SEI BELLA AGE-DEFYING LIQUID FOUNDATION BEIGE- octinoxate 7.5% lotion
Sei Bella Age-Defying Liquid Foundation by
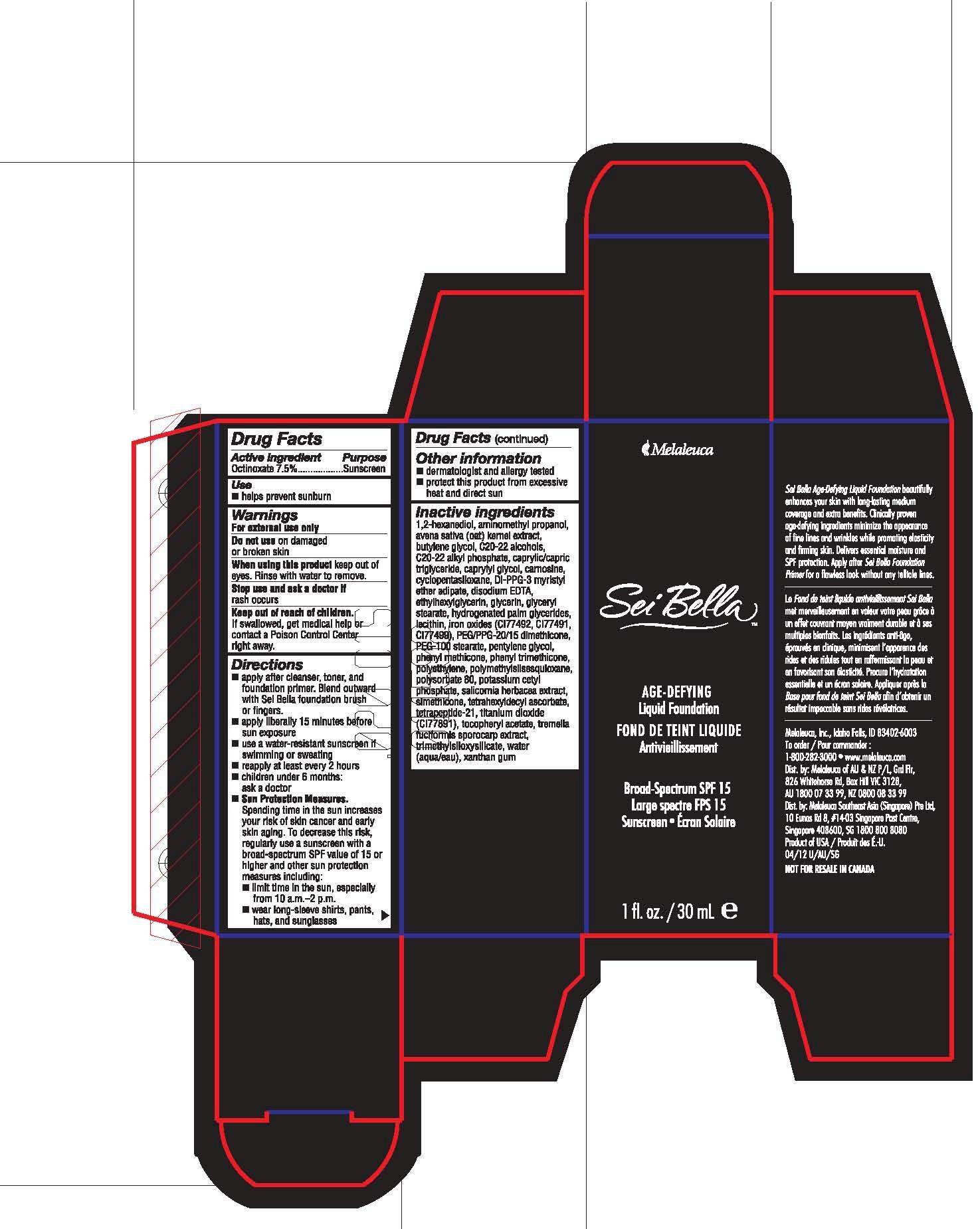
Drug Labeling and Warnings
Sei Bella Age-Defying Liquid Foundation by is a Otc medication manufactured, distributed, or labeled by Melaleuca Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active Ingredient
- Purpose
- Use
- Warnings
- WHEN USING
- STOP USE
- Keep out of reach of children.
-
Directions
■ apply after cleanser, toner, and foundation primer. Blend outward with Sei Bella foundation brush or fingers.
■ apply liberally 15 minutes before sun exposure
■ use a water-resistant sunscreen if swimming or sweating
■ reapply at least every 2 hours
■ children under 6 months: ask a doctor
■ Sun Protection Measures. Spending time in the sun increases you risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF value of 15 or higher and other sun protection measures including:
■ limit time in the sun, especially from 10 a.m.-2 p.m.
■ wear long-sleeve shirts, pants, hats, and sunglasses
-
Inactive ingredients
1,2-hexanediol, aminomethyl propanol, avena sativa (oat) kernel extract, butylene glycol, C20-22 alcohols, C20-22 alkyl phosphate, caprylic/capric triglyceride, caprylyl glycol, carnosine, cyclopentasiloxane, DI-PPG-3 myristyl ether adipate, disodium EDTA, ethylhexylglycerin, glycerin, glyceryl stearate, hydrogenated palm glycerides, lecithin, iron oxides (CI77492, CI77491, CI77499), PEG/PPG-20/15 dimethicone, PEG-100 stearate, pentylene glycol, phenyl methicone, phenyl trimethicone, polyethylene, polymethylsilsesquioxane, polysorbate 80, potassium cetyl phosphate, salicornia herbacea extract, simethicone, tetrahexyldecyl ascorbate, tetrapeptide-21, titanium dioxide (CI77891), tocopheryl acetate, tremella fuciformis sporocarp extract, trimethylsiloxysilicate, water (aqua/eau), xanthan gum
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SEI BELLA AGE-DEFYING LIQUID FOUNDATION BEIGE
octinoxate 7.5% lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 54473-228 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 2.25 g in 30 mL Inactive Ingredients Ingredient Name Strength .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) C20-22 ALCOHOLS (UNII: O4M0347C6A) C20-22 ALKYL PHOSPHATE (UNII: L4VKP0Y7RP) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNOSINE (UNII: 8HO6PVN24W) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) DI-PPG-3 MYRISTYL ETHER ADIPATE (UNII: T32481VTXW) EDETATE DISODIUM (UNII: 7FLD91C86K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) HYDROGENATED PALM GLYCERIDES (UNII: YCZ8EM144Q) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) OAT (UNII: Z6J799EAJK) PEG/PPG-20/15 DIMETHICONE (UNII: 06R6X77P9C) PEG-100 STEARATE (UNII: YD01N1999R) PENTYLENE GLYCOL (UNII: 50C1307PZG) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) SALICORNIA EUROPAEA (UNII: 6ADL50JAKW) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) TETRAPEPTIDE-21 (UNII: 179JUC43HU) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TREMELLA FUCIFORMIS FRUITING BODY (UNII: GG8N28393G) TRIMETHYLSILOXYSILICATE (M/Q 0.8-1.0) (UNII: 25LXE464L2) WATER (UNII: 059QF0KO0R) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 54473-228-01 1 in 1 BOX 01/01/2018 1 30 mL in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 10/01/2012 Labeler - Melaleuca Inc. (139760102) Registrant - Melaleuca Inc. (139760102) Establishment Name Address ID/FEI Business Operations Melaleuca Inc. 079711683 manufacture(54473-228)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.