SOMATULINE DEPOT- lanreotide acetate injection
SOMATULINE DEPOT by
Drug Labeling and Warnings
SOMATULINE DEPOT by is a Prescription medication manufactured, distributed, or labeled by Ipsen Biopharmaceuticals, Inc., IPSEN PHARMA BIOTECH. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use SOMATULINE DEPOT safely and effectively. See full prescribing information for SOMATULINE DEPOT.
SOMATULINE® DEPOT (lanreotide) injection, for subcutaneous use
Initial U.S. Approval: 2007RECENT MAJOR CHANGES
Warnings & Precautions, Cholelithiasis and Complications of Cholelithiasis (5.1) 4/2019 INDICATIONS AND USAGE
SOMATULINE DEPOT is a somatostatin analog indicated for:
- the long-term treatment of acromegalic patients who have had an inadequate response to or cannot be treated with surgery and/or radiotherapy. (1.1)
- the treatment of adult patients with unresectable, well- or moderately-differentiated, locally advanced or metastatic gastroenteropancreatic neuroendocrine tumors (GEP-NETs) to improve progression-free survival. (1.2)
- the treatment of adults with carcinoid syndrome; when used, it reduces the frequency of short-acting somatostatin analog rescue therapy. (1.3)
DOSAGE AND ADMINISTRATION
Administration (2.1):
- For deep subcutaneous injection only.
- Intended for administration by a healthcare provider.
- Administer in the superior external quadrant of the buttock.
- Alternate injection sites.
Recommended Dosage (2.1)
- Acromegaly: 90 mg every 4 weeks for 3 months. Adjust thereafter based on GH and/or IGF-1 levels. See full prescribing information for titration regimen.
- GEP-NETs: 120 mg every 4 weeks.
- Carcinoid Syndrome: 120 mg every 4 weeks. If patients are already being treated with SOMATULINE DEPOT for GEP-NET, do not administer an additional dose for carcinoid syndrome.
Dosage Adjustment:
DOSAGE FORMS AND STRENGTHS
Injection: 60 mg/0.2 mL, 90 mg/0.3 mL, and 120 mg/0.5 mL single-dose prefilled syringes (3)
CONTRAINDICATIONS
Hypersensitivity to lanreotide. (4)
WARNINGS AND PRECAUTIONS
- Cholelithiasis and Complications of Cholelithiasis: Monitor periodically. Discontinue if complications of cholelithiasis are suspected. Gallstones may occur; consider periodic monitoring. (5.1)
- Hyperglycemia and Hypoglycemia: Glucose monitoring is recommended and antidiabetic treatment adjusted accordingly. (5.2, 7.1)
- Cardiovascular Abnormalities: Decrease in heart rate may occur. Use with caution in at-risk patients. (5.3)
- Thyroid Function Abnormalities: Decreases in thyroid function may occur; perform tests where clinically indicated. (5.4)
ADVERSE REACTIONS
Most common adverse reactions are:
- Acromegaly (>5%): diarrhea, cholelithiasis, abdominal pain, nausea and injection site reactions. (6.1)
- GEP-NET (>10%): abdominal pain, musculoskeletal pain, vomiting, headache, injection site reaction, hyperglycemia, hypertension, and cholelithiasis. (6.1)
- Carcinoid Syndrome: (≥5% and at least 5% greater than placebo): headache, dizziness and muscle spasm. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Ipsen Biopharmaceuticals, Inc. at 1-855-463-5127 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
DRUG INTERACTIONS
- Cyclosporine: SOMATULINE DEPOT may decrease the absorption of cyclosporine. Dosage adjustment of cyclosporine may be needed. (7.2)
- Bromocriptine: SOMATULINE DEPOT may increase the absorption of bromocriptine. (7.3)
- Bradycardia-Inducing Drugs (e.g., beta-blockers): SOMATULINE DEPOT may decrease heart rate. Dosage adjustment of the coadministered drug may be necessary. (7.3)
USE IN SPECIFIC POPULATIONS
Lactation: Advise women not to breastfeed during treatment and for 6 months after the last dose. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 6/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Acromegaly
1.2 Gastroenteropancreatic Neuroendocrine Tumors
1.3 Carcinoid Syndrome
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
2.2 Recommended Dosage
2.3 Dosage Adjustment in Renal Impairment
2.4 Dosage Adjustment in Hepatic Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Cholelithiasis and Complications of Cholelithiasis
5.2 Hyperglycemia and Hypoglycemia
5.3 Cardiovascular Abnormalities
5.4 Thyroid Function Abnormalities
5.5 Monitoring: Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
6.3 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Insulin and Oral Hypoglycemic Drugs
7.2 Cyclosporine
7.3 Bromocriptine
7.4 Bradycardia-Inducing Drugs
7.5 Drug Metabolism Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Acromegaly
14.2 Gastroenteropancreatic Neuroendocrine Tumors
14.3 Carcinoid Syndrome
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Acromegaly
SOMATULINE DEPOT is indicated for the long-term treatment of acromegalic patients who have had an inadequate response to surgery and/or radiotherapy, or for whom surgery and/or radiotherapy is not an option.
The goal of treatment in acromegaly is to reduce growth hormone (GH) and insulin growth factor-1 (IGF-1) levels to normal.
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
- For deep subcutaneous injection only.
- SOMATULINE DEPOT is intended for administration by a healthcare provider.
Preparation
- Remove SOMATULINE DEPOT from the refrigerator 30 minutes prior to administration and allow to come to room temperature.
- Keep pouch sealed until just prior to injection.
- Product left in its sealed pouch at room temperature (not to exceed 104°F or 40°C) for up to 24 hours may be returned to the refrigerator for continued storage and use at a later time.
- Prior to administration, inspect the SOMATULINE DEPOT syringe visually for particulate matter and discoloration. Do not administer if particulate matter or discoloration is observed. The content of the prefilled syringe is a semi-solid phase having a gel-like appearance, with viscous characteristics and a color varying from white to pale yellow. The supersaturated solution can also contain micro bubbles that can clear up during injection. These differences are normal and do not interfere with the quality of the product.
2.2 Recommended Dosage
Acromegaly
The recommended starting dosage of SOMATULINE DEPOT is 90 mg given via the deep subcutaneous route, at 4-week intervals for 3 months.
After 3 months, the dosage may be adjusted as follows:
- GH greater than 1 ng/mL to less than or equal to 2.5 ng/mL, IGF-1 normal, and clinical symptoms controlled: maintain SOMATULINE DEPOT dosage at 90 mg every 4 weeks.
- GH greater than 2.5 ng/mL, IGF-1 elevated, and/or clinical symptoms uncontrolled: increase SOMATULINE DEPOT dosage to 120 mg every 4 weeks.
- GH less than or equal to 1 ng/mL, IGF-1 normal, and clinical symptoms controlled: reduce SOMATULINE DEPOT dosage to 60 mg every 4 weeks.
Thereafter, the dosage should be adjusted according to the response of the patient as judged by a reduction in serum GH and/or IGF-1 levels; and/or changes in symptoms of acromegaly.
Patients who are controlled on SOMATULINE DEPOT 60 or 90 mg may be considered for an extended dosing interval of SOMATULINE DEPOT 120 mg every 6 or 8 weeks. GH and IGF-1 levels should be obtained 6 weeks after this change in dosing regimen to evaluate persistence of patient response.
Continued monitoring of patient response with dosage adjustments for biochemical and clinical symptom control, as necessary, is recommended.
2.3 Dosage Adjustment in Renal Impairment
Acromegaly
The recommended starting dosage of SOMATULINE DEPOT in acromegalic patients with moderate or severe renal impairment (creatinine clearance less than 60 mL/min) is 60 mg via the deep subcutaneous route at 4-week intervals for 3 months followed by dosage adjustment [see Dosage and Administration (2.2), Use in Specific Populations (8.6)].
2.4 Dosage Adjustment in Hepatic Impairment
Acromegaly
The recommended starting dosage of SOMATULINE DEPOT in acromegalic patients with moderate or severe hepatic impairment (Child-Pugh Class B or C) is 60 mg via the deep subcutaneous route at 4-week intervals for 3 months followed by dosage adjustment [see Dosage and Administration (2.2), Use in Specific Populations (8.7)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
SOMATULINE DEPOT is contraindicated in patients with history of a hypersensitivity to lanreotide. Allergic reactions (including angioedema and anaphylaxis) have been reported following administration of lanreotide [see Adverse Reactions (6.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Cholelithiasis and Complications of Cholelithiasis
SOMATULINE DEPOT may reduce gallbladder motility and lead to gallstone formation; therefore, patients may need to be monitored periodically [see Adverse Reactions (6.1), Clinical Pharmacology (12.2)]. There have been postmarketing reports of cholelithiasis (gallstones) resulting in complications, including cholecystitis, cholangitis, and pancreatitis, and requiring cholecystectomy in patients taking SOMATULINE DEPOT. If complications of cholelithiasis are suspected, discontinue SOMATULINE DEPOT and treat appropriately.
5.2 Hyperglycemia and Hypoglycemia
Pharmacological studies in animals and humans show that lanreotide, like somatostatin and other somatostatin analogs, inhibits the secretion of insulin and glucagon. Hence, patients treated with SOMATULINE DEPOT may experience hypoglycemia or hyperglycemia. Blood glucose levels should be monitored when lanreotide treatment is initiated, or when the dose is altered, and antidiabetic treatment should be adjusted accordingly [see Adverse Reactions (6.1)].
5.3 Cardiovascular Abnormalities
The most common overall cardiac adverse reactions observed in three pooled SOMATULINE DEPOT cardiac studies in patients with acromegaly were sinus bradycardia (12/217, 5.5%), bradycardia (6/217, 2.8%), and hypertension (12/217, 5.5%) [see Adverse Reactions (6.1)].
In 81 patients with baseline heart rates of 60 beats per minute (bpm) or greater treated with SOMATULINE DEPOT in Study 3, the incidence of heart rate less than 60 bpm was 23% (19/81) as compared to 16% (15/94) of placebo treated patients; 10 patients (12%) had documented heart rates less than 60 bpm on more than one visit. The incidence of documented episodes of heart rate less than 50 bpm as well as the incidence of bradycardia reported as an adverse event was 1% in each treatment group. Initiate appropriate medical management in patients who develop symptomatic bradycardia.
In patients without underlying cardiac disease, SOMATULINE DEPOT may lead to a decrease in heart rate without necessarily reaching the threshold of bradycardia. In patients suffering from cardiac disorders prior to SOMATULINE DEPOT treatment, sinus bradycardia may occur. Care should be taken when initiating treatment with SOMATULINE DEPOT in patients with bradycardia.
5.4 Thyroid Function Abnormalities
Slight decreases in thyroid function have been seen during treatment with lanreotide in acromegalic patients, though clinical hypothyroidism is rare (less than 1%). Thyroid function tests are recommended where clinically indicated.
5.5 Monitoring: Laboratory Tests
Acromegaly: Serum GH and IGF-1 levels are useful markers of the disease and the effectiveness of treatment [see Dosage and Administration (2.2)].
-
6 ADVERSE REACTIONS
The following adverse reactions to SOMATULINE DEPOT are discussed in greater detail in other sections of the labeling:
- Cholelithiasis and Complications of Cholelithiasis [see Warnings and Precautions (5.1)]
- Hyperglycemia and Hypoglycemia [see Warnings and Precautions (5.2)]
- Cardiovascular Abnormalities [see Warnings and Precautions (5.3)]
- Thyroid Function Abnormalities [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Acromegaly
The data described below reflect exposure to SOMATULINE DEPOT in 416 acromegalic patients in seven studies. One study was a fixed-dose pharmacokinetic study. The other six studies were open-label or extension studies, one had a placebo-controlled, run-in period, and another had an active control. The population was mainly Caucasian (329/353, 93%) with a median age of 53 years of age (range 19 to 84 years). Fifty-four subjects (13%) were age 66 to 74 and 18 subjects (4.3%) were 75 years of age and older.
Patients were evenly matched for sex (205 males and 211 females). The median average monthly dose was 91.2 mg (e.g., 90 mg injected via the deep subcutaneous route every 4 weeks) over 385 days with a median cumulative dose of 1290 mg. Of the patients reporting acromegaly, severity at baseline (N=265), serum GH levels were less than 10 ng/mL for 69% (183/265) of the patients and 10 ng/mL or greater for 31% (82/265) of the patients.
The most commonly reported adverse reactions reported by greater than 5% of patients who received SOMATULINE DEPOT (N=416) in the overall pooled safety studies in acromegaly patients were gastrointestinal disorders (diarrhea, abdominal pain, nausea, constipation, flatulence, vomiting, loose stools), cholelithiasis, and injection site reactions.
Tables 1 and 2 present adverse reaction data from clinical studies with SOMATULINE DEPOT in acromegalic patients. The tables include data from a single clinical study and pooled data from seven clinical studies.
Adverse Reactions in Parallel Fixed-Dose Phase of Study 1
The incidence of treatment-emergent adverse reactions for SOMATULINE DEPOT 60, 90, and 120 mg by dose as reported during the first 4 months (fixed-dose phase) of Study 1 [see Clinical Studies (14.1)] are provided in Table 1.
Table 1: Adverse Reactions at an Incidence of Greater than 5% with SOMATULINE DEPOT Overall and Occurring at Higher Rate than Placebo: Placebo-Controlled and Fixed-Dose Phase of Study 1 By Dose Placebo-Controlled
Double-Blind Phase
Weeks 0 to 4Fixed-Dose Phase
Double-Blind + Single-Blind
Weeks 0 to 20Body System
Preferred TermPlacebo
(N=25)SOMATULINE DEPOT Overall
(N=83)SOMATULINE DEPOT 60 mg
(N=34)SOMATULINE DEPOT 90 mg
(N=36)SOMATULINE DEPOT 120 mg
(N=37)SOMATULINE DEPOT Overall
(N=107)N (%) N (%) N (%) N (%) N (%) N (%) A patient is counted only once for each body system and preferred term. Dictionary = WHOART. Gastrointestinal System Disorders 1 (4%) 30 (36%) 12 (35%) 21 (58%) 27 (73%) 60 (56%) Diarrhea 0 26 (31%) 9 (26%) 15 (42%) 24 (65%) 48 (45%) Abdominal pain 1 (4%) 6 (7%) 3 (9%) 6 (17%) 7 (19%) 16 (15%) Flatulence 0 5 (6%) 0 (0%) 3 (8%) 5 (14%) 8 (7%) Application Site Disorders 0 (0%) 5 (6%) 3 (9%) 4 (11%) 8 (22%) 15 (14%) (Injection site mass/ pain/ reaction/ inflammation) Liver and Biliary System Disorders 1 (4%) 3 (4%) 9 (26%) 7 (19%) 4 (11%) 20 (19%) Cholelithiasis 0 2 (2%) 5 (15%) 6 (17%) 3 (8%) 14 (13%) Heart Rate & Rhythm Disorders 0 8 (10%) 7 (21%) 2 (6%) 5 (14%) 14 (13%) Bradycardia 0 7 (8%) 6 (18%) 2 (6%) 2 (5%) 10 (9%) Red Blood Cell Disorders 0 6 (7%) 2 (6%) 5 (14%) 2 (5%) 9 (8%) Anemia 0 6 (7%) 2 (6%) 5 (14%) 2 (5%) 9 (8%) Metabolic & Nutritional Disorders 3 (12%) 13 (16%) 8 (24%) 9 (25%) 4 (11%) 21 (20%) Weight decrease 0 7 (8%) 3 (9%) 4 (11%) 2 (5%) 9 (8%) In Study 1, the adverse reactions of diarrhea, abdominal pain, and flatulence increased in incidence with increasing dose of SOMATULINE DEPOT.
Adverse Reactions in Long-Term Clinical Trials
Table 2 provides the most common adverse reactions (greater than 5%) that occurred in 416 acromegalic patients treated with SOMATULINE DEPOT pooled from 7 studies compared to those patients from the 2 efficacy studies (Studies 1 and 2). Patients with elevated GH and IGF-1 levels were either naive to somatostatin analog therapy or had undergone a 3-month washout [see Clinical Studies (14.1)].
Table 2: Adverse Reactions in SOMATULINE DEPOT-Treated Patients at an Incidence Greater than 5% in Overall Group Versus Adverse Reactions Reported in Studies 1 and 2 System Organ Class Number and Percentage of Patients Studies 1 & 2 Overall Pooled Data (N=170) (N=416) N % N % Dictionary = MedDRA 7.1 Patients with any Adverse Reactions 157 92 356 86 Gastrointestinal disorders 121 71 235 57 Diarrhea 81 48 155 37 Abdominal pain 34 20 79 19 Nausea 15 9 46 11 Constipation 9 5 33 8 Flatulence 12 7 30 7 Vomiting 8 5 28 7 Loose stools 16 9 23 6 Hepatobiliary disorders 53 31 99 24 Cholelithiasis 45 27 85 20 General disorders and administration site conditions 51 30 91 22 (Injection site pain /mass /induration/ nodule/pruritus) 28 17 37 9 Musculoskeletal and connective tissue disorders 44 26 70 17 Arthralgia 17 10 30 7 Nervous system disorders 34 20 80 19 Headache 9 5 30 7 In addition to the adverse reactions listed in Table 2, the following reactions were also seen:
- Sinus bradycardia occurred in 7% (12) of patients in the pooled Study 1 and 2 and in 3% (13) of patients in the overall pooled studies.
- Hypertension occurred in 7% (11) of patients in the pooled Study 1 and 2 and in 5% (20) of patients in the overall pooled studies.
- Anemia occurred in 7% (12) of patients in the pooled Study 1 and 2 and in 3% (14) of patients in the overall pooled studies.
Gastrointestinal Adverse Reactions
In the pooled clinical studies of SOMATULINE DEPOT therapy, a variety of gastrointestinal (GI) reactions occurred, the majority of which were mild to moderate in severity. One percent of acromegalic patients treated with SOMATULINE DEPOT in the pooled clinical studies discontinued treatment because of gastrointestinal reactions.
Pancreatitis was reported in less than 1% of patients.
Gallbladder Adverse Reactions
In clinical studies involving 416 acromegalic patients treated with SOMATULINE DEPOT, cholelithiasis and gallbladder sludge were reported in 20% of the patients. Among 167 acromegalic patients treated with SOMATULINE DEPOT who underwent routine evaluation with gallbladder ultrasound, 17% had gallstones at baseline. New cholelithiasis was reported in 12% of patients. Cholelithiasis may be related to dose or duration of exposure [see Warnings and Precautions (5.1)].
Injection Site Reactions
In the pooled clinical studies, injection site pain (4%) and injection site mass (2%) were the most frequently reported local adverse drug reactions that occurred with the administration of SOMATULINE DEPOT. In a specific analysis, 20 of 413 patients (5%) presented indurations at the injection site. Injection site adverse reactions were more commonly reported soon after the start of treatment and were less commonly reported as treatment continued. Such adverse reactions were usually mild or moderate but did lead to withdrawal from clinical studies in two subjects.
Glucose Metabolism Adverse Reactions
In the clinical studies in acromegalic patients treated with SOMATULINE DEPOT, adverse reactions of dysglycemia (hypoglycemia, hyperglycemia, diabetes) were reported by 14% (47/332) of patients and were considered related to study drug in 7% (24/332) of patients [see Warnings and Precautions (5.2)].
Cardiac Adverse Reactions
In the pooled clinical studies, sinus bradycardia (3%) was the most frequently observed heart rate and rhythm disorder. All other cardiac adverse drug reactions were observed in less than 1% of patients. The relationship of these events to SOMATULINE DEPOT could not be established because many of these patients had underlying cardiac disease [see Warnings and Precautions (5.3)].
A comparative echocardiography study of lanreotide and another somatostatin analog demonstrated no difference in the development of new or worsening valvular regurgitation between the 2 treatments over 1 year. The occurrence of clinically significant mitral regurgitation (i.e., moderate or severe in intensity) or of clinically significant aortic regurgitation (i.e., at least mild in intensity) was low in both groups of patients throughout the study.
Other Adverse Reactions
For the most commonly occurring adverse reactions in the pooled analysis, diarrhea, abdominal pain, and cholelithiasis, there was no apparent trend for increasing incidence with age. GI disorders and renal and urinary disorders were more common in patients with documented hepatic impairment; however, the incidence of cholelithiasis was similar between groups.
Gastroenteropancreatic Neuroendocrine Tumors
The safety of SOMATULINE DEPOT 120 mg for the treatment of patients with gastroenteropancreatic neuroendocrine tumors (GEP-NETs) was evaluated in Study 3, a double-blind, placebo-controlled trial. Patients in Study 3 were randomized to receive SOMATULINE DEPOT (N=101) or placebo (N=103) administered by deep subcutaneous injection once every 4 weeks. The data below reflect exposure to SOMATULINE DEPOT in 101 patients with GEP-NETs, including 87 patients exposed for at least 6 months and 72 patients exposed for at least 1 year (median duration of exposure 22 months). Patients treated with SOMATULINE DEPOT had a median age of 64 years (range 30 to 83 years), 53% were men and 96% were Caucasian. Eighty-one percent of patients (83/101) in the SOMATULINE DEPOT arm and 82% of patients (82/103) in the placebo arm did not have disease progression within 6 months of enrollment and had not received prior therapy for GEP-NETs. The rates of discontinuation due to treatment-emergent adverse reactions were 5% (5/101 patients) in the SOMATULINE DEPOT arm and 3% (3/103 patients) in the placebo arm.
Table 3 compares the adverse reactions reported with an incidence of 5% and greater in patients receiving SOMATULINE DEPOT 120 mg administered every 4 weeks and reported more commonly than placebo.
Table 3: Adverse Reactions Occurring in 5% and Greater of SOMATULINE DEPOT-Treated Patients and at a Higher Rate Than in Placebo-Treated Patients in Study 3 Adverse Reaction SOMATULINE DEPOT 120 mg
N=101Placebo
N=103Any (%) Severe* (%) Any (%) Severe* (%) - * Defined as hazardous to well-being, significant impairment of function or incapacitation
- † Includes preferred terms of abdominal pain, abdominal pain upper/lower, abdominal discomfort
- ‡ Includes one or more serious adverse events (SAEs) defined as any event that results in death, is life threatening, results in hospitalization or prolongation of hospitalization, results in persistent or significant disability, results in congenital anomaly/birth defect, or may jeopardize the patient and may require medical or surgical intervention to prevent one of the outcomes listed.
- § Includes preferred terms of myalgia, musculoskeletal discomfort, musculoskeletal pain, back pain
- ¶ Includes preferred terms of infusion site extravasation, injection site discomfort, injection site granuloma, injections site hematoma, injection site hemorrhage, injection site induration, injection site mass, injections site nodule, injection site pain, injection site pruritus, injection site rash, injection site reaction, injection site swelling
- # Includes preferred terms of diabetes mellitus, glucose tolerance impaired, hyperglycemia, type 2 diabetes mellitus
- Þ Includes preferred terms of hypertension, hypertensive crisis
- ß Includes preferred terms of depression, depressed mood
Any Adverse Reactions 88 26 90 31 Abdominal pain† 34‡ 6‡ 24‡ 4 Musculoskeletal pain§ 19‡ 2‡ 13 2 Vomiting 19‡ 2‡ 9‡ 2‡ Headache 16 0 11 1 Injection site reaction¶ 15 0 7 0 Hyperglycemia# 14‡ 0 5 0 HypertensionÞ 14‡ 1‡ 5 0 Cholelithiasis 14‡ 1‡ 7 0 Dizziness 9 0 2‡ 0 Depressionß 7 0 1 0 Dyspnea 6 0 1 0 Carcinoid Syndrome
The safety of SOMATULINE DEPOT 120 mg in patients with histopathologically confirmed neuroendocrine tumors and a history of carcinoid syndrome (flushing and/or diarrhea) was evaluated in Study 4, a double-blind, placebo-controlled trial. Patients were randomized to receive SOMATULINE DEPOT (N=59) or placebo (N=56) administered by deep subcutaneous injection once every 4 weeks. Patients in both arms of Study 4 had access to subcutaneous octreotide as rescue medication for symptom control.
Adverse reactions reported in Study 4 were generally similar to those reported in Study 3 for the GEP-NETs population shown in Table 3 above. Adverse reactions occurring in Study 4 in 5% and greater of SOMATULINE DEPOT-treated patients and occurring at least 5% more than in placebo-treated patients were headache (12% vs 5%, respectively), dizziness (7% vs 0%, respectively), and muscle spasm (5% vs 0%, respectively) by week 16.
6.2 Immunogenicity
As with all peptides, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to lanreotide in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
Laboratory investigations of acromegalic patients treated with SOMATULINE DEPOT in clinical studies show that the percentage of patients with putative antibodies at any time point after treatment is low (less than 1% to 4% of patients in specific studies whose antibodies were tested). The antibodies did not appear to affect the efficacy or safety of SOMATULINE DEPOT.
In Study 3, development of anti-lanreotide antibodies was assessed using a radioimmunoprecipitation assay. In patients with GEP NETs receiving SOMATULINE DEPOT, the incidence of anti-lanreotide antibodies was 4% (3 of 82) at 24 weeks, 10% (7 of 67) at 48 weeks, 11% (6 of 57) at 72 weeks, and 10% (8 of 84) at 96 weeks. Assessment for neutralizing antibodies was not conducted. In Study 4, less than 2% (2 of 108) of the patients treated with SOMATULINE DEPOT developed anti-lanreotide antibodies.
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of SOMATULINE DEPOT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary: steatorrhea; cholecystitis, cholangitis, pancreatitis, which have sometimes required cholecystectomy
Hypersensitivity: angioedema and anaphylaxis
Injection site reactions: injection site abscess -
7 DRUG INTERACTIONS
7.1 Insulin and Oral Hypoglycemic Drugs
Lanreotide, like somatostatin and other somatostatin analogs, inhibits the secretion of insulin and glucagon. Therefore, blood glucose levels should be monitored when SOMATULINE DEPOT treatment is initiated or when the dose is altered, and antidiabetic treatment should be adjusted accordingly [see Warnings and Precautions (5.2)].
7.2 Cyclosporine
Concomitant administration of cyclosporine with SOMATULINE DEPOT may decrease the absorption of cyclosporine, and therefore, may necessitate adjustment of cyclosporine dose to maintain therapeutic drug concentrations. [see Clinical Pharmacology (12.3)]
7.3 Bromocriptine
Limited published data indicate that concomitant administration of a somatostatin analog and bromocriptine may increase the absorption of bromocriptine [see Clinical Pharmacology (12.3)].
7.4 Bradycardia-Inducing Drugs
Concomitant administration of bradycardia-inducing drugs (e.g., beta-blockers) may have an additive effect on the reduction of heart rate associated with lanreotide. Dosage adjustments of concomitant drugs may be necessary.
7.5 Drug Metabolism Interactions
The limited published data available indicate that somatostatin analogs may decrease the metabolic clearance of compounds known to be metabolized by cytochrome P450 enzymes, which may be due to the suppression of growth hormone. Since it cannot be excluded that SOMATULINE DEPOT may have this effect, avoid other drugs mainly metabolized by CYP3A4 and which have a low therapeutic index (e.g., quinidine, terfenadine). Drugs metabolized by the liver may be metabolized more slowly during SOMATULINE DEPOT treatment and dose reductions of the concomitantly administered medications should be considered [see Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Limited available data based on postmarketing case reports with SOMATULINE DEPOT use in pregnant women are not sufficient to determine a drug-associated risk of adverse developmental outcomes. In animal reproduction studies, decreased embryo/fetal survival was observed in pregnant rats and rabbits at subcutaneous doses 5- and 2-times the maximum recommended human dose (MRHD) of 120 mg, respectively (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
A reproductive study in pregnant rats given 30 mg/kg of lanreotide by subcutaneous injection every 2 weeks (5 times the human dose, based on body surface area comparisons) resulted in decreased embryo/fetal survival. A study in pregnant rabbits given subcutaneous injections of 0.45 mg/kg/day (2 times the human therapeutic exposures at the maximum recommended dose of 120 mg, based on comparisons of relative body surface area) shows decreased fetal survival and increased fetal skeletal/soft tissue abnormalities.
8.2 Lactation
Risk Summary
There is no information available on the presence of lanreotide in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. Studies show that lanreotide acetate administered subcutaneously passes into the milk of lactating rats; however, due to specifies-specific differences in lactation physiology, animal data may not reliably predict drug levels in human milk. Because of the potential for serious adverse reactions in breastfed infants from SOMATULINE DEPOT, including effects on glucose metabolism and bradycardia, advise women not to breastfeed during treatment with SOMATULINE DEPOT and for 6 months (6 half-lives) following the last dose.
8.3 Females and Males of Reproductive Potential
Infertility
Females
Based on results from animal studies conducted in female rats, SOMATULINE DEPOT may reduce fertility in females of reproductive potential [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of SOMATULINE DEPOT in pediatric patients have not been established.
8.5 Geriatric Use
No overall differences in safety or effectiveness were observed between elderly patients with acromegaly compared with younger patients and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Studies 3 and 4, conducted in patients with neuroendocrine tumors, did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients.
Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
Acromegaly
Lanreotide has been studied in patients with end-stage renal function on dialysis, but has not been studied in patients with mild, moderate, or severe renal impairment. It is recommended that patients with moderate or severe renal impairment receive a starting dose of lanreotide of 60 mg. Caution should be exercised when considering patients with moderate or severe renal impairment for an extended dosing interval of SOMATULINE DEPOT 120 mg every 6 or 8 weeks [see Dosage and Administration (2.1) and Clinical Pharmacology (12.3)].
Neuroendocrine Tumors (NET) – Gastroenteropancreatic Neuroendocrine Tumors
No effect was observed in total clearance of lanreotide in patients with mild to moderate renal impairment receiving SOMATULINE DEPOT 120 mg. Patients with severe renal impairment were not studied [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Acromegaly
It is recommended that patients with moderate or severe hepatic impairment receive a starting dose of lanreotide of 60 mg. Caution should be exercised when considering patients with moderate or severe hepatic impairment for an extended dosing interval of SOMATULINE DEPOT 120 mg every 6 or 8 weeks [see Dosage and Administration (2.1) and Clinical Pharmacology (12.3)].
-
11 DESCRIPTION
SOMATULINE DEPOT (lanreotide) Injection 60 mg/0.2 mL, 90 mg/0.3 mL, and 120 mg/0.5 mL is a prolonged-release formulation for deep subcutaneous injection. It contains the drug substance lanreotide acetate, a synthetic octapeptide with a biological activity similar to naturally occurring somatostatin, water for injection and acetic acid (for pH adjustment).
SOMATULINE DEPOT is available as sterile, ready-to-use, single-dose prefilled syringes containing lanreotide acetate supersaturated bulk solution of 24.6% w/w lanreotide base.
Each syringe contains: SOMATULINE DEPOT
60 mg/0.2 mLSOMATULINE DEPOT
90 mg/0.3 mLSOMATULINE DEPOT
120 mg/0.5 mLLanreotide acetate 77.9 mg 113.6 mg 149.4 mg Acetic Acid q.s. q.s. q.s. Water for injection 186.6 mg 272.3 mg 357.8 mg Total Weight 266 mg 388 mg 510 mg Lanreotide acetate is a synthetic cyclical octapeptide analog of the natural hormone, somatostatin. Lanreotide acetate is chemically known as [cyclo S-S]-3-(2-naphthyl)-D-alanyl-L-cysteinyl-L-tyrosyl-D-tryptophyl-L-lysyl-L-valyl-L-cysteinyl-L-threoninamide, acetate salt. Its molecular weight is 1096.34 (base) and its amino acid sequence is:
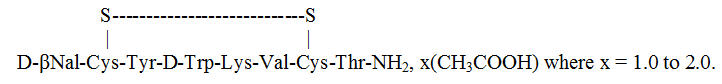
The SOMATULINE DEPOT in the prefilled syringe is a white to pale yellow, semi-solid formulation.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Lanreotide, the active component of SOMATULINE DEPOT is an octapeptide analog of natural somatostatin. The mechanism of action of lanreotide is believed to be similar to that of natural somatostatin.
12.2 Pharmacodynamics
Lanreotide has a high affinity for human somatostatin receptors (SSTR) 2 and 5 and a reduced binding affinity for human SSTR1, 3, and 4. Activity at human SSTR2 and 5 is the primary mechanism believed responsible for GH inhibition. Like somatostatin, lanreotide is an inhibitor of various endocrine, neuroendocrine, exocrine, and paracrine functions.
The primary pharmacodynamic effect of lanreotide is a reduction of GH and/or IGF-1 levels enabling normalization of levels in acromegalic patients [see Clinical Studies (14.1)]. In acromegalic patients, lanreotide reduces GH levels in a dose-dependent way. After a single injection of SOMATULINE DEPOT, plasma GH levels fall rapidly and are maintained for at least 28 days.
In Study 4, patients with carcinoid syndrome treated with SOMATULINE DEPOT 120 mg every 4 weeks had reduced levels of urinary 5-hydroxyindoleacetic acid (5-HIAA) compared with placebo [see Clinical Studies (14.3)].
Lanreotide inhibits the basal secretion of motilin, gastric inhibitory peptide, and pancreatic polypeptide, but has no significant effect on the secretion of secretin. Lanreotide inhibits postprandial secretion of pancreatic polypeptide, gastrin, and cholecystokinin (CCK). In healthy subjects, lanreotide produces a reduction and a delay in postprandial insulin secretion, resulting in transient, mild glucose intolerance.
Lanreotide inhibits meal-stimulated pancreatic secretions, and reduces duodenal bicarbonate and amylase concentrations, and produces a transient reduction in gastric acidity.
Lanreotide has been shown to inhibit gallbladder contractility and bile secretion in healthy subjects [see Warnings and Precautions (5.1)].
In healthy subjects, lanreotide inhibits meal-induced increases in superior mesenteric artery and portal venous blood flow, but has no effect on basal or meal-stimulated renal blood flow. Lanreotide has no effect on renal plasma flow or renal vascular resistance. However, a transient decrease in glomerular filtration rate (GFR) and filtration fraction has been observed after a single injection of lanreotide.
In healthy subjects, non-significant reductions in glucagon levels were seen after lanreotide administration. In diabetic non-acromegalic subjects receiving a continuous infusion (21-day) of lanreotide, serum glucose concentrations were temporarily decreased by 20% to 30% after the start and end of the infusion. Serum glucose concentrations returned to normal levels within 24 hours. A significant decrease in insulin concentrations was recorded between baseline and Day 1 only [see Warnings and Precautions (5.2)].
Lanreotide inhibits the nocturnal increase in thyroid-stimulating hormone (TSH) seen in healthy subjects. Lanreotide reduces prolactin levels in acromegalic patients treated on a long-term basis [see Warnings and Precautions (5.4)].
12.3 Pharmacokinetics
SOMATULINE DEPOT is thought to form a drug depot at the injection site due to the interaction of the formulation with physiological fluids. The most likely mechanism of drug release is a passive diffusion of the precipitated drug from the depot towards the surrounding tissues, followed by the absorption to the bloodstream.
After a single, deep subcutaneous administration, the mean absolute bioavailability of SOMATULINE DEPOT in healthy subjects was 73.4, 69.0, and 78.4% for the 60 mg, 90 mg, and 120 mg doses, respectively. Mean Cmax values ranged from 4.3 to 8.4 ng/mL during the first day. Single-dose linearity was demonstrated with respect to AUC and Cmax, and showed high inter-subject variability. SOMATULINE DEPOT showed sustained release of lanreotide with a half-life of 23 to 30 days. Mean serum concentrations were > 1 ng/mL throughout 28 days at 90 mg and 120 mg and > 0.9 ng/mL at 60 mg.
In studies evaluating excretion, <5% of lanreotide was excreted in urine and less than 0.5% was recovered unchanged in feces, indicative of some biliary excretion.
Acromegaly
In a repeat-dose administration pharmacokinetics (PK) study in acromegalic patients, rapid initial release was seen giving peak levels during the first day after administration. At doses of SOMATULINE DEPOT between 60 and 120 mg, linear pharmacokinetics were observed in acromegalic patients. At steady state, mean Cmax values were 3.8 ± 0.5, 5.7 ± 1.7, and 7.7 ± 2.5 ng/mL, increasing linearly with dose. The mean accumulation ratio index was 2.7, which is in line with the range of values for the half-life of SOMATULINE DEPOT. The steady-state trough serum lanreotide concentrations in patients receiving SOMATULINE DEPOT every 28 days were 1.8 ± 0.3; 2.5 ± 0.9 and 3.8 ± 1.0 ng/mL at 60 mg, 90 mg, and 120 mg doses, respectively. A limited initial burst effect and a low peak-to-trough fluctuation (81% to 108%) of the serum concentration at the plateau were observed.
For the same doses, similar values were obtained in clinical studies after at least four administrations (2.3 ± 0.9, 3.2 ± 1.1, and 4.0 ± 1.4 ng/mL, respectively).
Pharmacokinetic data from studies evaluating extended dosing use of SOMATULINE DEPOT 120 mg demonstrated mean steady-state, Cmin values between 1.6 and 2.3 ng/mL for the 8- and 6-week treatment interval, respectively.
Gastroenteropancreatic Neuroendocrine Tumors
In patients with GEP-NETs treated with SOMATULINE DEPOT 120 mg every 4 weeks, steady state concentrations were reached after 4 to 5 injections and the mean trough serum lanreotide concentrations at steady state ranged from 5.3 to 8.6 ng/mL.
Specific Populations
SOMATULINE DEPOT has not been studied in specific populations. However, the pharmacokinetics of lanreotide in renal impaired, hepatic impaired, and geriatric subjects were evaluated after IV administration of lanreotide immediate release formulation (IRF) at 7 mcg/kg dose.
Geriatric
Studies in healthy elderly subjects showed an 85% increase in half-life and a 65% increase in mean residence time (MRT) of lanreotide compared to those seen in healthy young subjects; however, there was no change in either AUC or Cmax of lanreotide in elderly as compared to healthy young subjects. Age has no effect on clearance of lanreotide based on population PK analysis in patients with GEP-NET which included 122 patients aged 65 to 85 years with neuroendocrine tumors.
Renal Impairment
An approximate 2-fold decrease in total serum clearance of lanreotide, with a consequent 2-fold increase in half-life and AUC was observed. Patients with acromegaly and with moderate to severe renal impairment should begin treatment with SOMATULINE DEPOT 60 mg. Caution should be exercised when considering patients with moderate or severe renal impairment for an extended dosing interval of SOMATULINE DEPOT 120 mg every 6 or 8 weeks.
Mild (CLcr 60-89 mL/min) or moderate (CLcr 30-59 mL/min) renal impairment has no effect on clearance of lanreotide in patients with GEP-NET based on population PK analysis which included 106 patients with mild and 59 patients with moderate renal impairment treated with SOMATULINE DEPOT. GEP-NET patients with severe renal impairment (CLcr < 30 mL/min) were not studied.
Hepatic Impairment
In subjects with moderate to severe hepatic impairment, a 30% reduction in clearance of lanreotide was observed. Patients with acromegaly and with moderate to severe hepatic impairment should begin treatment with SOMATULINE DEPOT 60 mg. Caution should be exercised when considering patients with moderate or severe hepatic impairment for an extended dosing interval of SOMATULINE DEPOT 120 mg every 6 or 8 weeks.
The effect of hepatic impairment on clearance of lanreotide has not been studied in patients with GEP-NET.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Standard lifetime carcinogenicity bioassays were conducted in mice and rats. Mice were given daily subcutaneous doses of lanreotide at 0.5, 1.5, 5, 10, and 30 mg/kg for 104 weeks. Cutaneous and subcutaneous tumors of fibrous connective tissues at the injection sites were observed at the high dose of 30 mg/kg/day. Fibrosarcomas in both genders and malignant fibrous histiocytomas were observed in males at 30 mg/kg/day resulting in exposures 3 times higher than the clinical therapeutic exposure at the maximum therapeutic dose of 120 mg given by monthly subcutaneous injection based on the AUC values. Rats were given daily subcutaneous doses of lanreotide at 0.1, 0.2, and 0.5 mg/kg for 104 weeks. Increased cutaneous and subcutaneous tumors of fibrous connective tissues at the injection sites were observed at the dose of 0.5 mg/kg/day resulting in exposures less than the clinical therapeutic exposure at 120 mg given by monthly subcutaneous injection. The increased incidence of injection site tumors in rodents is likely related to the increased dosing frequency (daily) in animals compared to monthly dosing in humans and therefore may not be clinically relevant.
Lanreotide was not genotoxic in tests for gene mutations in a bacterial mutagenicity (Ames) assay, or mouse lymphoma cell assay with or without metabolic activation. Lanreotide was not genotoxic in tests for the detection of chromosomal aberrations in a human lymphocyte and in vivo mouse micronucleus assay.
In a fertility study conducted with lanreotide in rats, reduced female fecundity was observed at estimated exposure corresponding to approximately 10-fold the plasma exposure at the MRHD of 120 mg. The fertility of male rats was unaffected by the treatment up to an estimated exposure corresponding to approximately 11-fold the plasma exposure at the MRHD of 120 mg.
-
14 CLINICAL STUDIES
14.1 Acromegaly
The effect of SOMATULINE DEPOT on reducing GH and IGF-levels and control of symptoms in patients with acromegaly was studied in 2 long-term, multiple-dose, randomized, multicenter studies.
Study 1
This 1-year study included a 4-week, double-blind, placebo-controlled phase; a 16-week single-blind, fixed-dose phase; and a 32-week, open-label, dose-titration phase. Patients with active acromegaly, based on biochemical tests and medical history, entered a 12-week washout period if there was previous treatment with a somatostatin analog or a dopaminergic agonist.
Upon entry, patients were randomly allocated to receive a single, deep subcutaneous injection of SOMATULINE DEPOT 60, 90, or 120 mg or placebo. Four weeks later, patients entered a fixed-dose phase where they received 4 injections of SOMATULINE DEPOT followed by a dose-titration phase of 8 injections for a total of 13 injections over 52 weeks (including the placebo phase). Injections were given at 4-week intervals. During the dose-titration phase of the study, the dose was titrated twice (every fourth injection), as needed, according to individual GH and IGF-1 levels.
A total of 108 patients (51 males, 57 females) were enrolled in the initial placebo-controlled phase of the study. Half (54/108) of the patients had never been treated with a somatostatin analog or dopamine agonist, or had stopped treatment for at least 3 months prior to their participation in the study and were required to have a mean GH level greater than 5 ng/mL at their first visit. The other half of the patients had received prior treatment with a somatostatin analog or a dopamine agonist before study entry and at study entry were required to have a mean GH concentration greater than 3 ng/mL and at least a 100% increase in mean GH concentration after washout of medication.
One hundred and seven (107) patients completed the placebo-controlled phase, 105 patients completed the fixed-dose phase, and 99 patients completed the dose-titration phase. Patients not completing withdrew due to adverse events (5) or lack of efficacy (4).
In the double-blind phase of Study 1, a total of 52 (63%) of the 83 lanreotide-treated patients had a greater than 50% decrease in mean GH from baseline to Week 4, including 52%, 44%, and 90% of patients in the 60, 90, and 120 mg groups, respectively, compared to placebo (0%, 0/25). In the fixed-dose phase at Week 16, 72% of all 107 lanreotide-treated patients had a decrease from baseline in mean GH of greater than 50%, including 68% (23/34), 64% (23/36), and 84% (31/37) of patients in the 60, 90, and 120 mg lanreotide treatment groups, respectively. Efficacy achieved in the first 16 weeks was maintained for the duration of the study (see Table 4).
Table 4: Overall Efficacy Results Based on GH and IGF-1 Levels by Treatment Phase in Study 1 Baseline Before Titration 1 Before Titration 2 Last Value Available* N=107 (16 weeks)
N=107(32 weeks)
N=105N=107 - * Last Observation Carried Forward
- † Age-adjusted
- ‡ n=105,
- § n=102,
GH ≤5.0 ng/mL Number of Responders (%) 20
(19%)72
(67%)76
(72%)74
(69%)≤2.5 ng/mL Number of Responders (%) 0
(0%)52
(49%)59
(56%)55
(51%)≤1.0 ng/mL Number of Responders (%) 0
(0%)15
(14%)18
(17%)17
(16%)Median GH ng/mL 10.27 2.53 2.20 2.43 GH Reduction Median % Reduction -- 75.5 78.2 75.5 IGF-1 Normal† Number of Responders (%) 9
(8%)58
(54%)57
(54%)62
(58%)Median IGF-1 ng/mL 775.0 332.0‡ 316.5§ 326.0 IGF-1 Reduction Median % Reduction -- 52.3‡ 54.5§ 55.4 IGF-1 Normal† + GH ≤2.5 ng/mL Number of Responders (%) 0
(0%)41
(38%)46
(44%)44
(41%)Study 2
This was a 48-week, open-label, uncontrolled, multicenter study that enrolled patients who had an IGF-1 concentration 1.3 times or greater than the upper limit of the normal age-adjusted range. Patients receiving treatment with a somatostatin analog (other than SOMATULINE DEPOT) or a dopaminergic agonist had to attain this IGF-1 concentration after a washout period of up to 3 months.
Patients were initially enrolled in a 4-month, fixed-dose phase where they received 4 deep subcutaneous injections of SOMATULINE DEPOT 90 mg, at 4-week intervals. Patients then entered a dose-titration phase where the dose of SOMATULINE DEPOT was adjusted based on GH and IGF-1 levels at the beginning of the dose-titration phase and, if necessary, again after another 4 injections. Patients titrated up to the maximum dose (120 mg) were not allowed to titrate down again.
A total of 63 patients (38 males, 25 females) entered the fixed-dose phase of the trial and 57 patients completed 48 weeks of treatment. Six patients withdrew due to adverse reactions (3), other reasons (2), or lack of efficacy (1).
After 48 weeks of treatment with SOMATULINE DEPOT at 4-week intervals, 43% (27/63) of the acromegalic patients in this study achieved normal age-adjusted IGF-1 concentrations. Mean IGF-1 concentrations after treatment completion were 1.3 ± 0.7 times the upper limit of normal compared to 2.5 ± 1.1 times the upper limit of normal at baseline.
The reduction in IGF-1 concentrations over time correlated with a corresponding marked decrease in mean GH concentrations. The proportion of patients with mean GH concentrations less than 2.5 ng/mL increased significantly from 35% to 77% after the fixed-dose phase and 85% at the end of the study. At the end of treatment, 24/63 (38%) of patients had both normal IGF-1 concentrations and a GH concentration of less than or equal to 2.5 ng/mL (see Table 5) and 17/63 patients (27%) had both normal IGF-1 concentrations and a GH concentration of less than 1 ng/mL.
Table 5: Overall Efficacy Results Based on GH and IGF-1 Levels by Treatment Phase in Study 2 Baseline Before Titration 1 (12 wks) Before Titration 2 (28 wks) Last Value Available* N=63 N=63 N=59 N=63 - * Last Observation Carried Forward
- † Age-adjusted,
- ‡ N= 62,
IGF-1 Normal† Number of Responders (%) 0
(0%)17
(27%)22
(37%)27
(43%)Median IGF-1 ng/mL 689.0 382.0 334.0 317.0 IGF-1 Reduction Median % Reduction -- 41.0 51.0 50.3 GH ≤5.0 ng/mL Number of Responders (%) 40
(64%)59
(94%)57
(97%)62
(98%)≤2.5 ng/mL Number of Responders (%) 21
(33%)47
(75%)47
(80%)54
(86 %)≤1.0 ng/mL Number of Responders (%) 8
(13%)19
(30%)18
(31%)28
(44%)Median GH ng/Ml 3.71 1.65 1.48 1.13 GH Reduction Median % Reduction -- 63.2 66.7 78.6‡ IGF-1 normal† + GH ≤2.5 ng/mL Number of Responders (%) 0
(0%)14
(22%)20
(34%)24
(38%)Examination of age and gender subgroups did not identify differences in response to SOMATULINE DEPOT among these subgroups. The limited number of patients in the different racial subgroups did not raise any concerns regarding efficacy of SOMATULINE DEPOT in these subgroups.
14.2 Gastroenteropancreatic Neuroendocrine Tumors
The efficacy of SOMATULINE DEPOT was established in a multicenter, randomized, double-blind, placebo-controlled trial of 204 patients with unresectable, well or moderately differentiated, metastatic or locally advanced, gastroenteropancreatic neuroendocrine tumors. Patients were required to have non-functioning tumors without hormone-related symptoms. Patients were randomized 1:1 to receive SOMATULINE DEPOT 120 mg (n=101) or placebo (n=103) every 4 weeks until disease progression, unacceptable toxicity, or a maximum of 96 weeks of treatment. Randomization was stratified by the presence or absence of prior therapy and by the presence or absence of disease progression within 6 months of enrollment. The major efficacy outcome measure was progression-free survival (PFS), defined as time to disease progression as assessed by central independent radiological review using the Response Evaluation Criteria in Solid Tumors (RECIST 1.0) or death.
The median patient age was 63 years (range 30 to 92 years) and 95% were Caucasian. Disease progression was present in nine of 204 patients (4.4%) in the 6 months prior to enrollment and 29 patients (14%) received prior chemotherapy. Ninety-one patients (45%) had primary sites of disease in the pancreas, with the remainder originating in the midgut (35%), hindgut (7%), or unknown primary location (13%). The majority (69%) of the study population had grade 1 tumors. Baseline prognostic characteristics were similar between arms with one exception; there were 39% of patients in the SOMATULINE DEPOT arm and 27% of patients in the placebo arm who had hepatic involvement by tumor of greater than 25%.
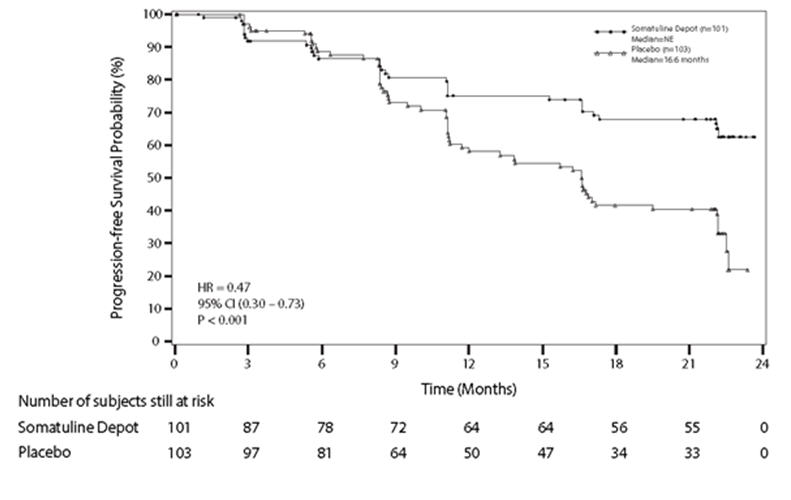
Patients on the SOMATULINE DEPOT arm had a statistically significant improvement in PFS compared to patients receiving placebo (see Table 6 and Figure 1).
Table 6: Efficacy Results in Study 3 SOMATULINE DEPOT Placebo n=101 n=103 - * NE = not reached at 22 months
- † Hazard Ratio is derived from a Cox stratified proportional hazards model
Number of Events (%) 32 (31.7%) 60 (58.3%) Median PFS (months)(95% CI) NE* (NE, NE) 16.6 (11.2, 22.1) HR (95% CI) 0.47 (0.30, 0.73)† Log-rank p-value <0.001 Figure 1: Kaplan-Meier Curves of Progression-Free Survival
14.3 Carcinoid Syndrome
Study 4 was a multicenter, randomized, 16-week, double-blind, placebo-controlled trial in 115 patients with histopathologically-confirmed neuroendocrine tumors and a history of carcinoid syndrome (flushing and/or diarrhea) who were treatment naïve or stable on another somatostatin analog and who were randomized 1:1 to receive SOMATULINE DEPOT 120 mg (n=59) or placebo (n=56) by deep subcutaneous injection every 4 weeks. Patients were instructed to self-administer a short-acting somatostatin analog (octreotide) as rescue medication as needed for symptom control. The use of rescue therapy and the severity and frequency of diarrhea and flushing symptoms were reported daily in electronic patient diaries. During the 16 week double-blind phase, the primary efficacy outcome measure was the percentage of days in which patients administered at least one injection of rescue medication for symptom control. Average daily frequencies of diarrhea and flushing events were assessed secondarily.
The patient population had a mean age of 59 years (range 27 to 85 years), 58% were female and 77% were Caucasian. Patients in the SOMATULINE DEPOT arm experienced 15% fewer days on rescue medication compared to patients in the placebo arm (34% vs. 49% of days, respectively; p=0.02). The average daily frequencies of diarrhea and flushing events in patients treated with SOMATULINE DEPOT (and rescue medication) were numerically lower relative to patients treated with placebo (and rescue medication), but were not statistically significantly different via hierarchical testing.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
SOMATULINE DEPOT is supplied in strengths of 60 mg/0.2 mL, 90 mg/0.3 mL, and 120 mg/0.5 mL as a white to pale yellow, semi-solid formulation in a single, sterile, prefilled, ready-to-use, polypropylene syringe fitted with an automatic safety system, a bromobutyl rubber plunger stopper and a 20 mm needle covered by a plastic cap.
Each prefilled syringe is placed in a plastic tray, sealed in a laminated pouch and packed in a carton.
NDC: 15054-1060-4 60 mg/0.2 mL, sterile, prefilled syringe NDC: 15054-1090-4 90 mg/0.3 mL, sterile, prefilled syringe NDC: 15054-1120-4 120 mg/0.5 mL, sterile, prefilled syringe -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Hypersensitivity Reactions
Advise patients to immediately contact their healthcare provider if they experience serious hypersensitivity reactions, such as angioedema or anaphylaxis [see Contraindications (4)].
Cholelithiasis and Complications of Cholelithiasis
Advise patients to contact their healthcare provider if they experience signs or symptoms of gallstones (cholelithiasis) or complications of gallstones (e.g., cholecystitis, cholangitis, or pancreatitis) [see Warnings and Precautions (5.1)].
Hyperglycemia and Hypoglycemia
Advise patients to immediately contact their healthcare provider if they experience signs or symptoms of hyper- or hypoglycemia [see Warnings and Precautions (5.2)].
Cardiovascular Abnormalities
Advise patients to immediately contact their healthcare provider if they experience bradycardia [see Warnings and Precautions (5.3)].
Thyroid Function Abnormalities
Advise patients to contact their healthcare provider if they experience signs or symptoms of hypothyroidism [see Warnings and Precautions (5.4)].
Laboratory Tests
Advise patients with acromegaly that response to SOMATULINE DEPOT should be monitored by periodic measurements of GH and IGF-1 levels, with a goal of decreasing these levels to the normal range [see Dosage and Administration (2.2)].
Lactation
Advise women not to breastfeed during treatment with SOMATULINE DEPOT and for 6 months after the last dose [see Use in Specific Populations (8.2)].
Infertility
Advise females of reproductive potential of the potential for reduced fertility from SOMATULINE DEPOT [see Use in Specific Populations (8.3)].
- SPL UNCLASSIFIED SECTION
-
PATIENT PACKAGE INSERT
Patient Information
SOMATULINE® DEPOT (So-mah-tu-leen Dee-Poh)
(lanreotide) injectionThis Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 6/2019 Read this Patient Information before you receive your first SOMATULINE DEPOT injection and before each injection. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. What is SOMATULINE DEPOT?
SOMATULINE DEPOT is a prescription medicine used for:
- the long-term treatment of people with acromegaly when:
- surgery or radiotherapy have not worked well enough or
- they are not able to have surgery or radiotherapy
- the treatment of adults with a type of cancer known as neuroendocrine tumors, from the gastrointestinal tract or the pancreas (GEP-NETs) that has spread or cannot be removed by surgery
- the treatment of adults with carcinoid syndrome to reduce the need for the use of short-acting somatostatin medicine
It is not known if SOMATULINE DEPOT is safe and effective in children.
Who should not receive SOMATULINE DEPOT?
Do not receive SOMATULINE DEPOT if you are allergic to lanreotide.
What should I tell my healthcare provider before receiving SOMATULINE DEPOT?
Before you receive SOMATULINE DEPOT, tell your healthcare provider about all of your medical conditions, including if you:
- have gallbladder problems
- have diabetes
- have heart problems
- have thyroid problems
- have kidney problems
- have liver problems
- are pregnant or plan to become pregnant. It is not known if SOMATULINE DEPOT will harm your unborn baby
- are breastfeeding or plan to breastfeed. It is not known if SOMATULINE DEPOT passes into your breast milk. You should not breastfeed if you receive SOMATULINE DEPOT and for 6 months after your last dose of SOMATULINE DEPOT
- are a female who can become pregnant. SOMATULINE DEPOT may affect fertility in females and may affect your ability to become pregnant. Talk to your healthcare provider if this is a concern for you
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. SOMATULINE DEPOT and other medicines may affect each other, causing side effects. SOMATULINE DEPOT may affect the way other medicines work, and other medicines may affect how SOMATULINE DEPOT works. Your dose of SOMATULINE DEPOT or your other medicines may need to be changed.
Especially tell your healthcare provider if you take:
- insulin or other diabetes medicines
- cyclosporine (Gengraf, Neoral, or Sandimmune)
- medicines that lower your heart rate such as beta blockers
How will I receive SOMATULINE DEPOT?
- You will receive a SOMATULINE DEPOT injection every 4 weeks in your healthcare provider's office
- Your healthcare provider may change your dose of SOMATULINE DEPOT or the length of time between your injections. Your healthcare provider will tell you how long you need to receive SOMATULINE DEPOT
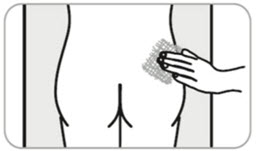
- SOMATULINE DEPOT is injected deep under the skin of the upper outer area of your buttock. Your injection site should change (alternate) between your right and left buttock from one injection of SOMATULINE DEPOT to the next
- During your treatment with SOMATULINE DEPOT for acromegaly, your healthcare provider may do certain blood tests to see if SOMATULINE DEPOT is working
What should I avoid while receiving SOMATULINE DEPOT?
SOMATULINE DEPOT can cause dizziness. If you have dizziness, do not drive a car or operate machinery.
What are the possible side effects of SOMATULINE DEPOT?
SOMATULINE DEPOT may cause serious side effects, including:
- Gallstones (cholelithiasis) and complications that can happen if you have gallstones. Gallstones are a serious but common side effect in people who take SOMATULINE DEPOT and have acromegaly and GEP-NET. Your healthcare provider may check your gallbladder before and during treatment with SOMATULINE DEPOT. Possible complications of gallstones include inflammation and infection of the gall bladder, and pancreatitis. Tell your healthcare provider if you get any symptoms of gallstones, including:
- sudden pain in your upper right stomach area (abdomen)
- yellowing of your skin and whites of your eyes
- nausea
- sudden pain in your right shoulder or between your shoulder blades
- fever with chills
-
Changes in your blood sugar (high blood sugar or low blood sugar). If you have diabetes, test your blood sugar as your healthcare provider tells you to. Your healthcare provider may change your dose of diabetes medicine especially when you first start receiving SOMATULINE DEPOT or if your dose of SOMATULINE DEPOT changes. High blood sugar is a common side effect in people with GEP-NET. Tell your healthcare provider right away if you have any signs or symptoms of high blood sugar or low blood sugar.
Signs and symptoms of high blood sugar may include:
- increased thirst
- increased appetite
- nausea
- weakness or tiredness
- urinating more often than normal
- your breath smells like fruit
Signs and symptoms of low blood sugar may include:
- dizziness or lightheadedness
- sweating
- confusion
- headache
- blurred vision
- slurred speech
- shakiness
- fast heartbeat
- irritability or mood changes
- hunger
- Slow heart rate. Tell your healthcare provider right away if you have slowing of your heart rate or if you have symptoms of a slow heart rate, including:
- dizziness or lightheadedness
- fainting or near-fainting
- chest pain
- shortness of breath
- confusion or memory problems
- weakness, extreme tiredness
- High blood pressure. High blood pressure can happen in people who receive SOMATULINE DEPOT and is a common side effect in people with GEP-NET.
- Changes in thyroid function. SOMATULINE DEPOT can cause the thyroid gland to not make enough thyroid hormones that the body needs (hypothyroidism) in people who have acromegaly. Tell your healthcare provider if you have signs and symptoms of low thyroid hormones levels, including:
- fatigue
- weight gain
- a puffy face
- being cold all of the time
- constipation
- dry skin
- thinning, dry hair
- decreased sweating
- depression
The most common side effects of SOMATULINE DEPOT in people with acromegaly include: - diarrhea
- stomach area (abdominal) pain
- nausea
- pain, itching, or a lump at the injection site
The most common side effects of SOMATULINE DEPOT in people with GEP-NET include: - stomach area (abdominal) pain
- muscle and joint aches
- vomiting
- headache
- pain, itching, or a lump at the injection site
The most common side effects of SOMATULINE DEPOT in people with carcinoid syndrome include: - headache
- dizziness
- muscle spasm
Tell your healthcare provider right away if you have signs of an allergic reaction after receiving SOMATULINE DEPOT, including: - swelling of your face, lips, mouth or tongue
- breathing problems
- fainting, dizziness, feeling lightheaded (low blood pressure)
- itching
- flushing or redness of your skin
- rash
- hives
These are not all the possible side effects of SOMATULINE DEPOT. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. General information about the safe and effective use of SOMATULINE DEPOT.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not receive SOMATULINE DEPOT for a condition for which it was not prescribed. You can ask your healthcare provider for information about SOMATULINE DEPOT that is written for health professionals.
What are the ingredients in SOMATULINE DEPOT?
Active ingredient: lanreotide acetate
Inactive ingredients: water for injection and acetic acid (for pH adjustment)
Manufactured by: Ipsen Pharma Biotech, Parc d'Activities du Plateau de Signes, 83870 Signes, France
Manufactured for: Ipsen Biopharmaceuticals, Inc., 1 Main Street, Unit 700, Cambridge, MA 02142 USA.
For more information, go to www.somatulinedepot.com or call Ipsen Biopharmaceuticals, Inc. at 1-855-463-5127.
- the long-term treatment of people with acromegaly when:
-
INSTRUCTIONS FOR USE
Somatuline®Depot
(lanreotide) InjectionInstructions for Use
IMPORTANT
If you have any questions about this medication or procedure, call 855-463-5127Manufactured By:
Ipsen Pharma Biotech
83870 Signes FranceDistributed By:
Ipsen Biopharmaceuticals, Inc.
Cambridge, MA 02142Before You Begin Injection
- 1. Please follow both sides of these Instructions for Use before using the device. FOLLOW THIS PROCEDURE EXACTLY, AS IT MAY DIFFER FROM YOUR PAST EXPERIENCE.
- 2. Somatuline Depot should be administered by a Healthcare Professional.
- 3. If this device is dropped or damaged in any way, please call 855-463-5127
- A.
Storage of Somatuline Depot
When you receive the medication, follow these steps for storing Somatuline Depot.
Important: Somatuline Depot MUST BE REFRIGERATED. DO NOT ALLOW IT TO REACH ROOM TEMPERATURE UNTIL READY TO USE.- A1.
Remove box from cold pack. Do not open box.
- A2. Check the following:
Note: Call 855-463-5127 if you have any questions.- A3. Place unopened box in your refrigerator.
- Do not place in freezer.
- A2. Check the following:
- A1.
Remove box from cold pack. Do not open box.
- B.
Prepare to Inject
- B1.
Confirm that date of this injection is as prescribed.

- B2. Remove box from refrigerator. Open box and remove contents.
- B3. Confirm that pouch is sealed and not damaged.
- B4. Check that the dose is as prescribed and the expiration date has not passed.

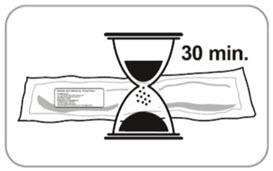
- B5. Let pouch sit for 30 minutes to reach room temperature. Do not open the pouch until ready to inject.
- Injection of cold medication may be painful.
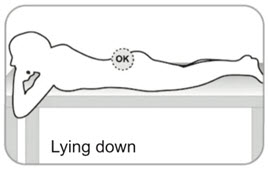
- B6. Find a clean, comfortable area for the patient to relax during procedure.
- It's important that the patient remains as still as possible during the injection.
OR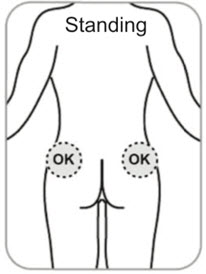
- B7. The person administering the injection must wash his/her hands with soap and water.
Follow the doctor or institution's policy on the use of surgical gloves during this procedure.
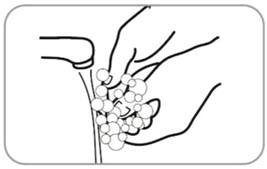
- B8. Tear open pouch along dotted line starting at the notch.
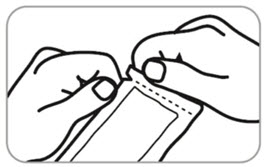
- B9. Do the following:
- Prior to administration, inspect the SOMATULINE DEPOT syringe visually for particulate matter and discoloration. Do not administer if particulate matter or discoloration is observed. The content of the prefilled syringe is a semi-solid phase having a gel-like appearance, with viscous characteristics and a color varying from white to pale yellow. The supersaturated solution can also contain micro bubbles that can clear up during injection. These differences are normal and do not interfere with the quality of the product.
- Confirm that expiration date on the device has not passed.
- Make sure it is the right dosage:
- 60 mg, or
- 90 mg, or
- 120 mg
- Set device aside on the empty pouch.
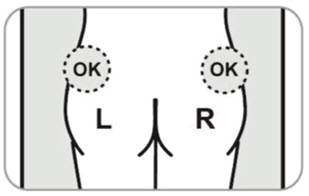
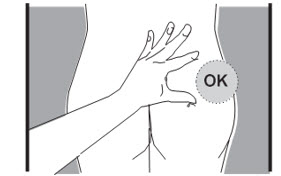
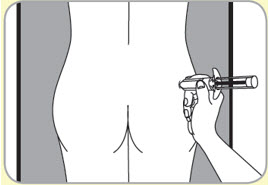
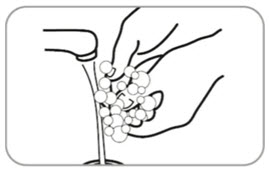
- B10. Choose which side of the buttocks to inject.
- Switch sides with each injection.
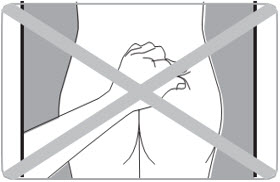
- Avoid areas with moles, scare tissue, reddened skin, or skin that feels bumpy.
- Only inject in the areas market with OK in the picture.
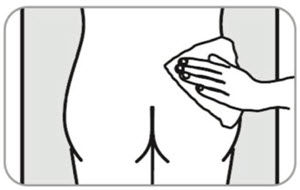
- B11. Clean area to be injected.
- B2. Remove box from refrigerator. Open box and remove contents.
- B1.
Confirm that date of this injection is as prescribed.
Turn page over for additional instructions
Somatuline®Depot
(lanreotide) InjectionInstructions for Use- Page 2
Article No. 1024297
IMPORTANT
If you have any questions about this medication or procedure, call 855-463-5127Ipsen Pharma Biotech
83870 Signes France- C.
Inject
Important: This is a single-use pre-filled syringe with a retractable needle.- ALL the medication must be injected during this use.
- If you drop or damage this device in any way, please call 855-463-5127.
- C1.
BEFORE injecting,
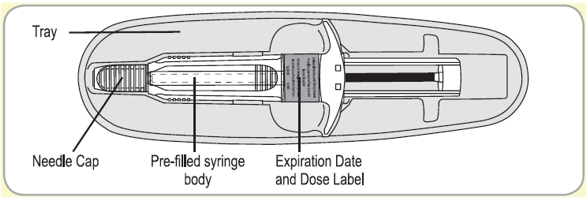
REMOVE the pre-filled syringe from its tray.
Discard tray.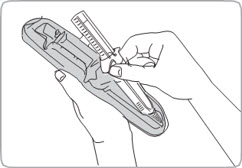
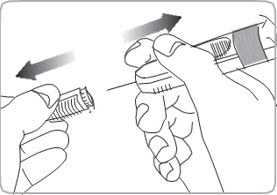
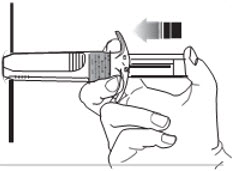
- C2. REMOVE NEEDLE CAP
- HOLD PRE-FILLED SYRINGE BY THE SYRINGE BODY and PULL OFF the needle cap to remove it.
- DO NOT HOLD the pre-filled syringe by the plunger
- Discard needle cap.
- C3. Hold pre-filled syringe by the syringe body. The pre-filled syringe is now ready for injection.
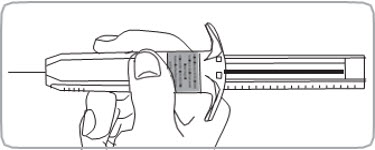
- C4. FLATTEN injection area using the thumb and index finger of your other hand to stretch the skin.
- Do NOT pinch skin.
- C5. Insert needle PERPENDICULAR to the skin (90 degree angle).
- Use a strong, straight, dart-like motion to quickly INSERT THE NEEDLE ALL THE WAY INTO THE SKIN.
- It is very important that you insert the needle COMPLETELY. You should not see any needle once it is fully inserted.
- Do not aspirate (do not draw back).
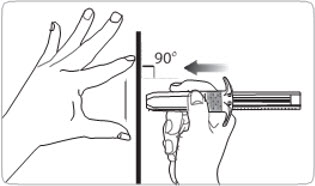
- Do not insert needle at an acute angle.
- Make sure needle is fully inserted.
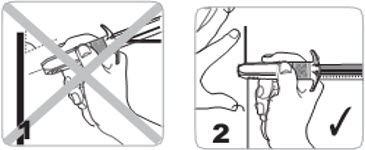
- C6. When needle is completely inserted, release injection site that has been flattened by your hand.
- C7. Push plunger with STEADY, VERY FIRM PRESSURE.
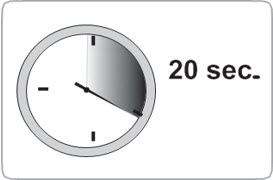
- The medication is thicker and harder to push than you might expect.
- While depressing plunger, slowly count to 20 and CONTINUE STEADY PRESSURE on the plunger. You may find it helpful to say:
a. "1 one-thousand"
b. "2 one-thousand"
c. "3 one-thousand"
- C8. GIVE PLUNGER A FINAL PUSH to make sure you cannot depress the plunger further.
Continue steady pressure with your thumb.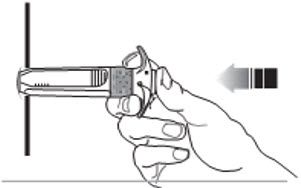
- C9. While continuing to hold down the plunger, remove the needle from the injection site (1), then allow the needle to retract by removing your thumb from the plunger (2) (3).
- If needle does not retract, push plunger again to engage safety mechanism. The needle will then retract.
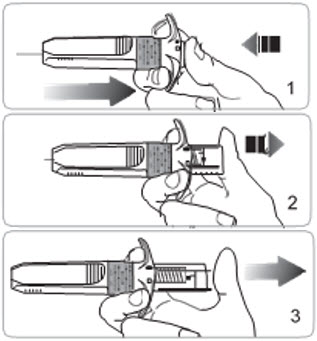
- C10. If needed, gently apply gauze pad to injection area.
- Important: Never rub or massage the injection site.
- C2. REMOVE NEEDLE CAP
- D. Dispose of Device
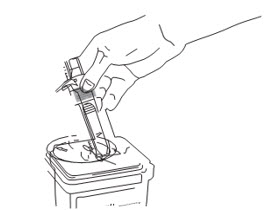
- D1.
Discard used device into a hard plastic container with a screw top (such as a detergent bottle) or sharps container as per your institutional policy.
- D2. Wash your hands.
- D2. Wash your hands.
-
PRINCIPAL DISPLAY PANEL - 60 mg/0.2 mL Syringe Carton
Rx only
Somatuline® Depot
(lanreotide) Injection
60 mg/0.2 mLNDC 15054 1060 3
For deep subcutaneous injection
For single use only. Discard unused portion.
Somatuline® Depot should be administered by a healthcare professional.Leave at room temperature for 30 minutes before administration.
CONTENTS: This box contains one (1) pre-filled syringe.
60 mg/0.2 mL
IPSEN

-
PRINCIPAL DISPLAY PANEL - 60 mg/0.2 mL Syringe Carton - NDC: 15054-1060-4
Rx only
Somatuline® Depot
(lanreotide) Injection
60 mg/0.2 mLNDC 15054 1060 4
For deep subcutaneous injection
For single use only. Discard unused portion.
Somatuline® Depot should be administered by a healthcare professional.Leave at room temperature for 30 minutes before administration.
CONTENTS: This box contains one (1) pre-filled syringe.
60 mg/0.2 mL
IPSEN
-
PRINCIPAL DISPLAY PANEL - 90 mg/0.3 mL Syringe Carton
Rx only
Somatuline® Depot
(lanreotide) Injection
90 mg/0.3 mLNDC 15054 1090 3
For deep subcutaneous injection
For single use only. Discard unused portion.
Somatuline® Depot should be administered by a healthcare professional.Leave at room temperature for 30 minutes before administration.
CONTENTS: This box contains one (1) pre-filled syringe.
90 mg/0.3 mL
IPSEN
-
PRINCIPAL DISPLAY PANEL - 90 mg/0.3 mL Syringe Carton - NDC: 15054-1090-4
Rx only
Somatuline® Depot
(lanreotide) Injection
90 mg/0.3 mLNDC 15054 1090 4
For deep subcutaneous injection
For single use only. Discard unused portion.
Somatuline® Depot should be administered by a healthcare professional.Leave at room temperature for 30 minutes before administration.
CONTENTS: This box contains one (1) pre-filled syringe.
90 mg/0.3 mL
IPSEN
-
PRINCIPAL DISPLAY PANEL - 120 mg/0.5 mL Syringe Carton
Rx only
Somatuline® Depot
(lanreotide) Injection
120 mg/0.5 mLNDC 15054 1120 3
For deep subcutaneous injection
For single use only. Discard unused portion.
Somatuline® Depot should be administered by a healthcare professional.Leave at room temperature for 30 minutes before administration.
CONTENTS: This box contains one (1) pre-filled syringe.
120 mg/0.5 mL
IPSEN

-
PRINCIPAL DISPLAY PANEL - 120 mg/0.5 mL Syringe Carton - NDC: 15054-1120-4
Rx only
Somatuline® Depot
(lanreotide) Injection
120 mg/0.5 mLNDC 15054 1120 4
For deep subcutaneous injection
For single use only. Discard unused portion.
Somatuline® Depot should be administered by a healthcare professional.Leave at room temperature for 30 minutes before administration.
CONTENTS: This box contains one (1) pre-filled syringe.
120 mg/0.5 mL
IPSEN

-
INGREDIENTS AND APPEARANCE
SOMATULINE DEPOT
lanreotide acetate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 15054-1060 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength lanreotide acetate (UNII: IEU56G3J9C) (lanreotide - UNII:0G3DE8943Y) lanreotide 60 mg in 0.2 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Acetic Acid (UNII: Q40Q9N063P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 15054-1060-3 1 in 1 CARTON 11/14/2007 1 1 in 1 POUCH 1 0.2 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC: 15054-1060-4 1 in 1 CARTON 09/01/2019 2 1 in 1 POUCH 2 0.2 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022074 11/14/2007 SOMATULINE DEPOT
lanreotide acetate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 15054-1090 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength lanreotide acetate (UNII: IEU56G3J9C) (lanreotide - UNII:0G3DE8943Y) lanreotide 90 mg in 0.3 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Acetic Acid (UNII: Q40Q9N063P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 15054-1090-3 1 in 1 CARTON 11/14/2007 1 1 in 1 POUCH 1 0.3 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC: 15054-1090-4 1 in 1 CARTON 09/01/2019 2 1 in 1 POUCH 2 0.3 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022074 11/14/2007 SOMATULINE DEPOT
lanreotide acetate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 15054-1120 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength lanreotide acetate (UNII: IEU56G3J9C) (lanreotide - UNII:0G3DE8943Y) lanreotide 120 mg in 0.5 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Acetic Acid (UNII: Q40Q9N063P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 15054-1120-3 1 in 1 CARTON 11/14/2007 1 1 in 1 POUCH 1 0.5 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC: 15054-1120-4 1 in 1 CARTON 09/01/2019 2 1 in 1 POUCH 2 0.5 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022074 11/14/2007 Labeler - Ipsen Biopharmaceuticals, Inc. (118461578) Establishment Name Address ID/FEI Business Operations IPSEN PHARMA BIOTECH 502570286 MANUFACTURE(15054-1060, 15054-1090, 15054-1120)
Trademark Results [SOMATULINE DEPOT]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SOMATULINE DEPOT 77863535 3920840 Live/Registered |
IPSEN PHARMA S.A.S. 2009-11-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.