Sodium Chloride Inhalation Solution
Sodium Chloride by
Drug Labeling and Warnings
Sodium Chloride by is a Other medication manufactured, distributed, or labeled by Lifestar Pharma LLC, Mankind Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
SODIUM CHLORIDE- sodium chloride
Lifestar Pharma LLC
----------
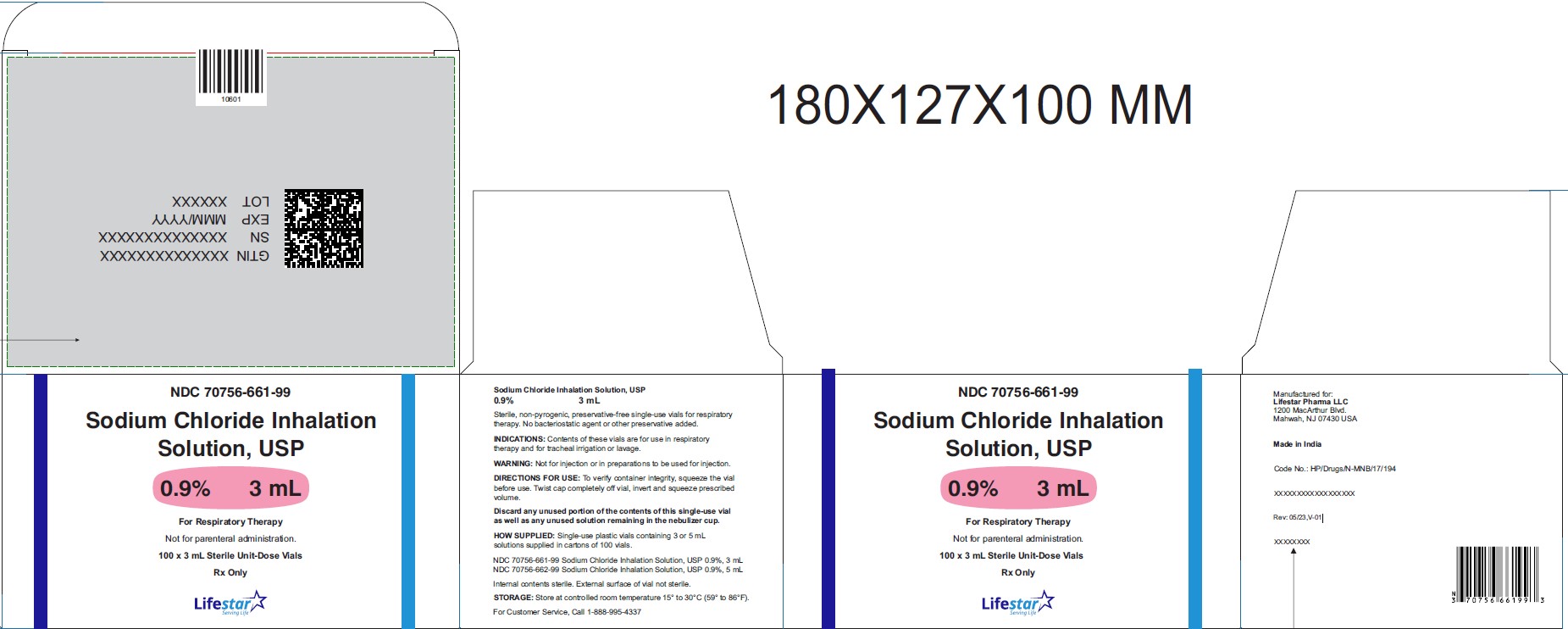
Sodium Chloride Inhalation Solution
Sodium Chloride Inhalation Solution 0.9% 3 mL
NDC: 70756-661-99
Sodium Chloride Inhalation
Solution, USP
0.9% 3 mL
For Respiratory Therapy
Not for Parenteral Administration.
100 x 3 mL Sterile Unit-Dose Vials
Rx Only
Sodium Chloride Inhalation Solution, USP
0.9% 3 mL
Sterile, non-pyrogenic, preservative-free single-use vials for respiratory therapy. No bacteriostatic agent or other preservative added.
INDICATIONS: Contents of these vials are for use in respiratory therapy and for tracheal irrigation or lavage.
WARNING: Not for injection or in preparations to be used for injection.
DIRECTIONS FOR USE: To verify container integrity, squeeze the vial before use. Twist cap completely off vial, invert and squeeze prescribed volume.
Discard any unused portion of the contents of this single-use vial as well as any unused solution remaining in the nebulizer cup.
HOW SUPPLIED: Single-use plastic vials containing 3 mL or 5 mL solutions supplied in cartons of 100 vials and 15 mL solution supplied in cartons of 50 vials.
NDC: 70756-661-99 Sodium Chloride Inhalation Solution, USP 0.9%, 3 mL
NDC: 70756-662-99 Sodium Chloride Inhalation Solution, USP 0.9%, 5 mL
Internal contents sterile. External surface of vial not sterile.
STORAGE: Store at controlled room temperature 15° to 30°C (59° to 86°F).
For Customer Service, Call 1-888-995-4337
Manufactured for:
Lifestar Pharma LLC
1200 MacArthur Blvd.
Mahwah, NJ 07430 USA
Made in India
Rev: 05/23,V-01
| SODIUM CHLORIDE
nebulizer (direct patient interface) |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Lifestar Pharma LLC (080268943) |
| Registrant - Mankind Pharma Limited (915834068) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Mankind Pharma Limited | 916512493 | manufacture, analysis, pack | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.