections of the perineum (Fournier's gangrene) have occurred with INVOKAMET/INVOKAMET XR. Counsel patients to promptly seek medical attention if they develop pain or tenderness, redness, or swelling of the genitals or the area from the genitals back to the rectum, along with a fever above 100.4°F or malaise [see Warnings and Precautions (5.8)].
Genital Mycotic Infections in Females (e.g., Vulvovaginitis):
Inform female patients that vaginal yeast infection (e.g., vulvovaginitis) may occur and provide them with information on the signs and symptoms of a vaginal yeast infection. Advise them of treatment options and when to seek medical advice [see Warnings and Precautions (5.9)].
Genital Mycotic Infections in Males (e.g., Balanitis or Balanoposthitis):
Inform male patients that yeast infection of penis (e.g., balanitis or balanoposthitis) may occur, especially in uncircumcised males and patients with prior history. Provide them with information on the signs and symptoms of balanitis and balanoposthitis (rash or redness of the glans or foreskin of the penis). Advise them of treatment options and when to seek medical advice [see Warnings and Precautions (5.9)].
Hypersensitivity Reactions:
Inform patients that serious hypersensitivity reactions, such as urticaria, rash, anaphylaxis, and angioedema, have been reported with canagliflozin. Advise patients to report immediately any signs or symptoms suggesting allergic reaction and to discontinue drug until they have consulted prescribing physicians [see Warnings and Precautions (5.10)].
Bone Fracture:
Inform patients that bone fractures have been reported in patients taking canagliflozin. Provide them with information on factors that may contribute to fracture risk [see Warnings and Precautions (5.11)].
Laboratory Tests:
Inform patients that they will test positive for glucose in their urine while on INVOKAMET/INVOKAMET XR [see Drug Interactions (7.2)].
Females of Reproductive Age:
Advise pregnant women, and females of reproductive potential of the potential risk to a fetus with treatment with INVOKAMET/INVOKAMET XR [see Use in Specific Populations (8.1)]. Instruct females of reproductive potential to report pregnancies to their physicians as soon as possible.
Inform females that treatment with INVOKAMET/INVOKAMET XR may result in ovulation in some premenopausal anovulatory women which may lead to unintended pregnancy [see Use in Specific Populations (8.3)].
Lactation:
Advise women that breastfeeding is not recommended during treatment with INVOKAMET/INVOKAMET XR [see Use in Specific Populations (8.2)].
Administration:
Instruct patients to keep INVOKAMET/INVOKAMET XR in the original bottle to protect from moisture. Advise patients that storage in a pill box or pill organizer is allowed for up to 30 days.
Instruct patients to take INVOKAMET only as prescribed twice daily with food. If a dose is missed, advise patients not to take two doses of INVOKAMET at the same time.
Instruct patients to take INVOKAMET XR only as prescribed once daily with the morning meal. If a dose is missed, advise patients to take it as soon as it is remembered unless it is almost time for the next dose, in which case patients should skip the missed dose and take the medicine at the next regularly scheduled time. Advise patients not to take more than two tablets of INVOKAMET XR at the same time.
Instruct patients that INVOKAMET XR must be swallowed whole and never crushed, cut, or chewed, and that the inactive ingredients may occasionally be eliminated in the feces as a soft mass that may resemble the original tablet.
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
Medication Guide
INVOKAMET® (in vok' a met)
(canagliflozin and metformin hydrochloride) tablets, for oral use
and
INVOKAMET® (in vok' a met) XR
(canagliflozin and metformin hydrochloride) extended-release tablets, for oral useThis Medication Guide has been approved by the U.S. Food and Drug Administration. Revised 01/2020 What is the most important information I should know about INVOKAMET or INVOKAMET XR?
INVOKAMET and INVOKAMET XR can cause serious side effects, including:-
Lactic Acidosis. Metformin, one of the medicines in INVOKAMET and INVOKAMET XR, can cause a rare but serious condition called lactic acidosis (a build-up of lactic acid in the blood) that can cause death. Lactic acidosis is a medical emergency and must be treated in the hospital.
Stop taking INVOKAMET or INVOKAMET XR and call your doctor right away if you have any of the following symptoms of lactic acidosis:
- feel cold in your hands or feet
- feel very weak or tired
- have trouble breathing
- have stomach pains, nausea, or vomiting
- have a slow or irregular heartbeat
- have unusual (not normal) muscle pain
- have unusual sleepiness or sleep longer than usual
- feel dizzy or lightheaded
Most people who have had lactic acidosis had other conditions that, in combination with metformin use, led to the lactic acidosis. Tell your doctor if you have any of the following, because you have a higher chance for getting lactic acidosis with INVOKAMET or INVOKAMET XR if you:
- have severe kidney problems or your kidneys are affected by certain x-ray tests that use injectable dye.
- have liver problems.
- drink alcohol very often or drink a lot of alcohol in short-term "binge" drinking.
- get dehydrated (lose a large amount of body fluids). This can happen if you are sick with a fever, vomiting, or diarrhea. Dehydration can also happen when you sweat a lot with activity or exercise and do not drink enough fluids.
- have surgery.
- have a heart attack, severe infection, or stroke.
- are 65 years of age or older.
The best way to keep from having a problem with lactic acidosis from metformin is to tell your doctor if you have any of the problems in the list above. Your doctor will decide to stop your INVOKAMET or INVOKAMET XR for a while if you have any of these things.
-
Amputations. INVOKAMET or INVOKAMET XR may increase your risk of lower limb amputations. Amputations mainly involve removal of the toe or part of the foot, however, amputations involving the leg, below and above the knee, have also occurred. Some people had more than one amputation, some on both sides of the body.
You may be at a higher risk of lower limb amputation if you:- have a history of amputation
- have heart disease or are at risk for heart disease
- have had blocked or narrowed blood vessels, usually in your leg
- have damage to the nerves (neuropathy) in your leg
- have had diabetic foot ulcers or sores
Call your doctor right away if you have new pain or tenderness, any sores, ulcers, or infections in your leg or foot. Your doctor may decide to stop your INVOKAMET or INVOKAMET XR for a while if you have any of these signs or symptoms.
Talk to your doctor about proper foot care.
INVOKAMET or INVOKAMET XR can have other serious side effects. See "What are the possible side effects of INVOKAMET or INVOKAMET XR?"
What is INVOKAMET or INVOKAMET XR?
- INVOKAMET contains 2 prescription medicines called canagliflozin (INVOKANA) and metformin hydrochloride (GLUCOPHAGE). INVOKAMET XR contains 2 prescription medicines called canagliflozin (INVOKANA) and metformin hydrochloride extended-release (GLUMETZA). INVOKAMET or INVOKAMET XR can be used:
- along with diet and exercise to lower blood sugar (glucose) in adults with type 2 diabetes.
- in adults with type 2 diabetes who have known cardiovascular disease and canagliflozin is needed to reduce the risk of major cardiovascular events such as heart attack, stroke, or death.
- in adults with type 2 diabetes and diabetic kidney disease (nephropathy) with a certain amount of protein in the urine, and canagliflozin is needed to reduce the risk of end stage kidney disease (ESKD), worsening of kidney function, cardiovascular death, and hospitalization for heart failure.
- INVOKAMET or INVOKAMET XR is not for people with type 1 diabetes.
- INVOKAMET or INVOKAMET XR is not for people with diabetic ketoacidosis (increased ketones in blood or urine).
- It is not known if INVOKAMET or INVOKAMET XR is safe and effective in children under 18 years of age.
Do not take INVOKAMET or INVOKAMET XR if you:
- have severe kidney problems
- are on kidney dialysis
- have a condition called metabolic acidosis or diabetic ketoacidosis (increased ketones in the blood or urine).
- are allergic to canagliflozin, metformin, or any of the ingredients in INVOKAMET or INVOKAMET XR. See the end of this Medication Guide for a list of ingredients in INVOKAMET and INVOKAMET XR. Symptoms of an allergic reaction to INVOKAMET and INVOKAMET XR may include:
- rash
- raised red patches on your skin (hives)
- swelling of the face, lips, mouth, tongue, and throat that may cause difficulty in breathing or swallowing
Before taking INVOKAMET or INVOKAMET XR, tell your doctor about all of your medical conditions, including if you:
- have a history of amputation.
- have heart disease or are at risk for heart disease.
- have had blocked or narrowed blood vessels, usually in your leg.
- have damage to the nerves (neuropathy) in your leg.
- have had diabetic foot ulcers or sores.
- have moderate to severe kidney problems.
- have liver problems.
- have a history of urinary tract infections or problems with urination.
- are on a low sodium (salt) diet. Your doctor may change your diet or your dose of INVOKAMET or INVOKAMET XR.
- have ever had an allergic reaction to INVOKAMET or INVOKAMET XR.
- are going to get an injection of dye or contrast agents for an x-ray procedure. INVOKAMET or INVOKAMET XR may need to be stopped for a short time. Talk to your doctor about when you should stop INVOKAMET or INVOKAMET XR and when you should start INVOKAMET or INVOKAMET XR again. See "What is the most important information I should know about INVOKAMET or INVOKAMET XR?"
- have heart problems, including congestive heart failure.
- are going to have surgery. Your doctor may stop your INVOKAMET or INVOKAMET XR before you have surgery. Talk to your doctor if you are having surgery about when to stop taking INVOKAMET or INVOKAMET XR and when to start it again.
- are eating less or there is a change in your diet.
- have or have had problems with your pancreas, including pancreatitis or surgery on your pancreas.
- drink alcohol very often or drink a lot of alcohol in the short-term ("binge" drinking).
- have low levels of vitamin B12 or calcium in your blood.
- are pregnant or plan to become pregnant. INVOKAMET or INVOKAMET XR may harm your unborn baby. If you become pregnant while taking INVOKAMET or INVOKAMET XR, tell your doctor as soon as possible. Talk with your doctor about the best way to control your blood sugar while you are pregnant.
- are a premenopausal woman (before the "change of life"), who does not have periods regularly or at all. INVOKAMET or INVOKAMET XR may increase your chance of becoming pregnant. Talk to your doctor about birth control choices while taking INVOKAMET or INVOKAMET XR, if you are not planning to become pregnant. Tell your doctor right away if you become pregnant while taking INVOKAMET or INVOKAMET XR.
- are breastfeeding or plan to breastfeed. INVOKAMET or INVOKAMET XR may pass into your breast milk and may harm your baby. Talk with your doctor about the best way to feed your baby if you are taking INVOKAMET or INVOKAMET XR. Do not breastfeed while taking INVOKAMET or INVOKAMET XR.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
INVOKAMET or INVOKAMET XR may affect the way other medicines work and other medicines may affect how INVOKAMET or INVOKAMET XR works. Especially tell your doctor if you take:
- diuretics (water pills)
- phenytoin or phenobarbital (used to control seizures)
- digoxin (used to treat heart problems)
- rifampin (used to treat or prevent tuberculosis)
- ritonavir (used to treat HIV infection)
Ask your doctor or pharmacist for a list of these medicines if you are not sure if your medicine is listed above.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist when you get a new medicine.
How should I take INVOKAMET or INVOKAMET XR?
- If you are prescribed INVOKAMET, take by mouth 2 times each day with meals exactly as your doctor tells you to take it. Taking INVOKAMET with meals may lower your chance of having an upset stomach.
- If you are prescribed INVOKAMET XR, take by mouth 1 time each day with the morning meal exactly as your doctor tells you to take it. Taking INVOKAMET XR with a meal may lower your chance of having an upset stomach.
- Swallow INVOKAMET XR whole. Do not crush, cut, or chew.
- You may sometimes pass a soft mass in your stools (bowel movement) that looks like INVOKAMET XR tablets. It is normal to see this in your stool.
- Your doctor may change your dose if needed.
- Your doctor may tell you to take INVOKAMET or INVOKAMET XR along with other diabetes medicines. Low blood sugar can happen more often when INVOKAMET or INVOKAMET XR is taken with certain other diabetes medicines. See "What are the possible side effects of INVOKAMET or INVOKAMET XR?"
- If you miss a dose of INVOKAMET, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose and take the medicine at the next regularly scheduled time. Do not take 2 tablets of INVOKAMET at the same time. Talk to your doctor if you have questions about a missed dose.
- If you miss a dose of INVOKAMET XR, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose and take the medicine at the next regularly scheduled time. Do not take more than 2 tablets of INVOKAMET XR at the same time. Talk to your doctor if you have questions about a missed dose.
- If you take too much INVOKAMET or INVOKAMET XR, call your doctor or go to the nearest hospital emergency room right away.
- When your body is under some types of stress, such as fever, trauma (such as a car accident), infection, or surgery, the amount of diabetes medicine you need may change. Tell your doctor right away if you have any of these conditions and follow your doctor's instructions.
- Stay on your prescribed diet and exercise program while taking INVOKAMET or INVOKAMET XR.
- Check your blood sugar as your doctor tells you to.
- INVOKAMET and INVOKAMET XR will cause your urine to test positive for glucose.
- Your doctor may do certain blood tests before you start INVOKAMET or INVOKAMET XR and during treatment as needed. Your doctor may change your dose of INVOKAMET or INVOKAMET XR based on the results of your blood tests.
- Your doctor will check your diabetes with regular blood tests, including your blood sugar levels and your hemoglobin A1C.
What should I avoid while taking INVOKAMET or INVOKAMET XR?
- Avoid drinking alcohol very often or drinking a lot of alcohol in a short period of time ("binge" drinking). It can increase your chances of getting serious side effects.
What are the possible side effects of INVOKAMET or INVOKAMET XR?
INVOKAMET or INVOKAMET XR may cause serious side effects including:
- See "What is the most important information I should know about INVOKAMET or INVOKAMET XR?"
- dehydration. INVOKAMET or INVOKAMET XR can cause some people to become dehydrated (the loss of too much body water). Dehydration may cause you to feel dizzy, faint, lightheaded, or weak, especially when you stand up (orthostatic hypotension).
You may be at higher risk of dehydration if you:
- have low blood pressure
- take medicines to lower your blood pressure, including diuretics (water pill)
- are on a low sodium (salt) diet
- have kidney problems
- are 65 years of age or older
Talk to your doctor about what you can do to prevent dehydration including how much fluid you should drink on a daily basis.
- ketoacidosis (increased ketones in your blood or urine). Ketoacidosis has happened in people who have type 1 diabetes or type 2 diabetes, during treatment with canagliflozin, one of the medicines in INVOKAMET and INVOKAMET XR. Ketoacidosis has also happened in people with diabetes who were sick or who had surgery during treatment with INVOKAMET or INVOKAMET XR. Ketoacidosis is a serious condition, which needs to be treated in a hospital. Ketoacidosis may lead to death. Ketoacidosis can happen with INVOKAMET or INVOKAMET XR, even if your blood sugar is less than 250 mg/dL. Stop taking INVOKAMET or INVOKAMET XR and call your doctor right away if you get any of the following symptoms:
- nausea
- vomiting
- stomach area (abdominal) pain
- tiredness
- trouble breathing
If you get any of these symptoms during treatment with INVOKAMET or INVOKAMET XR, if possible, check for ketones in your urine, even if your blood sugar is less than 250 mg/dL. -
kidney problems. Sudden kidney injury has happened to people taking INVOKAMET or INVOKAMET XR. Talk to your doctor right away if you:
- reduce the amount of food or liquid you drink for example, if you are sick or cannot eat or
- you start to lose liquids from your body for example, from vomiting, diarrhea or being in the sun too long.
- serious urinary tract infections. Serious urinary tract infections that may lead to hospitalization have happened in people who are taking canagliflozin, one of the medicines in INVOKAMET and INVOKAMET XR. Tell your doctor if you have any signs or symptoms of a urinary tract infection such as a burning feeling when passing urine, a need to urinate often, the need to urinate right away, pain in the lower part of your stomach (pelvis), or blood in the urine. Sometimes people may also have a fever, back pain, nausea, or vomiting.
- low blood sugar (hypoglycemia). If you take INVOKAMET or INVOKAMET XR with another medicine that can cause low blood sugar, such as a sulfonylurea or insulin, your risk of getting low blood sugar is higher. The dose of your sulfonylurea medicine or insulin may need to be lowered while you take INVOKAMET or INVOKAMET XR. Signs and symptoms of low blood sugar may include:
- headache
- confusion
- hunger
- shaking or feeling jittery
- drowsiness
- dizziness
- fast heartbeat
- weakness
- irritability
- sweating
- a rare but serious bacterial infection that causes damage to the tissue under the skin (necrotizing fasciitis) in the area between and around the anus and genitals (perineum). Necrotizing fasciitis of the perineum has happened in women and men who take canagliflozin, one of the medicines in INVOKAMET and INVOKAMET XR. Necrotizing fasciitis of the perineum may lead to hospitalization, may require multiple surgeries, and may lead to death. Seek medical attention immediately if you have a fever or you are feeling very weak, tired or uncomfortable (malaise) and you develop any of the following symptoms in the area between and around your anus and genitals:
- pain or tenderness
- swelling
- redness of the skin (erythema)
-
vaginal yeast infection. Women who take INVOKAMET or INVOKAMET XR may get vaginal yeast infections. Symptoms of a vaginal yeast infection include:
- vaginal odor
- white or yellowish vaginal discharge (discharge may be lumpy or look like cottage cheese)
- vaginal itching
- yeast infection of the penis (balanitis or balanoposthitis). Men who take INVOKAMET or INVOKAMET XR may get a yeast infection of the skin around the penis. Certain men who are not circumcised may have swelling of the penis that makes it difficult to pull back the skin around the tip of the penis. Other symptoms of yeast infection of the penis include:
- redness, itching, or swelling of the penis
- foul smelling discharge from the penis
- rash of the penis
- pain in the skin around the penis
Talk to your doctor about what to do if you get symptoms of a yeast infection of the vagina or penis. Your doctor may suggest you use an over-the-counter antifungal medicine. Talk to your doctor right away if you use an over-the-counter antifungal medication and your symptoms do not go away.
- serious allergic reaction. If you have any symptoms of a serious allergic reaction, stop taking INVOKAMET or INVOKAMET XR and call your doctor right away or go to the nearest hospital emergency room. See "Do not take INVOKAMET or INVOKAMET XR if you:". Your doctor may give you a medicine for your allergic reaction and prescribe a different medicine for your diabetes.
- broken bones (fractures). Bone fractures have been seen in patients taking canagliflozin. Talk to your doctor about factors that may increase your risk of bone fracture.
- low vitamin B12 (vitamin B12 deficiency). Using metformin for long periods of time may cause a decrease in the amount of vitamin B12 in your blood, especially if you have had low vitamin B12 blood levels before. Your doctor may do blood tests to check your vitamin B12 levels.
Other common side effects of INVOKAMET or INVOKAMET XR include:
- nausea and vomiting
- gas
- diarrhea
- upset stomach
- weakness
- indigestion
- headache
- changes in urination, including urgent need to urinate more often, in larger amounts, or at night
These are not all the possible side effects of INVOKAMET or INVOKAMET XR.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Janssen Pharmaceuticals, Inc. at 1-800-526-7736.
How should I store INVOKAMET or INVOKAMET XR?
- Store INVOKAMET or INVOKAMET XR at room temperature between 68°F to 77°F (20°C to 25°C).
- Store INVOKAMET or INVOKAMET XR in the original container to protect from moisture. Storage in a pill box or pill organizer is allowed for up to 30 days.
Keep INVOKAMET and INVOKAMET XR and all medicines out of the reach of children.
General information about the safe and effective use of INVOKAMET or INVOKAMET XR.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use INVOKAMET or INVOKAMET XR for a condition for which it was not prescribed. Do not give INVOKAMET or INVOKAMET XR to other people, even if they have the same symptoms you have. It may harm them.
You can ask your pharmacist or doctor for information about INVOKAMET or INVOKAMET XR that is written for healthcare professionals.
For more information about INVOKAMET or INVOKAMET XR, call 1-800-526-7736 or visit our websites at www.invokamet.com or www.invokametxr.com.
What are the ingredients of INVOKAMET?
Active ingredients: canagliflozin and metformin hydrochloride
Inactive ingredients: The tablet core contains croscarmellose sodium, hypromellose, magnesium stearate, and microcrystalline cellulose. The magnesium stearate is vegetable-sourced. In addition, the tablet coating contains Macrogol/PEG3350, polyvinyl alcohol (partially hydrolyzed), talc, titanium dioxide, iron oxide yellow (50 mg/1,000 mg and 150 mg/500 mg tablets only), iron oxide red (50 mg/1,000 mg, 150 mg/500 mg and 150 mg/1,000 mg tablets only), and iron oxide black (150 mg/1,000 mg tablets only).
What are the ingredients of INVOKAMET XR?
Active ingredients: canagliflozin and metformin hydrochloride
Inactive ingredients: The tablet core contains croscarmellose sodium, hydroxypropyl cellulose, hypromellose, lactose anhydrous, magnesium stearate (vegetable-sourced), microcrystalline cellulose, polyethylene oxide, and silicified microcrystalline cellulose (50 mg/500 mg and 50 mg/1,000 mg tablets only). In addition, the tablet coating contains macrogol/PEG3350, polyvinyl alcohol (partially hydrolyzed), talc, titanium dioxide, iron oxide red, iron oxide yellow, and iron oxide black (50 mg/1,000 mg and 150 mg/1,000 mg tablets only).
Manufactured for: Janssen Pharmaceuticals, Inc., Titusville, NJ 08560. Licensed from Mitsubishi Tanabe Pharma Corporation. © 2014 – 2020 Janssen Pharmaceutical Companies
For more information about INVOKAMET or INVOKAMET XR, call 1-800-526-7736 or visit our websites at www.invokamet.com or www.invokametxr.com.
-
Lactic Acidosis. Metformin, one of the medicines in INVOKAMET and INVOKAMET XR, can cause a rare but serious condition called lactic acidosis (a build-up of lactic acid in the blood) that can cause death. Lactic acidosis is a medical emergency and must be treated in the hospital.
- PRINCIPAL DISPLAY PANEL - 50 mg/500 mg Tablet Bottle Label - 540
- PRINCIPAL DISPLAY PANEL - 50 mg/1,000 mg Tablet Bottle Label - 541
- PRINCIPAL DISPLAY PANEL - 150 mg/500 mg Tablet Bottle Label - 542
- PRINCIPAL DISPLAY PANEL - 150 mg/1,000 mg Tablet Bottle Label - 543
- PRINCIPAL DISPLAY PANEL - 50 mg/500 mg Tablet Bottle Label - 940
- PRINCIPAL DISPLAY PANEL - 50 mg/1000 mg Tablet Bottle Label - 941
- PRINCIPAL DISPLAY PANEL - 150 mg/500 mg Tablet Bottle Label - 942
- PRINCIPAL DISPLAY PANEL - 150 mg/1000 mg Tablet Bottle Label - 943
-
INGREDIENTS AND APPEARANCE
INVOKAMET
canagliflozin and metformin hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-540 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 50 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 500 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) water (UNII: 059QF0KO0R) polyethylene glycol 3350 (UNII: G2M7P15E5P) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) Product Characteristics Color WHITE Score no score Shape OVAL (capsule shaped) Size 17mm Flavor Imprint Code CM;155 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-540-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 2 NDC: 50458-540-91 10 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 08/09/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204353 08/08/2014 INVOKAMET
canagliflozin and metformin hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-541 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 50 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 1000 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) water (UNII: 059QF0KO0R) polyethylene glycol 3350 (UNII: G2M7P15E5P) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide yellow (UNII: EX438O2MRT) ferric oxide red (UNII: 1K09F3G675) Product Characteristics Color BROWN (beige) Score no score Shape OVAL (capsule shaped) Size 21mm Flavor Imprint Code CM;551 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-541-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 2 NDC: 50458-541-91 10 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 08/09/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204353 08/08/2014 INVOKAMET
canagliflozin and metformin hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-542 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 150 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 500 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) water (UNII: 059QF0KO0R) polyethylene glycol 3350 (UNII: G2M7P15E5P) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide yellow (UNII: EX438O2MRT) ferric oxide red (UNII: 1K09F3G675) Product Characteristics Color YELLOW Score no score Shape OVAL (capsule shaped) Size 18mm Flavor Imprint Code CM;215 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-542-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 2 NDC: 50458-542-91 10 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 08/09/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204353 08/08/2014 INVOKAMET
canagliflozin and metformin hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-543 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 150 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 1000 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) water (UNII: 059QF0KO0R) polyethylene glycol 3350 (UNII: G2M7P15E5P) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) ferrosoferric oxide (UNII: XM0M87F357) Product Characteristics Color PURPLE Score no score Shape OVAL (capsule shaped) Size 22mm Flavor Imprint Code CM;611 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-543-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 2 NDC: 50458-543-91 10 in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2014 08/09/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204353 08/08/2014 INVOKAMET XR
canagliflozin and metformin hydrochloride tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-940 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 50 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 500 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) hypromellose, unspecified (UNII: 3NXW29V3WO) anhydrous lactose (UNII: 3SY5LH9PMK) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) ferric oxide yellow (UNII: EX438O2MRT) Product Characteristics Color WHITE (almost white to light orange) Score no score Shape OVAL (oblong, biconvex) Size 21mm Flavor Imprint Code CM1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-940-01 60 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 2 NDC: 50458-940-02 10 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 09/21/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205879 09/20/2016 INVOKAMET XR
canagliflozin and metformin hydrochloride tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-941 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 50 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 1000 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) hypromellose, unspecified (UNII: 3NXW29V3WO) anhydrous lactose (UNII: 3SY5LH9PMK) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) ferric oxide yellow (UNII: EX438O2MRT) ferrosoferric oxide (UNII: XM0M87F357) Product Characteristics Color PINK Score no score Shape OVAL (oblong, biconvex) Size 22mm Flavor Imprint Code CM3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-941-01 60 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 2 NDC: 50458-941-02 10 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 09/21/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205879 09/20/2016 INVOKAMET XR
canagliflozin and metformin hydrochloride tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-942 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 150 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 500 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) hypromellose, unspecified (UNII: 3NXW29V3WO) anhydrous lactose (UNII: 3SY5LH9PMK) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) ferric oxide yellow (UNII: EX438O2MRT) Product Characteristics Color ORANGE Score no score Shape OVAL (oblong, biconvex) Size 21mm Flavor Imprint Code CM2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-942-01 60 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 2 NDC: 50458-942-02 10 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 09/21/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205879 09/20/2016 INVOKAMET XR
canagliflozin and metformin hydrochloride tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-943 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength canagliflozin (UNII: 0SAC974Z85) (canagliflozin anhydrous - UNII:6S49DGR869) canagliflozin anhydrous 150 mg metformin hydrochloride (UNII: 786Z46389E) (metformin - UNII:9100L32L2N) metformin hydrochloride 1000 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) hypromellose, unspecified (UNII: 3NXW29V3WO) anhydrous lactose (UNII: 3SY5LH9PMK) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) polyvinyl alcohol, unspecified (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) ferric oxide yellow (UNII: EX438O2MRT) ferrosoferric oxide (UNII: XM0M87F357) Product Characteristics Color BROWN (reddish brown) Score no score Shape OVAL (oblong, biconvex) Size 22mm Flavor Imprint Code CM4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-943-01 60 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 2 NDC: 50458-943-02 10 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 09/21/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205879 09/20/2016 Labeler - Janssen Pharmaceuticals, Inc. (063137772) Establishment Name Address ID/FEI Business Operations Janssen Ortho LLC 805887986 MANUFACTURE(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) , ANALYSIS(50458-540, 50458-940, 50458-941, 50458-942, 50458-943) , PACK(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) , LABEL(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Janssen Pharmaceutica NV 400345889 API MANUFACTURE(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) , ANALYSIS(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Farmhispania 465582486 API MANUFACTURE(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) , ANALYSIS(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Granules India Limited 918457644 API MANUFACTURE(50458-540, 50458-541, 50458-542, 50458-543) , ANALYSIS(50458-540, 50458-541, 50458-542, 50458-543) Establishment Name Address ID/FEI Business Operations Janssen Pharmaceutica NV 370005019 ANALYSIS(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Janssen Pharmaceuticals Inc 063137772 ANALYSIS(50458-540, 50458-541, 50458-542, 50458-543, 50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Ajinomoto OmniChem 400344443 API MANUFACTURE(50458-540, 50458-541, 50458-542, 50458-543) Establishment Name Address ID/FEI Business Operations Janssen Cilag SpA 542797928 MANUFACTURE(50458-540, 50458-541, 50458-542, 50458-543) , PACK(50458-540, 50458-541, 50458-542, 50458-543) Establishment Name Address ID/FEI Business Operations Rolabo Outsourcing S.L. (Spain) 513603225 API MANUFACTURE(50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Johnson & Johnson Private Limited (Mumbai) 677603030 ANALYSIS(50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations Patheon Puerto Rico, Inc. (Manati) 143814544 MANUFACTURE(50458-940, 50458-941, 50458-942, 50458-943) , API MANUFACTURE(50458-940, 50458-941, 50458-942, 50458-943) Establishment Name Address ID/FEI Business Operations PHAST Gesellschaft für Pharmazeutische Qualitätsstandards mbH 331156161 ANALYSIS(50458-940, 50458-941, 50458-942, 50458-943)
Trademark Results [INVOKAMET XR]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 INVOKAMET XR 86277042 5134045 Live/Registered |
JOHNSON & JOHNSON 2014-05-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.