LIFYORLI- relacorilant kit
Lifyorli by
Drug Labeling and Warnings
Lifyorli by is a Prescription medication manufactured, distributed, or labeled by Corcept Therapeutics Incorporated. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LIFYORLI safely and effectively. See full prescribing information for LIFYORLI.
LIFYORLITM (relacorilant) capsules, for oral use
Initial U.S. Approval: 2026INDICATIONS AND USAGE
LIFYORLI is a glucocorticoid receptor antagonist indicated in combination with nab-paclitaxel for the treatment of adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior systemic treatment regimens, at least one of which included bevacizumab. (1, 14)
DOSAGE AND ADMINISTRATION
The recommended dosage of LIFYORLI is 150 mg orally once on the day before, the day of, and the day after each nab-paclitaxel infusion. (2)
DOSAGE FORMS AND STRENGTHS
Capsules: 25 mg, 100 mg (3)
CONTRAINDICATIONS
Concurrent systemic glucocorticoid therapy for a lifesaving indication. (4)
WARNINGS AND PRECAUTIONS
- Neutropenia and Severe Infections: Monitor complete blood counts prior to each weekly treatment with LIFYORLI in combination with nab-paclitaxel and as clinically indicated. Withhold, reduce the dose, or permanently discontinue LIFYORLI based on severity. (2.3, 5.1)
- Adrenal insufficiency: Monitor for signs and symptoms of adrenal insufficiency. (5.2)
- Exacerbation of Conditions Treated with Glucocorticoids: LIFYORLI makes systemic glucocorticoids less effective in patients who have an ongoing requirement for systemic glucocorticoids. (5.3)
- Embryo-Fetal Toxicity: LIFYORLI can cause fetal harm. Advise females of reproductive potential of the risk to a fetus and to use effective contraception. (5.4)
ADVERSE REACTIONS
Most common adverse reactions (incidence > 20%), including laboratory abnormalities, that occurred in patients treated with LIFYORLI in combination with nab-paclitaxel were decreased hemoglobin, decreased neutrophils, fatigue, nausea, diarrhea, decreased platelets, rash, and decreased appetite. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Corcept Therapeutics at 1-855-844-3270 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Strong CYP3A Inducers: Avoid coadministration with LIFYORLI in combination with nab-paclitaxel. (7.1)
- CYP2C8 Inducers and Moderate CYP3A Inducers: Monitor for reduced effectiveness for LIFYORLI in combination with nab-paclitaxel. (7.1)
- CYP2C8 Inhibitors: Monitor for increased adverse reactions and modify the dosage for adverse reactions as recommended. (7.1)
- CYP3A Substrates: Avoid coadministration unless otherwise recommended. (7.2)
- Certain CYP2C8 Substrates: Avoid coadministration unless otherwise recommended. (7.2)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Information
2.2 Recommended Dosage and Administration
2.3 Dosage Modifications for Adverse Reactions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Neutropenia and Severe Infections
5.2 Adrenal Insufficiency
5.3 Exacerbation of Conditions Treated with Glucocorticoids
5.4 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on LIFYORLI in Combination with Nab-Paclitaxel
7.2 Effect of LIFYORLI on Other Drugs
7.3 Potential for Reduced Effectiveness of Glucocorticoids or LIFYORLI with Coadministration
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
LIFYORLI is indicated in combination with nab-paclitaxel for the treatment of adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior systemic treatment regimens, at least one of which included bevacizumab [see Clinical Studies (14)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Information
LIFYORLI
Follow LIFYORLI dosing instructions provided on the blister card.
Take LIFYORLI with food.
Swallow capsules whole. Do not crush, chew, dissolve, or split the capsules.
If a dose of LIFYORLI is missed by less than 12 hours, take the missed dose. If a dose of LIFYORLI is missed by 12 hours or more, skip the missed dose and take the next dose at the regularly scheduled time. Do not take 2 doses at the same time to make up for a missed dose.
If vomiting occurs after taking LIFYORLI, do not take an additional dose.
Nab-Paclitaxel
The recommended dosage and dosage modifications for nab-paclitaxel when administered in combination with LIFYORLI differ from those for other nab-paclitaxel indications [see Dosage and Administration (2.2 and 2.3) and Clinical Studies (14)].
Do not substitute with other paclitaxel formulations.
2.2 Recommended Dosage and Administration
The recommended dosage of LIFYORLI is 150 mg orally once on the day before, the day of, and the day after each nab-paclitaxel infusion until disease progression or unacceptable toxicity.
The recommended dosage for nab-paclitaxel is 80 mg/m2 administered as an intravenous infusion on Days 1, 8 and 15 of each 28-day cycle until disease progression or unacceptable toxicity [see Clinical Pharmacology (12.3), Clinical Studies (14)]. Refer to the Prescribing Information for nab-paclitaxel for administration.
2.3 Dosage Modifications for Adverse Reactions
Dose reduction levels are summarized in Table 1 and Table 2.
Table 1: Recommended Dosage Reductions for Adverse Reactions for Nab-Paclitaxel Dose Reduction Nab-Paclitaxel First 60 mg/m2 on Days 1, 8 and 15 of each 28-day cycle Second 60 mg/m2 on Days 1 and 15 of
each 28-day cycleThird Permanently discontinue nab-paclitaxel if unable to tolerate after two dose reductions. Table 2: Recommended Dosage Reductions for Adverse Reactions for LIFYORLI Dose Reduction LIFYORLI First 125 mg once the day before, the day of and the day after the nab-paclitaxel infusion Second Permanently discontinue LIFYORLI in patients unable to tolerate after one dose reduction. The recommended dosage modifications for adverse reactions are provided in Tables 3 and 4. Interrupt or discontinue LIFYORLI whenever nab-paclitaxel is interrupted or discontinued.
Table 3: Dosage Modifications for Hematologic Adverse Reactions Adverse Reaction Severitya Dosage Modification
a Unless otherwise specified, Grade per National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 5.0.
b Supportive short acting G-CSF administered 24 hours after nab-paclitaxel for 2 days in accordance with clinical practice.
c If the delay in nab-paclitaxel dosing exceeds 7 days, omit the nab-paclitaxel dose.
Neutropenia
Day 1
[see Warnings and Precautions (5.1)]ANC 1,000 to < 1,500/mm3 Nab-paclitaxel:
Withhold until ≥ 1,500/mm3; resume at same dose
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumes
ANC < 1,000/mm3Nab-paclitaxel:
Withhold until ≥ 1,500/mm3; resume at reduced dose
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumes
Neutropenia
Day 8 or 15
[see Warnings and Precautions (5.1)]ANC < 1,000/mm3 Nab-paclitaxel:
Omit dose; resume at reduced dose or continue at the same dose with short acting G-CSFb,c
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumesFebrile neutropenia
[see Warnings and Precautions (5.1)]Grade 3 or 4 Nab-paclitaxel:
Withhold until fever resolves and ANC ≥ 1,500/mm3; resume at reduced dosec
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumesThrombocytopenia
Day 1
[see Adverse Reactions (6.1)]Platelets < 100,000/mm3 Nab-paclitaxel:
Withhold until ≥ 100,000/mm3; resume at the same dose
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumesThrombocytopenia
Day 8 or 15
[see Adverse Reactions (6.1)]Platelets < 50,000/mm3 Nab-paclitaxel:
Omit dose; resume at reduced dose
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumesOther hematologic adverse reaction (excluding laboratory abnormalities that are not clinically relevant)
[see Adverse Reactions (6.1)]Grade 3 or 4 Nab-paclitaxel:
Withhold until Grade ≤ 1; resume at reduced dosec
LIFYORLI:
Withhold until Grade ≤ 1; resume at reduced doseTable 4: Dosage Modifications for non-Hematologic Adverse Reactions Adverse Reaction Severitya Dosage Modification
a Unless otherwise specified, Grade per National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 5.0.
b If the delay in nab-paclitaxel dosing exceeds 7 days, omit the nab-paclitaxel dose.
Peripheral neuropathy
[see Adverse Reactions (6.1)]Intolerable Grade 2 Nab-paclitaxel:
Reduce dose
LIFYORLI:
No changeGrade 3 Nab-paclitaxel:
Withhold until Grade ≤ 2; resume at reduced doseb
LIFYORLI:
Withhold; resume at the same dose once nab-paclitaxel resumesGrade 4 Nab-paclitaxel:
Permanently discontinue
LIFYORLI:
Permanently discontinueCutaneous toxicity
[see Adverse Reactions (6.1)]Grade 3 Nab-paclitaxel:
Withhold until Grade ≤ 1; resume at reduced doseb
LIFYORLI:
Resume at the same dose once nab-paclitaxel resumesGrade 4 Nab-paclitaxel:
Permanently discontinue
LIFYORLI:
Permanently discontinueMucositis or Diarrhea
[see Adverse Reactions (6.1)]Grade 3 or 4 Nab-paclitaxel:
Withhold until Grade ≤ 1; resume at reduced doseb
LIFYORLI:
Resume at the same dose once nab-paclitaxel resumes.Other non-hematologic adverse reactions (excluding laboratory abnormalities that are not clinically relevant)
[see Adverse Reactions (6.1)]Grade 3 Nab-paclitaxel:
Withhold until Grade ≤ 1; resume at reduced doseb
LIFYORLI:
Withhold until Grade ≤ 1; resume at reduced doseGrade 4 Nab-paclitaxel:
Permanently discontinue
LIFYORLI:
Permanently discontinue - 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
LIFYORLI is contraindicated in patients receiving systemic glucocorticoids for lifesaving purposes (e.g., immunosuppression after organ transplantation) because LIFYORLI antagonizes the effect of glucocorticoids [see Warnings and Precautions (5.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Neutropenia and Severe Infections
LIFYORLI in combination with nab-paclitaxel can cause neutropenia, including febrile neutropenia and severe infections.
In ROSELLA, decreased neutrophil count occurred in 74% of patients treated with LIFYORLI in combination with nab-paclitaxel, 30% Grade 3, 15% Grade 4, and 3.7% febrile neutropenia. The median time to onset of neutropenia was 15 days (range: 8 to 329). Instances of neutropenia were temporally associated with infection in 16% of patients. There was one fatal event of septic shock with febrile neutropenia. Thirty-eight percent of patients initiated granulocyte colony-stimulating factor (G-CSF) during the first or second cycle of therapy.
Monitor complete blood counts prior to each weekly treatment with LIFYORLI in combination with nab-paclitaxel and as clinically indicated. Based on the severity of neutropenia, delay, reduce dose or permanently discontinue LIFYORLI in combination with nab-paclitaxel. Consider short-acting G-CSF administration as applicable [see Dosage and Administration (2.3)]. Consider the possibility of concurrent adrenal insufficiency, particularly in the setting of serious infection [see Warnings and Precautions (5.2)]. Inform patients to promptly report any episodes of fever to their healthcare provider.
5.2 Adrenal Insufficiency
LIFYORLI is a reversible glucocorticoid receptor antagonist and can cause adrenal insufficiency. Adrenal insufficiency can occur at any time during treatment with LIFYORLI. The risk of adrenal insufficiency is increased in situations of stress, such as acute illness, infection, or surgery. Consider whether supplemental glucocorticoids are required in the perioperative period in patients who have received LIFYORLI within 30 days of surgery.
Monitor patients receiving LIFYORLI for signs and symptoms of adrenal insufficiency. Serum cortisol levels do not provide an accurate assessment of adrenal insufficiency in patients receiving LIFYORLI. Withhold LIFYORLI and administer glucocorticoid therapy if adrenal insufficiency is suspected. High doses of supplemental glucocorticoids may be needed to overcome the glucocorticoid receptor antagonism produced by LIFYORLI. When deciding on the duration of glucocorticoid treatment, consider the long half-life of relacorilant (27.5 hours) and that glucocorticoid receptor antagonism may occur for up to 6 days after the last dose of LIFYORLI [see Clinical Pharmacology (12.3)]. After resolution of adrenal insufficiency, resume previous dose, reduce dose or permanently discontinue LIFYORLI based on severity [see Dosage and Administration (2.3)].
Educate patients on the symptoms associated with adrenal insufficiency and advise them to contact a healthcare provider if they occur.
5.3 Exacerbation of Conditions Treated with Glucocorticoids
Use of LIFYORLI in patients who are taking systemic glucocorticoids for other conditions (e.g., autoimmune disorders) may exacerbate these conditions. LIFYORLI is a glucocorticoid receptor antagonist which may make systemic glucocorticoids less effective. Similarly, coadministration of systemic glucocorticoids may make LIFYORLI less effective. Monitor patients for reduced effectiveness of LIFYORLI and glucocorticoids in patients receiving both [see Drug Interactions (7.3)]. Patients who require chronic or frequent use of glucocorticoids were excluded from clinical trials of LIFYORLI in combination with nab-paclitaxel.
LIFYORLI is contraindicated in patients receiving systemic glucocorticoids for lifesaving purposes, [see Contraindications (4)].
5.4 Embryo-Fetal Toxicity
Based on data from animal studies, LIFYORLI can cause fetal harm when administered to a pregnant woman. Oral administration of relacorilant to pregnant rabbits resulted in adverse developmental outcomes, including embryo-fetal mortality.
Advise pregnant women of the potential risk to a fetus. Verify pregnancy status of females of reproductive potential prior to initiating LIFYORLI treatment. Advise females of reproductive potential to use effective contraception during treatment with LIFYORLI and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with LIFYORLI and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3)].
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Neutropenia and Severe Infections [see Warnings and Precautions (5.1)].
- Adrenal Insufficiency [see Warnings and Precautions (5.2)]
- Exacerbation of Conditions Treated with Glucocorticoids [see Warnings and Precautions (5.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of LIFYORLI in combination with nab-paclitaxel was evaluated in patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer in ROSELLA [see Clinical Studies (14)].
Patients received LIFYORLI (150 mg orally on the day before, the day of, and the day after each administration of nab-paclitaxel 80 mg/m2 intravenous infusion on Days 1, 8 and 15 of each 28-day cycle (n=188) or nab-paclitaxel 100 mg/m2 intravenous infusion (n=190) until disease progression or unacceptable toxicity. LIFYORLI dosing was interrupted whenever nab-paclitaxel was interrupted. The median duration of LIFYORLI treatment was 4.7 months (range: 0.2 to 24).
Serious adverse reactions occurred in 35% of patients who received LIFYORLI in combination with nab-paclitaxel. Serious adverse reactions in ≥2% of patients were neutropenia (4%), pneumonia (3.2%), pleural effusion (3.2%), febrile neutropenia (2.1%), and fatigue (2.1%). Fatal adverse reactions occurred in 2.1% of patients who received LIFYORLI in combination with nab-paclitaxel including septic shock (0.5%), cardiac arrest (0.5%), ischemic stroke (0.5%), and intestinal perforation (0.5%).
Permanent discontinuation of LIFYORLI in combination with nab-paclitaxel due to adverse reactions occurred in 9% of patients. The adverse reaction which resulted in permanent discontinuation of LIFYORLI in >2% of patients was intestinal obstruction (2.6%).
Dosage interruptions of LIFYORLI due to an adverse reaction occurred in 72% of patients. Adverse reactions which required dosage interruptions of LIFYORLI in combination with nab-paclitaxel in ≥5% of patients included neutropenia (44%), anemia (12%), and fatigue (7%).
Dose reductions of LIFYORLI due to an adverse reaction occurred in 7.4% of patients and dose reductions of nab-paclitaxel occurred in 48% of patients. Adverse reactions which required dose reductions of LIFYORLI included fatigue (1.6%), decreased appetite (1.2%), abdominal pain (0.5%), neutropenia (0.5%), edema (0.5%), and sciatica (0.5%).
The most common (>20%) adverse reactions, including laboratory abnormalities, were decreased hemoglobin, decreased neutrophils, fatigue, nausea, diarrhea, decreased platelets, rash, and decreased appetite.
Table 5 and 6 summarize adverse reactions and laboratory abnormalities, respectively, occurring in ≥10% of patients who received LIFYORLI in combination with nab-paclitaxel in ROSELLA.
Table 5: Adverse Reactions Occurring in ≥10% of Patients Who Received LIFYORLI in Combination with Nab-Paclitaxel in ROSELLA a Includes multiple related terms
Adverse Reaction LIFYORLI +
Nab-Paclitaxel
(n=188)Nab-Paclitaxel
(n=190)All Grades (%) Grades 3 or 4 (%) All Grades (%) Grades 3 or 4 (%) General disorders Fatiguea 54 9 45 1.6 Edemaa 19 1.1 12 0.5 Pyrexiaa 14 0.5 9 0 Gastrointestinal disorders Nausea 44 4 35 3 Diarrheaa 40 3.7 27 1.6 Stomatitisa 19 3.2 9 1.1 Skin and subcutaneous tissue disorders Rasha 24 3.7 10 0.5 Nail disordersa 19 0 12 2 Metabolism and nutrition disorders Decreased appetite 22 1.6 12 0.5 Respiratory disorders Cougha 19 0.5 10 0 Nervous system disorders Dizzinessa 13 0.5 4 0.5 Dysgeusiaa 13 0 5 0 Clinically relevant adverse reactions occurring in <10% of patients who received LIFYORLI in combination with nab-paclitaxel in ROSELLA included dry eye (8.5%), hypotension (8.5%), hypertension (4.8%), acute kidney injury (4.3%), febrile neutropenia (3.7%), and syncope (2.7%).
Table 6: Select Laboratory Abnormalities ≥10% for All Grades, in Patients Who Received LIFYORLI in Combination with Nab-Paclitaxel in ROSELLA Laboratory Abnormality LIFYORLI +
Nab-Paclitaxel (n=188)Nab-Paclitaxel (n=190) All Grades
(%)Grades 3 or 4
(%)All Grades
(%)Grades 3 or 4
(%)Hemoglobin decreased 89 18 79 10 Leukocytes decreased 82 25 77 16 Neutrophils decreased 74 44 65 27 Platelets decreased 28 2 14 1 -
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on LIFYORLI in Combination with Nab-Paclitaxel
Strong CYP3A Inducers
Avoid coadministration of LIFYORLI plus nab-paclitaxel with strong CYP3A inducers.
Both relacorilant and paclitaxel are CYP3A substrates. Coadministration of strong CYP3A inducers can decrease concentrations of relacorilant and paclitaxel, which may reduce their effectiveness [see Clinical Pharmacology (12.3)].
CYP2C8 Inducers and Moderate CYP3A Inducers
Monitor for reduced effectiveness of LIFYORLI plus nab-paclitaxel with CYP2C8 inducers and moderate CYP3A inducers.
Paclitaxel is a substrate of CYP2C8 and CYP3A and relacorilant is a CYP3A substrate. Coadministration of CYP2C8 inducers and moderate CYP3A inducers can decrease concentrations of paclitaxel and relacorilant, which may reduce their effectiveness.
CYP2C8 Inhibitors
Monitor for increased adverse reactions and modify the dosage for adverse reactions as recommended [see Dosage and Administration (2.3)].
Paclitaxel is a substrate of CYP2C8. Coadministration of CYP2C8 inhibitors may increase concentrations of paclitaxel, which may increase the risk of adverse reactions.
7.2 Effect of LIFYORLI on Other Drugs
CYP3A Substrates
Avoid concomitant use unless otherwise recommended in the Prescribing Information for CYP3A substrates.
Relacorilant is a strong CYP3A inhibitor. Relacorilant increases exposure of CYP3A substrates [see Clinical Pharmacology (12.3)], which may increase the risk for adverse reactions related to these substrates.
Certain CYP2C8 Substrates
Avoid concomitant use unless otherwise recommended in the Prescribing Information for CYP2C8 substrates where minimal concentration changes may lead to reduced effectiveness. Relacorilant is a weak CYP2C8 inducer. Relacorilant decreases exposure of CYP2C8 substrates [see Clinical Pharmacology (12.3)], which may decrease the effectiveness related to these substrates.
7.3 Potential for Reduced Effectiveness of Glucocorticoids or LIFYORLI with Coadministration
LIFYORLI is contraindicated in patients receiving systemic glucocorticoids for lifesaving purposes (e.g., immunosuppression in organ transplantation) [see Contraindications (4)].
Avoid coadministration in patients with other medical conditions requiring systemic glucocorticoids when possible. If coadministration cannot be avoided, monitor patients for reduced effectiveness of LIFYORLI and glucocorticoids.
LIFYORLI is a glucocorticoid receptor antagonist, which may make systemic glucocorticoids less effective. Coadministration of systemic glucocorticoids may make LIFYORLI less effective.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
LIFYORLI is used in combination with nab-paclitaxel. Refer to the Prescribing Information of nab-paclitaxel for pregnancy information.
Based on findings in animals, LIFYORLI can cause fetal harm when administered to a pregnant woman. There are no available data on relacorilant use in pregnant women to inform drug-associated risk. In animal embryo-fetal development studies, oral administration of relacorilant to pregnant rabbits during the period of organogenesis resulted in embryo-fetal mortality and structural abnormalities at maternal doses of ≥ 10 mg/kg/day (0.6 times the human exposure based on area under the curve (AUC) at the recommended dose). Oral administration of relacorilant to pregnant rats during the period of organogenesis did not result in fetal malformations [see Data].
Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is between 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Relacorilant is a partial agonist of the glucocorticoid receptor in rats. In an embryo-fetal development study, relacorilant was administered to pregnant rats at oral doses of 1, 2.5, and 10 mg/kg/day during the period of organogenesis. Maternal toxicity (decreased body weight and food consumption) was observed at a dose of 10 mg/kg/day (3.8 times the human exposure based on AUC at the recommended dose). No developmental toxicity was noted at doses up to 10 mg/kg/day.
In an embryo-fetal development study, relacorilant administered to pregnant rabbits at oral doses of 1, 3, and 10 mg/kg/day during the period of organogenesis resulted in embryo-fetal mortality (increased post-implantation loss, fetal resorptions, and a decrease in litter size) at 10 mg/kg/day (0.6 times the human exposure based on AUC at the recommended dose). Fetal malformations (abnormal flexure of the forepaw, microcephaly, malrotated hindlimbs, absent interparietal bone, and fused sternebra in sternum) were observed at a dose of 10 mg/kg/day (0.6 times the human exposure based on AUC at the recommended dose). Additional adverse effects included skeletal variations of bipartite interparietal bone of the skull observed at doses ≥ 1 mg/kg/day (lower than the human exposure based on AUC at the recommended dose).
8.2 Lactation
Risk Summary
LIFYORLI is used in combination with nab-paclitaxel. Refer to the Prescribing Information of nab-paclitaxel for lactation information.
There are no data on the presence of relacorilant or its metabolites in animal or human milk, the effects on the breastfed child, or the effects on milk production. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with LIFYORLI and for one week after the last dose.
8.3 Females and Males of Reproductive Potential
LIFYORLI is used in combination with nab-paclitaxel. Refer to the Prescribing Information of nab-paclitaxel for contraception and infertility information.
LIFYORLI can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status in women of reproductive potential prior to initiating LIFYORLI.
8.4 Pediatric Use
The safety and effectiveness of LIFYORLI in pediatric patients has not been established.
8.5 Geriatric Use
Of the 188 patients with platinum-resistant epithelial ovarian cancer who received LIFYORLI, 38% were 65 years or older and 9.6% were 75 years or older. A higher incidence of grade 3-4 adverse events and dosage modification occurred in patients 65 years or older when compared to younger adult patients. No overall differences in effectiveness were observed between patients in ≥65 years of age and younger patients.
8.6 Hepatic Impairment
Avoid LIFYORLI in combination with nab-paclitaxel in patients with moderate or severe hepatic impairment (total bilirubin >1.5 to 10x ULN and any AST) because of the potential for increased relacorilant and paclitaxel exposures [see Clinical Pharmacology (12.3)].
-
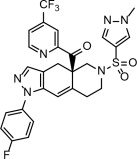
11 DESCRIPTION
LIFYORLI (relacorilant) is a glucocorticoid receptor antagonist antineoplastic agent.
Relacorilant chemical names:
- (R)-(1-(4-fluorophenyl)-6-((1-methyl-1H-pyrazol-4-yl)sulfonyl)-4,4a,5,6,7,8-hexahydro-1H-pyrazolo[3,4-g]isoquinolin-4a-yl)(4-trifluoromethyl)(pyridin-2-yl)methanone
- [(4aR)-1-(4-fluorophenyl)-6-(1-methyl-1H-pyrazole-4-sulfonyl)-1,4,5,6,7,8-hexahydro-4aH-pyrazolo[3,4-g]isoquinolin-4a-yl][4-(trifluoromethyl)pyridin-2-yl]methanone
The molecular formula is C27H22F4N6O3S and the molecular weight is 586.57 g/mol. The chemical structure of relacorilant is:
Relacorilant is an off-white to light yellow solid that is practically insoluble in aqueous media.
LIFYORLI capsules for oral use are supplied as immediate release, printed, soft gelatin capsules containing 25 mg or 100 mg of relacorilant. Each capsule contains the following excipients: butylated hydroxytoluene, lauroyl polyoxyl-32 glycerides, and propylene glycol caprylate. The capsule shell for the 25 mg strength contains black iron oxide, gelatin, red iron oxide, sorbitol special glycerin blend, titanium dioxide, and yellow iron oxide. The capsule shell for the 100 mg strength contains gelatin, sorbitol special glycerin blend, titanium dioxide, and yellow iron oxide. The printing ink contains ammonium hydroxide, black iron oxide, ethanol, ethyl acetate, isopropyl alcohol, macrogol/polyethylene glycol, polyvinyl acetate phthalate, propylene glycol, and purified water.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Relacorilant is a reversible glucocorticoid receptor (GR) antagonist. In functional in vitro assays with the mineralocorticoid receptor, relacorilant showed no agonist or antagonist activity. In human cell-line derived xenograft models, relacorilant enhanced apoptosis and antitumor activity when administered with paclitaxel.
Cortisol binding to the GR is immunosuppressive, decreasing secretion of pro‑inflammatory cytokines. GR antagonism may indirectly activate the immune system; relacorilant inhibited the cortisol-induced reduction of tumor necrosis factor alpha and interferon gamma in stimulated peripheral blood mononuclear cells.
12.2 Pharmacodynamics
12.3 Pharmacokinetics
Relacorilant pharmacokinetics were observed in adult patients at the approved recommended dosage and are presented as mean (CV%), unless otherwise specified. Relacorilant maximum concentration (Cmax) is 720 (68%) ng/mL and systemic exposure (AUC) is 5,686 (84%) ng*h/mL following the third consecutive daily dose.
Absorption
Relacorilant median (min, max) time to maximum plasma concentration (Tmax) is 2.5 hours (1.2, 5.5).
Effect of Food
Relacorilant Cmax increased 1.7-fold, AUC increased 2-fold, and Tmax was delayed 0.5 hours following administration with a low-fat meal (approximately 400 to 500 calories, 25% fat content).
Relacorilant Cmax increased 1.9-fold, AUC increased 2.4-fold, and Tmax was delayed 0.25 hours following administration with a high-fat meal (approximately 800 to 1,000 calories, 50% fat content).
Distribution
Relacorilant apparent volume of distribution (Vz/F) is 2490 (54%) L after a single 150 mg dose under fasted conditions. Protein binding of relacorilant in human plasma is >99% in vitro.
Elimination
Relacorilant terminal half-life is 27 (30%) hours with an apparent total clearance of 52 (69%) L/h under fasted conditions.
Specific Populations
No clinically significant differences in the pharmacokinetics of relacorilant were observed based on age (26 to 85 years), body weight (35 to 128 kg), race (White [81%], Asian [9%]), ethnicity (Non-Hispanic [93%], Hispanic [7%]), CLcr 30 to <90 mL/min, or mild hepatic impairment (total bilirubin > ULN to ≤ 1.5 × ULN or AST > ULN and total bilirubin ≤ ULN).
The effect of CLcr <30 mL/min or end-stage renal disease undergoing hemodialysis or severe hepatic impairment (total bilirubin > 3 to 10 × ULN and any AST) on the pharmacokinetics of relacorilant is unknown.
Drug Interaction Studies
Clinical Studies
Nab-paclitaxel: Dose-normalized paclitaxel (CYP2C8 and CYP3A4 substrate) Cmax increased 2-fold and dose-normalized AUC increased 1.7-fold following coadministration of nab-paclitaxel with LIFYORLI 150 mg.
No clinically significant differences in the pharmacokinetics of relacorilant were observed when given in combination with nab-paclitaxel.
Strong CYP3A Inhibitors: Relacorilant (300 mg once daily for 10 days under fasted conditions) steady state Cmax increased 1.2-fold and steady state AUC increased 1.5-fold following coadministration of itraconazole (strong CYP3A inhibitor) 200 mg once daily for 10 days.
CYP3A Substrates: Midazolam (CYP3A substrate) Cmax increased 3.1-fold and AUC increased 8.9-fold following coadministration of LIFYORLI 350 mg under fasted conditions (1.8 times the relacorilant exposure at the recommended dosage) once daily for 10 days.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a 6-month carcinogenicity study, transgenic rasH2 mice were orally administered up to 100 mg/kg/day relacorilant. In a 2-year carcinogenicity study, rats were orally administered up to 10 mg/kg/day in male rats (1.3 times the human exposure based on AUC at the recommended dose) and 3 mg/kg/day in female rats (0.7 times the human exposure based on AUC at the recommended dose). There was no evidence of relacorilant-induced carcinogenicity in either study.
Mutagenesis
Relacorilant was not genotoxic in the bacterial reverse mutation (Ames) assay, an in vitro human lymphocyte micronucleus assay, or an in vivo micronucleus assay in rats.
Impairment of Fertility
In a fertility study, relacorilant was administered orally to male rats prior to and throughout mating and to female rats prior to mating and up to the implantation day (gestation day 7). Relacorilant had no effect on fertility or reproductive function in male or female rats at doses up to 40 mg/kg/day (≥4.5 times the human exposure based on AUC at the recommended dose).
-
14 CLINICAL STUDIES
The efficacy of LIFYORLI in combination with nab-paclitaxel was evaluated in ROSELLA (NCT05257408), a multicenter, open-label, active-controlled, randomized, two-arm, trial in patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer. Patients were permitted to receive up to three prior lines of systemic therapy and prior bevacizumab was required. The trial excluded patients who required chronic or frequent use of glucocorticoids.
A total of 381 patients were randomized (1:1) to receive:
- LIFYORLI 150 mg orally the day before, the day of, and the day after administration of nab-paclitaxel 80 mg/m2 intravenous infusion on Days 1, 8 and 15 of each 28-day cycle (188)
or - nab-paclitaxel 100 mg/m2 intravenous infusion on Days 1, 8 and 15 of each 28-day cycle (193)
Treatment was administered until disease progression or unacceptable toxicity. Tumor response assessments occurred every 8 weeks for the first 40 weeks and every 12 weeks thereafter. Randomization was stratified by the following factors: number of prior lines of therapy (1 vs. >1) and region (North America vs. Europe vs. Korea/Australia/Latin America).
The major efficacy outcome measures were progression-free survival (PFS) assessed by blinded independent central review (BICR) and overall survival (OS). PFS was evaluated according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.
The median age was 62 years (range: 26 to 86); 71% were White, 13% were Asian, 1.3% were Black or African American, and 13% did not have race reported. Nine percent of patients were Hispanic or Latino, 76% were non-Hispanic, and 15% had ethnicity not reported or unknown. Nearly all patients had an Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 (69%) or 1 (30%). Nine percent of patients had received 1 prior line of systemic therapy, 48% of patients had received 2 prior lines of systemic therapy, and 44% of patients had received 3 prior lines of systemic therapy. Thirty-nine percent of patients received prior systemic therapy for platinum-resistant disease. All patients received prior bevacizumab, 99.5% had received a prior taxane (19% in the last line of therapy and 4% in the platinum-resistant setting) and 61% had received a prior PARP inhibitor. Of patients who had received a prior PARP inhibitor, 78% had radiographic progression while receiving the PARP inhibitor.
LIFYORLI demonstrated a statistically significant improvement in PFS and OS for patients randomized to LIFYORLI in combination with nab-paclitaxel compared to nab-paclitaxel monotherapy.
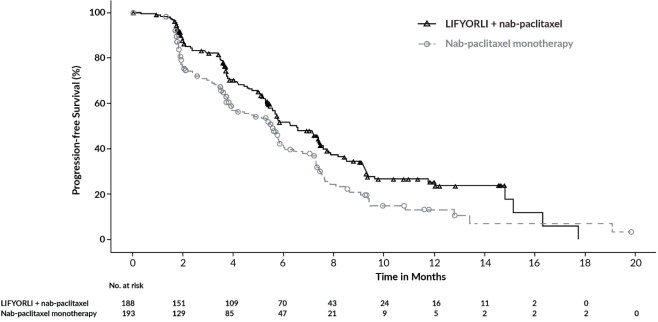
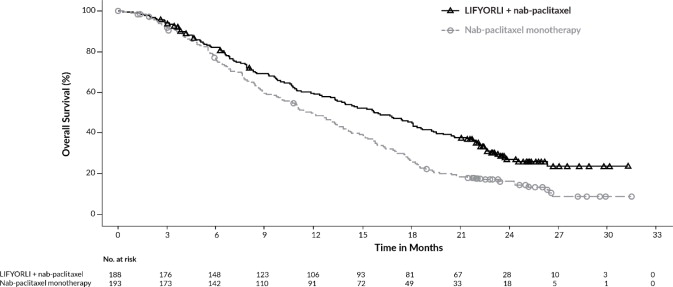
Efficacy results for ROSELLA are summarized in Table 7, Figure 1 and Figure 2.
Table 7: Efficacy Results in ROSELLA BICR = Blinded Independent Central Review; CI = Confidence Interval
a Results at the pre-specified final PFS analysis.
b Based on the stratified Cox proportional hazard model.
c Two-sided p-value based on stratified log-rank test (compared to a significance level of 0.04).
d Results at the pre-specified final OS analysis.
e Two-sided p-value based on stratified log-rank test (compared to a significance level of 0.0499).
LIFYORLI and
Nab-Paclitaxel
N=188Nab-Paclitaxel
N=193Progression-free Survival (PFS) by BICR a Number (%) of patients with events 113 (60) 121 (63) Median, months (95% CI) 6.5 (5.6, 7.4) 5.5 (3.9, 5.9) Hazard ratio (95% CI)b 0.70 (0.54, 0.91) p-valuec 0.0076 Overall Survival (OS)d Number (%) of patients with events 129 (69) 159 (82) Median, months (95% CI) 16.0 (13.0, 18.3) 11.9 (10.0, 13.8) Hazard ratio (95% CI)b 0.65 (0.51, 0.83) p-valuee 0.0004 Figure 1: Kaplan-Meier Curve for Progression-Free Survival in ROSELLA
Figure 2: Kaplan-Meier Curve for Overall Survival in ROSELLA
- LIFYORLI 150 mg orally the day before, the day of, and the day after administration of nab-paclitaxel 80 mg/m2 intravenous infusion on Days 1, 8 and 15 of each 28-day cycle (188)
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
LIFYORLI is available as soft gelatin capsules containing 25 mg or 100 mg of relacorilant.
LIFYORLI 25 mg are opaque dark brown, oval soft gelatin capsules with “CR25” printed in black.
LIFYORLI 100 mg are opaque yellow, oblong soft gelatin capsules with “CR100” printed in black.
LIFYORLI capsules are supplied as follows:
Dose Each Carton Contains Each Blister Card Contains NDC 150 mg One blister card of 3 capsules.
One 100 mg capsule, and
Two 25 mg capsules.NDC: 76346-450-01 One blister card of 9 capsules.
Three 100 mg capsules, and
Six 25 mg capsules.NDC: 76346-550-03 Three cartons each containing one blister card of 9 capsules.
(27 capsules total).Three 100 mg capsules, and
Six 25 mg capsules.NDC: 76346-550-09 125 mg One blister card of 2 capsules.
One 100 mg capsule, and
One 25 mg capsule.NDC: 76346-425-01 One blister card of 6 capsules.
Three 100 mg capsules, and
Three 25 mg capsules.NDC: 76346-525-03 Three cartons each containing one blister card of 6 capsules.
(18 capsules total).Three 100 mg capsules, and
Three 25 mg capsules.NDC: 76346-525-09 -
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Patient Information).
Neutropenia and Severe Infections
Inform patients about the risk of neutropenia and infection. Instruct patients to immediately report any fever or symptoms of infection to their healthcare provider [see Warnings and Precautions (5.1)].
Adrenal Insufficiency
Inform patients that LIFYORLI can cause adrenal insufficiency, a potentially life-threatening condition. Advise patients to immediately report signs or symptoms of adrenal insufficiency to their healthcare provider [see Warnings and Precautions (5.2)].
Exacerbation of Conditions Treated with Glucocorticoids
Advise patients who require chronic or frequent use of glucocorticoids that glucocorticoids may be less effective and to contact their healthcare provider for any worsening symptoms during concomitant use [see Warnings and Precautions (5.3)].
Embryo-Fetal Toxicity
Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
Advise females to inform their healthcare provider of a known or suspected pregnancy. Advise females of reproductive potential to use effective contraception during treatment with LIFYORLI and for 1 week after the last dose.
Advise male patients with female partners of reproductive potential to use effective contraception during treatment with LIFYORLI and for 1 week after the last dose [see Warnings and Precautions (5.4) and Use in Specific Population (8.1, 8.3)].
Lactation
Advise women not to breastfeed during treatment with LIFYORLI and for 1 week after the last dose [see Use in Specific Populations (8.2)].
Administration
Advise patients to follow the LIFYORLI dosing instructions on the blister card. Advise patients to take LIFYORLI with food, how to make up a missed dose, and to swallow capsules whole and to not crush, chew, dissolve or split the capsules [see Dosage and Administration (2.1, 2.2)].
Manufactured for:
Corcept Therapeutics Incorporated
Redwood City, CA 94065LIFYORLITM is a trademark of Corcept Therapeutics Incorporated.
Copyright 2026 Corcept Therapeutics Incorporated. All rights reserved.
USPI-220641-v01 03/2026 -
PATIENT PACKAGE INSERT
This Patient Information has been approved by the U.S. Food and Drug Administration.
Issued: 03/2026
PATIENT INFORMATION
LIFYORLITM (lif-YOR-lee)
(relacorilant)
capsulesWhat is LIFYORLI?
LIFYORLI is a prescription medicine used with a chemotherapy medicine called nab-paclitaxel to treat adults with ovarian, fallopian tube, or primary peritoneal cancer who:
- have not responded to or are no longer responding to treatment with platinum-based chemotherapy, and
- have received one to three prior systemic treatments, at least one of which included bevacizumab.
It is not known if LIFYORLI is safe and effective in children. Who should not take LIFYORLI?
Do not take LIFYORLI if you are receiving or taking a steroid medicine by mouth or injection (systemic glucocorticoid) for lifesaving purposes. LIFYORLI can make glucocorticoid medicines less effective.Before you take LIFYORLI, tell your healthcare provider about all your medical conditions, including if you:
- plan to have surgery or have had surgery within the past 30 days
- have liver problems
- are pregnant or plan to become pregnant. LIFYORLI can harm your unborn baby.
Females who are able to become pregnant:- Your healthcare provider should do a pregnancy test before you start treatment with LIFYORLI.
- Use effective birth control (contraception) during treatment with LIFYORLI and for 1 week after your last dose. Talk with your healthcare provider about birth control methods you can use during this time.
- Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with LIFYORLI.
- Use effective birth control (contraception) during treatment with LIFYORLI and for 1 week after your last dose.
- are breastfeeding or plan to breastfeed. It is not known if LIFYORLI passes into your breast milk. Do not breastfeed during treatment with LIFYORLI and for 1 week after your last dose.
Tell your healthcare provider about all medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking LIFYORLI with certain other medicines may affect how LIFYORLI or the other medicines work and may cause side effects.
Especially tell your healthcare provider if you are taking steroid medicines called glucocorticoids. See “What are the possible side effects of LIFYORLI?” Ask your healthcare provider if you are not sure if you take a glucocorticoid.How should I take LIFYORLI?
- Take LIFYORLI exactly as your healthcare provider tells you.
- Follow the LIFYORLI dosing instructions on the blister card.
- Take LIFYORLI 1 time on the day before, the day of, and the day after each nab-paclitaxel infusion.
- Take LIFYORLI with food.
- Swallow LIFYORLI capsules whole. Do not crush, chew, dissolve, or split the capsules
- If you miss a dose of LIFYORLI by less than 12 hours, take it as soon as you remember. If you miss a dose by more than 12 hours, skip the missed dose and take the next dose at your regular time. Do not take 2 doses at the same time to make up for a missed dose.
- If you vomit after taking LIFYORLI, do not take an extra dose.
What are the possible side effects of LIFYORLI?
LIFYORLI can cause serious side effects, including:- low white blood cell count (neutropenia) and severe infections. LIFYORLI in combination with nab-paclitaxel can cause low white blood cell counts, which can lead to a fever and severe infections and cause death. Your healthcare provider will check your blood counts before each weekly treatment with LIFYORLI in combination with nab-paclitaxel and as needed. Tell your healthcare provider right away if you have a fever, chills, weakness, confusion, or other signs or symptoms of an infection.
-
reduced effects of the hormone cortisol (adrenal insufficiency). LIFYORLI reduces the effect of an adrenal hormone called cortisol, which may be life-threatening and require hospitalization. This can happen at any time during treatment. Your risk of adrenal insufficiency is increased when your body is under stress, such as when you are sick, have an infection, or have surgery. Tell your healthcare provider right away if you develop the following symptoms during treatment with LIFYORLI:
- feeling dizzy or lightheaded
- low blood pressure (hypotension)
- low blood sugar (hypoglycemia)
- worsening weakness or tiredness
- worsening nausea or vomiting
- worsening loss of appetite or weight loss
- worsening of conditions treated with glucocorticoids. Treatment with LIFYORLI in people who also take glucocorticoids for other conditions may worsen symptoms of these conditions. LIFYORLI makes glucocorticoids less effective, and use of glucocorticoids makes LIFYORLI less effective. If you take a glucocorticoid medicine during treatment with LIFYORLI, tell your healthcare provider if symptoms of your other condition get worse.
The most common side effects of LIFYORLI in combination with nab-paclitaxel include: - decreased red blood cell, white blood cell, and platelet counts
- tiredness
- nausea
- diarrhea
- rash
- decreased appetite
Your healthcare provider may change your dose of LIFYORLI, delay treatment, or completely stop treatment with LIFYORLI if you have certain side effects.
These are not all of the possible side effects of LIFYORLI.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store LIFYORLI?
- Store LIFYORLI at room temperature between 68°F to 77°F (20°C to 25°C).
- Store LIFYORLI in the original carton.
Keep LIFYORLI and all medicines out of the reach of children. General information about the safe and effective use of LIFYORLI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LIFYORLI for a condition for which it was not prescribed. Do not give LIFYORLI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LIFYORLI that is written for health professionals.What are the ingredients in LIFYORLI?
Active ingredient: relacorilant
Inactive ingredients: butylated hydroxytoluene, lauroyl polyoxyl-32 glycerides, and propylene glycol caprylate. The 25mg capsule shell contains: black iron oxide, gelatin, red iron oxide, sorbitol special glycerin blend, titanium dioxide, and yellow iron oxide. The 100 mg capsule shell contains: gelatin, sorbitol special glycerin blend, titanium dioxide, and yellow iron oxide. The printing ink contains: ammonium hydroxide, black iron oxide, ethanol, ethyl acetate, isopropyl alcohol, macrogol/polyethylene glycol, polyvinyl acetate phthalate, propylene glycol, and purified water.
Manufactured for Corcept Therapeutics Incorporated, Redwood City, CA 94065
LIFYORLITM is a trademark of Corcept Therapeutics Incorporated.
Copyright 2026 Corcept Therapeutics Incorporated. All rights reserved.
PPI-220641-v01 03/2026
For more information, go to www.LIFYORLI.com or call 1-85-LIFYORLI (1-855-439-6754). -
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 125 mg Outer Carton (1-Dose Supply)
NDC 76346-425-01 Rx ONLY Lifyorli ™ (relacorilant) capsules 100 mg per capsule 25 mg per capsule 125 mg Dose
(Take one 100 mg capsule and one 25 mg capsule)
1-Dose Supply:
Contents: One blister card of 2 capsules:
one 100 mg capsules and one 25 mg capsule.
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 125 mg Outer Carton (3-Dose Supply)
NDC 76346-525-03 Rx ONLY Lifyorli ™ (relacorilant) capsules 100 mg per capsule 25 mg per capsule 125 mg Dose
(Take one 100 mg capsule and one 25 mg capsule)
3-Dose Supply:
Contents: One blister card of 6 capsules:
three 100 mg capsules and three 25 mg capsules.
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 125 mg Outer Carton (9-Dose Supply)
NDC 76346-525-09 Rx ONLY Lifyorli ™ (relacorilant) capsules 100 mg per capsule 25 mg per capsule 125 mg Dose
(Take one 100 mg capsule and one 25 mg capsule)
9-Dose Supply:
Contents: Three cartons each containing one blister card
of 6 capsules: three 100 mg capsules and three 25 mg capsules.
18 capsules total.
-
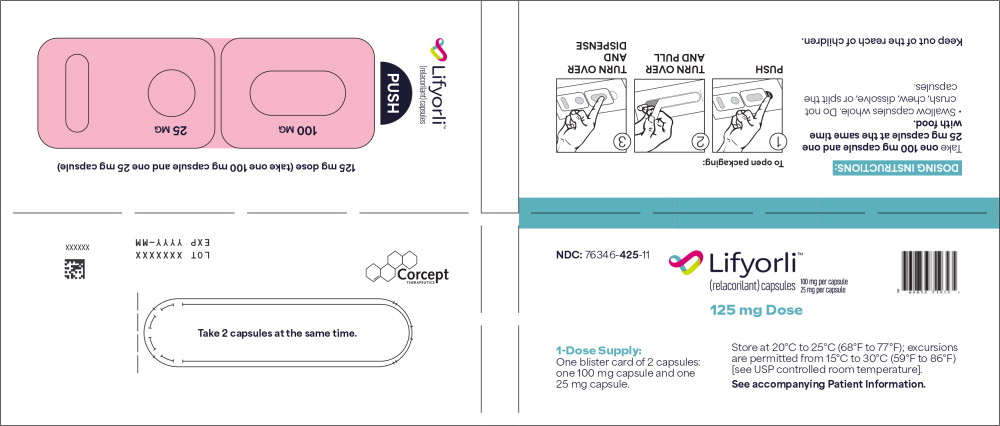
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 125 mg Blister Packs (1-Dose Supply)
NDC 76346-425-11 Lifyorli ™ 100 mg per capsule (relacorilant) capsules 25 mg per capsule 125 mg Dose
1-Dose Supply:
One blister card of 2 capsules:
one 100 mg capsules and one
25 mg capsule.Store at 20°C to 25°C (68°F to 77°F); excursions
are permitted from 15°C to 30°C (59°F to 86°F)
[see USP controlled room temperature].
See accompanying Patient Information. -
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 125 mg Blister Packs (3-Dose Supply)
NDC 76346-525-33 Lifyorli ™ 100 mg per capsule (relacorilant) capsules 25 mg per capsule 125 mg Dose
3-Dose Supply:
One blister card of 6 capsules:
three 100 mg capsules and three
25 mg capsule.Store at 20°C to 25°C (68°F to
77°F); excursions are permitted
from 15°C to 30°C (59°F to 86°F)
[see USP controlled room
temperature]. Store in the
original carton.
Keep out of the reach of children.
See accompanying Patient Information.
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 150 mg Outer Carton (1-Dose Supply)
NDC 76346-450-01 Rx ONLY Lifyorli ™ (relacorilant) capsules 100 mg per capsule 25 mg per capsule 150 mg Dose
(Take one 100 mg capsule and two 25 mg capsules) 1-Dose Supply:
Contents: One blister card of 3 capsules:
one 100 mg capsules and two 25 mg capsules.
-
PRINCIPAL DISPLAY PANEL
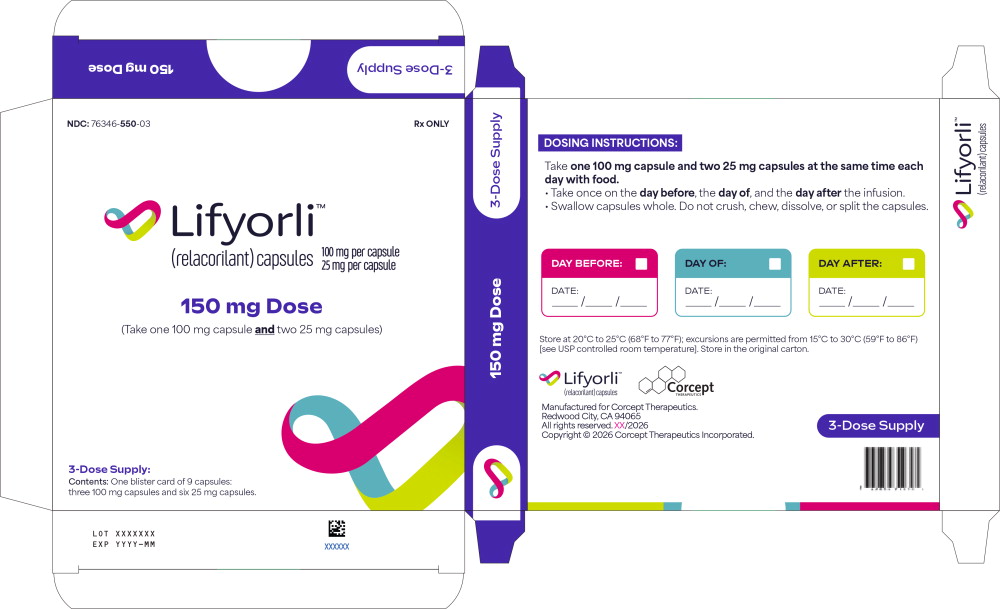
Principal Display Panel – for 150 mg Outer Carton (3-Dose Supply)
NDC 76346-550-03 Rx ONLY Lifyorli ™ (relacorilant) capsules 100 mg per capsule 25 mg per capsule 150 mg Dose
(Take one 100 mg capsule and two 25 mg capsules) 3-Dose Supply:
Contents: One blister card of 9 capsules:
three 100 mg capsules and six 25 mg capsules.
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 150 mg Outer Carton (9-Dose Supply)
NDC 76346-550-09 Rx ONLY Lifyorli ™ (relacorilant) capsules 100 mg per capsule 25 mg per capsule 150 mg Dose
(Take one 100 mg capsule and two 25 mg capsules) 9-Dose Supply:
Contents: Three cartons each containing one blister card
of 9 capsules: three 100 mg capsules and six 25 mg capsules.
27 capsules total.
-
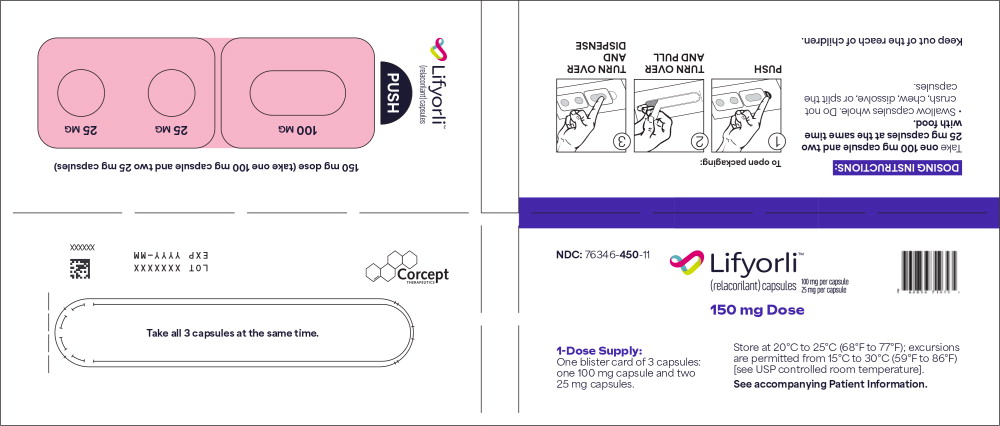
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 150 mg Blister Packs (1-Dose Supply)
NDC 76346-450-11 Lifyorli ™ 100 mg per capsule (relacorilant) capsules 25 mg per capsule 150 mg Dose 1-Dose Supply:
One blister card of 3 capsules:
one 100 mg capsules and two
25 mg capsule.Store at 20°C to 25°C (68°F to 77°F); excursions
are permitted from 15°C to 30°C (59°F to 86°F)
[see USP controlled room temperature].
See accompanying Patient Information. -
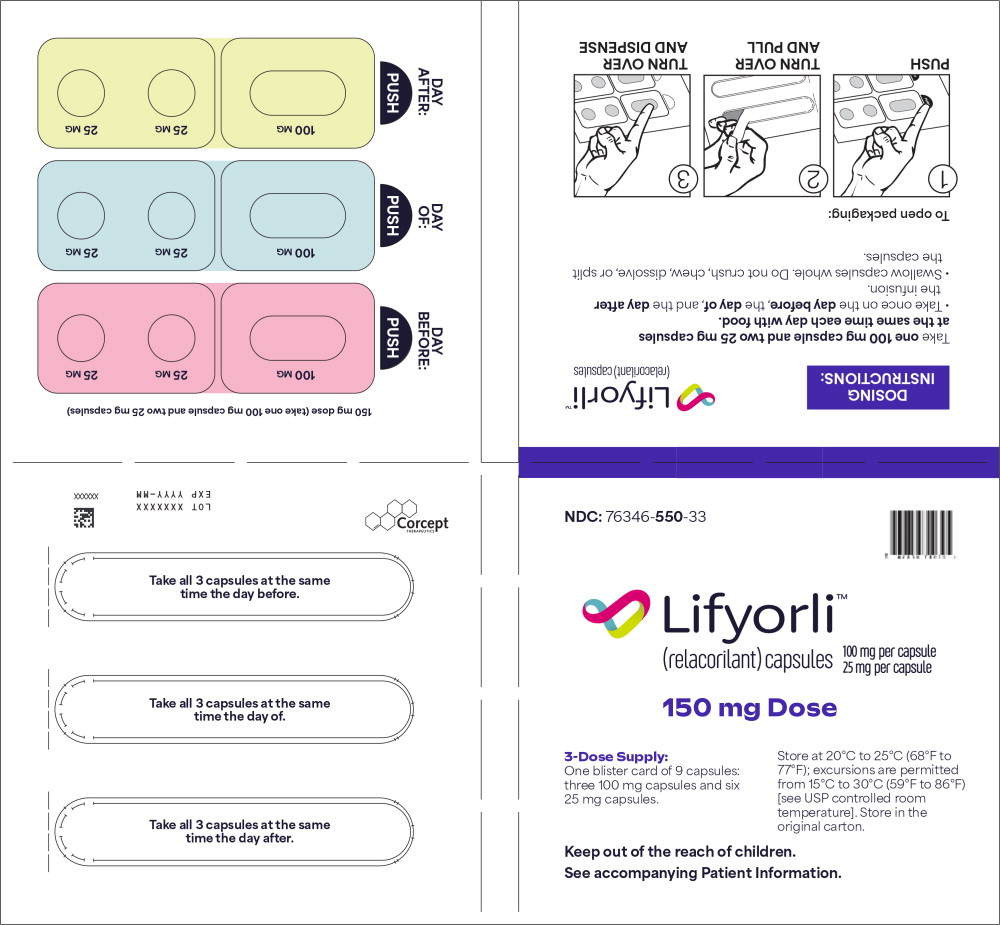
PRINCIPAL DISPLAY PANEL
Principal Display Panel – for 150 mg Blister Packs (3-Dose Supply)
NDC 76346-550-33 Lifyorli ™ 100 mg per capsule (relacorilant) capsules 25 mg per capsule 150 mg Dose 3-Dose Supply:
One blister card of 9 capsules:
three 100 mg capsules and six
25 mg capsule.Store at 20°C to 25°C (68°F to
77°F); excursions are permitted
from 15°C to 30°C (59°F to 86°F)
[see USP controlled room
temperature]. Store in the
original carton.
Keep out of the reach of children.
See accompanying Patient Information.
-
INGREDIENTS AND APPEARANCE
LIFYORLI
relacorilant kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76346-425 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-425-01 1 in 1 CARTON 03/25/2026 1 NDC: 76346-425-11 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 PACKAGE, COMBINATION 1 Part 2 1 PACKAGE, COMBINATION 1 Part 1 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-025 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 25 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color brown (dark brown) Score no score Shape OVAL (OVAL) Size 8mm Flavor Imprint Code CR25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-025-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Part 2 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 100 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color yellow (yellow) Score no score Shape CAPSULE (oblong) Size 20mm Flavor Imprint Code CR100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-100-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 LIFYORLI
relacorilant kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76346-525 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-525-03 1 in 1 CARTON 03/25/2026 1 NDC: 76346-525-33 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 76346-525-09 3 in 1 CARTON 03/25/2026 2 NDC: 76346-525-03 1 in 1 CARTON 2 NDC: 76346-525-33 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 3 PACKAGE, COMBINATION 3 Part 2 3 PACKAGE, COMBINATION 3 Part 1 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-025 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 25 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color brown (dark brown) Score no score Shape OVAL (OVAL) Size 8mm Flavor Imprint Code CR25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-025-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Part 2 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 100 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color yellow (yellow) Score no score Shape CAPSULE (oblong) Size 20mm Flavor Imprint Code CR100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-100-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 LIFYORLI
relacorilant kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76346-450 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-450-01 1 in 1 CARTON 03/25/2026 1 NDC: 76346-450-11 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 2 PACKAGE, COMBINATION 2 Part 2 1 PACKAGE, COMBINATION 1 Part 1 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-025 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 25 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color brown (dark brown) Score no score Shape OVAL (OVAL) Size 8mm Flavor Imprint Code CR25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-025-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Part 2 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 100 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color yellow (yellow) Score no score Shape CAPSULE (oblong) Size 20mm Flavor Imprint Code CR100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-100-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 LIFYORLI
relacorilant kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76346-550 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-550-03 1 in 1 CARTON 03/25/2026 1 NDC: 76346-550-33 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 76346-550-09 3 in 1 CARTON 03/25/2026 2 NDC: 76346-550-03 1 in 1 CARTON 2 NDC: 76346-550-33 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 6 PACKAGE, COMBINATION 6 Part 2 3 PACKAGE, COMBINATION 3 Part 1 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-025 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 25 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color brown (dark brown) Score no score Shape OVAL (OVAL) Size 8mm Flavor Imprint Code CR25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-025-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Part 2 of 2 LIFYORLI
relacorilant capsuleProduct Information Item Code (Source) NDC: 76346-100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RELACORILANT (UNII: 2158753C7E) (RELACORILANT - UNII:2158753C7E) RELACORILANT 100 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) LAUROYL PEG-32 GLYCERIDES (UNII: H5ZC52369M) PROPYLENE GLYCOL MONO- AND DICAPRYLATE (UNII: QJL3I5WV4F) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color yellow (yellow) Score no score Shape CAPSULE (oblong) Size 20mm Flavor Imprint Code CR100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76346-100-01 1 in 1 PACKAGE, COMBINATION; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220641 03/25/2026 Labeler - Corcept Therapeutics Incorporated (069540610)
Trademark Results [Lifyorli]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 LIFYORLI 98858236 not registered Live/Pending |
Corcept Therapeutics Incorporated 2024-11-18 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.