HISTEX™ Chewable Tablets
HISTEX by
Drug Labeling and Warnings
HISTEX by is a Otc medication manufactured, distributed, or labeled by Allegis Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
HISTEX- triprolidine hydrochloride tablet, chewable
Allegis Pharmaceuticals, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
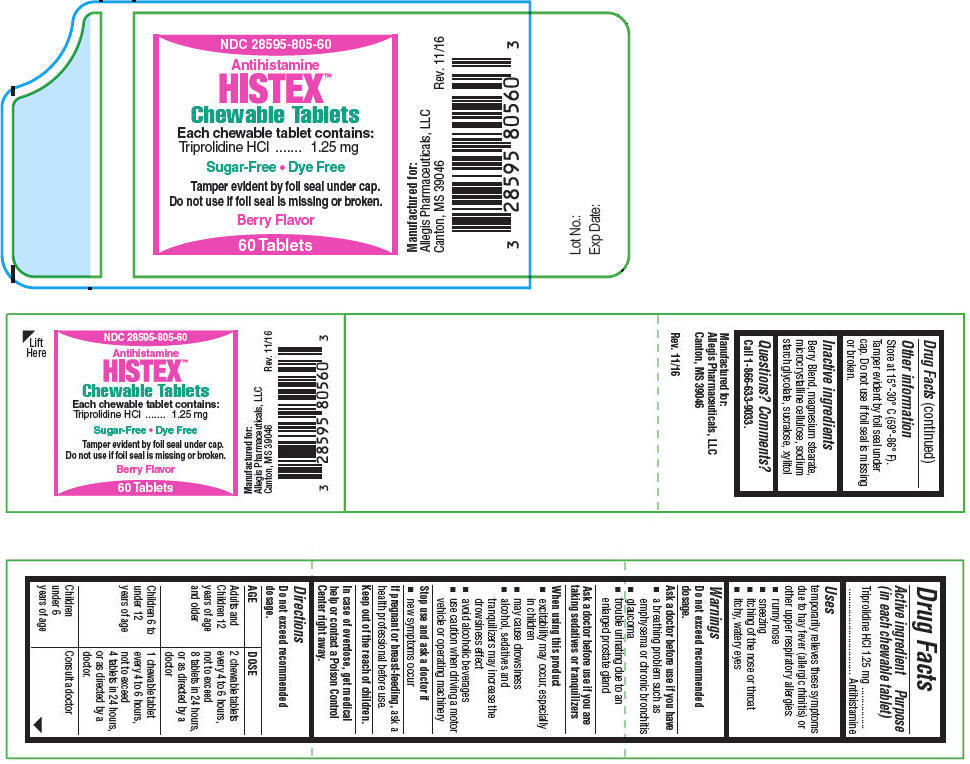
HISTEX™ Chewable Tablets
Uses
temporarily relieves these symptoms due to hay fever (allergic rhinitis) or other upper respiratory allergies:
- runny nose
- sneezing
- itching of the nose or throat
- itchy, watery eyes
Warnings
Do not exceed recommended dosage.
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Directions
Do not exceed recommended dosage.
| AGE | DOSE |
|---|---|
| Adults and Children 12 years of age and older | 2 chewable tablets every 4 to 6 hours, not to exceed 8 tablets in 24 hours, or as directed by a doctor |
| Children 6 to under 12 years of age | 1 chewable tablet every 4 to 6 hours, not to exceed 4 tablets in 24 hours, or as directed by a doctor. |
| Children under 6 years of age | Consult a doctor |
Other information
Store at 15°-30° C (59°-86° F).
Tamper evident by foil seal under cap. Do not use if foil seal is missing or broken.
Inactive ingredients
Berry Blend, magnesium stearate, microcrystalline cellulose, sodium starch glycolate, sucralose, xylitol
PRINCIPAL DISPLAY PANEL - 60 Tablet Bottle Label
NDC: 28595-805-60
Antihistamine
HISTEX™
Chewable Tablets
Each chewable tablet contains:
Triprolidine HCl
1.25 mg
Sugar-Free Dye Free
Tamper evident by foil seal under cap.
Do not use if foil seal is missing or broken.
Berry Flavor
60 Tablets
| HISTEX
triprolidine hydrochloride tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Allegis Pharmaceuticals, LLC (792272861) |