Artificial Tears Ointment Drug Facts
Artificial Tears by
Drug Labeling and Warnings
Artificial Tears by is a Otc medication manufactured, distributed, or labeled by Rugby Laboratories Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ARTIFICIAL TEARS- white petrolatum mineral oil ointment
Rugby Laboratories Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
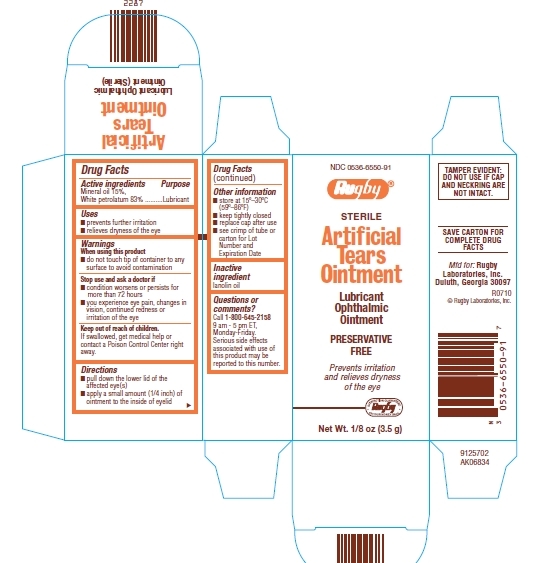
Artificial Tears Ointment Drug Facts
Warnings
When using this product
- do not touch tip of container to any surface to avoid contamination
Stop use and ask a doctor if
- condition worsens or persists for more than 72 hours
- you experience eye pain, changes in vision, continued redness or irritation of the eye
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- pull down the lower lid of the affected eye (s)
- apply a small amount (1/4 inch) of ointment to the inside of eyelid
Other information
- store at 15° - 30°C (59° - 86°F)
- keep tightly closed
- replace cap after use
- see crimp of tube or carton for Lot Number and Expiration Date
Questions or comments?
Call 1-800-645-2158
9 am - 5 pm ET, Monday-Friday.
Serious side effects associated with use of this product may be reported to this number.
Package/Label Principal Display Panel
NDC: 0536-6550-91
Rugby®
STERILE
Artificial Tears Ointment
Lubricant Ophthalmic Ointment
PRESERVATIVE FREE
Prevents irritation and relieves dryness of the eye
Rugby®
Net Wt. 1/8 oz (3.5 g)
| ARTIFICIAL TEARS
white petrolatum mineral oil ointment |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Rugby Laboratories Inc. (079246066) |