TERAZOL 7- terconazole cream TERAZOL 3- terconazole cream TERAZOL 3- terconazole suppository
Terazol 3 by
Drug Labeling and Warnings
Terazol 3 by is a Prescription medication manufactured, distributed, or labeled by Janssen Pharmaceuticals, Inc., Janssen Cilag Manufacturing, LLC, Noramco, Inc., Janssen Ortho LLC, Janssen Pharmaceuticals, Inc, Jubilant HollisterStier General Partnership. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
TERAZOL® 7 (terconazole) Vaginal Cream 0.4% is a white to off-white, water washable cream for intravaginal administration containing 0.4% of the antifungal agent terconazole, cis-1-[p-[[2-(2,4-Dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-4-isopropylpiperazine, compounded in a cream base consisting of butylated hydroxyanisole, cetyl alcohol, isopropyl myristate, polysorbate 60, polysorbate 80, propylene glycol, stearyl alcohol, and purified water.
TERAZOL® 3 (terconazole) Vaginal Cream 0.8% is a white to off-white, water washable cream for intravaginal administration containing 0.8% of the antifungal agent terconazole, cis-1-[p-[[2-(2,4-Dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-4-isopropylpiperazine, compounded in a cream base consisting of butylated hydroxyanisole, cetyl alcohol, isopropyl myristate, polysorbate 60, polysorbate 80, propylene glycol, stearyl alcohol, and purified water.
TERAZOL® 3 (terconazole) Vaginal Suppositories are white to off-white suppositories for intravaginal administration containing 80 mg of the antifungal agent terconazole, cis-1-[p-[[2-(2,4-Dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-4-isopropylpiperazine, in triglycerides derived from coconut and/or palm kernel oil (a base of hydrogenated vegetable oils) and butylated hydroxyanisole.
The structural formula of terconazole is as follows:
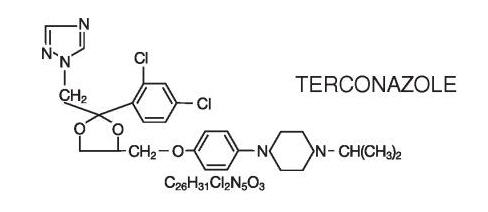
Terconazole, a triazole derivative, is a white to almost white powder with a molecular weight of 532.47. It is insoluble in water; sparingly soluble in ethanol; and soluble in butanol.
-
CLINICAL PHARMACOLOGY
Absorption
Following a single intravaginal application of a suppository containing 240 mg 14C-terconazole to healthy women, approximately 70% (range: 64–76%) of terconazole remains in the vaginal area during the suppository retention period (16 hours); approximately 10% (range: 5–16%) of the administered radioactivity was absorbed systemically over 7 days. Maximum plasma concentrations of terconazole occur 5 to 10 hours after intravaginal application of the cream or suppository. Systemic exposure to terconazole is approximately proportional to the applied dose, whether as the cream or suppository. The rate and extent of absorption of terconazole are similar in patients with vulvovaginal candidiasis (pregnant or non-pregnant) and healthy subjects.
Distribution
Terconazole is highly protein bound (94.9%) in human plasma and the degree of binding is independent of drug concentration over the range of 0.01 to 5.0 mcg/mL.
Elimination
Across various studies in healthy women, after single or multiple intravaginal administration of terconazole as the cream or suppository/ovule, the mean elimination half-life of unchanged terconazole ranged from 6.4 to 8.5 hours. Following a single intravaginal administration of a suppository containing 240 mg 14C-terconazole to hysterectomized or tubal ligated women, approximately 3 to 10% (mean ± SD: 5.7 ± 3.0%) of the administered radioactivity was eliminated in the urine and 2 to 6% (mean ± SD: 4.2 ± 1.6%) was eliminated in the feces during the 7-day collection period.
Multiple Dosing
There is no significant increase in maximum plasma concentration or overall exposure (AUC) after multiple daily applications of the cream for 7 days or suppositories for 3 days.
Photosensitivity reactions were observed in some normal volunteers following repeated dermal application of terconazole 2.0% and 0.8% creams under conditions of filtered artificial ultraviolet light.
Photosensitivity reactions have not been observed in U.S. and foreign clinical trials in patients who were treated with terconazole suppositories or vaginal cream (0.4% and 0.8%).
Microbiology
Mechanism of action
Terconazole, an azole antifungal agent, inhibits fungal cytochrome P-450-mediated 14 alpha-lanosterol demethylase enzyme. This enzyme functions to convert lanosterol to ergosterol. The accumulation of 14 alpha-methyl sterols correlates with the subsequent loss of ergosterol in the fungal cell wall and may be responsible for the antifungal activity of terconazole. Mammalian cell demethylation is less sensitive to terconazole inhibition.
Activity in vitro
Terconazole exhibits antifungal activity in vitro against Candida albicans and other Candida species. The MIC values of terconazole against most Lactobacillus spp. typically found in the human vagina were ≥128 mcg/mL; therefore these beneficial bacteria are not affected by drug treatment.
-
INDICATIONS AND USAGE
TERAZOL® 7 (terconazole) Vaginal Cream 0.4%, TERAZOL® 3 (terconazole) Vaginal Cream 0.8% and TERAZOL® 3 (terconazole) Vaginal Suppositories 80 mg are indicated for the local treatment of vulvovaginal candidiasis (moniliasis). As these products are effective only for vulvovaginitis caused by the genus Candida, the diagnosis should be confirmed by KOH smears and/or cultures.
- CONTRAINDICATIONS
- WARNINGS
-
PRECAUTIONS
General
For vulvovaginal use only. TERAZOL® is not for ophthalmic or oral use. Discontinue use and do not retreat with terconazole if sensitization, irritation, fever, chills or flu-like symptoms are reported during use.
The base contained in the suppository formulation may interact with certain rubber or latex products, such as those used in vaginal contraceptive diaphragms or latex condoms; therefore concurrent use is not recommended.
Laboratory Tests
If there is lack of response to terconazole, appropriate microbiologic studies (standard KOH smear and/or cultures) should be repeated to confirm the diagnosis and rule out other pathogens.
Drug Interactions
The therapeutic effect of terconazole is not affected by oral contraceptive usage.
The levels of estradiol and progesterone did not differ significantly when 0.8% terconazole vaginal cream was administered to healthy female volunteers established on a low dose oral contraceptive.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Studies to determine the carcinogenic potential of terconazole have not been performed.
Pregnancy
Teratogenic Effects
There was no evidence of teratogenicity when terconazole was administered orally up to 40 mg/kg/day (25× the recommended intravaginal human dose of the suppository formulation, 50× the recommended intravaginal human dose of the 0.8% vaginal cream formulation, and 100× the intravaginal human dose of the 0.4% vaginal cream formulation) in rats, or 20 mg/kg/day in rabbits, or subcutaneously up to 20 mg/kg/day in rats.
Dosages at or below 10 mg/kg/day produced no embryotoxicity; however, there was a delay in fetal ossification at 10 mg/kg/day in rats. There was some evidence of embryotoxicity in rabbits and rats at 20–40 mg/kg. In rats, this was reflected as a decrease in litter size and number of viable young and reduced fetal weight. There was also delay in ossification and an increased incidence of skeletal variants.
The no-effect dose of 10 mg/kg/day resulted in a mean peak plasma level of terconazole in pregnant rats of 0.176 mcg/mL which exceeds by 44 times the mean peak plasma level (0.004 mcg/mL) seen in normal subjects after intravaginal administration of terconazole 0.4% vaginal cream, by 30 times the mean peak plasma level (0.006 mcg/mL) seen in normal subjects after intravaginal administration of terconazole 0.8% vaginal cream, and by 17 times the mean peak plasma level (0.010 mcg/mL) seen in normal subjects after intravaginal administration of terconazole 80 mg vaginal suppository. This safety assessment does not account for possible exposure of the fetus through direct transfer to terconazole from the irritated vagina by diffusion across amniotic membranes.
Since terconazole is absorbed from the human vagina, it should not be used in the first trimester of pregnancy unless the physician considers it essential to the welfare of the patient.
Terconazole may be used during the second and third trimester if the potential benefit outweighs the possible risks to the fetus.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Animal studies have shown that rat offspring exposed via the milk of treated (40 mg/kg/orally) dams showed decreased survival during the first few post-partum days, but overall pup weight and weight gain were comparable to or greater than controls throughout lactation. Because many drugs are excreted in human milk, and because of the potential for adverse reaction in nursing infants from terconazole, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
-
ADVERSE REACTIONS
Adverse Reactions from Clinical Trials
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
TERAZOL® 7 (terconazole) Vaginal Cream 0.4%
During controlled clinical studies conducted in the United States, 521 patients with vulvovaginal candidiasis were treated with terconazole 0.4% vaginal cream. Based on comparative analyses with placebo, the adverse experiences considered most likely related to terconazole 0.4% vaginal cream were headache (26% vs. 17% with placebo) and body pain (2.1% vs. 0% with placebo). Fever (1.7% vs. 0.5% with placebo) and chills (0.4% vs. 0.0% with placebo), vulvovaginal burning, itching and irritation have also been reported. The adverse drug experience on terconazole most frequently causing discontinuation was vulvovaginal itching.
TERAZOL® 3 (terconazole) Vaginal Cream 0.8%
During controlled clinical studies conducted in the United States, patients with vulvovaginal candidiasis were treated with terconazole 0.8% vaginal cream for three days. Based on comparative analyses with placebo and a standard agent, the adverse experiences considered most likely related to terconazole 0.8% vaginal cream were headache (21% vs. 16% with placebo) and dysmenorrhea (6% vs. 2% with placebo). Other adverse experiences reported with terconazole 0.8% vaginal cream were abdominal pain (3.4% vs. 1% with placebo) and fever (1% vs. 0.3% with placebo). The adverse drug experience most frequently causing discontinuation of therapy was vulvovaginal itching, 0.7% with the terconazole 0.8% vaginal cream group and 0.3% with the placebo group.
TERAZOL® 3 (terconazole) Vaginal Suppositories 80 mg
During controlled clinical studies conducted in the United States, 284 patients with vulvovaginal candidiasis were treated with terconazole 80 mg vaginal suppositories. Based on comparative analyses with placebo (295 patients), the adverse experiences considered adverse reactions most likely related to terconazole 80 mg vaginal suppositories were headache (30.3% vs. 20.7% with placebo) and pain of the female genitalia (4.2% vs. 0.7% with placebo). Adverse reactions that have also been reported but were not statistically significantly different from placebo were burning (15.2% vs. 11.2% with placebo) and body pain (3.9% vs. 1.7% with placebo). Fever (2.8% vs. 1.4% with placebo) and chills (1.8% vs. 0.7% with placebo) have also been reported. The adverse drug experience on terconazole most frequently causing discontinuation was burning (2.5% vs. 1.4% with placebo) and pruritus (1.8% vs. 1.4% with placebo).
Post-marketing Experience
The following adverse drug reactions have been first identified during post-marketing experience with TERAZOL®. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
General: Asthenia, Influenza-Like Illness consisting of multiple listed reactions including fever and chills, nausea, vomiting, myalgia, arthralgia, malaise
Immune: Hypersensitivity, Anaphylaxis, Face Edema
Nervous: Dizziness
Respiratory: Bronchospasm
Skin: Rash, Toxic Epidermal Necrolysis, Urticaria
-
OVERDOSAGE
In the rat, the oral LD 50 values were found to be 1741 and 849 mg/kg for the male and female, respectively. The oral LD 50 values for the male and female dog were ≅1280 and ≥640 mg/kg, respectively.
In the event of oral ingestion of suppository or cream, supportive and symptomatic measures should be carried out. If the cream is accidentally applied to the eyes, wash with clean water or saline and seek medical attention if symptoms persist.
-
DOSAGE AND ADMINISTRATION
TERAZOL® 7 (terconazole) Vaginal Cream 0.4%
One full applicator (5 g) of TERAZOL® 7 Vaginal Cream (20 mg terconazole) should be administered intravaginally once daily at bedtime for seven consecutive days.
TERAZOL® 3 (terconazole) Vaginal Cream 0.8%
One full applicator (5 g) of TERAZOL® 3 Vaginal Cream (40 mg terconazole) should be administered intravaginally once daily at bedtime for three consecutive days.
TERAZOL® 3 (terconazole) Vaginal Suppositories 80 mg
One TERAZOL® 3 Vaginal Suppository (80 mg terconazole) should be administered intravaginally once daily at bedtime for three consecutive days.
Before prescribing another course of therapy, the diagnosis should be reconfirmed by smears and/or cultures and other pathogens commonly associated with vulvovaginitis ruled out. The therapeutic effect of these products is not affected by menstruation.
-
HOW SUPPLIED
TERAZOL® 7 (terconazole) Vaginal Cream 0.4% is available in 45 g (NDC: 50458-535-01) tubes with an ORTHO1 Measured-Dose Applicator. Store at Controlled Room Temperature 15–30°C (59–86°F).
TERAZOL® 3 (terconazole) Vaginal Cream 0.8% is available in 20 g (NDC: 50458-536-01) tubes with an ORTHO1 Measured-Dose Applicator. Store at Controlled Room Temperature 15–30°C (59–86°F).
TERAZOL® 3 (terconazole) Vaginal Suppositories 80 mg are available as 2.5 g, elliptically-shaped white to off-white suppositories in packages of three (NDC: 50458-531-01) with a vaginal applicator. Store at Controlled Room Temperature 15–30°C (59–86°F).
- 1 Trademark
-
SPL UNCLASSIFIED SECTION
Manufactured by:
Janssen Ortho, LLC, Manati, Puerto Rico 00674 (for the Vaginal Cream)
Jubilant HollisterStier General Partnership, Kirkland, Quebec, Canada H9H 4J4 (for the Vaginal Cream and Vaginal Suppositories)
Manufactured for:
(logo)
Janssen Pharmaceuticals, Inc. Titusville, New Jersey 08560
© Janssen Pharmaceuticals, Inc. 1998
Revised May 2018
-
PATIENT PACKAGE INSERT
TERAZOL® 7 VAGINAL CREAM 0.4%
(terconazole)TERAZOL® 3 VAGINAL CREAM 0.8%
(terconazole)PATIENT INSTRUCTIONS
Filling the applicator:
- 1.
Remove the cap from the tube.
- 2. Use the pointed tip on the top of the cap to puncture the seal on the tube.
- 3. Screw the applicator onto the tube.
- 4. Squeeze the tube from the bottom and fill the applicator until the plunger stops.
- 5. Unscrew the applicator from the tube.
- 2. Use the pointed tip on the top of the cap to puncture the seal on the tube.
Using the applicator:
- 1.
Lie on your back with your knees drawn up toward your chest.
- 2. Holding the applicator by the ribbed end of the barrel, insert the filled applicator into the vagina as far as it will comfortably go.
- 3. Slowly press the plunger of the applicator to release the cream into the vagina.
- 4. Remove the applicator from the vagina.
- 5. Apply one applicatorful each night for as many days at bedtime, as directed by your doctor.
- 2. Holding the applicator by the ribbed end of the barrel, insert the filled applicator into the vagina as far as it will comfortably go.
Cleaning the applicator: (Does not apply to sample applicators, which are for one time use only)
After each use, you should thoroughly clean the applicator by following the procedure below:
- 1.
Pull the plunger out of the barrel.
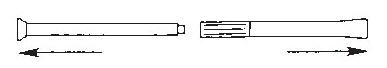
- 2. Wash the pieces with lukewarm, soapy water, and dry them thoroughly.
- 3. Put the applicator back together by gently pushing the plunger into the barrel as far as it will go.
- 2. Wash the pieces with lukewarm, soapy water, and dry them thoroughly.
NOTE: Store the cream at Controlled Room Temperature 15–30°C (59–86°F). See end flap for lot number and expiration date.
TERAZOL® 3 VAGINAL SUPPOSITORIES 80 mg
(terconazole)Three oval suppositories, for use inside the vagina only.
Designed to be inserted into the vagina.
HOW TO USE:
Place one suppository into the vagina each night at bedtime, for 3 nights, as directed by your doctor. The TERAZOL® Suppository is self-lubricating and may be inserted with or without the applicator.
- A.
Insertion with the applicator
- 1.
Filling the applicator
- Break off suppository from the plastic strip.
- Pull the plastic completely apart at the notched end.
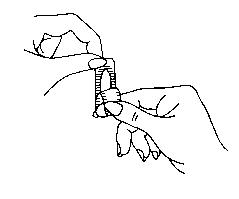
- Place the flat end of the suppository into the open end of the applicator as shown. You are now ready to insert the suppository into the vagina.
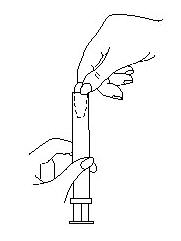
- 2. Using the applicator
- Lie on your back with your knees drawn up toward your chest.
- Holding the applicator by the ribbed end of the barrel, gently insert it into the vagina as far as it will comfortably go.
- Press the plunger to release the suppository into the vagina.
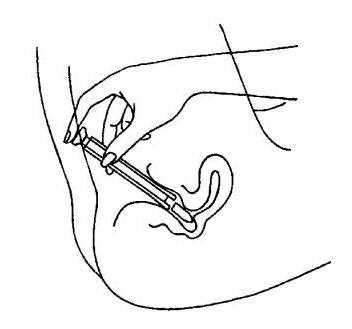
- Remove the applicator from the vagina.
- 3. Cleaning the applicator (Does not apply to sample applicators, which are for one time use only)
After each use, you should thoroughly clean the applicator by following the procedure below:
- Pull the plunger out of the barrel.
- Wash both pieces with lukewarm, soapy water, and dry them thoroughly.
- Put the applicator back together by gently pushing the plunger into the barrel as far as it will go.
- Break off suppository from the plastic strip.
- Lie on your back with your knees drawn up toward your chest.
- Place the suppository on the tip of your finger as shown.
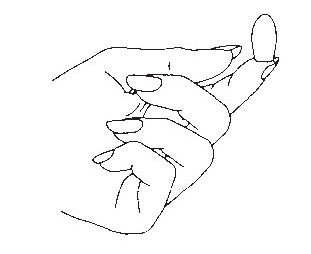
- Insert the suppository gently into the vagina as far as it will comfortably go.
- 1.
Filling the applicator
NOTE: Store the suppositories at Controlled Room Temperature 15–30°C (59–86°F). See end flap for lot number and expiration date.
A WORD ABOUT YEAST INFECTIONS
Why do yeast infections occur?
Yeast infections are caused by an organism called Candida (KAN di duh). It may be present in small and harmless amounts in the mouth, digestive tract, and vagina. Sometimes the natural balance of the vagina becomes upset. This may lead to rapid growth of Candida, which results in a yeast infection. Symptoms of a yeast infection include itching, burning, redness, and an abnormal discharge.
Your doctor can make the diagnosis of a yeast infection by evaluating your symptoms and looking at a sample of the discharge under the microscope.
How can I prevent yeast infections?
Certain factors may increase your chance of developing a yeast infection. These factors don't actually cause the problem, but they may create a situation that allows the yeast to grow rapidly.
- Clothing: Tight jeans, nylon underwear, pantyhose, and wet bathing suits can hold in heat and moisture (two conditions in which yeast organisms thrive). Looser pants or skirts, 100% cotton underwear, and stockings may help avoid this problem.
- Diet: Cutting down on sweets, milk products, and artificial sweeteners may reduce the risk of yeast infections.
- Antibiotics: Antibiotics work by eliminating disease-causing organisms. While they are helpful in curing other problems, antibiotics may lead to an overgrowth of Candida in the vagina.
- Pregnancy: Hormonal changes in the body during pregnancy encourage the growth of yeast. This is a very common time for an infection to occur. Until the baby is born, it may be hard to completely eliminate yeast infections. If you believe you are pregnant, tell your doctor.
- Menstruation: Sometimes monthly changes in hormone levels may lead to yeast infections.
- Diabetes: In addition to heat and moisture, yeast thrives on sugar. Because diabetics often have sugar in their urine, their vaginas are rich in this substance. Careful control of diabetes may help prevent yeast infection.
Controlling these factors can help eliminate yeast infections and may prevent them from coming back.
Some other helpful tips:
- 1.
For best results, be sure to use the medication as prescribed by your doctor, even if you feel better quickly.
- 2. Avoid sexual intercourse, if your doctor advises you to do so. The suppository formulation (not the cream) may damage the diaphragm or latex condom. Therefore, use of the diaphragm or latex condom during therapy with the suppository is not recommended. Consult your physician.
- 3. If your partner has any penile itching, redness, or discomfort, he should consult his physician and mention that you are being treated for a yeast infection.
- 4. You can use the medication even if you are having your menstrual period. However, you should not use tampons because they may absorb the medication. Instead, use external pads or napkins until you have finished your medication. You may also wish to wear a sanitary napkin if the vaginal medication leaks.
- 5. Dry the genital area thoroughly after showering, bathing, or swimming. Change out of a wet bathing suit or damp exercise clothes as soon as possible. A dry environment is less likely to encourage the growth of yeast.
- 6. Wipe from front to rear (away from the vagina) after a bowel movement.
- 7. Don't douche unless your doctor specifically tells you to do so. Douching may disturb the vaginal balance.
- 8. Don't scratch if you can help it. Scratching can cause more irritation and spread the infection.
- 9. Discuss with your physician any medication you are already taking. Certain types of medication can make your vagina more susceptible to infection.
- 10. Eat nutritious meals to promote your general health.
- 2. Avoid sexual intercourse, if your doctor advises you to do so. The suppository formulation (not the cream) may damage the diaphragm or latex condom. Therefore, use of the diaphragm or latex condom during therapy with the suppository is not recommended. Consult your physician.
Manufactured by:
- Janssen Ortho, LLC, Manati, Puerto Rico 00674 (for the Vaginal Cream)
- Jubilant HollisterStier General Partnership, Kirkland, Quebec, Canada H9H 4J4 (for the Vaginal Cream and Vaginal Suppositories)
Manufactured for:
(logo)
Janssen Pharmaceuticals, Inc. Titusville, New Jersey 08560
© Janssen Pharmaceuticals, Inc. 1998
Revised July 2014 - 1.
Remove the cap from the tube.
-
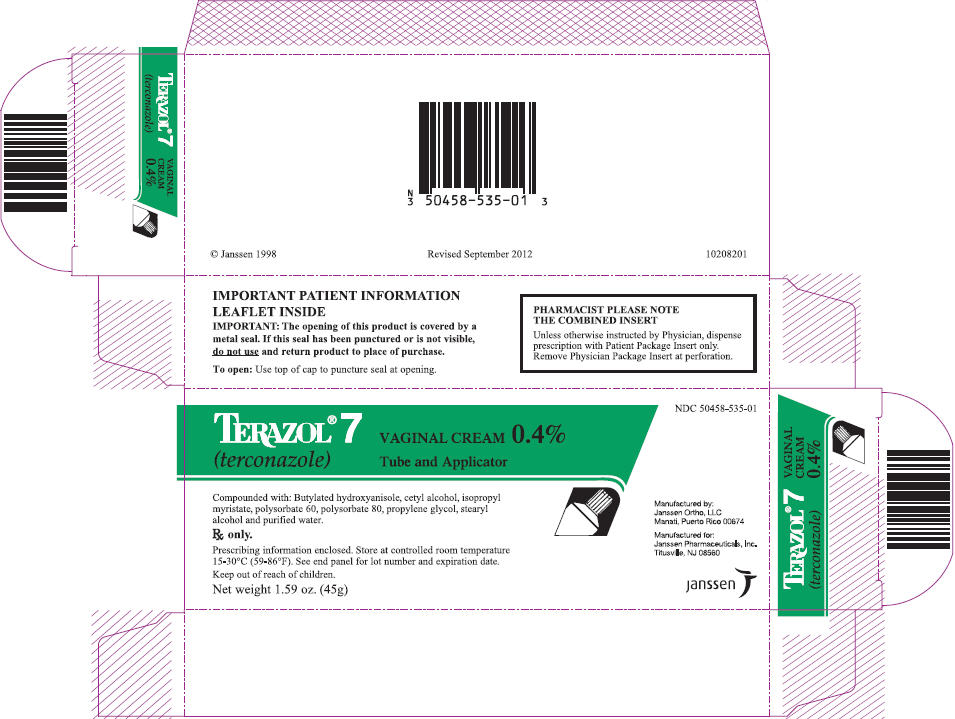
PRINCIPAL DISPLAY PANEL - 45g Tube Carton
NDC: 50458-535-01
TERAZOL® 7
(terconazole)VAGINAL CREAM 0.4%
Tube and ApplicatorCompounded with: Butylated hydroxyanisole, cetyl alcohol, isopropyl
myristate, polysorbate 60, polysorbate 80, propylene glycol, stearyl
alcohol and purified water.Rx only.
Prescribing information enclosed. Store at controlled room temperature
15-30°C (59-86°F). See end panel for lot number and expiration date.Keep out of reach of children.
Net weight 1.59 oz. (45g)
Manufactured by:
Janssen Ortho, LLC
Manati, Puerto Rico 00674Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, NJ 08560janssen
-
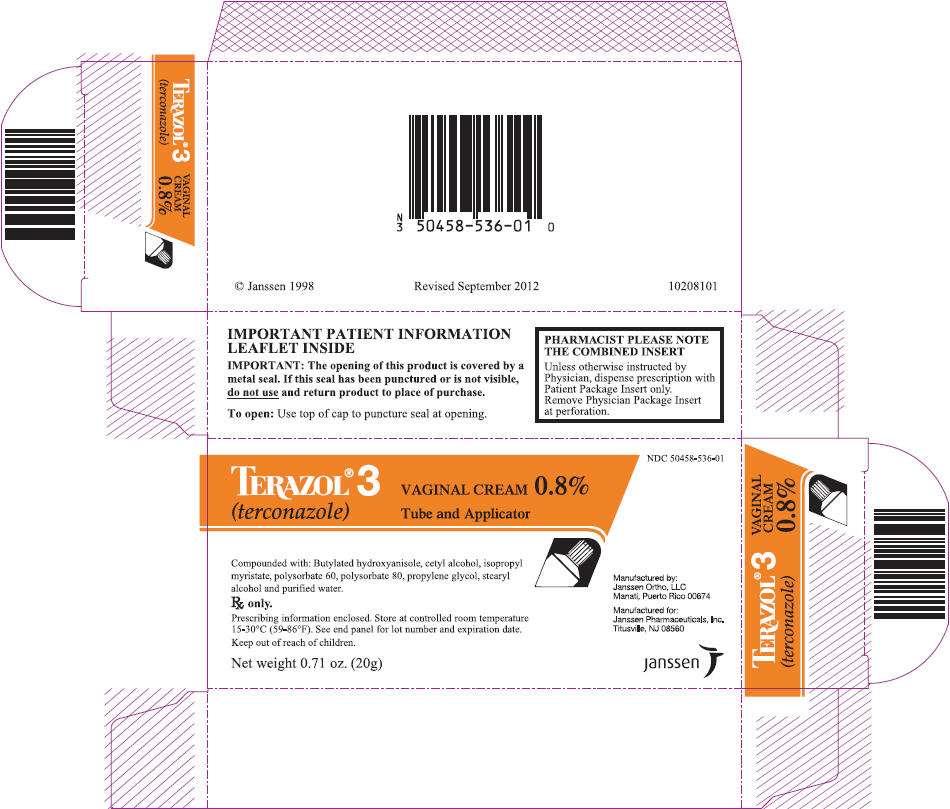
PRINCIPAL DISPLAY PANEL - 20g Tube Carton
NDC: 50458-536-01
TERAZOL® 3
(terconazole)VAGINAL CREAM 0.8%
Tube and ApplicatorCompounded with: Butylated hydroxyanisole, cetyl alcohol, isopropyl
myristate, polysorbate 60, polysorbate 80, propylene glycol, stearyl
alcohol and purified water.Rx only.
Prescribing information enclosed. Store at controlled room temperature
15-30°C (59-86°F). See end panel for lot number and expiration date.Keep out of reach of children.
Net weight 0.71 oz. (20g)
Manufactured by:
Janssen Ortho, LLC
Manati, Puerto Rico 00674Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, NJ 08560janssen
-
PRINCIPAL DISPLAY PANEL - 80mg Dose Pack Carton
TERAZOL® 3
(terconazole)VAGINAL SUPPOSITORIES 80mg
With ApplicatorNDC: 0062-5351-01
Each suppository contains terconazole 80 mg compounded with
triglycerides derived from coconut and/or palm kernel oil (a base
of hydrogenated vegetable oils) and butylated hydroxyanisole.Rx only.
Prescribing information enclosed.
Store at 15-30°C (59-86°F).
See end panel for lot number and expiration date.
Keep out of reach of children.3 suppositories with vaginal applicator.
Manufactured by:
DRAXIS Specialty Pharmaceuticals Inc.
Kirkland, Quebec, Canada
H9H 4J4ORTHO WOMEN'S HEALTH & UROLOGY™
DIVISION OF ORTHO-MCNEIL-JANSSEN PHARMACEUTICALS, INC.Manufactured for:
Ortho Women's Health & Urology,
Division of Ortho-McNeil-Janssen
Pharmaceuticals, Inc.
Raritan, New Jersey 08869
-
INGREDIENTS AND APPEARANCE
TERAZOL 7
terconazole creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-535 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TERCONAZOLE (UNII: 0KJ2VE664U) (TERCONAZOLE - UNII:0KJ2VE664U) TERCONAZOLE 4 mg in 1 g Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) CETYL ALCOHOL (UNII: 936JST6JCN) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) POLYSORBATE 60 (UNII: CAL22UVI4M) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) WATER (UNII: 059QF0KO0R) Product Characteristics Color WHITE (white to off-white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-535-01 1 in 1 CARTON 12/31/1987 06/30/2018 1 45 g in 1 TUBE, WITH APPLICATOR; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA019579 12/31/1987 TERAZOL 3
terconazole creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-536 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TERCONAZOLE (UNII: 0KJ2VE664U) (TERCONAZOLE - UNII:0KJ2VE664U) TERCONAZOLE 8 mg in 1 g Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) CETYL ALCOHOL (UNII: 936JST6JCN) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) POLYSORBATE 60 (UNII: CAL22UVI4M) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) WATER (UNII: 059QF0KO0R) Product Characteristics Color WHITE (white to off-white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-536-01 1 in 1 CARTON 02/21/1991 10/31/2016 1 20 g in 1 TUBE, WITH APPLICATOR; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA019964 02/21/1991 10/31/2016 TERAZOL 3
terconazole suppositoryProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-531 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TERCONAZOLE (UNII: 0KJ2VE664U) (TERCONAZOLE - UNII:0KJ2VE664U) TERCONAZOLE 80 mg Inactive Ingredients Ingredient Name Strength COCONUT OIL (UNII: Q9L0O73W7L) PALM KERNEL OIL (UNII: B0S90M0233) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) Product Characteristics Color WHITE (white to off-white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-531-01 1 in 1 CARTON 05/24/1988 05/25/1988 1 3 in 1 DOSE PACK; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA019641 05/24/1988 05/25/1988 Labeler - Janssen Pharmaceuticals, Inc. (063137772) Establishment Name Address ID/FEI Business Operations Janssen Cilag Manufacturing, LLC 963971374 API MANUFACTURE(50458-535, 50458-536, 50458-531) Establishment Name Address ID/FEI Business Operations Noramco, Inc. 057234486 API MANUFACTURE(50458-535, 50458-536, 50458-531) Establishment Name Address ID/FEI Business Operations Janssen Ortho LLC 084894661 MANUFACTURE(50458-535, 50458-536, 50458-531) Establishment Name Address ID/FEI Business Operations Janssen Ortho LLC 805887986 ANALYSIS(50458-535, 50458-536, 50458-531) Establishment Name Address ID/FEI Business Operations Janssen Pharmaceuticals, Inc 063137772 ANALYSIS(50458-535, 50458-536, 50458-531) Establishment Name Address ID/FEI Business Operations Jubilant HollisterStier General Partnership 246762764 ANALYSIS(50458-531) , MANUFACTURE(50458-531)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.