PENTOXIFYLLINE tablet, film coated, extended release
Pentoxifylline by
Drug Labeling and Warnings
Pentoxifylline by is a Prescription medication manufactured, distributed, or labeled by NCS HealthCare of KY, Inc dba Vangard Labs. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION:
Each extended-release tablet, for oral administration, contains 400 mg of pentoxifylline, USP and the following inactive ingredients: D&C Red No. 30 Aluminum Lake, FD&C Blue No. 2 Aluminum Lake, FD&C Yellow No. 6 Aluminum Lake, hydroxyethyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, povidone, titanium dioxide and triacetin in an extended-release formulation. Pentoxifylline is a tri-substituted xanthine derivative designated chemically as 1-(5-oxohexyl)-3,7-dimethylxanthine that, unlike theophylline, is a hemorrheologic agent, i.e. an agent that affects blood viscosity. Pentoxifylline is soluble in water and ethanol, and sparingly soluble in toluene.
The structural formula is:
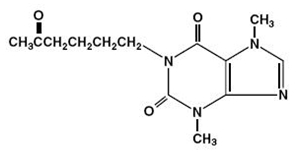
C13H18N4O3 M.W. 278.31
Meets USP Dissolution Test 6.
-
CLINICAL PHARMACOLOGY:
Mode of Action:
Pentoxifylline and its metabolites improve the flow properties of blood by decreasing its viscosity. In patients with chronic peripheral arterial disease, this increases blood flow to the affected microcirculation and enhances tissue oxygenation. The precise mode of action of pentoxifylline and the sequence of events leading to clinical improvement are still to be defined. Pentoxifylline administration has been shown to produce dose-related hemorrheologic effects, lowering blood viscosity, and improving erythrocyte flexibility. Leukocyte properties of hemorrheologic importance have been modified in animal and in vitro human studies. Pentoxifylline has been shown to increase leukocyte deformability and to inhibit neutrophil adhesion and activation. Tissue oxygen levels have been shown to be significantly increased by therapeutic doses of pentoxifylline in patients with peripheral arterial disease.
Pharmacokinetics and Metabolism:
After oral administration in aqueous solution pentoxifylline is almost completely absorbed. It undergoes a first-pass effect and the various metabolites appear in plasma very soon after dosing. Peak plasma levels of the parent compound and its metabolites are reached within 1 hour. The major metabolites are Metabolite l (1-[5-hydroxyhexyl]-3,7-dimethylxanthine) and Metabolite V (1-[3-carboxypropyl]-3,7-dimethylxanthine), and plasma levels of these metabolites are 5 and 8 times greater, respectively, than pentoxifylline.
Following oral administration of aqueous solutions containing 100 mg to 400 mg of pentoxifylline, the pharmacokinetics of the parent compound and Metabolite l are dose-related and not proportional (non-linear), with half-life and area under the blood-level time curve (AUC) increasing with dose. The elimination kinetics of Metabolite V are not dose-dependent. The apparent plasma half-life of pentoxifylline varies from 0.4 to 0.8 hours and the apparent plasma half-lives of its metabolites vary from 1 to 1.6 hours. There is no evidence of accumulation or enzyme induction (Cytochrome P450) following multiple oral doses.
Excretion is almost totally urinary; the main biotransformation product is Metabolite V. Essentially no parent drug is found in the urine. Despite large variations in plasma levels of parent compound and its metabolites, the urinary recovery of Metabolite V is consistent and shows dose proportionality. Less than 4% of the administered dose is recovered in feces. Food intake shortly before dosing delays absorption of an immediate release dosage form but does not affect total absorption. The pentoxifylline AUC was increased and elimination rate decreased in an older population (60 to 68 years, n = 6) compared to younger individuals (22 to 30 years, n = 6). See PRECAUTIONS: Geriatric Use.)
After administration of the 400 mg pentoxifylline extended-release tablet, plasma levels of the parent compound and its metabolites reach their maximum within 2 to 4 hours and remain constant over an extended period of time. Coadministration of pentoxifylline extended-release tablets with meals resulted in an increase in mean AUC and Cmax of about 1.1- and 1.3-fold for pentoxifylline, respectively. Cmax for Metabolite 1 also increased about 1.2-fold. The extended-release of pentoxifylline from the tablet eliminates peaks and troughs in plasma levels for improved gastrointestinal tolerance.
Patients with Hepatic Impairment:
In patients with mild to moderate liver impairment AUC and Cmax of pentoxifylline increased 6.5- and 7.5-fold, respectively, after a single 400 mg dose of pentoxifylline. AUC and Cmax of the active Metabolite l also increased 6.9- and 8.2-fold, respectively, in hepatic impaired subjects.
Pentoxifylline has not been studied in patients with severe hepatic failure.
Patients with Renal Impairment:
In patients with mild, moderate, or severe renal impairment the exposure to pentoxifylline and its active Metabolite l are not increased. In contrast, AUC0-tss and Cmax of the active Metabolite V in patients with mild to moderate renal impairment increased 2.4- and 2.1-fold, respectively, with a 400 mg three times daily regimen of pentoxifylline. In severe renal impairment AUC0-tss and Cmax of the active Metabolite V increased 12.9- and 10.6-fold, respectively, with a 400 mg pentoxifylline three times daily regimen. The increase in exposure to Metabolite V is only slightly smaller in both renal impairment groups if pentoxifylline is administered twice daily.
-
INDICATIONS AND USAGE:
Pentoxifylline extended-release tablets, USP are indicated for the treatment of patients with intermittent claudication on the basis of chronic occlusive arterial disease of the limbs. Pentoxifylline extended-release tablets can improve function and symptoms but is not intended to replace more definitive therapy, such as surgical bypass, or removal of arterial obstructions when treating peripheral vascular disease.
- CONTRAINDICATIONS:
-
PRECAUTIONS:
General:
At the first sign of anaphylactic/anaphylactoid reaction, pentoxifylline must be discontinued.
Patients with chronic occlusive arterial disease of the limbs frequently show other manifestations of arteriosclerotic disease. Pentoxifylline has been used safely for treatment of peripheral arterial disease in patients with concurrent coronary artery and cerebrovascular diseases, but there have been occasional reports of angina, hypotension, and arrhythmia. Controlled trials do not show that pentoxifylline causes such adverse effects more often than placebo, but, as it is a methylxanthine derivative, it is possible some individuals will experience such responses. Patients on warfarin should have more frequent monitoring of prothrombin times, while patients with other risk factors complicated by hemorrhage (e.g., recent surgery, peptic ulceration, cerebral and/or retinal bleeding) should have periodic examinations for bleeding including, hematocrit and/or hemoglobin.
In patients with hepatic or renal impairment, the exposure to pentoxifylline and/or active metabolites is increased. The consequences of the increase in drug exposure are not known (see CLINICAL PHARMACOLOGY: Pharmacokinetics and Metabolism and DOSAGE AND ADMINISTRATION).
Drug Interactions:
Although a causal relationship has not been established, there have been reports of bleeding and/or prolonged prothrombin time in patients treated with pentoxifylline with and without anticoagulants or platelet aggregation inhibitors. Patients on warfarin should have more frequent monitoring of prothrombin times, while patients with other risk factors complicated by hemorrhage (e.g., recent surgery, peptic ulceration) should have periodic examinations for bleeding including hematocrit and/or hemoglobin.
Concomitant administration of pentoxifylline and theophylline-containing drugs leads to increased theophylline levels and theophylline toxicity in some individuals. Such patients should be closely monitored for signs of toxicity and have their theophylline dosage adjusted as necessary.
Concomitant administration of strong CYP1A2 inhibitors (including e.g., ciprofloxacin or fluvoxamine) may increase the exposure to pentoxifylline (see ADVERSE REACTIONS).
Pentoxifylline has been used concurrently with antihypertensive drugs, beta blockers, digitalis, diuretics, and antiarrhythmics, without observed problems. Small decreases in blood pressure have been observed in some patients treated with pentoxifylline; periodic systemic blood pressure monitoring is recommended for patients receiving concomitant antihypertensive therapy. If indicated, dosage of the antihypertensive agents should be reduced.
Postmarketing cases of increased anticoagulant activity have been reported in patients concomitantly treated with pentoxifylline and vitamin K antagonists. Monitoring of anticoagulant activity in these patients is recommended when pentoxifylline is introduced or the dose is changed.
Carcinogenesis, Mutagenesis and Impairment of Fertility:
Long-term studies of the carcinogenic potential of pentoxifylline were conducted in mice and rats by dietary administration of the drug at doses up to 450 mg/kg (approximately 19 times the maximum recommended human daily dose [MRHD] in both species when based on body weight; 1.5 times the MRHD in the mouse and 3.3 times the MRHD in the rat when based on body surface area). In mice, the drug was administered for 18 months, whereas in rats, the drug was administered for 18 months followed by an additional 6 months without drug exposure. In the rat study, there was a statistically significant increase in benign mammary fibroadenomas in females of the 450 mg/kg group. The relevance of this finding to human use is uncertain. Pentoxifylline was devoid of mutagenic activity in various strains of Salmonella (Ames test) and in cultured mammalian cells (unscheduled DNA synthesis test) when tested in the presence and absence of metabolic activation. It was also negative in the in vivo mouse micronucleus test.
Pregnancy:
Teratogenic Effects. Category C:
Teratogenicity studies have been performed in rats and rabbits using oral doses up to 576 and 264 mg/kg, respectively. On a weight basis, these doses are 24 and 11 times the maximum recommended human daily dose (MRHD); on a body-surface-area basis, they are 4.2 and 3.5 times the MRHD. No evidence of fetal malformation was observed. Increased resorption was seen in rats of the 576 mg/kg group. There are no adequate and well controlled studies in pregnant women. Pentoxifylline should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers:
Pentoxifylline and its metabolites are excreted in human milk. Because of the potential for tumorigenicity shown for pentoxifylline in rats, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
Geriatric Use:
Clinical studies of pentoxifylline did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
The active Metabolite V is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
-
ADVERSE REACTIONS:
Clinical trials were conducted using either extended-release pentoxifylline tablets for up to 60 weeks or immediate-release pentoxifylline capsules for up to 24 weeks. Dosage ranges in the tablet studies were 400 mg bid to tid and in the capsule studies, 200 to 400 mg tid.
The table summarizes the incidence (in percent) of adverse reactions considered drug related, as well as the numbers of patients who received extended-release pentoxifylline tablets, immediate-release pentoxifylline capsules, or the corresponding placebos. The incidence of adverse reactions was higher in the capsule studies (where dose related increases were seen in digestive and nervous system side effects) than in the tablet studies. Studies with the capsule include domestic experience, whereas studies with the extended-release tablets were conducted outside the U.S.
The table indicates that in the tablet studies few patients discontinued because of adverse effects.
INCIDENCE (%) OF SIDE EFFECTS Extended-Release Tablets Immediate-Release Capsules Commercially Available Used only for Controlled Clinical Trials Pentoxifylline Placebo Pentoxifylline Placebo (Numbers of Patients at Risk) (321) (128) (177) (138) Discontinued for Side Effect 3.1 0 9.6 7.2 CARDIOVASCULAR SYSTEM Angina/Chest Pain 0.3 - 1.1 2.2 Arrhythmia/Palpitation - - 1.7 0.7 Flushing - - 2.3 0.7 DIGESTIVE SYSTEM Abdominal Discomfort - - 4 1.4 Belching/Flatus/Bloating 0.6 - 9 3.6 Diarrhea - - 3.4 2.9 Dyspepsia 2.8 4.7 9.6 2.9 Nausea 2.2 0.8 28.8 8.7 Vomiting 1.2 - 4.5 0.7 NERVOUS SYSTEM Agitation/Nervousness - - 1.7 0.7 Dizziness 1.9 3.1 11.9 4.3 Drowsiness - - 1.1 5.8 Headache 1.2 1.6 6.2 5.8 Insomnia - - 2.3 2.2 Tremor 0.3 0.8 - - Blurred Vision - - 2.3 1.4 Pentoxifylline has been marketed in Europe and elsewhere since 1972. In addition to the above symptoms, the following have been reported spontaneously since marketing or occurred in other clinical trials with an incidence of less than 1%; the causal relationship was uncertain:
Cardiovascular: dyspnea, edema, hypotension.
Digestive: anorexia, cholecystitis, constipation, dry mouth/thirst.
Nervous: anxiety, confusion, depression, seizures, aseptic meningitis.
Respiratory: epistaxis, flu-like symptoms, laryngitis, nasal congestion.
Skin and Appendages: brittle fingernails, pruritus, rash, urticaria, angioedema.
Special Senses: blurred vision, conjunctivitis, earache, scotoma.
Miscellaneous: bad taste, excessive salivation, leukopenia, malaise, sore throat/swollen neck glands, weight change.
A few rare events have been reported spontaneously worldwide since marketing in 1972. Although they occurred under circumstances in which a causal relationship with pentoxifylline could not be established, they are listed to serve as information for physicians: Cardiovascular: angina, arrhythmia, tachycardia; Digestive: hepatitis, jaundice, cholestasis, increased liver enzymes; and Hemic and Lymphatic: decreased serum fibrinogen, pancytopenia, aplastic anemia, leukemia, purpura, thrombocytopenia; Immune system disorders: anaphylactic reaction, anaphylactoid reaction, anaphylactic shock.
-
OVERDOSAGE:
Overdosage with pentoxifylline has been reported in pediatric patients and adults. Symptoms appear to be dose related. A report from a poison control center on 44 patients taking overdoses of enteric-coated pentoxifylline extended-release tablets noted that symptoms usually occurred 4 to 5 hours after ingestion and lasted about 12 hours. The highest amount ingested was 80 mg/kg; flushing, hypotension, convulsions, somnolence, loss of consciousness, fever, and agitation occurred. All patients recovered. In addition to symptomatic treatment and gastric lavage, special attention must be given to supporting respiration, maintaining systemic blood pressure, and controlling convulsions. Activated charcoal has been used to absorb pentoxifylline in patients who have overdosed.
-
DOSAGE AND ADMINISTRATION:
The usual dosage of pentoxifylline in extended-release tablet form is one tablet (400 mg) three times a day with meals.
While the effect of pentoxifylline extended-release tablets may be seen within 2 to 4 weeks, it is recommended that treatment be continued for at least 8 weeks. Efficacy has been demonstrated in double-blind clinical studies of 6 months duration.
Digestive and central nervous system side effects are dose related. If patients develop these effects it is recommended that the dosage be lowered to one tablet twice a day (800 mg/day). If side effects persist at this lower dosage, the administration of pentoxifylline extended-release tablets should be discontinued.
In patients with severe renal impairment (creatinine clearance below 30 mL/min) reduce dose to 400 mg once a day.
Dosing information cannot be provided for patients with hepatic impairment.
-
HOW SUPPLIED:
Pentoxifylline Extended-release Tablets, USP are available containing 400 mg of pentoxifylline, USP. The 400 mg tablet is a lavender, film- coated, modified capsule-shaped, unscored tablet debossed with MYLAN on one side of the tablet and 357 on the other side. It is available as follows:
NDC: 0615-4523-39 blisterpacks of 30 tablets
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.REVISED APRIL 2015
PENX:R9 - PACKAGE LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PENTOXIFYLLINE
pentoxifylline tablet, film coated, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0615-4523(NDC: 0378-0357) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PENTOXIFYLLINE (UNII: SD6QCT3TSU) (PENTOXIFYLLINE - UNII:SD6QCT3TSU) PENTOXIFYLLINE 400 mg Inactive Ingredients Ingredient Name Strength D&C RED NO. 30 (UNII: 2S42T2808B) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYDROXYETHYL CELLULOSE (140 MPA.S AT 5%) (UNII: 8136Y38GY5) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) POVIDONES (UNII: FZ989GH94E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color PURPLE (Lavender) Score no score Shape OVAL (Capsule shaped) Size 16mm Flavor Imprint Code MYLAN;357 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0615-4523-39 30 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074425 09/21/2009 Labeler - NCS HealthCare of KY, Inc dba Vangard Labs (050052943) Establishment Name Address ID/FEI Business Operations NCS HealthCare of KY, Inc dba Vangard Labs 050052943 REPACK(0615-4523)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
