CYCLOSPORINE/CHONDROITIN PF- cyclosporine/chondroitin sulfate pf emulsion
Cyclosporine/Chondroitin PF by
Drug Labeling and Warnings
Cyclosporine/Chondroitin PF by is a Prescription medication manufactured, distributed, or labeled by Imprimis NJOF, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- STORAGE AND HANDLING
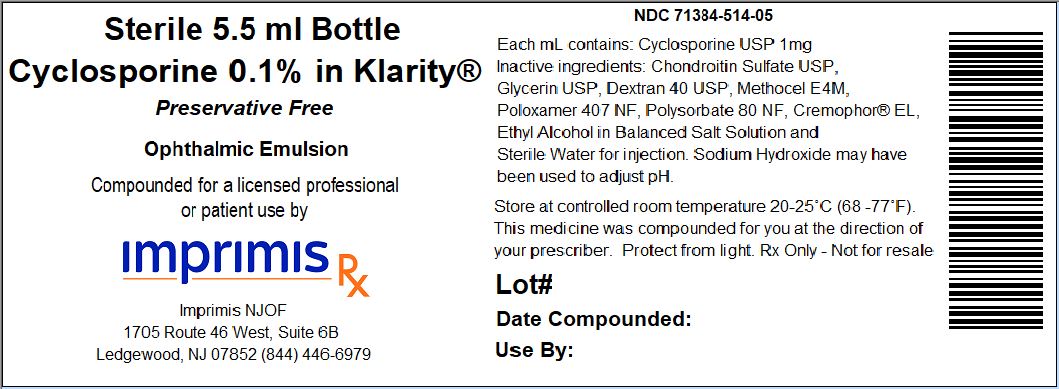
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CYCLOSPORINE/CHONDROITIN PF
cyclosporine/chondroitin sulfate pf emulsionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71384-514 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CYCLOSPORINE (UNII: 83HN0GTJ6D) (CYCLOSPORINE - UNII:83HN0GTJ6D) CYCLOSPORINE 1 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71384-514-05 5.5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 07/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/01/2018 Labeler - Imprimis NJOF, LLC (080431967)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.